Abstract
Introduction
Osteosarcoma is a malignant mesenchymal tumor with a double-peak distribution. It predominantly occurs in children and young adults, comprising approximately 60% of all cases, whereas a significant second incidence peak of osteosarcoma is observed in elderly patients.1,2 Due to the rapid increase in the proportion of the elderly population worldwide, the proportion of patients aged over 40 years with osteosarcoma has been reported to be approximately 13% to 30%.3,4 However, the current proportion is increasing gradually. It is well known that improvements in surgical, neoadjuvant, and adjuvant chemotherapy techniques have led to significantly improved predictions for osteosarcoma in adolescents and young adults. However, notably, not much is known about the clinical outcomes of patients aged over 40 years with osteosarcoma.
Published literature has suggested that the clinicopathological characteristics of osteosarcoma in adolescent patients are well documented,5-7 whereas there are only a few reports regarding osteosarcoma in patients aged older than 40 years. Although osteosarcoma is often found in patients aged older than of 40 years, it is also commonly attributed to Paget's disease, irradiated bone, or bone infarct.3,8 Therefore, for patients with osteosarcoma over the age of 40 years, if they are received by a doctor who cannot make a clear diagnosis, it is likely to lead to misdiagnosis. 9 Moreover, because there is no standard treatment strategy established for osteosarcoma, the outcomes for patients in this age group remain poor even with multidisciplinary treatment. Interestingly, advancements in neoadjuvant chemotherapy improve the 5-year disease-specific survival by greater than 60% for patients with osteosarcoma.10,11 However, because all trials included all age groups, these data do not apply to these specific populations of patients aged older than 40 years. Notably, the value of chemotherapy for the survival benefits of patients aged older than 40 years that have osteosarcoma has remained controversial.4,10,12-14 Some reports have stated to have found no difference between patients that are receiving chemotherapy and those that are not,4,15 whereas others have suggested that receiving chemotherapy was more beneficial than not receiving chemotherapy for patients with osteosarcoma aged older than 40 years.16,17 In recent years, the incidence of patients with osteosarcoma aged older than 40 years has also increased due to the rapid increase in the number of elderly people and the decline in birth rates globally, and this trend is especially noticeable in Japan.18,19 Therefore, there is a constantly increasing demand for research on this specific age group, especially for the assessment of survival prognosis.
This study aimed to verify whether chemotherapy exerts a significant survival benefit in patients over the age of 40 years that have osteosarcoma. Compared with the previous small sample studies, we analyzed a large sample database of all patients from 1975 to 2018 from the National Cancer Institute's Surveillance, Epidemiology, and End Results (SEER) program with the aim to produce more reliable results. Comparisons between patients who have received chemotherapy and those who have not were used to evaluate overall survival (OS) and cancer-specific survival (CSS). In addition, we identified the prognostic factors for these patients, highlighting stratification that may be more appropriate for the comprehensive evaluation of patients over 40 years old with osteosarcoma who have received chemotherapy, providing clinical recommendations for future patients in this specific age group to receive chemotherapy or not.
Patients and Methods
Patient Selection
All data used within this study were derived from the SEER 18 registries’ custom data (with additional treatment fields). We used SEER*Stat version 8.3.9 software to obtain patient data. Because we have signed a data use agreement with the SEER program, we are permitted to have public access to the SEER database. Given that the database is publicly available, informed patient consent is not required for the release of data. Our study was exempted from review by the ethics institutional review board and met the ethical standards. The inclusion criteria for the patients were (1) patients diagnosed as having osteosarcoma between 1975 and 2018, (2) patients over the age of 40 at the time of initial diagnosis, and (3) patients histologically diagnosed with osteosarcoma (ICD-O-3 9180-9186 and 9192-9194). In addition, the exclusion criteria for the patients include (1) patients with unknown grade, stage, and surgery data; (2) patients diagnosed with osteosarcoma at autopsy only or via death certificate only; and (3) patients with unknown survival data or survival time equal to 0 month. The flowchart of patient data extraction is shown in Figure 1.

The flowchart of patient data extraction procedure.
Data Collection
The following patient information was collected in this study: baseline demographics, inclusive of the year of diagnosis, their age at diagnosis, race, and sex. Tumor features included tumor location, tumor grade, and tumor stage. Therapy information included surgery, radiation, and chemotherapy. Follow-up records included the SEER cause-specific death classification, vital status, and survival months. Based on statistical and clinical considerations, ages were classified as 40 to 60 and >60 years. Tumor grade was classified as the following: well differentiated (I), moderately differentiated (II), poorly differentiated (III), and undifferentiated (IV). Grade I and II tumors fall into the category of low-grade tumors, whereas grades III and IV tumors were categorized as high-grade tumors. Tumor stage was classified as localized, regional, and distant. In addition, localized or regional tumors were classified as nonmetastatic, whereas distant tumors were classified as metastatic. The endpoints of the current study were OS and CSS, which were defined as the death of any cause and death of osteosarcoma, respectively.
Statistical Analyses
The differences in baseline demographics, tumor features, and treatment characteristics between the two groups, namely nonchemotherapy and chemotherapy, were compared using the Pearson squared test. This study performs univariate and multivariate logistic regression analyses to investigate the predictors associated with receiving chemotherapy for patients with osteosarcoma over the age of 40. The odds ratios (ORs) and corresponding 95% confidence intervals (CIs) were also presented. The Kaplan–Meier method was used to plot OS and CSS survival curves of the 2 groups and further comparison was carried out using the log-rank test. The prognostic predictors associated with multiple variables for OS and CSS were determined using the univariate and multivariate Cox proportional hazards models. Moreover, hazard ratios (HRs) and 95% CIs were presented. Stratified analysis by tumor grade and stage was performed between the nonchemotherapy and chemotherapy groups using Kaplan–Meier plots with log-rank tests. All tests were two-tailed, and this study considers P < .05 to be significant. All analyses performed within this study used the Statistical Package for the Social Sciences (SPSS) statistical software version 26.0 (SPSS, Chicago, IL, USA).
Results
Patient Characteristics
A total of 1032 eligible patients were enrolled in the analysis, in which, 586 (56.8%) patients received chemotherapy, and the remaining 446 (43.2%) patients did not receive chemotherapy. The baseline demographics, tumor features, and treatment characteristics of each group are shown in Table 1. No significant difference was found in race and surgery between the patients in the chemotherapy and nonchemotherapy groups (P > .05). However, a significant difference was identified in age, sex, location, grade, stage, and radiation between the group that did receive chemotherapy and the group that did not (P < .05). Demographically, in comparison to those in the nonchemotherapy group, patients categorized in the chemotherapy group were younger (aged between 40 and 60 years) (66.7% vs 42.4%) and more predominantly male (57.7% vs 47.1%). Regarding tumor features, the chemotherapy group had more patients with osteosarcoma located in the extremity (63% vs 57%), grade III/IV (92.2% vs 69.1%), and regional (47.1% vs 43.3%) and distant stage (24.6% vs 18.8%). In terms of treatment, the chemotherapy group received less radiation than the nonchemotherapy group (17.9% vs 24%).
Baseline Characteristics of Patients With or Without Receiving Chemotherapy Enrolled in this study (n = 1032).

Factors Associated With Receipt of Chemotherapy
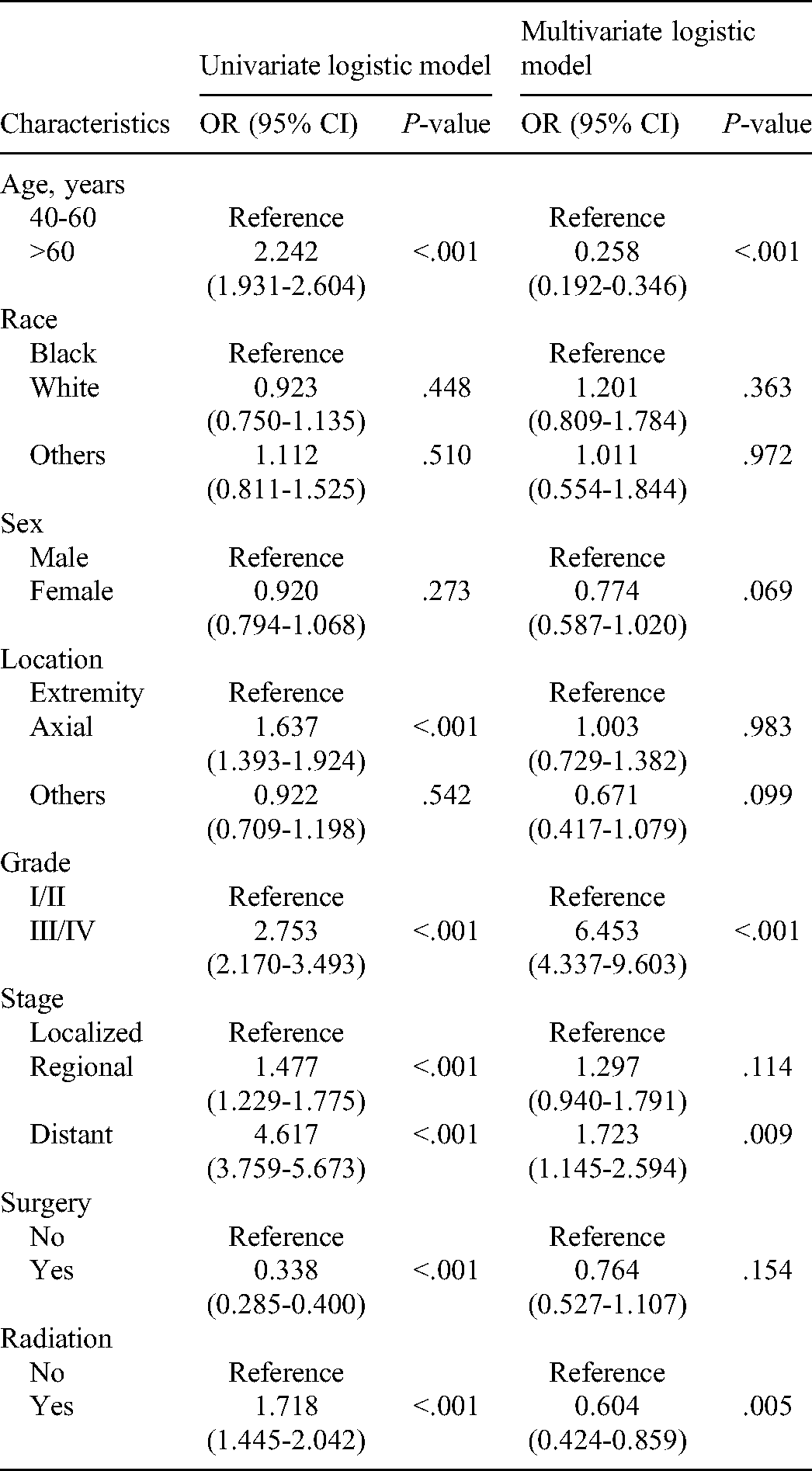
We performed univariate logistic analysis to identify the clinical characteristics that are associated with the receipt of chemotherapy. Table 2 shows the OR for receiving chemotherapy in the light of all variables in the univariate logistic regression model. Increased OR of receiving chemotherapy was associated with age > 60 years (OR = 2.242, 95% CI = 1.931-2.604, P < .001), axial location of osteosarcoma (OR = 1.637, 95% CI = 1.393-1.924, P < .001), grade III/IV (OR = 2.753, 95% CI = 2.170-3.493, P < .001), regional stage (OR = 1.477, 95% CI = 1.229-1.775, P = .009), distant stage (OR = 4.617, 95% CI = 3.759-5.673, P < .001), and receiving radiation (OR = 1.718, 95% CI = 1.445-2.042, P < .001). Multivariate logistic analysis indicated that age > 60 years (OR = 0.258, 95% CI = 0.192-0.346, P < .001), grade III/IV (OR = 6.453, 95% CI = 4.337-9.603, P < .001) and distant stage (OR = 1.723, 95% CI = 1.145-2.594, P = .009) were also associated with receiving chemotherapy.
Factors Associated With Receiving Chemotherapy (n = 1032).
Survival Analysis of all Patients
To explore whether receiving chemotherapy could benefit survival in patients with osteosarcoma that were over 40 years old, Kaplan–Meier plots were applied to screen survival in the chemotherapy and nonchemotherapy groups according to OS and CSS. The survival curves for OS and CSS are exhibited in Figure 2, and there was no significant difference (P > .05) in survival between the 2 groups.
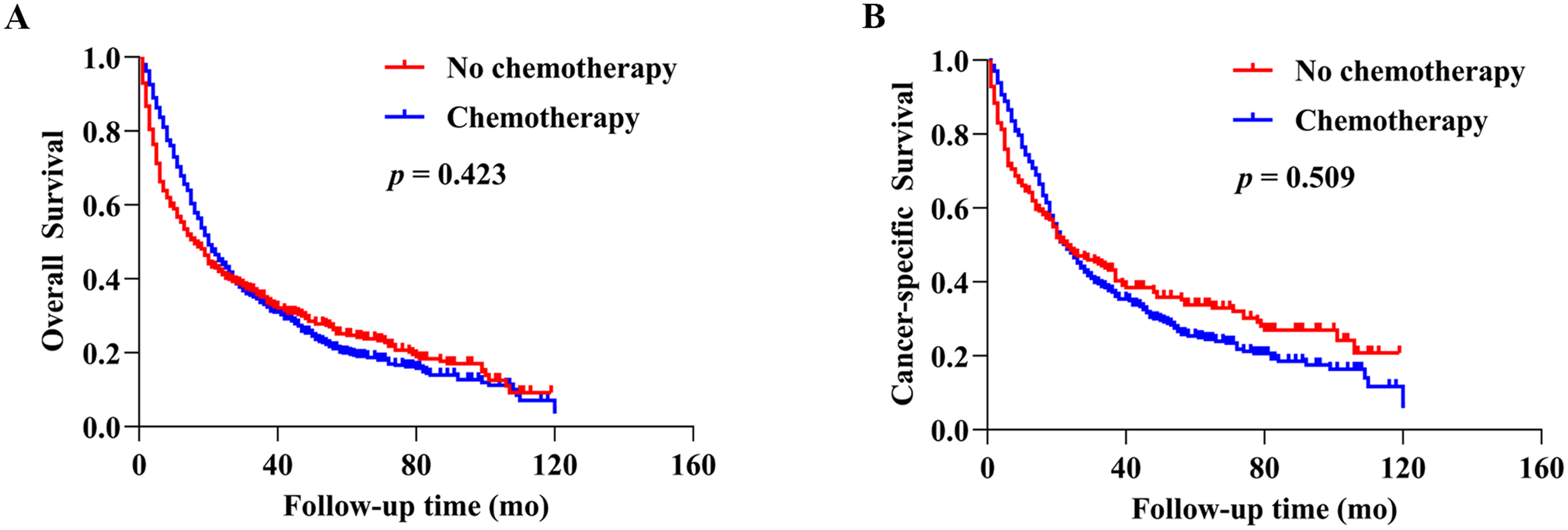
Kaplan–Meier curves with the log-rank tests of (A) overall survival (P > .05) and (B) cancer-specific survival (P > .05).
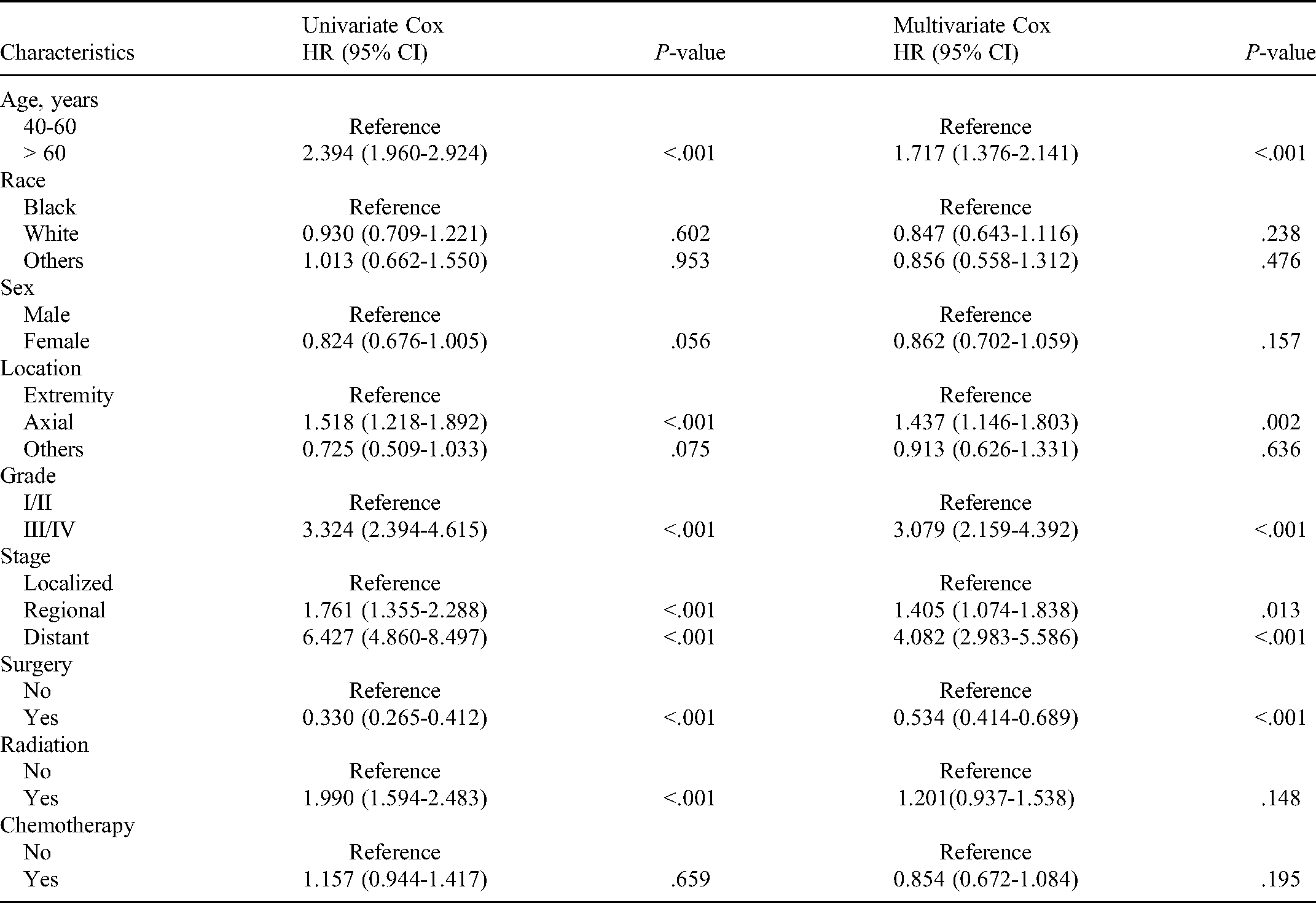
Subsequently, univariate and multivariate Cox proportional hazards analyses were performed to determine the prognostic factors associated with OS and CSS. In univariate analysis of OS (Table 3), the significant variables were age > 60 years, axial location of osteosarcoma, grade III/IV, regional stage, distant stage, receiving surgery, and receiving radiation (P < .05). Similarly, univariate analysis of CSS obtained the same results as the above variables, which were significant for OS (Table 4). All variables were subsequently adjusted and involved in the multivariate Cox analysis. As shown in Tables 3 and 4, our study considers age > 60 years, axial location of osteosarcoma, grade III/IV, regional stage, and distant stage to be independent risk factors for poor prognosis in both OS and CSS (HR > 1, P < .05). In contrast, this analysis further indicated that receiving surgery was an independent protective factor for favorable prognosis in both OS and CSS (HR < 1, P < .05).
Prognostic Factors Associated With Overall Survival (OS) Based On Univariate Cox Analysis Model and Multivariate Cox Analysis Model.

Prognostic Factors Associated With Cancer-Specific Survival (CSS) Based on Univariate Cox Analysis Model and Multivariate Cox Analysis Model.
Survival Analysis Stratified by Tumor Grade and Stage
As mentioned above, tumor grade and stage were not only highly associated with receiving chemotherapy but also important prognostic factors affecting OS and CSS in patients with osteosarcoma aged over 40 years (Tables 3 and 4). To identify the role of survival benefit from chemotherapy in these patients, we stratified the patients by tumor grade and stage. For both OS and CSS, Kaplan–Meier plots displayed that receiving chemotherapy exerted survival benefits for patients with grade III/IV and distant stage (Figures 3 and 4). However, it failed to provide a survival benefit for patients receiving chemotherapy with grade I/II and localized and regional stage, whereas it provided a survival benefit in patients that have not yet received chemotherapy (Figures 3 and 4).

Kaplan–Meier curves with the log-rank tests of overall survival for (A) I/II population, (B) III/IV population, (C) localized population, (D) regional population, and (E) distant population.

Kaplan–Meier curves with the log-rank tests of cancer-specific survival for (A) I/II population, (B) III/IV population, (C) localized population, (D) regional population, and (E) distant population.
Discussion
This study reviews the medical records of large population data amounting to 1032 patients aged older than 40 years with osteosarcoma collected from the SEER database to examine the clinicopathological characteristics and survival benefits of chemotherapy for patients in this age group. To the best of our understanding, this study represents the largest study conducted to date on the survival benefit of chemotherapy in this age group. Our results suggest that for patients aged older than 40 years with osteosarcoma, chemotherapy did not improve their OS or CSS. Stratified analysis showed that receiving chemotherapy may have survival benefits in patients with grade III/IV tumor and distant stage metastasis.
It can be found in some prospective studies that adjuvant chemotherapy combined with methotrexate, cisplatin, and doxorubicin has a survival benefit in young patients with osteosarcoma.11,20,21 However, these studies did not include the age group of patients over 40 years old. The role of chemotherapy in patients aged over 40 years with osteosarcoma remains controversial. Bacci et al 22 have reported that 34 patients aged between 40 and 60 years with nonmetastatic osteosarcoma of the extremities that have received chemotherapy were treated with a survival benefit. Moreover, a retrospective study by the European Musculoskeletal Oncology Society mentioned that 481 patients aged older than 40 years with osteosarcoma have also shown that treatment could be intensified by chemotherapy. 3 However, the above studies were conducted on small populations, so the power of these analyses is limited. In contrast, our study recruited a larger sample study population; thus, the power of the analysis in our study appears to be more compelling. In contrast, Joo et al 23 have analyzed the prognostic factors in a multinational, multiinstitutional platform studying patients with osteosarcoma aged older than 40 years in an Asian population and showed that receiving chemotherapy did not affect OS. Furthermore, Carsi et al 24 found chemotherapy to have no significant effect on disease-free survival in 47 patients aged older than 40 years. The findings of Joo and Cars et al.‘s study are consistent with our results, and our study used the Kaplan–Meier plot and Cox regression analysis reflecting no survival benefit on OS in patients with osteosarcoma who received chemotherapy and were aged older than of 40 years. Notably, our study showed no significant effect of chemotherapy on OS or CSS. In addition, we analyzed the clinicopathological factors associated with chemotherapy treatment using univariate logistic regression and further multivariate logistic regression. Our results suggested that grade III/IV and distant stage were independent factors associated with the receipt of chemotherapy, which suggests that osteosarcoma with high-grade tumors and metastasis was associated with receiving chemotherapy treatment. The findings of this study may help us to better identify the potential individuals that would benefit from receiving chemotherapy and facilitate clinician decisions.
We next aimed to highlight the effect of chemotherapy on survival in patients over the age of 40 years with osteosarcoma by basing this study on a large population data retrieved from the SEER database. Receiving chemotherapy was shown to have no benefit on survival of patients that fall within in this age group by univariate Cox regression analysis. However, undergoing surgery is considered to have a potential survival benefit. To balance the covariates of baseline demographics, clinicopathological characteristics, and treatment characteristics and to reduce selection bias, multivariate Cox regression analysis was performed in our study. Our results suggest that patients aged older than 40 years with osteosarcoma who received chemotherapy did not show better OS and CSS than the nonchemotherapy group, but surgical intervention achieved a significant survival benefit. Therefore, our findings may differ from that of Grimer et al.'s study, 3 which claimed a survival benefit of surgery plus chemotherapy treatment in patients aged 40 years and older with osteosarcoma. Our study showed a significant survival benefit after surgery but not after receiving chemotherapy treatment.
Although receiving chemotherapy has no survival benefit for patients aged older than 40 years that have osteosarcoma, we aimed to identify potential subgroups that would benefit from receiving chemotherapy. Logistic regression analysis showed that the underlying factors that determine chemotherapy options were highly associated with grade III/IV and stage distant. Therefore, we stratified patients in this age group for tumor grade and stage for further survival analysis. Interestingly, we found OS and CSS benefitted survival for patients over the age of 40 years with osteosarcoma who were treated with chemotherapy with grade III/IV tumor. In contrast, patients with grade I/II tumor who did not receive chemotherapy benefitted survival to OS and CSS. Our results allow clinicians and patients to consider chemotherapy for patients over 40 years old that have high-grade osteosarcoma, and patients with low-grade osteosarcoma may not receive chemotherapy. In addition, our results showed that receiving chemotherapy exerted a significant survival benefit on OS and CSS only for distant stage population, whereas there was no survival benefit for localized and regional stage populations.
Several limitations in the present study need to be acknowledged. First, this study was retrospective, and we cannot exclude selection bias. Second, this study lacked a precise analysis regarding the type, dose, and intensity of chemotherapy, which are thought of as essential considering that they affect patients’ chemotherapy sensitivity. Finally, we were unable to assess the effects chemotherapy treatment has on patient relapse-free survival because of the failure to include relapse data in the SEER database. However, this studying being the largest population-based analysis to date, our results suggest that chemotherapy did not significantly improve OS or CSS in patients aged over 40 years old with osteosarcoma but may improve tumor grade III/IV and distant stage population in these patients.
Conclusion
In conclusion, we found that chemotherapy did not improve OS or CSS in patients over the age of 40 with osteosarcoma. Survival may benefit from receiving chemotherapy in patients within this age group that had high-grade (grade III/IV) and metastasis (distant stage) tumors. Certainly, future multicentered, larger scaled, more prospective studies with long-term follow-up are still necessary to validate our findings.
Footnotes
Abbreviations
Acknowledgments
I sincerely thank my girlfriend Wu Weiwei for helping me collect materials I needed and put forward many priceless suggestions, and encourage me when I am confused.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the National Natural Science Foundation (grant no. 81860404) and the Foundation of Health Commission of Jiangxi Province (grant no. 20203120).
