Abstract
Introduction
Multiple myeloma (MM) is a bone marrow malignancy, characterized by abnormal amplification of plasma cells, elevated immunoglobulin, lytic skeletal lesions, hypercalcemia, anemia, and renal failure. 1 The survival time of MM is highly variable, ranging from less than 1 month to more than 10 years. 2 Risk stratification is very important for early recognizing high-risk patients with poor prognosis, who could benefit from different therapeutic strategies and intensive surveillance.
Both International Staging System (ISS) and Revised International Staging System (R-ISS) are widely used for risk stratification in MM. R-ISS was created by incorporating serum lactate dehydrogenase (LDH) and cytogenetic abnormality into the original ISS and could improve the predictive power. 3 However, under the existing staging system, there were still numerous patients with poor prognoses who had not been early identified. 4 Moreover, the cytogenetic abnormality could only be evaluated in rather limited institutions where gene analysis could be carried out. Therefore, it is still urgent to establish more convenient and precise methods that could be used for risk stratification in MM patients.
Low triiodothyronine (T3) syndrome often occurs in severe illness, and typically presents with decreased serum free T3 (FT3) concentration, increased reverse T3 level, and normal or decreased free thyroxine (FT4), and thyrotropin (TSH) levels. 5 Low T3 syndrome has been considered an adaptive response of the body to save energy. It is not caused by intrinsic abnormalities in thyroid function, hence is also named euthyroid sick syndrome or non-thyroidal illness syndrome. 6 Recent studies have demonstrated that low T3 syndrome was related to the severity of the disease and was a prognostic marker in various diseases including trauma, 7 sepsis, 8 heart diseases,9,10 stroke, 11 liver diseases,12,13 kidney diseases, 14 HIV infection, 15 and respiratory diseases.16,17 Moreover, it has been demonstrated that low T3 syndrome could predict survival in patients with lung carcinoma, 18 brain carcinoma, 19 diffused large B cell lymphoma, 20 or lymphocytic leukemia. 21
However, to our knowledge, the predictive value of low T3 syndrome for survival in MM patients has not been reported. The present study was performed to investigate the clinical characteristics of low T3 syndrome in newly diagnosed and untreated MM patients, and to evaluate the association between baseline low T3 syndrome and survival.
Materials and Methods
Participants
A total of 229 patients with newly diagnosed and untreated MM, who underwent a thyroid function examination at our hospital from April 2009 to December 2019, were consecutively reviewed in this retrospective study. This study has obtained approval from the Ethics Committee of our institution (approval number: 2020-ke-414). The diagnosis was made according to the definition by the International Working Group on myeloma (IMWG). 2 The exclusion criteria were: patients with the previous history of abnormal thyroid function; patients who had received treatments that would affect thyroid function, including anti-thyroid drugs, levothyroxine, or amiodarone; patients with comorbidities that could directly lead to low T3 syndromes, such as severe infection, recent myocardial infarction, recent stroke or heart failure; patients whose thyroid function had met the diagnostic criteria of hypothyroidism or hyperthyroidism. Thus, 28 patients were excluded from this study. A total of 201 MM patients were finally enrolled. All patients or their families gave verbal informed consent to the use of data regarding this study. Before use, the data were anonymized, and all patient details were de-identified. The reporting of this study conformed to STROBE guidelines. 22
Data Extraction and Laboratory Evaluation
Baseline clinical characteristics were collected, including age, sex, body mass index, blood hemoglobin, neutrophil, monocyte, lymphocyte, albumin, creatinine, calcium, β2-microglobulin (β2-MG), and LDH levels, ISS and RISS stages. Blood samples for laboratory analyses were collected within 24 h after admitted to a hospital. Chromosomal abnormalities were evaluated by fluorescence in-situ hybridization. The existence of at least one of t (4;14), t (14; 16), or del (17p) was judged as a high-risk cytogenetic abnormality. Serum thyroid hormones were assessed via chemiluminescent immunoassay (Siemens Healthcare Diagnostics, USA). The normal concentrations of FT3, FT4, and TSH were 2.3 to 4.2 pg/mL, 0.89 to 1.76 ng/Dl, and 0.55 to 4.78 μIU/mL, respectively, in this study. Because the total thyroid hormone in MM patients has been spuriously elevated in previous studies,23–25 we did not include total T3 (TT3) and total T4 (TT4) for analyses in this study.
Treatment
All patients received induction therapy. A total of 139 patients received bortezomib-based treatments, 16 patients received lenalidomide-based treatments, 25 patients received bortezomib combined lenalidomide-based therapy, and 21 patients received thalidomide-based therapy. After induction therapy, 41 patients (20.40%) received autologous stem cell transplantation (ASCT).
Grouping
The patients were divided into 2 groups based on serum FT3 level: the low T3 syndrome group (FT3 below the normal range, FT3 < 2.3 pg/mL) and the non-low T3 syndrome group (FT3 ≥ 2.3 pg/mL).
We ran subgroup analyses according to different principles. First, patients were divided into 2 subgroups according to whether having received novel agents treatment (bortezomib and/or lenalidomide-based therapy) or not. Secondly, 2 subgroups according to whether having received ASCT or not. Third, 3 subgroups according to ISS stage. Fourth, 3 subgroups according to the R-ISS stage.
Survival Evaluation
Overall survival (OS) was calculated as the months from diagnosis until death due to any cause. Progression-free survival (PFS) was defined as the months from diagnosis until relapse or death due to any cause. Participants without an event until the last follow-up were classified as censored. The information was retrieved from medical records, or through interviewing patients in outpatient clinics or their families by telephone. The follow-up was started from the date of diagnosis to August 2021 or when the death occurred.
Statistical Analysis
Continuous variables with normal distribution were shown as mean ± standard deviation (SD) and compared by t-test between the low T3 syndrome group and the non-low T3 syndrome group. Continuous variables with skewed distribution, including blood creatinine, β2-MG, LDH, TSH, neutrophil/lymphocyte, and (neutrophil + monocyte)/lymphocyte ratio were shown as median (upper and lower quartiles) and compared by the Mann-Whitney test. Categorical variables were expressed as numbers and percentages and compared by Chi-square test. Pearson's or Spearman's correlation was used to analyzing the associations between thyroid hormones and baseline clinical parameters. Curves of OS and PFS were generated by the Kaplan-Meier method and compared using a log-rank test. The Cox regression was used to find independent predictive factors of OS and PFS. The predictive ability of FT3, FT4, or TSH for death or one-year death was analyzed using the receiver operator characteristic curve (ROC). All data were analyzed using IBM SPSS 21.0 software. P-value less than .05 was considered to be statistically significant.
Results
Baseline Characteristics of MM Patients With and Without Low T3 Syndrome
In total 201 patients, 80 (39.8%) patients were diagnosed with low T3 syndrome. Age, sex, body mass index, serum calcium, and LDH, and incidence of high-risk cytogenetic abnormality were similar between the 2 groups. Patients in the low T3 syndrome group had significantly lower blood hemoglobin (P = .001) and albumin (P < .001), higher serum creatinine and β2-MG concentrations (both P < .001), the higher inflammatory status shown as neutrophil/lymphocyte ratio (P = .001) and (neutrophil + monocyte)/lymphocyte ratio (P = .001), and more advanced ISS (P = .01) and R-ISS stages (P < .001, Table 1).
Clinical Characteristics of MM Patients With and Without Low T3 Syndrome.

Note: Data are presented as mean ± SD, median (upper and lower quartiles), or n (%). High-risk cytogenetic abnormality: t(4;14), t(14;16), or del(17p).
Abbreviations: BMI, body mass index; LDH, lactate dehydrogenase; ISS, international staging system; R-ISS, revised international staging system; MM, multiple myeloma.
Correlation Between Thyroid Hormones and Baseline Clinical Parameters
The relationship between serum thyroid hormones and potential associated baseline factors was explored (Table 2). Serum FT3 was positively correlated with albumin and hemoglobin levels (both P < .001), and inversely related to creatinine, β2-MG, neutrophil/lymphocyte, and (neutrophil + monocyte)/lymphocyte ratios (all P < .01).
Correlations Between Thyroid Hormone and Baseline Clinical Parameters.

Abbreviations: BMI, body mass index; LDH, lactate dehydrogenase; ISS, international staging system; R-ISS, revised international staging system.
Low T3 Syndrome and Survival
At the end of follow-up, 51 (63.75%) cases had died in the low T3 syndrome group, while 34 (28.57%) cases had died in the non-low T3 syndrome group (P < .001). OS time was significantly shorter in the low T3 syndrome group, as compared to that in the non-low T3 syndrome group (median OS: 34 months vs 85 months, P < .001, Figure 1A). A similar result was demonstrated in PFS with a median of 20 months in the low T3 syndrome group versus a median of 52 months in the non-low T3 syndrome group (P < .001, Figure 1B).
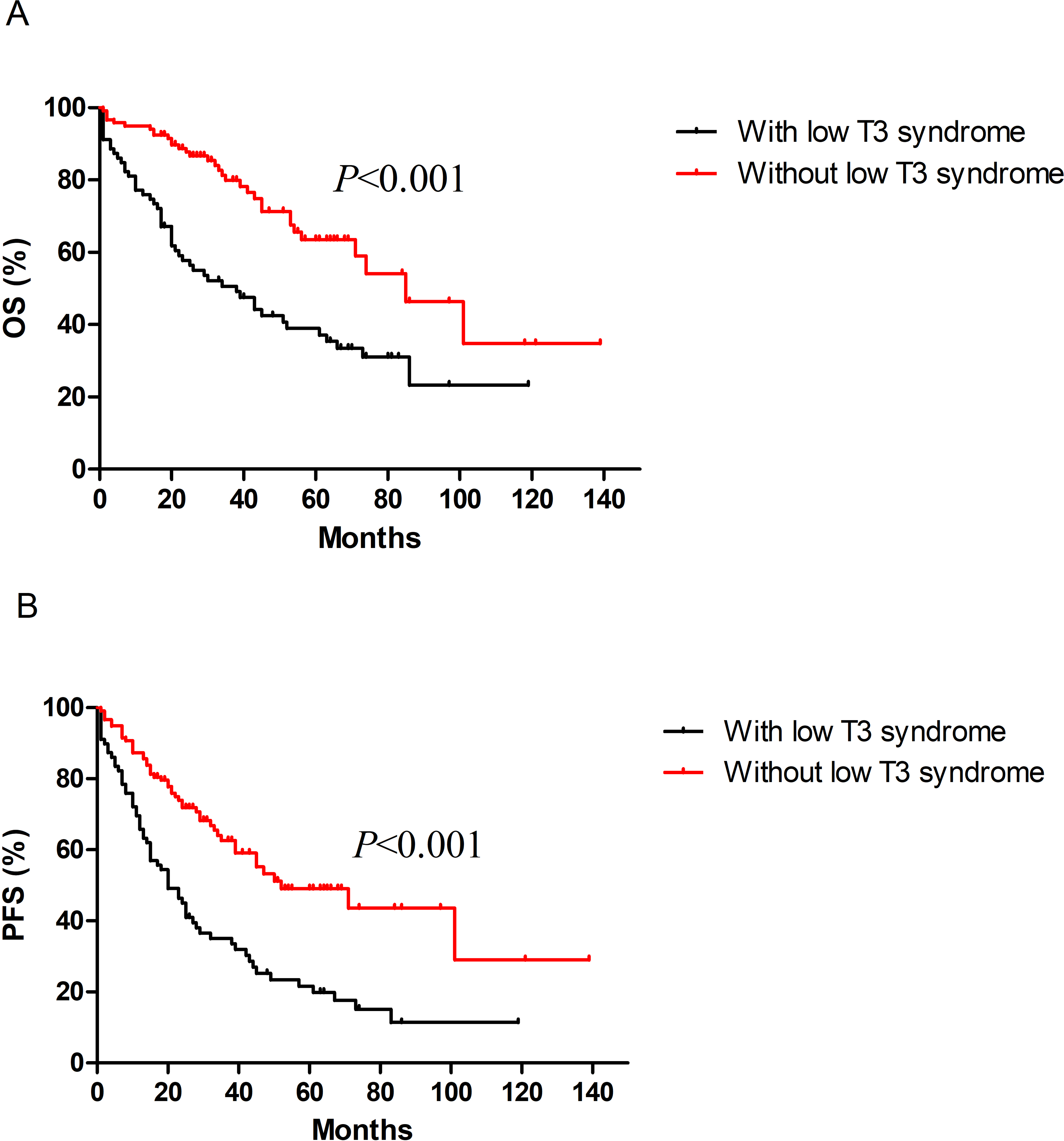
Kaplan-Meier curves of OS (A) and PFS (B) were compared between MM patients with and without T3 syndrome.
Subgroup Analyses of the Association Between Low T3 Syndrome and Survival
In patients not having received ASCT, those with low T3 syndrome had significantly shorter OS and PFS times (both P < .001, Figure 2A and B). In 41 patients having received ASCT, we did not find a significant difference in OS or PFS time between the low T3 syndrome group and the non-low T3 syndrome group (OS, P = .382; PFS, P = .173, Figure 2C and D). Patients with low T3 syndrome had significantly shorter OS and PFS times than patients without low T3 syndrome, regardless of having received novel agents treatment or not (all P < .05, Figure 2E to H). In patients with ISS II, ISS III, R-ISS II, or R-ISS III stage, those with low T3 syndrome had significantly shorter OS and PFS times (all P < .05, Supplemental Figures 1 and 2). In patients with ISS I or R-ISS I stage, there was no significant differences in OS or PFS time between the low T3 syndrome group and the non-low T3 syndrome group (Supplemental Figures 1 and 2).

Subgroup analyses of OS and PFS in relation to low T3 syndrome in patients having or not having received autologous stem cell transplantation (ASCT) (A to D), in patients not having received novel agents (thalidomide based therapy) (E, F), or in patients having received novel agents (bortezomib and/or lenalidomide based therapy) (G, H).
Univariate and Multivariate Cox Regression for Survival
In univariate analyses, age, albumin, creatinine, LDH, ISS stage, R-ISS stage, and low T3 syndrome were significantly associated with OS; age, R-ISS stage, and low T3 syndrome were significantly associated with PFS (Table 3). Accordingly, age, albumin, creatinine, LDH, ISS stage (or R-ISS stage), and low T3 syndrome were included as covariates in multivariate Cox regression for analyzing independent factors associated with OS, while age, R-ISS stage, and low T3 syndrome were included for analyzing independent factors associated with PFS (Table 4). Multivariate Cox regression revealed that low T3 syndrome and age were independent factors associated with both OS and PFS (all P < .05).
Univariate Cox Analyses of Baseline Parameters Associated With OS and PFS.

Abbreviations: OS, overall survival; PFS, progression-free survival; β2-MG, β2-microglobulin; LDH, lactate dehydrogenase; ISS, international staging system; R-ISS, revised international staging system.
Multivariate Cox Analyses of Baseline Parameters Associated With OS and PFS.

Model a: age, albumin, creatinine, LDH, ISS stage, and low T3 syndrome were included as covariates for analyzing independent factors associated with OS. Model b: age, albumin, creatinine, LDH, ISS stage, and low T3 syndrome were included as covariates for analyzing independent factors associated with OS. Age, R-ISS stage, and low T3 syndrome were included as covariates in multivariate Cox regression for analyzing independent factors associated with PFS.
Abbreviations: OS, overall survival; PFS, progression-free survival; β2-MG, β2-microglobulin; LDH, lactate dehydrogenase; ISS, international staging system; R-ISS, revised international staging system.
ROC Analyses of Thyroid Hormones for Predicting Death
We plotted the ROC to evaluate the predictive abilities of FT3, FT4, and TSH for death during the entire follow-up period and during 1 year (Figure 3). FT3 had the largest area under the curve (AUC) and had statistical significance (AUC = 0.720 for death, P < .001; AUC = 0.747 for one-year death, P < .001). The optimal cut-off of FT3 for predicting death was 2.36 pg/mL and that for predicting one-year death was 2.34 pg/mL.

Receiver operator characteristic analyses of FT3, FT4, and TSH for predicting death during the entire follow-up period (A) and during one year (B) in MM patients. FT3 had the largest area under the curve (AUC) and had statistical significance (AUC = 0.720, 95% CI 0.647-0.792 for death, P < .001; AUC = 0.747, 95% CI 0.665-0.829 for one-year death, P < .001). The optimal cut-off value of FT3 for predicting death was 2.36 pg/mL and that for predicting one-year death was 2.34 pg/mL.
Discussion
In this study, 39.8% of patients with newly diagnosed MM had low T3 syndrome. Patients with low T3 syndrome had lower hemoglobin and albumin, higher creatinine and β2-MG levels, higher inflammatory status, and more advanced ISS and R-ISS stages. Serum FT3 was positively correlated with albumin and hemoglobin, and inversely related to creatinine, β2-MG, and inflammatory status. Low T3 syndrome was a strong prognostic factor for both OS and PFS. Furthermore, patients with low T3 syndrome had inferior OS and PFS in ISS II, ISS III, R-ISS II, and R-ISS III subgroups. ROC analyses revealed that serum FT3 was a significant predictor of death and one-year death. To our knowledge, this study firstly showed the association between low T3 syndrome and survival in MM patients.
The incidence of low T3 syndrome reported in malignant tumors varied greatly. Low T3 syndrome incidence was identified to be about 43% in newly diagnosed lung cancer, 18 74% in brain tumor, 19 and 16.5% in breast cancer. 26 Regarding hematological tumors, 12.8% of patients with diffuse large B cell lymphoma, 20 and 14.34% of patients with chronic lymphocytic leukemia 21 had low T3 syndrome. Among 201 MM patients included in the present study, 80 (39.8%) had low T3 syndrome. This incidence was moderate compared with those in solid tumors shown in the above studies but was higher than those in 2 previously reported hematological tumor studies.
Inflammation, malnutrition, and hypoxia have been shown to play a crucial role in the pathogenesis of low T3 syndrome.6,27 Several cytokines have been reported to inhibit the expression of type 1 deiodinase and therefore reduce conversion from T4 to T3. 28 Continuous infusions of both interleukin (IL)-1 and IL-6 could suppress hypothalamic-pituitary-thyroid axis in rats. 29 In this study, low T3 syndrome patients had lower hemoglobin and albumin levels, higher creatinine, higher inflammatory status shown by neutrophil/lymphocyte ratio and (neutrophil + monocyte)/lymphocyte ratio. Moreover, serum FT3 was positively correlated with protein nutrition (albumin) and anemia indicator (hemoglobin), and negatively correlated with renal function marker (creatinine) and inflammatory status. These evidences indicated that multiple factors, consisting of elevated inflammation, protein malnutrition, anemia (which might render hypoxia), and renal dysfunction, were probably connected with the occurrence of low T3 syndrome in MM.
We showed that low T3 syndrome was an independent prognostic factor for both OS and PFS. In subgroup analyses, we also found that low T3 syndrome conferred inferior OS and PFS, regardless of having received novel agents treatment or not. In patients not having received ASCT, those with low T3 syndrome also had significantly shorter OS and PFS times. Moreover, low T3 syndrome could further discriminate survival outcomes in patients with ISS II, ISS III, R-ISS II, and R-ISS III stages. ROC analyses showed that FT3 was a significant predictor for both death and one-year death. The optimal cut-off values of FT3 for predicting death and one-year death were 2.36 pg/mL and 2.34 pg/mL, respectively. Both above values were close to the lower limit of the FT3 normal range (2.3 pg/mL), which also was the value used to define low T3 syndrome in this study. Low T3 syndrome might be useful in risk stratification in future clinical practice of MM.
As to why low T3 syndrome at baseline was strongly associated with survival in MM patients, we suggested that it was probably due to the correlations of low T3 syndrome with not one but several known prognostic factors. Recent clinical studies revealed that inflammation was related to shorter survival in MM.30–32 Blood β2-MG, albumin, and hemoglobin levels were associated with the prognosis in MM. 33 Several studies have found inferior OS in patients with renal dysfunction.34–36 The higher inflammation state, serum β2-MG, and creatinine levels, lower hemoglobin and albumin levels, and more advanced ISS and R-ISS stages in low T3 syndrome patients in this study suggested that elevated inflammation, malnutrition, anemia, renal dysfunction, and more advanced disease severity stage probably could explain the predictive ability of low T3 syndrome for the prognosis in MM.
In the acute phase of illness, low T3 syndrome was considered as the body's protective response, which could reduce excessive catabolism and energy requirement. However, when diseases entered a chronic phase, low T3 syndrome has been reported to become disadvantageous.37,38 In MM (as a prolonged consuming disease), persistent low T3 syndrome might affect the recovery of malnutrition and anemia. Moreover, corticosteroid has been shown to suppress serum TSH value and induce a low thyroid hormone state. 39 Large dose corticosteroid, which was commonly used in the therapy of MM, might aggravate the low T3 state during subsequent treatment. In addition, it has been reported that thalidomide therapy could induce hypothyroidism. 40 Therefore, the treatment of MM might induce further lowering of thyroid hormone and aggravate poor prognosis in patients with low T3 syndrome.
The strengths of this study included (1) the description of clinical characteristics of low T3 syndrome in untreated MM patients; (2) the novel observation of the relationship between the presence of low T3 syndrome and elevated inflammation, malnutrition, anemia, renal dysfunction, and more advanced disease severity stage in untreated MM patients; (3) the finding of the association between baseline low T3 syndrome and inferior survival in MM.
There were several limitations in the present study. The main limitation was the single-center retrospective nature of this study. Second, the sample size estimation for this study was not performed. As the number of patients having received ASCT or patients with ISS I or R-ISS I stage was small, we might not have sufficient power to get a reliable statistical result in subgroup analyses in these patients. Third, the wide distribution of diagnosis time and the variable induction therapies might induce much heterogeneity. In addition, we only assessed low T3 syndrome at the time of diagnosis and did not continuously get information on thyroid function during follow-up.
Conclusions
We found that in newly diagnosed MM patients, those with low T3 syndrome had lower hemoglobin and albumin, higher creatinine and β2-MG, higher inflammatory status, and more advanced ISS and R-ISS stages. MM patients with low T3 syndrome at baseline had inferior PFS and OS times. Considering that thyroid hormone could be easily measured, low T3 syndrome might be a useful prognostic marker in future clinical practices of MM.
Supplemental Material
sj-docx-1-tct-10.1177_15330338221094422 - Supplemental material for The Association Between Low T3 Syndrome and Survival in Patients With Newly Diagnosed Multiple Myeloma: A Retrospective Study
Supplemental material, sj-docx-1-tct-10.1177_15330338221094422 for The Association Between Low T3 Syndrome and Survival in Patients With Newly Diagnosed Multiple Myeloma: A Retrospective Study by Qingrong Pan, Yuan Jian, Yeqing Zhang, Wenkai Zhang, Zhe Chen, Yanna Yang, Aijun Liu and Guang Wang in Technology in Cancer Research & Treatment
Footnotes
Abbreviations
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was funded by a grant from the Chinese National Natural Science Foundation (no. 81770792) and the Beijing Municipal Administration of Hospitals Clinical Medicine Development of Special Funding Support (Yangfan Program, no. DFL20180301).
Ethics Approval
This study has obtained approval from the Ethics Committee of Beijing Chao-yang Hospital (approval number: 2020-ke-414). All patients or their families gave oral informed consent to the use of data regarding this study.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
