Abstract
Inspired by nature, superwettable material-based biosensors have aroused wide interests due to their potential in cancer biomarker detection. This mini review mainly summarized the superwettable materials as novel biosensing substrates for the development of evaporation-induced enrichment-based signal amplification and visual biosensing method. Biosensing applications based on the superhydrophobic surfaces, superwettable micropatterned surfaces, and slippery lubricant-infused porous surfaces for various cancer biomarker detections were described in detail. Finally, an insight of remaining challenges and perspectives of superwettable biosensor is proposed.
Introduction
Early diagnosis is crucial for the effectiveness of cancer treatment.1,2 The level of biomarkers in the body fluids offers possibility to distinguish the development of cancer in the early-, middle-, or late-stages.3,4 Biomarkers including proteins, mRNA, exosomes, metabolites, circulating DNA, and tumor cells can be collected from tissues, blood, and urine.5,6 Therefore, developing highly selective and sensitive methods for biomarkers is desired to be accurate and promising approach for the screening, diagnosis, prognosis, and recovery on treatment of cancer.
In recent years, substantial and significant progress has been made for cancer biomarker detection. Different promising detection methods have been developed, such as enzyme-linked immunosorbent assay, 7 polymerase chain reaction, 8 electrophoresis, 9 surface enhanced Raman spectroscopy (SERS), 10 fluorescence methods, 11 electrochemical assay,12,13 colorimetric assay, 14 etc. As one kind of emerging method, the bio-inspired superwettable surfaces are used to develop biosensors with advantages of simple, portability, high flexibility, and easy-to-use.15,16
Learning from nature is an eternal theme from the past to the present. Special wetting behaviors have drawn a lot of attention as a prevalent phenomenon. 17 According to the findings of Barthlott and colleagues, lotus leaves can always maintain themselves clean in unclean situations due to self-cleaning and superhydrophobicity properties generated by the microstructures and low surface energy material of wax. 18 Jiang et al discovered that micro/nano hierarchical structures and a waxy coating lead to superhydrophobicity on lotus leaves, where water droplets slide easily and randomly on such surfaces to clean contaminations. 19 Since then, springtails, bacterial (Bacillus subtilis) biofilm colonies, Namib Desert beetles (Stenocara sp.), pellicles, leafhoppers, and Nepenthes pitcher plant have all been examined.20,21 Meanwhile, bioinspired wettability-based applications such as antiicing, liquid separation, self-cleaning, antifogging, antifouling, and fog harvesting have emerged in recent decades.22–25 Here, we mainly summarize the Lotus leaf-inspired superhydrophobic surfaces, beetle-inspired patterned superwettable surfaces, and Nepenthes-inspired slippery lubricant-infused porous surfaces (SLIPS) (Figure 1) for cancer biomarker detection. Their superwettability is utilized for developing new biosensing substrates or methods. Compared to the traditional solution-based sensing approaches, superwettable biosensing mainly use droplet for the detection, which holds great potential for point-of-care testing applications.
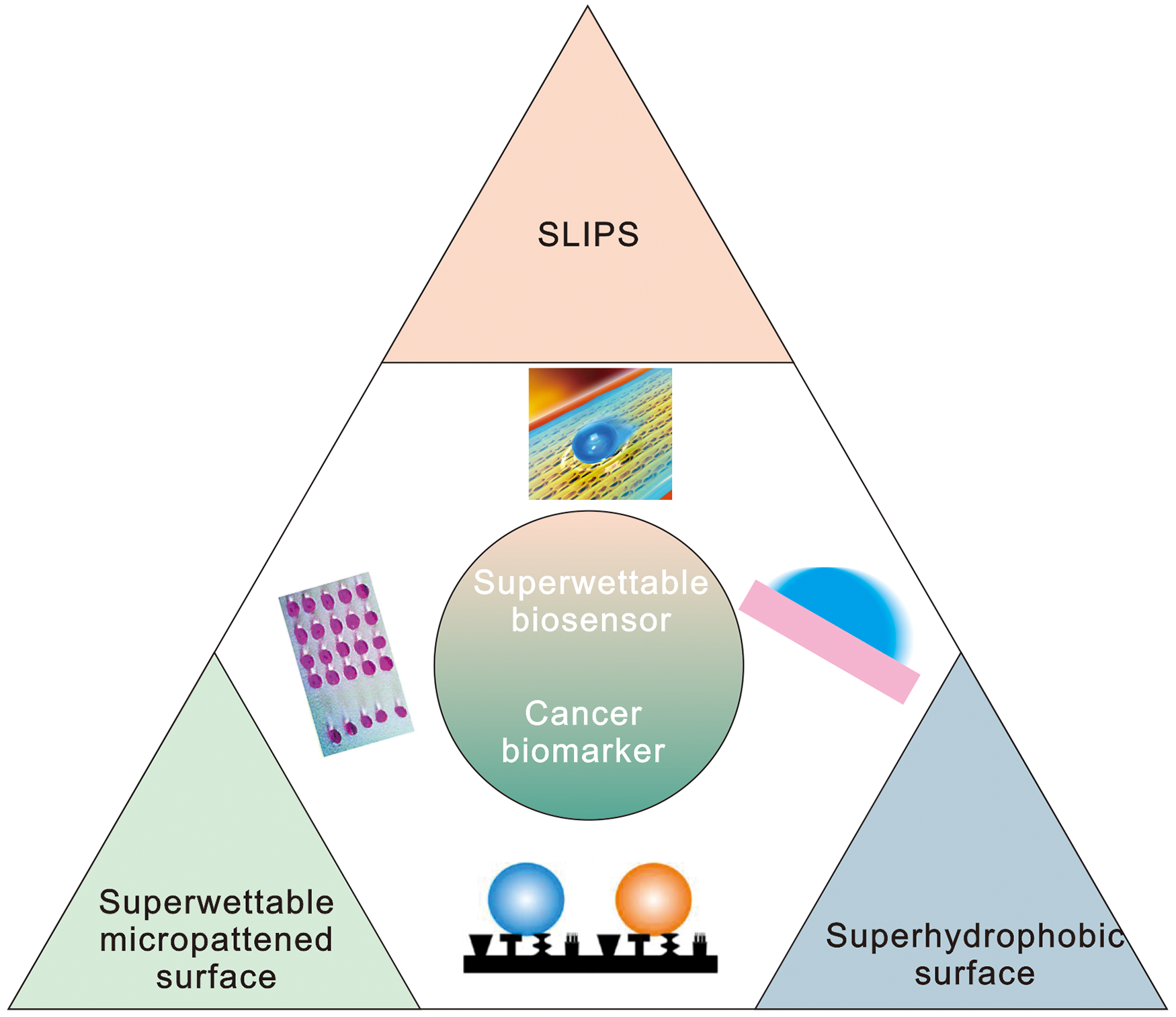
Bioinspired superwettable biosensor for the detection of cancer biomarker.
Superwettable Biosensors
Evaporation-Induced Enrichment-Based Signal Amplification
The spread, evaporation, deposition of droplets has significant influences on the biosensing performance. When a droplet is placed on a common (hydrophilic or hydrophobic) surface, it spreads rapidly and leaves coffee-ring stain after the droplet completely evaporated.26,27 To eliminate this problem, superhydrophobic surface is used as a powerful substrate for biosensing. Water droplet on the superhydrophobic surface shows a high contact angle (CA) larger than 150° with small contact area between the droplet and surface. Thus, it can be used as evaporation-induced enrichment-based signal amplification for sensitive detection. The Marangoni convection inside the droplet allows the solutes to circulate rapidly, thus, accelerating the reaction, improving the homogenization, and ultimately producing a uniform deposited pattern without coffee-ring stain. On superwettable surfaces, there are 2 traditional mechanisms of droplet evaporation. (1) Mode of constant contact area. The 3-phase contact line is pinned to the surface, and the CA steadily lowers while the solid–liquid contact area stays constant. This mode is predicted for droplets with an initial CA smaller than 90°. (2) Mode of constant CA. The CA stays unchanged, and the droplet remains spherical, although the radius and height decrease. This mode is predicted when the initial CA is larger than 90°. However, these are 2 extreme situations. One mode might change to the other at any moment during the complicated evaporation process, or 2 modes might coexist at the completion of droplet evaporation. The existence and duration of these 2 unique evaporation modes are determined by liquid content and solid surface qualities, such as chemical heterogeneity, thermal conductivity, and microstructure.
Suarasan and co-workers used soft lithography method to fabricate the polydimethylsiloxanes (PDMS) based superhydrophobic substrate with micro/nano structures. 28 Silver nanoparticles were in-situ grown on the surface to improve SERS enhancing properties. This superhydrophobic plasmonic biosensor was used for the detection of exosomes with only 0.5 μL testing sample. Di Fabrizio et al developed superhydrophobic surfaces decorated with photonics structures for the detection of exosomes. 29 Exosomes were concentrated and carried onto the active domains of the substrate, whereby they were analyzed and characterized by SERS. Li's group established a refreshable electrochemical biosensor by casting superhydrophobic conductive PDMS and multiwalled carbon nanotubes nanocomposite onto a glassy carbon electrode (Figure 2A). 30 This self-cleaning electrochemical biosensor has excellent self-cleaning property. A tumor marker, carcinoembryonic antigen, has been successfully detected by analyzing horseradish peroxidase-dependent electrochemical signals with a wide dynamic range from 0.1 to 100 mg/mL and low limit of detection (LOD) of 0.041 ng/mL. The current responsiveness of self-cleaning electrode rises 5 ± 2.4 times when compared to bare electrode due to the high conductivity and effective electrode area of the self-cleaning electrode.

(A) The preparation of self-cleaning electrode for electrochemical biosensing. 30 (B) The superwettable micropatterned microchip for the detection of free PSA. 31 (C) Non-faradaic impedance sensing approach for the detection of DNA. 35 (D) AIE-induced fluorescence enhancement for the detection of matrix metalloproteinase-2 on SLIPS. 36
As the droplet easily rolls away from the superhydrophobic surface with high uncontrollable ability, superwettable micropatterned wetting samples that possess superhydrophilic microwells on a superhydrophobic surface were designed to anchor droplets. On the superwettable micropatterned surface, the CA of the droplet on the microwell is related to the volume of the droplet and the area of the microwell. The analytes are generally dispersed homogenously in the microwells after the evaporation process due to the Marangoni effect.
Taking the advantage of evaporation-induced enrichment of fluorescence, various biomarkers can be detected by superwettable micropatterned biosensors with a high signal-to-noise ratio as compared to the conventional fluorescent probes. As shown in Figure 2B, Chen et al reported a superwettable micropatterned microchip for the detection of free prostate-specific antigen (PSA). 31 It demonstrated excellent performance with a LOD of 10 fg/mL. Superwettable micropatterned biosensors can also be applied for the detection of other kinds of biomarkers, such as miRNA32,33 and mRNA. 34
Apart from constant CA mode evaporation on superhydrophobic surface, the constant contact area mode evaporation showed significant biosensing performance as well. Alam's group developed a non-faradaic impedance sensing approach for the DNA detection by evaporating a microliter sample over a nanotextured superhydrophobic electrode array (Figure 2C). Continuous monitoring of each droplet impedance as a function of evaporation time is used to greatly enhance sensitivity to attomolar levels. 35 As the effect of evaporation-induced enrichment is a universal strategy, it can be used for the development of electrochemical, SERS, and colorimetric biosensor.
Instead of air, the SLIPS filling with lubricant fluid in the microstructures avoids the pinning effect of the 3-phase contact line and shows unique liquid-repellent ability. After droplet evaporation, analytes inside are concentrated into a small pattern. Therefore, SLIPS has significant potential in biosensing. It is worth noting that the aggregation-induced quenching (ACQ) effect might present after the droplet evaporated, resulting in the inaccurate or false positive feedback. To eliminate the ACQ problem of traditional fluorophores, Lou et al combined the evaporation-enrichment effect with the aggregation-induced emission induced fluorescence enhancement for the detection of tumor marker matrix metalloproteinase-2 on SLIPS (Figure 2D). This SLIPS-based biosensor achieved a low LOD of 3.7 ng/mL and significant selectivity. 36 The fluorescent signal on SLIPS showed the strongest enhancement: 1.25-fold higher than that of superhydrophobic substrates, 1.44-fold higher than that of hydrophobic substrates, and 4.59-fold higher than that of hydrophilic substrates.
Taken together, these superwettable biosensors demonstrate a great perspective in evaporation-induced enrichment-based signal amplification. However, most of them cannot be reused. To address this issue, nucleic acid probes with regenerated conformations can be used to develop sensitive superwettable biosensors.
Visual Biosensing Strategy Based on Superwettability
Various visual biosensing strategies were developed for the quantitative detection of cancer biomarkers. As a classic visual strategy, colorimetric assay is widely adopted due to its simple, equipment-free, and rapid advantages. However, hundreds of millions of individuals have vision problems worldwide. For example, color blindness influences as many as 8% of men and 0.5% of women. 37 Thus, colorimetric methods are not suitable for the user with color blindness or color weakness. The wetting performances, including CA and rolling/sliding angle, are the most obvious and straightforward parameters of the superwettability, these characters have been demonstrated as a powerful and promising visual strategy for cancer biomarker detection. For example, pH-sensitive superwettable biosensor was developed for the detection of PSA by direct reading the change of droplet CA (Figure 3A). 38 The pH-responsive superwettable biosensor was prepared by coating the self-assembled organosilanes functionalized silica nanoparticles onto the glass. The pH-sensitive organosilanes, 3-[2-(2-amino ethyl amino) ethyl amino] propyl trimethoxy silane, was used to impart hydrophilicity at the acidic solution due to the protonation of the pH-sensitive amino groups and octyl trimethoxy silane was employed as hydrophobic coating regardless of pH. The sensitivity was down to 3.2 pg/mL, which was significantly improved compared with traditional enzyme linked immunosorbent assays.
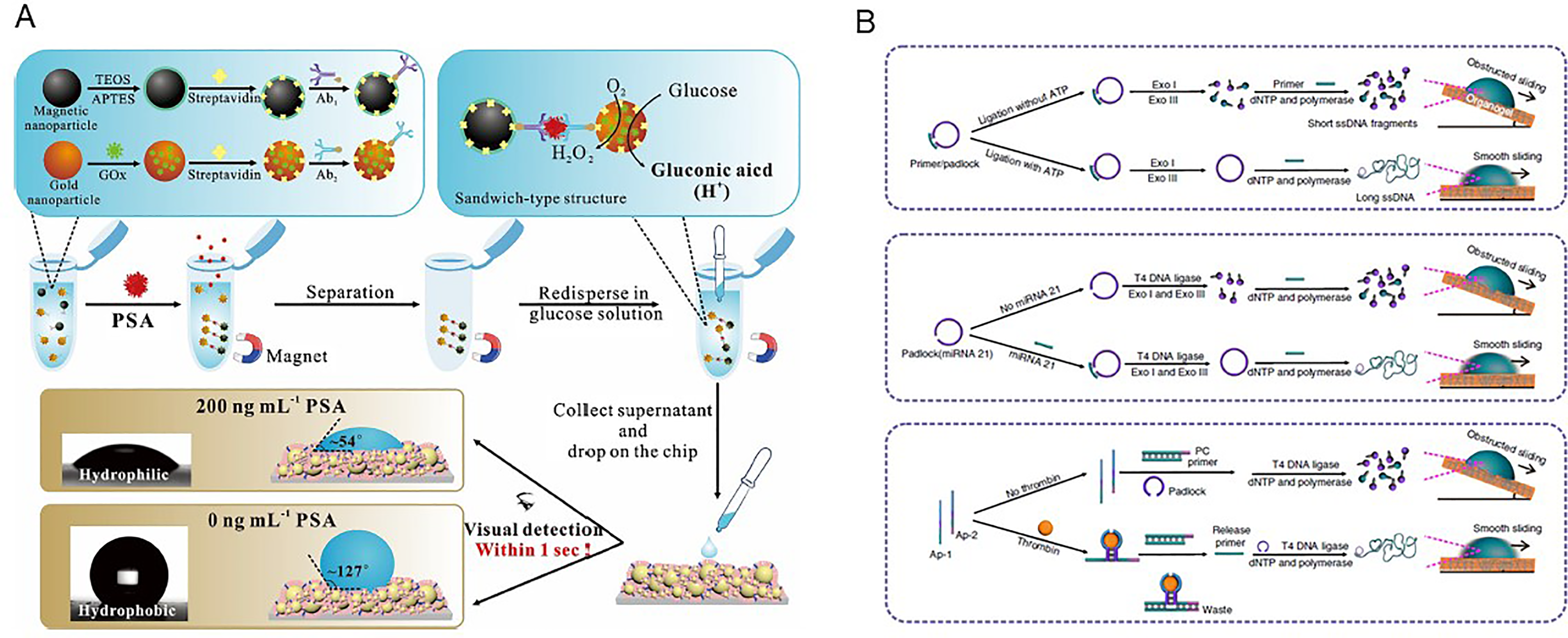
Using the target-induced rolling circle amplification to tune the interfacial adhesion between the droplet and the surface, miRNA can be detected by recording the droplet sliding angle on the SLIPS. 39 The target, such as ATP, miRNA, and thrombin, were designed to assist the completion of rolling circle amplification (Figure 3B), changing the short ssDNA fragments to long ssDNA. Thus, the hydrophobicity of DNA was reduced, leading to the weak interfacial adhesion between the droplet and the surface. According to the droplet wetting behavior, such as droplet sliding angle, speed, and displacement, various targets can be visually detected with high sensitivity. The droplet wetting behavior was hardly sensitive to external influences, such as temperature, elevation, and droplet color, indicating a great potential to numerous users including color-blind/weak people.
Taken together, these present wetting behavior-based visual diagnostics have obvious implications for creating efficient, simple, and low-cost biomarker detection techniques. The naked eye, however, cannot distinguish between minor variations in rolling/sliding angle and CA. As a result, it is preferable to create a smartphone-based digital approach for visual detection.
Conclusion
In conclusion, as a rising star of sensor, superwettable biosensor exhibited its unique performance for different kinds of cancer biomarkers. Although biosensing platforms utilize similar principles, they might show considerable differences when detecting different biomarkers. For example, most strategies require separation and enrichment step prior to detection of circulating proteins, RNA, or cells; exosomes always require pre-purification. Despite the progress of superwettable biosensors by leaps and bounds, some important issues should be addressed. Firstly, high-throughput superwettable biosensors for simultaneous detection of multiple biomarkers are challenging and promising for future application. Secondly, highly stable surfaces with robust superwettability are urgently desired for real sample application under complex and extreme biomedical conditions. Finally, current methods for fabrication require several steps, which are tedious and expensive, thus simple, easily compatible, and commercial-scale manufacturing of superwettable biosensors is advocated. Although many issues need to be solved, there are still several chances in this area. Because of the variety of substrates, droplet-based biosensors have the potential to be highly promising in point-of-care detection and wearable biosensors. It might open the door for high-throughput biosensing by merging with the liquid handling robot approach. We hope that this review attracts more attention in the scientific community and delivers fresh perspectives into superwettable biosensors.
Footnotes
Abbreviations
Author Contributions
YJY wrote the manuscript. ZFG reviewed and edited the manuscript. All authors revised the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (22176080 and 31800829) and the Natural Science Foundation of Shandong Province (ZR2018BB054).
