Abstract
Multiple myeloma (MM) is a malignant hematological disease originating from plasma cells that remains incurable. Autologous stem cell transplantation (ASCT) is an important treatment method for MM. With the development of new drugs, the treatment of MM patients who meet the ASCT criteria has significantly improved, and the median survival time has increased by 8–10 years. The current treatment for MM patients who meet the ASCT criteria consists mainly of the following stages: induction therapy, stem cell collection, stem cell transplantation, and consolidation and maintenance therapy. Even today, long-term disease control remains the goal of MM treatment in clinical practice. In the era of new drugs, early ASCT still results in longer progression-free survival (PFS) and is currently the standard treatment method for young newly diagnosed multiple myeloma (NDMM) patients. Moreover, tandem transplantation can be considered for MM patients with high-risk cytogenetics. This review discusses mainly the role of ASCT in MM, the conditions for patient transplantation, the induction chemotherapy regimen before transplantation, the conditioning regimen, the timing of transplantation, and the effectiveness of tandem transplantation, including maintenance and salvage ASCT after transplantation.
Keywords
Introduction
Multiple myeloma (MM) remains an incurable plasma cell-related malignancy characterized by uncontrolled proliferation of clonal plasma cells and organ damage, including hypercalcemia, renal insufficiency, anemia, and bone destruction (CRAB). 1 MM accounts for approximately 10% of all malignant hematological tumors and ranks second in incidence. The malignant plasma cells are primarily found in the bone marrow, but they can also be observed in peripheral blood and other extramedullary sites, such as soft tissues and organs, especially in the later stages of the disease.
The advent of new drugs, including proteasome inhibitors (PIs), immunomodulatory drugs (IMiDs), and CD38 monoclonal antibodies, as well as the widespread use of autologous stem cell transplantation (ASCT), has greatly improved the prognosis of MM patients, with more than 50% of patients achieving a complete response (CR). 2
Two-thirds of multiple myeloma (MM) patients are over the age of 65 at the time of diagnosis. 3 With an increasingly aging population, this proportion is expected to increase further. In addition, there is a high incidence of clonal proliferation of hematopoietic stem cells leading to clonal hematopoiesis in bone marrow over the age of 60 years. Clonal hematopoiesis (CH) results from the excessive contribution of hematopoietic stem and progenitor cell (HSPC) expansion clones to hematopoiesis.CH is a precancerous state; somatic mutations detected in CH are the initiating mutations for hematologic malignancies, and CH is a strong predictor of blood cancers. 4
Over the last three decades, ASCT has been a primary therapeutic modality for MM. However, despite the advent of novel agents for MM, their efficacy has been subject to scrutiny. Nevertheless, in newly diagnosed multiple myeloma (NDMM) patients meeting transplant the eligibility criteria, ASCT continues to be the standard recommended treatment according to international guidelines such as the American Society of Clinical Oncology 5 and the European Society for Medical Oncology. 6
Currently, the main treatment options for newly diagnosed multiple myeloma patients are shown in Figure 1. For patients who are not suitable for ASCT are not considered for the time being, double or triple treatment regimens are usually adopted until disease progression; for transplant patients, after double, triple or even quadruple induction therapy, more intensive pre-treatment, ASCT is followed by single or double treatment. 7 For high-risk patients, tandem ASCT can bring more benefits. In a phase III trial, 8 700 patients were randomized into a transplant group versus a VRD group, and after 1 year of lenalidomide maintenance therapy, there was a significant improvement in the negative rate of minimal residual disease (79% vs 65%; P < .001) in the transplant versus VRD alone, and the progression-free survival was significantly longer in the transplant group than in the VRD group. This shows that drug combined with ASCT brings more benefits compared with drug therapy alone.
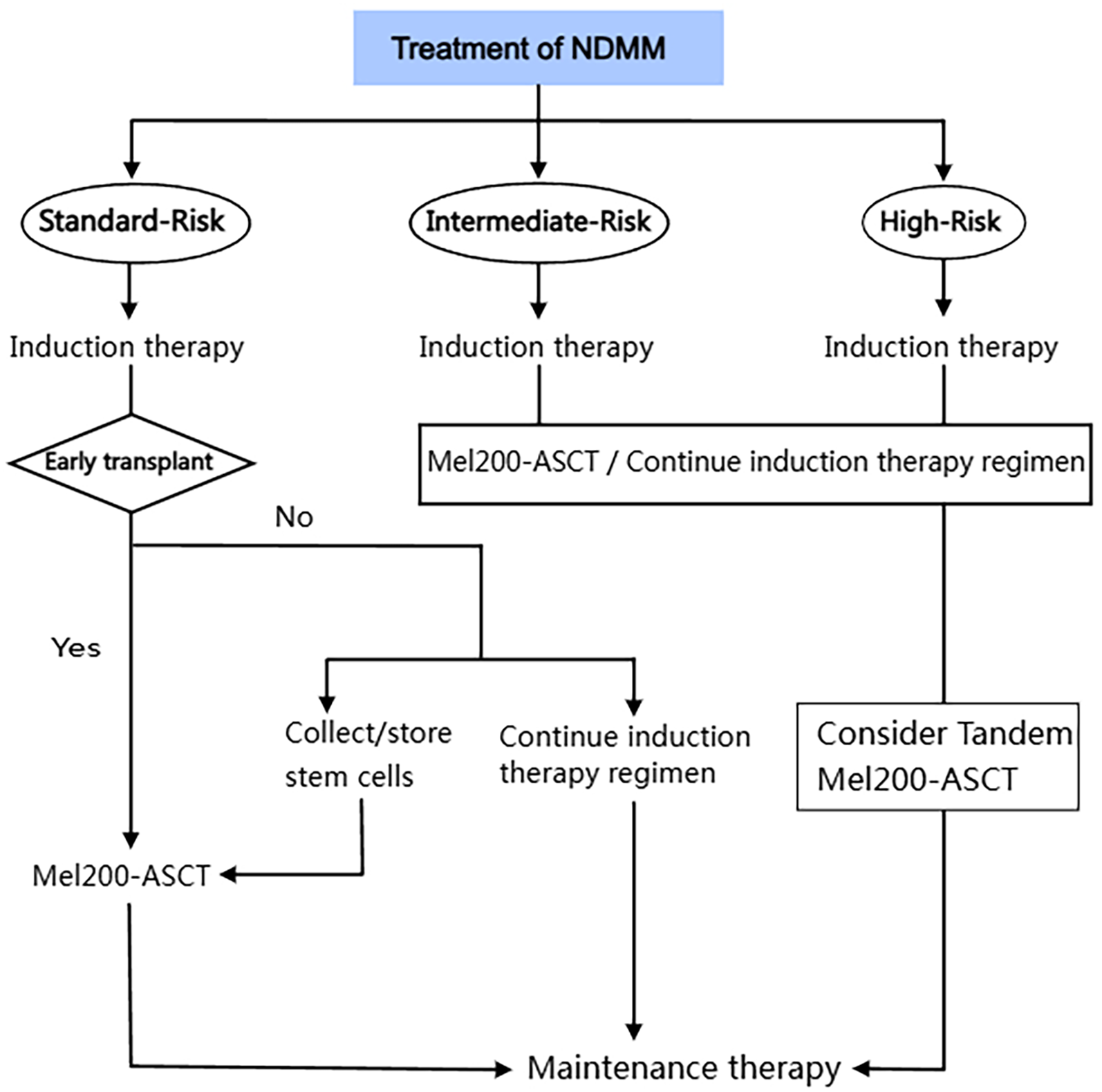
The treatment of NDMM.
The standard treatment protocol for young NDMM patients currently involves induction, high-dose melphalan, ASCT, and sequential maintenance therapy. In the last decade, the median progression-free survival (PFS) time for MM patients who meet the transplant criteria has increased from 3–5 to 8–10 years; 9 however, the PFS and overall survival (OS) have since increased, and the treatment efficacy has significantly improved. Together with the extension of PFS and improvement in OS, there has been a marked improvement in treatment efficacy. However, with advancements in medical technology, there has been a realization that even in patients who achieve CR, the number of residual malignant cells may be extremely small and undetectable, and this is known as minimal residual disease (MRD). Reports indicate that patients who are MRD-negative have better PFS and OS rates.10,11 Therefore, MRD is a better predictor of PFS and OS than cell morphology. 11 Despite the good efficacy of treatment achieved in MM patients, relapse due to MRD that cannot be detected by traditional serological (like serum M protein, serum quantitative immunoglobulins, serum protein electrophoresis, serum immunofixation electrophoresis, etc) or morphological methods in the bone marrow still occurs.12,13 Therefore, various methods have been developed to detect deeper levels of response. CR refers to the situation wherein there are no detectable malignant cells in the patient's blood or bone marrow after treatment, and there should be no evidence of new bone lesions or progression of existing bone lesions.
Assessment of ASCT Eligibility
Currently, autologous stem cell transplantation following high-dose melphalan treatment is the standard therapy for MM. 14 For MM patients who are candidates for transplantation, there is no current consensus on the age limit. Therefore, different institutions and countries have different criteria. Most clinical trials have evaluated the effectiveness and safety of ASCT for MM patients when the age of the patient is limited to less than 65 years, and this age limit is still the inclusion criterion for ASCT in most European countries. 15 Analysis by the European Society for Blood and Marrow Transplantation (EBMT) and the Center for International Blood and Marrow
Transplant Research (CIBMTR) registries clearly revealed that the use of ASCT in elderly patients aged 65 years or over increased from 1991 to 2010.16,17 As shown in Figure 2, the number of patients receiving ASCT has increased in all age groups, and in recent years, the survival rate of elderly patients has been significantly higher than that of younger patients. From 2006 to 2010, the median survival rates at 2 and 5 years after transplantation ranged from 85.9% and 61.5% in patients aged 40 to 80.2% and 49.7% in patients aged 70. The results of this study suggest that the use and safety of ASCT have increased in Europe in recent years, and post-transplant survival has improved, especially in elderly MM patients, and age should not be a limiting factor in considering ASCT modalities. 17

The efficacy of ASCT in the differ age groups.
Although most clinical trials limit the patient age to under 65 years, this does not mean that patients over 65 years old cannot undergo ASCT. Many studies have confirmed that age itself is not a limiting factor for ASCT; for MM patients, the results of ASCT for patients over 70 years of age and elderly patients are comparable to those of young patients under 50 years of age.3,18 These findings confirm the feasibility of using high-dose melphalan in combination with ASCT in elderly patients and provide evidence of its safety and efficacy as a first-line treatment for elderly patients with MM.
The latest expert consensus in China currently recommends that MM patients undergo ASCT at an age of ≤65 years or that patients over 65 years old have a good overall health status, as determined by assessing the patient's status in terms of organ function, comorbidities (using the Charlson index), frailty, and disability. 19 It is not determined solely on the basis of age or the presence of renal insufficiency; rather, eligibility should be evaluated on the basis of the patient's performance status (PS) and comorbidities to ensure the safety and effectiveness of the treatment.
Induction Chemotherapy Regimen
Induction Chemotherapy Regimens
Induction chemotherapy is a treatment method used before ASCT to reduce the number of tumor cells, improve the depth of remission, increase the success rate of transplantation, and minimize the impact on normal hematopoietic function while maintaining patient tolerance. The induction therapy regimen suitable for MM patients undergoing hematopoietic stem cell transplantation should meet the following goals: rapid control of the disease and maximum possible remission; restoration of disease-related damage (such as renal dysfunction); minimizing treatment side effects; reducing early mortality; and minimizing the impact on stem cell collection. 20 The efficacy of induction therapy directly affects the efficacy of subsequent ASCT. Achieving MRD negativity after induction can be considered the optimal response depth, which is closely related to the long-term survival of the patient.8,11,21
Currently, for transplant-eligible patients, four to six cycles of induction therapy prior to myeloablative high-dose chemotherapy (HDT)/ASCT are recommended, depending on several factors, including the patient's response to the induction regimen and its depth of response and tolerability and the availability of ASCT infrastructure. In the era of new drugs, the induction therapy drugs for NDMM patients include PIs (eg, bortezomib, ixazomib, carfilzomib), IMiDs (eg, thalidomide, lenalidomide), monoclonal antibodies (eg, anti-CD38 monoclonal antibodies), cytotoxic drugs (eg, cyclophosphamide, liposomal doxorubicin), and glucocorticoids (eg, dexamethasone, prednisone). Currently, three drug combinations, namely, PIs, IMiDs, and glucocorticoids, are considered the gold standard regimens. 22 The superiority of triple therapy over dual therapy has long been established.23-25 The selection of induction drugs for MM patients undergoing ASCT should consider avoiding the accumulation of toxic effects on hematopoietic stem cells and avoiding effects on the collection of hematopoietic stem cells and hematopoietic reconstruction. The guidelines for ASCT in MM in China recommend the following three drug combination regimens for newly diagnosed patients who are eligible for transplantation: bortezomib + lenalidomide + dexamethasone (VRD), bortezomib + thalidomide + dexamethasone (VTD), bortezomib + cyclophosphamide + dexamethasone (VCD), ixazomib + lenalidomide + dexamethasone (IRD), bortezomib + liposomal doxorubicin + dexamethasone (PAD), thalidomide + doxorubicin + dexamethasone (TAD), and thalidomide + cyclophosphamide + dexamethasone (TCD). 26 Moreover, researchers have reported that the use of polyethylene glycol liposomal doxorubicin significantly reduces cardiotoxicity. 27 Therefore, doxorubicin-containing regimens can be further optimized. To assess whether triple induction therapy could further increase MRD negative rates and improve outcomes (Table 1).
Summary of Clinical Trials Evaluating pre-ASCT Induction Chemotherapy Regimens and Their Efficacy.

Abbreviations: AEs adverse events; ASCT autologous stem cell transplant; VRd lenalidomide, bortezomib, dexamethasone; Mel melphalan; VTd bortezomib, thalidomide, dexamethasone; VCd bortezomib, thalidomide, dexamethasone; KRD carfilzomib, lenalidomide, dexamethasone; Dara-VTd daratumumab, bortezomib, thalidomide, dexamethasone; OS overall survival; CR complete response; sCR stringent complete response; EFS event-free survival; VGPR very good partial response; PFS progression-free survival; ORR overall response rate; MRD, minimal residual disease; GI gastrointestinal; NR not reported; PD progressive disease; PN peripheral neuropathy ; R lenalidomide; RR relative risk; D day(s); yrs years.
VRD
VRD regimen-induced sequential ASCT is the standard first-line treatment for NDMM patients who meet the transplantation criteria. A three-phase trial enrolled 700 patients who were randomly assigned to either the VRD group (8 cycles; 350 patients) or the transplant group (3 cycles of VRD, followed by high-dose melphalan and ASCT, followed by 2 additional cycles of VRD; 350 patients), and the patients in both of these groups received lenalidomide maintenance therapy for 1 year. 28 In the transplant group, compared with those in the VRD group, the complete response rate (CRR, 59% vs 48%; P = .006) and the MRD-negative rate (79% vs 65%; P < .001) were both increased, and the PFS time was significantly prolonged by VRD combined with transplantation compared with VRD alone.
A PETHEMA/GEM2012 trial conducted in Spain in 2019 enrolled 458 NDMM patients aged up to 65 years who received 6 cycles of VRD induction chemotherapy and then underwent ASCT followed by 2 cycles of VRD after transplantation. The responses were grouped according to induction, transplantation, and consolidation, and treatment response deepened over time. Among the 426 patients who completed 6 cycles of induction, the rates of very good partial response (VGPR) were 55.6% at cycle 3, 63.8% at cycle 4, 68.3% at cycle 5, and 70.4% after induction was completed. 29 In another real-world clinical study, the MRD-negative data from 317 patients who were subjected to next-generation sequencing (NGS) technology showed a similar pattern of gradual improvement, with MRD-negative rates of 35%, 54%, and 58% after 6 cycles of induction chemotherapy, ASCT, and 2 consolidation cycles, respectively. 33 This study indicates that VRD is an effective and well-tolerated NDMM induction regimen that leads to increased responses throughout the induction process and during treatment.
VRD regimens containing lenalidomide have been demonstrated in several studies to be a particularly effective induction regimen for transplant- and non-transplant-suitable NDMM and are approved by the NCCN guidelines as the first-line treatment of choice for MM.29,34,35
VTD Versus VCD
A randomized trial in France compared the induction effects of VTD and VCD in NDMM patients before high-dose chemotherapy and ASCT, with 340 patients randomized to receive either VTD or VCD treatment. 30 In the VTD group, approximately 66.3% of patients achieved at least VGPR, whereas in the VCD treatment group, the rate was 56.2%. There was no significant difference in the CRR between the two groups, but the overall response rate (ORR) of the VTD group was significantly higher than that of the VCD group (92.3% vs 83.4%). The study results show that both VTD and VCD are viable induction regimens. The 10% difference in VGPR also suggests that the combination of PIs, IMiDs, and dexamethasone is a better choice for intensification before ASCT. These trial results support the use of VTD as an efficient induction regimen before ASCT.30,36 VTD and VCD triple therapy have shown high response rates in prospective phase 237-39 and phase 340,41 clinical trials and are the two most commonly used induction regimens prior to ASCT and recommended by international guidelines. 35
KRD
Recently, the introduction of a new generation of PIs, including carfilzomib, into induction therapy regimens has been evaluated. 42 The combination of carfilzomib, lenalidomide, and dexamethasone (KRD) therapy was associated with early response and had an ORR of 95%; in particular, the percentage of those who achieved a CR or higher on efficacy assessment was 20%,31,43 and a high percentage of MRD-negative patients in the NDMM group were MRD-negative, which translated into longer PFS in MRD-negative patients. Therefore, the KRD regimen has become a new option for inducing treatment in transplantable NDMM patients.
Daratumumab Combined Three-Drug Regimen
Building on the three-drug regimen, the next step will be to evaluate whether a four-drug regimen-based induction therapy can further improve response rates and improve outcomes. Daratumumab is a humanised IgG-κ monoclonal antibody that targets CD38, and its emergence has changed the prospects of treatment for MM. The effectiveness of daratumumab as a single agent or in combination regimens in relapsed patients, as well as in combination with bortezomib, melphalan, and prednisone (VMP) as a conditioning regimen for MM patients who are not eligible for ASCT, provides compelling reasons to introduce it into the first-line treatment of NDMM patients with ASCT eligibility. The CASSIOPEIA study revealed that adding daratumumab to VTD three-drug induction therapy is beneficial for eligible NDMM patients, improving the depth of response and PFS before and after ASCT, and that the safety is acceptable. 44
The phase 3 CASSIOPEIA study included 1085 NDMM patients aged 18–65 years and assessed whether the addition of daratumumab to VTd (Dara-VTd) before and after ASCT improved outcomes. 32 A quadruple regimen with Daratumumab on top of a triple regimen is a new direction to explore in the current era of new drugs. Prior to the advent of quadruple regimens, tandem ASCT has been beneficial for myeloma patients, especially among high-risk patients. 32 The next step will be to assess whether induction therapy based on quadruple therapy can further increase response rates and improve efficacy. In the CASSIOPEIA study, 32 the proportion of patients obtaining a CR or sCR was significantly higher in the D-VTd group than in the VTd group (211 [39%] vs 141 [26%], p < .0001), and the proportion of patients obtaining ≥ VGPR was also significantly higher than in the VTd group (453 [83%] vs 423 [78%], p = .024). The proportion of MRD-negative patients was higher in the D-VTd group than in the VTd group (346 [64%] out of 543 vs 236 [44%] out of 542, p < .0001). Addition of daratumumab to VTd does not increase overall toxicity, and provides a significant and clinically meaningful benefit.
Cellular Immunotherapy from Cell Immunotherapy
Chimeric antigen receptor T cell(CAR-T) cell therapy and Bispecific T-cell engager (BiTEs) are emerging models for treating multiple myeloma, although not yet used in first-line therapy.
Providing treatment for patients with relapsed and refractory multiple myeloma (RRMM) who have failed to respond to standard therapies such as IMiDs, PIs, and anti-CD38 monoclonal antibodies, either in early relapse or late relapse, is a major challenge. 45 Treatments for RRMM are evolving to overcome tumor immune evasion mechanisms and improve patient survival, and CART cell therapy has shown clinical benefit for patients with blood cancers. 46 CAR-T therapy reprograms a patient's T cells to attack specific cell antigens, such as CD19 for B cell malignancies and B cell maturation antigen (BCMA) for MM. 47 CART cell therapy has shown good efficacy in treating hematological cancers, although it has not been successful in solid tumors. CAR-T cells targeting CD19 are the most successful and widely studied to date. 48 A National Cancer Institute human Phase I clinical trial in RRMM patients showed that, 48 CAR-T cells are a better therapy at this point in time, especially in relapsed refractory MM, showing better efficacy and prolonging the patient's PFS.
BiTEs therapy is a therapy that links endogenous T-cells to tumor-expressed antigens to activate the cytotoxic potential of the patient's own T-cells, thereby eliminating the cancer without the need to genetically alter the T-cells or expand/manipulate them in vitro. 47
Considering the current low use of first-line therapies, most BiTEs molecules are still in the clinical trial stage and show better potential in targeting BCMA as a target. As clinical trials progress, BiTEs therapies are expected to provide more treatment options for multiple myeloma patients.
Hematopoietic Stem Cell Mobilization and Collection
The key and essential step prior to ASCT is mobilizing hematopoietic stem cells from the bone marrow and collecting them from the peripheral blood, as shown in Figure 3. The minimum number of CD34 + stem cells considered sufficient for successful engraftment is 2 × 106/kg, but the optimal target is usually set at 5 × 106/kg. 49 For patients who undergo sequential ASCT, higher targets should be set. This can be achieved through steady-state mobilization and chemotherapy mobilization, which can optimize the number of mobilized stem cells. 50

Autologous hematopoietic stem cell transplantation.
Granulocyte-Colony-Stimulating Factor
Methods of mobilizing peripheral blood hematopoietic stem cells include granulocyte‒colony-stimulating factor (G-CSF) monotherapy or combination therapy with plerixafor and high-dose chemotherapy with G-CSF. For many years, the most widely used chemotherapy mobilization regimen has been high-dose cytosine arabinoside (2-4 g/m2), followed by G-CSF (5 μg/kg/day) given five days after chemotherapy mobilization until the last stem cell collection day. 51 However, cyclophosphamide plus G-CSF regimens have been more widely used in recent years. 52 Cyclophosphamide plus G-CSF can increase the number of circulating stem cells by 24-fold; thus, for some patients whose stem cells are difficult to collect, the use of cyclophosphamide in combination with G-CSF can increase the success rate of collection. 53 Although the time required for the combined regimen is shorter and more stem cells are collected, due to the destruction of the bone marrow microenvironment by chemotherapy drugs such as cyclophosphamide, there are defects in the stem cell implantation environment, causing a longer time for neutrophil and platelet implantation than when G-CSF is used alone.54,55 Whether to apply cyclophosphamide should be based on the individualization of the patient by an experienced physician to make an accurate judgment.
Plerixafor
Plerixafor is a selective, reversible inhibitor of the chemokine receptor CXCR4 that disrupts the interaction between CXCR4 and SDF-1, causing stem cells to be released from the bone marrow, enhances the stem cell mobilization action of G-CSF and can improve stem cell collection in patients with failed mobilization when it is added.49,56 Plerixafor mobilized stem cells also increased the expression of VLA-4 and CXCR4, 57 as well as the expression of genes promoting cell adhesion, cell motility, cell cycle, and anti-apoptosis. 58 These features suggest that plerixafor-mobilized cell products may have a greater ability to repopulate the bone marrow and rebuild the immune system compared to cell products mobilized only by G-CSF and have been demonstrated in animal models such as mice.59,60
Sickle cell disease (SCD) is the most common single-gene inherited disease in the UK, and as the life expectancy of people with SCD increases, so does their risk of developing malignant tumors. 61 Given the age of onset of MM, patients with SCD often already have severe organ dysfunction by the time they develop the disease. For ASCT, the minimum threshold for CD34 + cell collection is currently defined as 2.0 × 106 cells/kg6 or CFU-GM >20 × 104 cells/kg (as implemented in our center). In 3 patients with SCD and MM, 62 HPC mobilization was successfully achieved with plerixafor alone. However, the HPC yield was modest, allowing only enough cells for a single ASCT. but this may limit optimal treatment of patients with high-risk disease requiring tandem transplantation.
Plerixafor can be used as salvage therapy for patients who have failed mobilization, increasing the number of patients who reach collection targets, but it is usually not used in the initial phase owing to the high cost of the drug. Studies have suggested that the use of lenalidomide during the induction phase is one of the factors affecting the successful mobilization of stem cells, mainly because lenalidomide can lead to the upregulation of CXCR4, increasing the binding of stem cells to the matrix. 63 Additionally, plerixafor can counteract the effects of stem cell mobilization caused by prolonged exposure to lenalidomide. 64 For patients who have been using lenalidomide for a long period of time, the addition of pralatrexate can increase the success rate of stem cell mobilization. The most commonly regimen used for chemotherapy-based mobilization or solitary steady-state mobilization is still high-dose cyclophosphamide plus G-CSF. When mobilization is poor, pralatrexate is preferred, but new drug combinations are actively being explored. 65
Conditioning Regimen Prior to ASCT
Patients who have a better response to pretransplant chemotherapy can achieve better PFS and OS times. Melphalan is the most commonly used drug in the ASCT pretreatment regimen for MM patients, and the standard ASCT pretreatment regimen is high-dose melphalan (200 mg/m2). To reduce transplant-related complications and mortality, the dose for patients over 65–70 years old or with renal insufficiency (glomerular filtration rate less than 60 ml/min) can be reduced to 140 mg/m². Current studies compare the effects of different doses of melphalan on PFS and OS to enhance the pretreatment chemotherapy regimen, improve the patient's transplant remission rate and reduce recurrence. 3
Currently, the main conditioning regimen options consist mainly of a single use of melphalan, melphalan combined with new drugs, or melphalan combined with traditional chemotherapy drugs. Some studies66-68 on the use of oral busulfan and melphalan combined have shown that, compared with the single use of high-dose melphalan, although the incidence of venous thromboembolic disease is higher, PFS is longer. Compared with oral busulfan, the introduction of intravenous busulfan formulations with linear pharmacokinetics and more reproducible systemic exposure may reduce drug toxicity and produce better therapeutic effects. 69 In one phase III trial, 70 202 NDMM patients aged ≤70 years who had undergone at least 2 cycles of induction therapy and minimally had stable disease (SD) were randomly assigned to the busulfan–melphalan group or the group receiving melphalan alone at a 1:1 ratio. In the group receiving busulfan-melphalan, the median PFS was 64.7 months (IQR: 32.9, 64.7 months), and in the group receiving melphalan alone, the median PFS was 43.5 months (IQR: 19.9 months, not estimated), with a hazard ratio (HR) of 0.53, 95% confidence interval (CI) [0.30, 0.91] (p = .022). The trial showed PFS was higher with the combination of intravenous busulfan with melphalan pretreatment than with melphalan alone.
In addition, other regimens, such as CEB (cyclophosphamide 50 mg/kg, once daily, −3∼−2 d; etoposide 10 mg/kg, once daily, −5∼−4 d; busulfan 0.8 mg/kg, every 6 h, −8∼−6 d) and BUCY (busulfan 0.8 mg/kg, every 6 h, −7∼−4 d; cyclophosphamide 60 mg/kg, once daily, −3∼−2 d), are also selected for clinical use. A phase II trial used bortezomib combined with high-dose melphalan (bortezomib 1 mg/m2 × 4 + melphalan 200 mg/m2) as a pretreatment regimen; approximately 70% of patients achieved VGPR, and 32% achieved CR, with no long-term hematological toxicity. 71 The combination of melphalan with new drugs has also become a new research direction.
The Best Time for Transplantation
Early transplant refers to transplantation performed immediately after disease relief by induction therapy, which means that the transplant is performed within one year of diagnosis. 56 Late transplantation involves the collection of hematopoietic stem cells for storage and subsequent use after induction therapy, followed by maintenance therapy with drugs until the first recurrence, and then transplantation is performed. Fermand conducted a randomized trial in which 185 MM patients were assigned to receive ASCT as either first-line or second-line therapy in the era of conventional chemotherapy. Compared with late ASCT, early ASCT was associated with significantly higher response rates, PFS, median time without symptoms, treatment-free survival, and treatment-related toxicity; however, no OS benefit was observed. 72 A recent trial conducted by the Intergroupe Francophone du Myelome compared the efficacy and survival outcomes of early or delayed ASCT after VRD treatment, with lenalidomide maintenance, in patients who were randomly assigned to two groups. 73 One group received VRD (3 cycles) + ASCT + VRD consolidation (2 cycles), whereas the other group received VRD (8 cycles), with ASCT reserved for relapse. Both groups received lenalidomide maintenance treatment for one year. Early ASCT can significantly improve PFS, but whether it can translate into a significant improvement in OS remains to be further studied.
Gay conducted a pooled analysis of the RV-MM209 and EMN-441 phase 3 trials; regardless of the prognostic group, early ASCT significantly prolonged PFS 1 and PFS 2 as well as OS. 74 Additionally, another important aspect is estimating the likelihood of a patient being eligible for delayed ASCT in the future, as well as the patient's relevant characteristics (such as advanced age, comorbidities, and frailty) and myeloma-related variables (such as response to second-line therapy) that may be associated with this. In the relevant literature, the proportion of RRMM patients who were not eligible for delayed ASCT ranged from 11% to 46%.8,22,74 Overall, the available data support ASCT as a first-line treatment, but delayed ASCT remains a viable option.
Single ASCT Versus Tandem ASCT
Tandem ASCT refers to the second ASCT within six months after the initial ASCT. This concept first appeared in the 1990s, primarily aimed at further improving the depth of response and long-term efficacy in the era when traditional chemotherapy drugs were the mainstay. 75 Several phase III studies have shown that tandem ASCT can improve event-free survival (EFS); however, only one study has shown an improvement in OS, and this was only for patients who did not achieve VGPR after the first ASCT.76-78 Previous randomized trials have shown that tandem transplantation improves survival and quality of life, even in patients who do not achieve VGPR after the first transplantation. 79
In the EMN02/HO95 phase 3 study, 415 NDMM patients were randomly assigned to receive either single or tandem ASCT after 3–4 cycles of bortezomib, cyclophosphamide, and dexamethasone induction therapy. 80 The use of tandem ASCT resulted in a 25% increase in the depth of response, with more than half of the patients achieving or surpassing CR. In terms of PFS and OS extension for the overall patient population and the R-ISS-defined high-risk subgroups with high-risk cytogenetic features, the tandem ASCT group performed better than did the single-ASCT group, with a reduction in the risk of death and disease progression of approximately 30%. 80
In a prospective randomized phase III trial conducted in the United States by the Bone Marrow Transplantation Clinical Trial Network (BMT-CTN0702), 81 758 patients with NDMM were randomly assigned to receive induction therapy with tandem ASCT and lenalidomide consolidation and maintenance therapy; ASCT and 4-cycle VRD and lenalidomide maintenance therapy for consolidation; or ASCT and lenalidomide maintenance therapy. A second ASCT after the first ASCT or additional VRD consolidation therapy did not improve PFS or OS in patients at high risk. In this trial, half of the patients received VRD as induction therapy, and it was evenly distributed among each treatment group. The BMT-CTN 0702 StaMINA study revealed no significant difference in survival outcomes between single and tandem autologous transplantation. The trial results suggest that single ASCT and lenalidomide maintenance therapy should remain the standard approach, as more than 30% of patients who are randomized to receive tandem grafts do not receive a second transplant. 81 An integrated analysis of patient-level data from European Phase III studies, 82 for patients with high-risk cytogenetics [t(4;14) and/or del17p] and who did not achieve a complete response after bortezomib induction therapy, tandem ASCT resulted in PFS (41 months vs 20 months;HR, 0.52;P5.003) and OS advantages (67 months vs 31.5 months;HR, 0.32;P,.001), respectively, compared with single ASCT.
In summary, compared with drug alone, both single ASCT and tandem ASCT showed more significant improvements in PFS and OS for patients. Despite tandem ASCT conflicting data, we can conclude from the available data that tandem ASCT is beneficial for high-risk patients (including those with high-risk cytogenetic and R-ISS3 disease). 81
Salvage ASCT After Transplant
The concept of salvage ASCT refers to a second ASCT after a relapse has been induced by a chemotherapy regimen following a recurrence. Despite consolidation and maintenance therapy after ASCT, most patients still experience relapse. 83 A prospective trial from the UK, in which traditional chemotherapy was used as a control, revealed that salvage ASCT improved PFS. 84 Studies have shown that salvage ASCT provides significant survival benefits to patients who have responded to the initial transplant, regardless of whether maintenance therapy is given. In a prospective, phase III study known as ReLApsE, relapsed MM patients were randomized to two groups: one group received lenalidomide–dexamethasone (Rd) reinduction, followed by salvage ASCT and Rd maintenance, and the other group continued to receive Rd. The ReLApsE trial is the first RCT of salvage ASCT versus continuous new drug therapy, making it a milestone. The analysis revealed that patients who received salvage ASCT had significant PFS/OS benefits, and the low-risk patients benefited the most.85,86 MM patients who have relapsed after not receiving maintenance therapy for more than 18 months after the first ASCT or after maintenance therapy for more than 36 months, especially those with good disease characteristics, should consider salvage ASCT. 15 Salvage ASCT can effectively control potential tumors and improve the cell structure and function of the bone marrow, as well as reduce the hematological toxicity associated with the drugs. 87
Maintenance Therapy
The role of maintenance therapy is to provide long-term, low-intensity treatment with minimal toxicity to prevent relapse and prolong survival. In the EU and US, lenalidomide monotherapy for the maintenance treatment of NDMM after ASCT has been established as the global standard of care. A meta-analysis of several prospective studies confirmed the survival advantage of lenalidomide maintenance therapy. 8 Lenalidomide maintenance therapy can reduce the risk of disease progression by 52% and the risk of death by 25%, has been shown to be superior to other treatment regimens based on bortezomib or thalidomide, and is considered the best choice.8,88 Induction chemotherapy and ASCT followed by MRD negativity are strong predictors of long-term survival for patients,11,21 and MRD status after ASCT can guide treatment decisions regarding consolidation therapy, as well as the type and duration of maintenance therapy.
On the basis of the consideration of the efficacy and toxicity of the treatment regimens for MM, the National Comprehensive Cancer Network (NCCN) 35 expert group classified all the treatment regimens into “preferred”, “additional recommendations”, and “useful in certain circumstances”. For maintenance treatment, the guidelines recommend the application of PIs combination regimens for high-risk patients and PIs monotherapy for standard-risk patients. On the basis of the findings of the phase II FORTE study, 89 there is a trend toward a PFS benefit for PIs combination regimens compared with PIs monotherapy in high-risk newly diagnosed MM patients.
In a meta-analysis several phase III trials (such as IFM-2005-02, 90 GIMEMA RV-MM-PI-209 91 and CALGB 100104 92 ), the PFS of the patients who received maintenance therapy with lenalidomide after ASCT was significantly longer than that of patients who did not receive maintenance therapy.8,93 Patients who received maintenance therapy with lenalidomide had significantly improved PFS and OS compared with those who received placebo or no treatment. On the basis of the evidence from several phase III trials.94,95 The NCCN Multiple Myeloma Expert Panel has listed lenalidomide as one of the preferred maintenance therapy options for MM. 35
There have been no prospective studies comparing lenalidomide and bortezomib in maintenance therapy after ASCT. However, in a retrospective analysis of a GMMG trial, 96 lenalidomide and bortezomib had no effect on OS after two years of treatment, but a significant improvement in PFS was observed in the lenalidomide maintenance treatment group after the effects of different sequential ASCT rates were excluded. In the phase III TOURMALINE–MM3 study, the oral drug ixazomib was explored as maintenance therapy after ASCT, with a 28% lower risk of progression or death for patients taking ixazomib than for those in the placebo group (median PFS of 26.5 months [95% CI 23.7-33.8] vs 21.3 months [18.0-24.7]; hazard ratio (HR) of 0.72, 95% CI 0.58–0.89; p = .0023) 97 However, bortezomib and ixazomib have not yet been approved by the European Medicines Agency for maintenance therapy after ASCT.
Conclusion
For NDMM patients who meet the transplantation criteria, even in the era of new drugs, early ASCT remains the standard of care. To date, research data have confirmed that ASCT has significant therapeutic effects, and it remains the cornerstone of treatment for eligible patients. As people continue to explore new drugs, induction therapy, stem cell mobilization protocols, consolidation, and maintenance therapies have been continuously improved, resulting in the gradual introduction of carfilzomib and daratumumab into frontline treatment regimens to achieve the goals of deepening responses, prolonging PFS, and improving MRD negativity. ASCT can increase the depth of response and provide significant PFS benefits to MM patients. Although it is usually performed in the initial phase of treatment, ASCT is multifaceted: early or delayed, single or tandem, and even salvage therapy. The OS of early ASCT is similar to that of delayed ASCT but ASCT has PFS benefits. The role of tandem ASCT is not yet clear. Previous research results have been inconsistent, but in MM patients with high-risk cytogenetics and unfavorable prognostic factors, consideration should be given to performing tandem ASCT in the initial phase. Despite improvements in this situation, the treatment outcomes for very high-risk MM patients remain poor, so there is an urgent need to explore innovative therapies beyond quadruple therapy and ASCT for this group of patients. In addition, the development of MRD detection technologies and methods has led to revolutionary changes in the treatment and management of patients with blood malignancies, allowing doctors to monitor treatment effects more precisely, predict disease progression, and provide more targeted treatment plans for patients. Considering the high cost of new drugs and treatments, whether MRD assessment can be conducted through NGS and flow cytometry analysis of bone marrow to guide the enhancement or reduction of treatment intensity, avoid drug toxicity, protect the quality of life of patients, and guide maintenance therapy on the basis of MRD status is another challenge being addressed in ongoing research.
Footnotes
Abbreviations
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the The Natural Science Foundations of Shandong Province, (grant number ZR2020MH124).
