Abstract
Introduction
Esophageal squamous cell carcinoma (ESCC) is a devastating malignant disease causing a significant public health problem worldwide, with an approximate incidence of 570,000 new cases and 500,000 deaths in 2018. 1 In China, the rate of esophageal cancer incidence has been increasing due to the lack of suitable selective therapies to enhance overall survival, and progression free survival.2,3
Concurrent chemoradiotherapy (CCRT) is significantly a preferred method of treatment against locally advanced stage III and IV ESCC. 5 Advances in the radiotherapy, especially the invention of Intensity-modulated radiation therapy (IMRT) and TOMOTherapy Hi·Art, have been proven to promote a better mitigation in tumor growth in several patients. However, the improvement in OS and PFS of ESCC patients’ is poor. 4 The 5-year survival rate of advanced ESCC patients is still only 23% to 34%, 6 which typically delineates the requirement of efficient treatment strategies to enhance OS and PFS in patients’ to improve clinical outcomes. Improvements in comprehensive application of new technologies such as genomics, next-generation sequencing, big data cloud computing, artificial intelligence and biopharmaceuticals, led to the significant implications of precision medicine for the treatment of cancer.
Tumor progression can be fostered by the angiogenesis.7,8 A plethora of studies delineated that receptor tyrosine kinases (RTKs), typically involved in mediating tumor cell progression, as they can act as ‘high-affinity cell surface receptors’ for the binding of polypeptide growth factors, cytokines, and hormones.10,11 Modulating the activity of RTKs [VEGF, EGFR, FGFR, DDR, RET receptor, Eph receptor] could be considered as an attractive therapeutic target due to their involvement in tumor angiogenesis in promoting a variety of solid tumors, such as breast cancer, 12 lung cancer, 13 colorectal cancer 14 and head and neck cancer.15,16 Therapeutic efficacy of RTKs inhibitors17,18 have been examined including reversibility (erlotinib and gefitinib) and irreversibility (afatinib, dacomitinib, and osimertinib), 11 which further signify the need to decipher the combinatorial efficacy of these drugs in the treatment of ESCC. However, the performance of the targeted RTKs inhibitors is typically mixed; for instance, Dutton et al. reported the failure of gefitinib treatment in enhancing the overall survival time in advanced oesophageal cancer. 20 Conversely, another study performed a phase 2, single-arm, multicenter trial and proved the efficacy of icotinib in the patients with advanced ESCC. 21 Anlotinib is a multi-target receptor tyrosine kinase (RTK) inhibitor which can inhibit VEGFR1, VEGFR2, VEGFR3, c-kit, PDGFRβ kinase activity. 22 Anlotinib exhibits dual pharmacological effects by impairing angiogenesis and tumor cell proliferation. 23 Considering the great potential of anlotinib in clinical application, we conducted this retrospective study to evaluate the therapeutic efficacy and clinical safety of anlotinib in combinatorial regimen with concurrent chemoradiotherapy when treating advanced ESCC patients.
Methods
Eligibility Criteria
Patients with stage III and IV ESCC who were enrolled & received combinatorial regimen of ‘CCRT and anlotinib therapy’ at the third People's hospital of Zhengzhou, the First Affiliated Hospital of Zhengzhou University, Anyang Cancer Hospital, and the Affiliated Hospital of Qingdao University during August 2018 to April 2019 were considered in this retrospective study. This study was approved by the Joint Ethics Approval Committee of the third People's hospital of Zhengzhou. We obtained informed consent in written format for all the patients and de-indentified patient details, and we guarantee that the patient data will be kept confidential. Work was reported as per the requirements of the STROBE guidelines. Eligibility criteria were selected according to individual age between 18–70 years, a Karnofsky Performance Score (KPS) of 70 or higher, the stage III or IV ESCC was confirmed by pathology or endoscopy. Patients were excluded if they exhibit the following: history of radiotherapy, chemotherapy or surgical treatment, high risk of tumor bleeding, esophageal perforation and hoarseness, the expected survival time < 6 months; heart, or pulmonary, or liver and kidney dysfunction, myelosuppression (ie, neutrophil count ≤1500/mm3, platelet count ≤10 × 104/mm3, hemoglobin ≤8.0 g/dL, creatinine ≤1.2 mg/dL); missing clinical information.
A total of 45 patients were met the above criteria and received combinatorial regimen of anlotinib and CCRT, named as ‘anlotinib + CCRT group’. 45 patients with stage III and IV ESCC who received CCRT alone at the same time and also met the above criteria were selected from the four hospitals as ‘CCRT group’.
Treatment
The treatment of the CCRT group (45 patients) was as follows: (1) Radiotherapy: all patients treated with IMRT, (Varian TrueBeam linear accelerator), and 6MV-X rays were used as the radiation energy. The patients were in the supine position, and their arms extended on to the side or alternately the elbows placed in front of forehead; afterwards, the thermoplastic phantom was in a fixed position, the neck and upper segment in patients were used the head, neck and shoulder masks for fixation, whereas body masks for middle and lower patients. Subsequently, the enhanced CT simulation recommended scanning from the top of the head to the lower edge of the costal arch, and eclipse treatment planning system was performed for conventionally fractionated radiotherapy. Later, 95% PTV 60~64Gy/1.8~2.0Gy, once a day, 5 times a week, or 95% PTV 50Gy/1.8~2.0 Gy, sequential 95% PGTV 10~14Gy/1.8~2.0 Gy, once a day, 5 times a week was preferred as the treatment regimen. After the radiotherapy plan was formulated, the target area plan was verified, confirmed and reviewed by the radiation oncologists, doctors, physicists, and technicians. (2) Chemotherapy: Paclitaxel 45~60 mg/m2, d1; Carboplatin AUC 2, was given in a combinatorial regimen with nedaplatin 20–25 mg/m2, d1, in total a cycle of 7 days for 5 to 6 cycles.
Another group of 45 patients treated with combinatorial regimen of anlotinib with CCRT group: In this group, the oral anlotinib hydrochloride capsules (Chia-tai Tianqing Pharmaceutical Co., Ltd [China], Specification: 12 mg) was administered during concurrent radiotherapy and chemotherapy. The recommended dose was 12 mg each time, once in a day, orally prior to the breakfast for 2 consecutive weeks and withdrawal for 1 week, 3 weeks as a cycle of treatment, during the occurrence of unexpected disease progression or intolerable adverse events.
Toxicity Monitoring
Toxicities were monitored every week during treatment according to Common Terminology Criteria for Adverse Events (CTCAE) version 5.0 (2017); both acute (<90 days from the start of treatment) and late (>90 days from the beginning of treatment) side effects were recorded. Toxicities include bone marrow suppression, esophagitis, radiation pneumonitis, gastrointestinal disorders, hypertension, and hand-foot skin reaction.
Clinical Responses & Outcomes
Short-term responses were evaluated one month after the treatment termination. According to the response evaluation criteria in solid tumors (RECIST 1.1), short-term responses were divided into complete remission (CR), partial remission (PR), stable disease (SD), and disease progression (PD). 20
The primary outcome and secondary outcome were overall survival (OS) time, and progression-free survival (PFS) evaluated.
Statistical Methods
SPSS 21.0 was used for all data processing, and two-sided tests were performed for all statistical tests. Significance was defined as P < 0.05. The univariate relationship of patients’ general information, adverse reactions, and short-term curative effect were tested by χ2 test, Fisher's exact test and rank-sum test. This study was a retrospective type and the sample size depends on the statistical methods. For χ2 test, the minimum sample size requirement was that the expected frequency of any cell in the Table is greater than 5. If expected frequency of any cell was less than 5, then we apply the Fisher's exact test in our methods. So, the sample size was enough and executed the sample analysis for this study. Survival analysis was performed by the Kaplan-Meier method, and comparison between the two groups was executed using the log-rank test.
Results
Patient Characteristics
The patients’ characteristics were listed in Table 1. General information include: gender (χ2 = 0.403, P = 0.672), age (χ2 = 0.741, P = 0.519), clinical stage (χ2 = 0.799, P = 0.699), grade (χ2 = 2.195, P = 0.352), site (χ2 = 0.450, P = 0.739), KPS (χ2 = 1.515, P = 0.705). All differences were significant between the CCRT group and the ‘group receiving a combination of anlotinib with CCRT’.
Baseline Characteristics for CCRT and ‘Anlotinib in Combination with CCRT Group’.

Abbreviations: CCRT, Concurrent Chemoradiotherapy; KPS, Karnofsky.
Efficacy Evaluation
All the patients were recorded for efficacy evaluation. The cutoff date was April 2021. The PFS in the CCRT group (2 months, 95% CI, 1.8-3.1) was shorter than the group receiving a combination of ‘anlotinib with CCRT’ (5 months, 95% CI, 3.1-7.0). The hazard ratio (HR) of PFS was 0.577 (95% CI, 0.333-0.902). The difference was statistically significant (P = 0.014), as shown in Figure 1.

Kaplan-Meier plot for progression free survival (PFS) in the CCRT group and ‘Anlotinib + CCRT group; Abbreviations: CI- Confidence Interval; HR- Hazard Ratio.
The median OS time was 5 months (95% CI, 4.1-7.5) for the CCRT group, whereas 9 months (95% CI, 7.3-18.0) for the group, which received combination of ‘anlotinib with CCRT’, the difference was statistically significant (HR = 0.578, 95% CI, 0.337-0.924, P = 0.021) as shown in Figure 2.
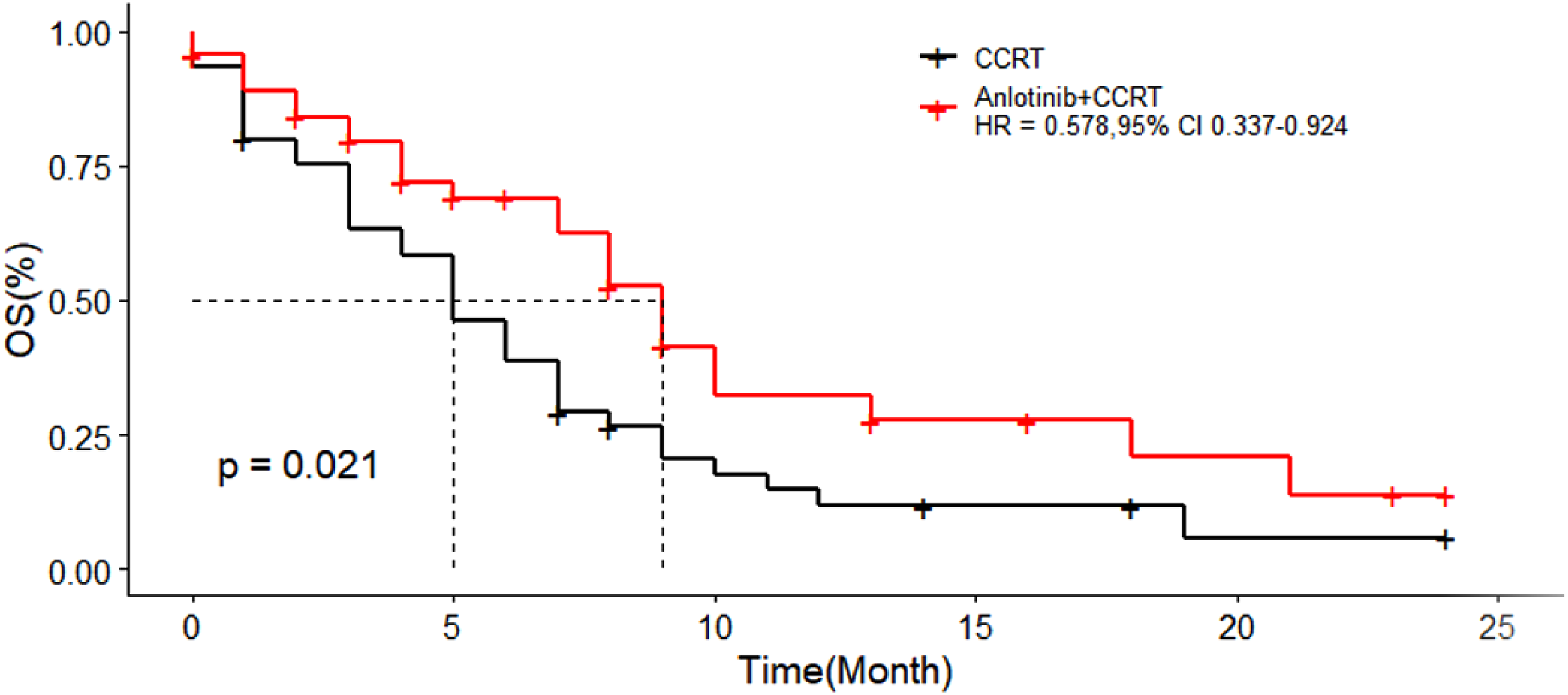
Kaplan-Meier plot for overall survival (OS) in the CCRT group and the Anlotinib + CCRT group; Abbreviations: OS-Overall Survival; CI-confidence interval; HR-hazard ratio.
The objective response was evaluated at 3, 6, and 12 months after the treatment respectively. Total 35 patients exhibited complete responses at 12 months, whereas no patient exhibited a partial response, and 1 patient exhibited a stable disease, and 9 patients exhibited progressive disease in the CCRT group. Total 41 patients exhibited a complete response, 2 patients exhibited a partial response, whereas 1 patient exhibited a stable disease, and 1 patient exhibited progressive disease in the group received ‘anlotinib with CCRT’ (Table 2); Therefore, 35 patients exhibited response, and 10 patients exhibited non-response in CCRT group whereas 43 patients exhibited a response, whereas 2 patients exhibited non-response in the group received ‘anlotinib with CCRT’ (Table 3). However, there was no statistically significant difference between CCRT and the group that received ‘anlotinib with CCRT’ at 3 months (P1 = 0.895, P2 = 1). However, there was a statistical difference between the two groups at 6 months (P1 = 0.027, P2 = 0.015) and 12 months (P1 = 0.012, P2 = 0.027).
Comparative ‘Objective Responses’ in the CCRT Group and the Group Received the Combinatorial Regimen of Anlotinib and CCRT.

Response or Non-Response in CCRT and Anlotinib + CCRT Group.

Safety Evaluation
All the 90 patients were evaluated for adverse events (Table 4). In the CCRT group, the incidence of leucopenia, thrombocytopenia, anemia, gastrointestinal disorders, radiation esophagitis, radiation pneumonitis, and hypertension were 55.55%, 26.67%, 11.11%, 35.56%, 22.22%, 46.67%, 11.11%, respectively; and no patients were experienced any hand-foot skin reaction. In the group received combination of ‘anlotinib with CCRT’, the incidence of leucopenia, thrombocytopenia, anemia, gastrointestinal disorders, radiation esophagitis, radiation pneumonitis, hypertension, and hand-foot skin reaction were 57.77%, 26.67%, 15.56%, 46.67%, 31.11%, 48.89%, 44.44%, and 28.89% respectively. There was no significant difference observed in the incidence of leukopenia, thrombocytopenia, anemia, gastrointestinal disorders, radiation esophagitis and radiation pneumonitis between the CCRT group and the group received combination of ‘anlotinib with CCRT’. However, the incidence of hypertension in the group receiving anlotinib with CCRT was higher than the CCRT group (P = 0.023). There were no patients who experienced hand-foot skin reactions in the CCRT group, while 13 patients who exhibited this adverse effect in the group received anlotinib in combination with CCRT.
Toxicity of Patients in ‘Combinatorial Group Received Anlotinib and CCRT Group’ than CCRT Group.

Abbreviations: NA, not application
Discussion
Although concurrent chemoradiotherapy is minimally effective for treating the advanced ESCC patients, there are still lapses for delivering effective clinical outcomes by treating locally advanced ESCC patients. 24 A phase II trial of chemoradiotherapy in advanced ESCC patients has been reported that the median survival time was only 305.5 days, whereas the 2-year survival rate was 31.5%. 25 Hence, it is crucial to increase the efficacy and reduce recurrence and metastasis of advanced ESCC patients with immunotherapy and concurrent chemoradiotherapy.
In recent years, significant progress has been made in the targeted immunotherapy of ESCC, which provides a new option for ESCC patients. For instance, 26 the higher disease control rate was associated typically due to the modulated EGFR expression in ESCC patients treated with gefitinib in a phase 2 trial. Another phase 3 trial by Janmaat, ML et al. reported that gefitinib did not prolong overall survival time for advanced esophageal cancer but produced palliative benefits in the patients with short life expectancy.20 Subsequently, Lorenzen S et al. carried out a randomized phase II study to compare the efficacy of ‘cisplatin and 5-fluorouracil (CF) combinatorial regimen with cetuximab’ and CF alone in the ESCC patients with advanced stages. The results of this study delineated that cetuximab could be considered as safer and enhanced the clinical efficacy of ‘cisplatin and 5-fluorouracil’ chemotherapy. 27 Lu et al. reported that administration of nimotuzumab to the standard paclitaxel (T)/cisplatin (P) regimen could significantly improve objective response rate (ORR), duration of disease control (DDC) and OS in the locally advanced ESCC patients. 28 However, due to the limited efficacy of the targeted immunotherapy, only minimal clinical evidence is currently available pertinent to the combination of molecular immunotherapy and concurrent chemoradiotherapy.
Anlotinib administration has been proven effective to act against lung cancer, advanced soft tissue sarcoma (STS), thyroid cancer, metastatic renal cell carcinoma (mRCC). 29 A multicenter phase II trial suggested that stage IV ESCC patients treated with anlotinib typically exhibited a better PFS and DDC than those treated with placebo. 30 Another multicenter phase III study [NCT02388919] delineated the efficacy of anlotinib in 437 patients with advanced NSCLC and this drug facilitated the prolongation of OS (9.6 vs 6.3 months) and PFS (5.4 vs 1.4 months) when compared to placebo. 31 Our studies are in line with the above reports.
Shi et al. demonstrated the strongest anti-tumor responses of ‘anlotinib combination with chemoradiotherapy’ when compared to other groups using patient-derived xenografts.4 Hence, anlotinib was recommended to treat advanced ESCC patients by the Chinese Society of Clinical Oncology guidelines in 2019 due to its substantial therapeutic efficacy. 32 Our present study assessed the safety and efficacy of anlotinib as complementary drugs for concurrent chemoradiotherapy with advanced ESCC patients. The results showed that the PFS (5 months, 95% CI, 3.1-7.0 vs 2 months, 95% CI, 1.8-3.1, P = 0.014), OS (9 months, 95% CI, 7.3-18.0 vs 5 months, 95% CI, 4.1-7.59 months, 95% CI, 7.3-18.0, P = 0.021) were comparatively better in the group received ‘combinatorial regimen of anlotinib with CCRT’ group when compared to CCRT group. In case of the objective responses, there was no significant difference between the two groups at 3 months after the treatment termination, but comparatively a higher number of patients were shown ‘complete response’ in the group received combination of anlotinib and CCRT than the CCRT group at 6 months and 12 months after the treatment termination respectively. Therefore, our study is inferring that it could typically require at least 3 months for anlotinib to show its therapeutic efficacy after the administration.
The incidence of adverse events during therapeutic regimen is considered as the critical factor that can induce patients’ compliance. The adverse events reported during of anlotinib treatment were hypertension, elevated thyroid-stimulating hormone, thyroglobulin, total cholesterol elevation, diarrhea, and hand-foot skin reaction. 33
Jingzhen Shi et al. (2020) reported the efficacy of anlotinib in mitigating the tumor growth by apoptosis induction and angiogenesis by fostering the apoptosis using patient tumor-derived xenografts (PDX) models. 4 This study concluded the angiogenesis blockade can have a significant role in ESCC treatment. 4 In our study, among 45 patients who received combinatorial regimen of anlotinib and CCRT treatment, total 10 patients were experienced ‘hand-foot skin reaction’ in grade 1∼2, whereas 3 patients experienced in grade 3∼4 hand-foot skin reaction; there were no patients experienced hand-foot skin reaction in the CCRT group. However, there was no adverse events-related mortality in the groups. Other adverse events in the group receiving anlotinib with CCRT were not increased when compared to the CCRT group. Hence, it can be concluded that anlotinib can exhibit its manageable tolerability in our study.
Conclusion
Past reports delineated the efficacy of immune checkpoint inhibitors against advanced esophageal carcinoma as multiline therapeutic modality. The combination of anlotinib with concurrent chemoradiotherapy exhibited significant clinical efficacy, safety and may benefit for treating the locally advanced ESCC patients.
Supplemental Material
sj-pdf-1-tct-10.1177_15330338221080939 - Supplemental material for Comparative Clinical Efficacy of ‘Concurrent Chemoradiotherapy (CCRT) and Anlotinib’ Than CCRT in Patients with Locally Advanced ESCC
Supplemental material, sj-pdf-1-tct-10.1177_15330338221080939 for Comparative Clinical Efficacy of ‘Concurrent Chemoradiotherapy (CCRT) and Anlotinib’ Than CCRT in Patients with Locally Advanced ESCC by Gang Wang, Narasimha M. Beeraka, Wenjing Xiao, Yaowen Zhang, Nannan Xue, Gongan Chen, Junqi Liu and Yang Liu in Technology in Cancer Research & Treatment
Footnotes
Abbreviations
Funding
This study was supported by National Natural Science Foundation of China (No. 81700729), and Henan Province Medical Science and Technology Research Project (No. LHGJ20190249).
Conflict of Interests
The authors declare no conflict of interest.
Availability of Data and Materials
Supplemental material will be provided upon request by journal office or reviewers.
Ethics Approval and Consent to Participate
This study was approved by the Joint Ethics Approval Committee of First affiliated Hospital of Zhengzhou University. We obtained informed consent for all patients, and we guarantee that the patient data will be kept confidential.
Consent for Publication
Not applicable.
Author Contributions
Gang Wang (GW), Narasimha M. Beeraka (NMB), Junqi Liu (JL), Yang Liu (YL) conceptualized and designed the study Wenjing Xiao (WX), Yaowen Zhang (YZ), Nannan Xue (NX), Gongan Chen (GC), GW, WX, YZ, NX and GC collected clinical data. GW, NMB, JL, YL, XL, and GC performed the literature analysis, and wrote the original manuscript draft. NMB, JL, YZ revised, edited, and extended the final draft. All authors have reviewed and approved the manuscript before submission.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
