Abstract
MicroRNAs are highly conserved noncoding RNA that negatively modulate protein expression at a posttranscriptional and/or translational level. MicroRNAs play an important role in the development and progression of human cancers, including osteosarcoma. Recent studies have shown that miR-100 was downregulated in many cancers; however, the role of miR-100 in human osteosarcoma has not been totally elucidated. In this study, we demonstrate that the expression of miR-100 was significantly downregulated in human osteosarcoma tissues compared to the adjacent tissues. Enforced expression of miR-100 inhibited cell proliferation, migration, and invasion abilities of osteosarcoma cells, U-2OS, and MG-63. Additionally, miR-100 also sensitized osteosarcoma cells to cisplatin and promoted apoptosis. Furthermore, overexpression of miR-100 decreased the expression of insulin-like growth factor I receptor and inhibited PI3K/AKT and MAPK/ERK signaling. In human clinical specimens, insulin-like growth factor I receptor was inversely correlated with miR-100 in osteosarcoma tissues. Collectively, our results demonstrate that miR-100 is a tumor suppressor microRNA and indicate its potential application for the treatment of osteosarcoma in future.
Introduction
Osteosarcoma is one of the most common primary bone tumor and occurs predominantly in adolescents and young adults. The poor prognosis and high lethality of the disease is largely due to its complex pathogenesis and resistance to conventional treatments. In recent years, the 5-year survival for patients with localized osteosarcoma remains 50% to 80% due to chemotherapy together with surgical techniques. 1,2 However, a part of the patients still response poor to a series of treatment, and the current treatment strategies have a limited efficacy in the treatment of metastatic and recurrent osteosarcoma. Hence, it’s urgent to develop new strategies and innovative therapeutics to further improve survival in patients with osteosarcoma. With the understanding of the pathogenesis, progression, and prognosis in osteosarcoma at the molecular level, the identification of the effector molecules and/or signal pathways responsible for regulating tumor development and chemotherapy-resistant is crucial for improving the osteosarcoma treatment level.
MicroRNAs (miRNAs), a short conserved noncoding RNA with 22 to 24 nucleotides long, function in posttranscriptional regulation of gene expression. 3 More than 30% of the human genome is regulated by miRNAs simultaneously targeting multiple genes. MicroRNAs have been shown to participate in a variety of physiological and pathological processes including tumor growth, differentiation, metastasis, cell apoptosis, and angiogenesis. 4 -6 Studies have reported that several miRNAs were dysregulated in osteosarcoma tissues or cell lines, including miR-17, miR-26a, miR-126, and miR-145. 7 -9 MicroRNAs also play a role in chemoresistance of osteosarcoma. Recent studies found that miR-33a is upregulated in chemoresistant osteosarcoma and could promote osteosarcoma cell resistance to cisplatin via downregulating twist. 10 Similarly, it was reported that miR-215 increases the chemoresistance to methotrexate and Tomudex by suppressing the expression of DTL. 11 These findings suggest that miRNAs may have a unique potential as a novel biomarker candidate for osteosarcoma. However, as only a few miRNAs were reported to be involved in osteosarcoma, not much is known about the expression and regulatory mechanism of miRNAs in osteosarcoma.
Recently, miR-100 has been reported to be downregulated in some types of cancer, such as lung, colorectal, prostate, and ovarian cancer. 12 -15 In these malignancies, forced expression of miR-100 inhibits cancer cell growth, indicating that miR-100 plays an important role in tumorigenesis and cancer progression. Previous studies have shown that miR-100 was significantly downregulated in osteosarcoma tissues and inhibited osteosarcoma cell proliferation by targeting Cyr61. 16 However, the role and mechanism of miR-100 in regulating tumorigenesis are still to be further elucidated. In this study, we will investigate (1) what is the role of miR-100 in osteosarcoma cell growth, migration, and invasion; (2) what is the direct target of miR-100 that is associated with cancer development; (3) whether there is a relationship between miR-100 and its direct target; and (4) whether miR-100 and its target are responsible for the resistance to cisplatin treatment in osteosarcoma. These results will provide new insights into the molecular mechanism of osteosarcoma development as well as provide potential new therapeutic strategy for osteosarcoma treatment in the future.
Methods and Materials
Tumor Specimens
Human osteosarcoma tumor samples were obtained from 20 patients from the Department of Orthopedics, Huaihe Hospital of Henan University. The identities of all tumor, normal muscle, and bone samples were confirmed by an experienced pathologist. This study was approved by the Research Ethics Committee of Huaihe Hospital of Henan University. All patients were given written informed consent. Tissue samples were collected at surgery, immediately frozen in liquid nitrogen, and stored until total RNAs or proteins were extracted.
Cell Culture and Reagents
Human osteosarcoma cell lines HOS, U-2OS, Saos-2, MG-63, and normal osteoblast cell (NHOst) were purchased from the Type Culture Collection of the Chinese Academy of Sciences. Cells were cultured in Dulbecco modified Eagle medium (DMEM) supplemented with 10% fetal bovine serum (FBS) and antibiotics (100 units/mL penicillin and 100 mg/mL streptomycin). Cells were incubated at 37°C in a humidified atmosphere of 5% CO2 in air. Antibodies against phosphorylated RAC-alpha serine/threonine-protein kinase (p-AKT) (Ser473), AKT, phosphorylated extracellular-signal-regulated kinases (p-ERK)1/2, and ERK1/2 were purchased from Cell Signaling Technology (Danvers, Massachusetts). Antibodies against insulin-like growth factor I receptor (IGFIR), hypoxia-inducible factor-1α (HIF-1a), and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) were from Bioworld Technology (Atlanta, Georgia). The growth factor-reduced Matrigel were from BD Biosciences (Bedford, Massachusetts).
Lentiviral Packaging and Stable Cell Line Establishment
To stably overexpress miR-100 in osteosarcoma cells, the lentiviral packaging kit was used (Thermo Fisher Scientific, Shanghai, China). Lentivirus carrying miR-100 or miR-100-negative control (miR-NC) was packaged using HEK293 T cells following the manufacturer’s manual. The lentiviral vector has red fluorescent protein tag, which can be used to check the efficiency of packaging using microscope. Cells were infected by lentivirus carrying miR-100 or miR-NC in the presence of polybrene (Sigma-Aldrich, Shanghai, China) and selected by puromycin (Sigma-Aldrich, Shanghai, China) for 2 weeks to obtain stable cell lines.
RNA Extraction, Reverse Transcription Polymerase Chain Reaction, and Quantitative Real-Time Polymerase Chain Reaction
RNA was isolated from harvested cells or human tissues with Trizol reagent according to the manufacturer’s instruction (Invitrogen, California). To determine the mRNA levels of vascular endothelial growth factor (VEGF), total RNAs were reversely transcribed by oligo-dT primer using PrimeScript RT Reagent Kit (Takara, Dalian, China). Housekeeping gene GAPDH was used as an internal control. The complementary DNAs were amplified by quantitative real-time polymerase chain reaction (qRT-PCR) using SYBR Premix DimerEraser (Takara, Dalian, China) on a 7900HT system, and fold changes were calculated by relative quantification (2−△△Ct).
Cell Proliferation Assay
U-2OS or MG-63 cells (3000 cells per well) were seeded onto 96-well plates and incubated in corresponding medium supplemented with 10% FBS. After 24, 48, 72, and 96-hour incubation, we added CCK-8 into each well, followed by 2-hour incubation. Absorbance value at 450 nm was then measured. Experiments were carried out in triplicate.
Invasion and Migration Assay
Migration and invasion assays were determined using 24-well BD Matrigel invasion chambers (BD Biosciences, Cowley, United Kingdom) in accordance with the manufacturer’s instructions. Cells were seeded at 5 × 104 cells/well in the upper well of the migration chamber in DMEM without serum, and the lower chamber well contained DMEM supplemented with 10% FBS, which aim to stimulate cell invasion. The chamber of invasion assay was treated with growth factor-reduced Matrigel. After incubation for 24 hours, noninvading cells were removed from the top well with a cotton swab, whereas the bottom cells were fixed with 3% paraformaldehyde, stained with 0.1% crystal violet, and extracted with 33% acetic acid. Then, cells were detected quantitatively using a standard microplate reader (optical density at 570 nm). Three independent experiments were conducted in triplicate.
Western Blotting
Total proteins were extracted by radioimmunoprecipitation assay buffer lysis buffer (100 mmol/L Tris, pH 7.4, 150 mmol/L NaCl, 5 mmol/L EDTA, 1% Triton X-100, 1% deoxycholate acid, 0.1% sodium dodecyl sulfate (SDS), 2 mmol/L phenylmethylsulfonyl fluoride, 1 mmol/L sodium orthovanadate, 2 mmol/L dithiothreitol, 2 mmol/L leupeptin, and 2 mmol/L pepstatin), and protein concentration was determined by bicinchoninic acid protein assay (Pierce, Shanghai, China). Proteins were then separated by 10% SDS-polyacrylamide gel electrophoresis and transferred to polyvinylidene fluoride membranes (Millipore, Shanghai, China). Membranes were incubated with primary antibodies overnight at 4°C and then with horseradish peroxidase-conjugated secondary antibodies. Signals were then visualized by ChemiDoc MP Imaging System (Bio-Rad, California).
Chemosensitivity Array
Cancer cells were seeded at a density of 4000 cells/well in a 96-well plate. Twenty-four hours later, freshly prepared cisplatin (Sigma-Aldrich, St Louis, Missouri) was added with the final concentration ranging from 1.25 to 80 μmol/L. Forty-eight hours later, cell viability was assayed by CCK8 kit.
Flow Cytometry Assay
Apoptosis was measured by flow cytometry. For annexin V staining, 5 μL phycoerythrin-annexin V, 5 μL propidium iodide (BD Pharmingen, Shanghai, China), and 400 μL 1 × binding buffer were added to the samples, which were incubated for 15 minutes at room temperature in the dark. Then, the samples were analyzed by flow cytometry (FACSCanto II; BD Biosciences) within 1 hour. The data were analyzed using FlowJo software. Three experiments were performed in triplicate.
Caspase-3 Activity Assay
The activity of caspase-3 was determined using the Beyotime caspase-3 activity kit. Cell lysates were prepared and incubated with reaction buffer containing caspase-3 substrate (L-Asparagine, N-acetyl-L-a-aspartyl-L-a-glutamyl-L-valyl-N-(4-nitrophenyl)-(9CI) [Ac-DEVD-pNA]) after the treatment as indicated. Caspase-3 activity assay was performed on 96-well plates by incubating 10 μL protein of cell lysate per sample in 80 μL reaction buffer containing 10 μL caspase-3 substrate (Ac-DEVD-pNA; 2 mmol/L) at 37°C for 2 hours according to the manufacturer’s protocol. The reaction was then measured at 405 nm for absorbance.
Statistical Analysis
All experiments were performed 3 times, and data were analyzed with GraphPad Prism 5 (La Jolla, California). The correlation between miR-100 expression and IGFIR levels in osteosarcoma tissues was analyzed using Spearman rank test. Statistical evaluation for data analysis was determined by t test. The differences were considered to be statistically significant at P < .05.
Results
MiR-100 Was Downregulated in Osteosarcoma Tissues and Cell Lines
To investigate the role of miR-100 in osteosarcoma, we first evaluated the expression levels of miR-100 in normal tissues and osteosarcoma tissues by qRT-PCR (Figure 1A). To compare the miR-100 expression levels among different clinical stages, we found that the miRNA expression levels in tumor tissues were correlated with the clinical stages of patients with osteosarcoma. The expression levels of miR-100 in high-grade tumors (World Health Organization [WHO] grade III) were downregulated compared with those in low-grade tumors (WHO grades I and II; Supplemental Figure 1). The results showed that the expression of miR-100 was consistently lower in the osteosarcoma tissues than in normal tissues. In addition, the expression of miR-100 in 4 osteosarcoma cell lines, HOS, Saos-2, MG-63, and U-2OS, was significantly decreased compared with the NHOst (Figure 1B). Our results indicated that miR-100 was downregulated in osteosarcoma tissues and cell lines.
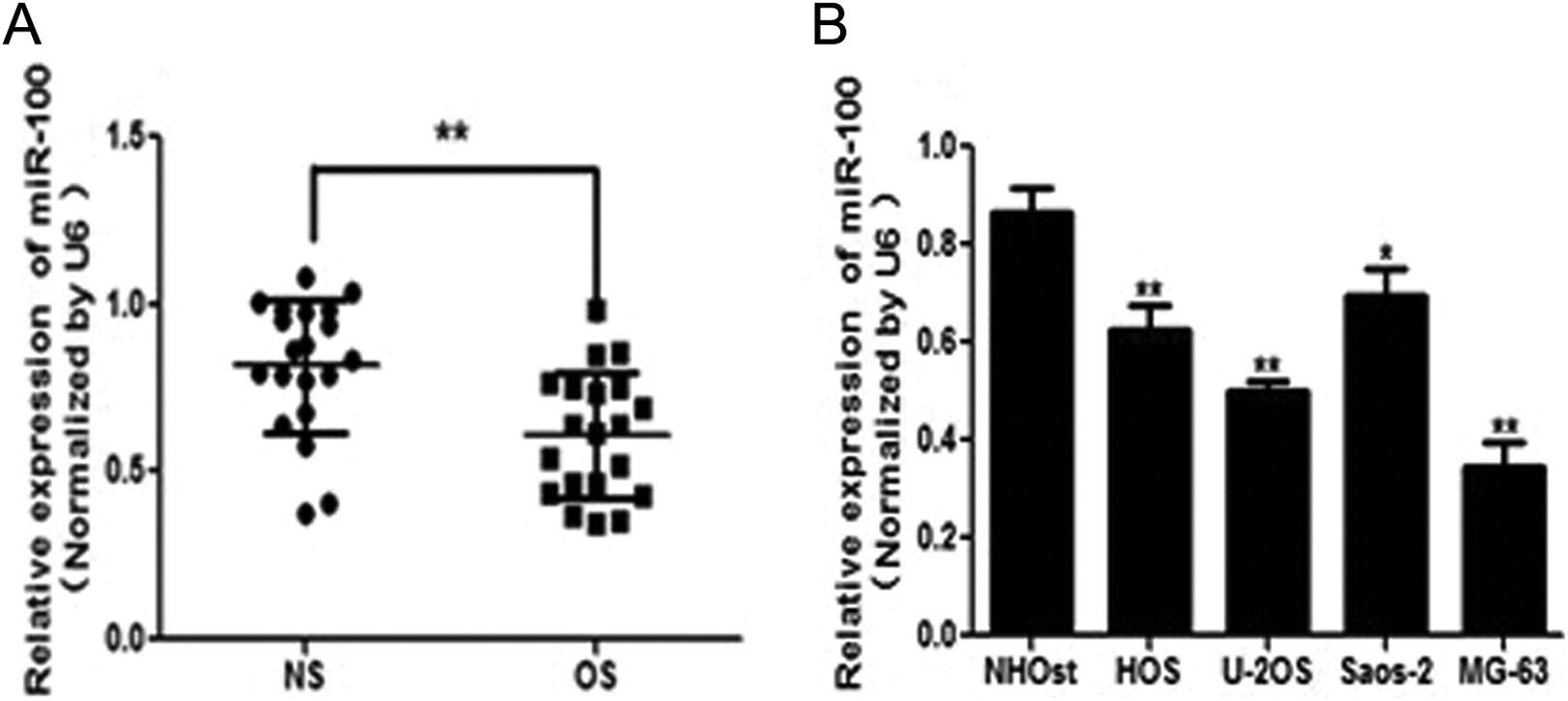
MiR-100 expression is downregulated in osteosarcoma tissues and cell lines. A, Relative miR-100 expression levels were analyzed by quantitative real-time polymerase chain reaction (qRT-PCR) in 20 paired osteosarcoma (OS) tissues compared with adjacent noncancerous tissues (NS). The U6 RNA level was used as an internal control. B, Relative miR-100 expression was analyzed in normal osteoblast cells (NHOst) and 4 OS cell lines, HOS, Saos-2, MG-63, and U-2OS. Data represent mean ± standard deviation of 3 replicates. *Significant difference at P < .05; **significant difference at P < .01.
Overexpression of miR-100 Suppresses Osteosarcoma Cell Proliferation, Migration, and Invasion
To study the role of miR-100 in osteosarcoma carcinogenesis, stable cell lines were established (Supplemental Figure 2A and B). The miR-100-overexpressing osteosarcoma cells were used to analyze cell growth. The results showed that cell growth was attenuated in miR-100-overexpressing osteosarcoma cells compared with osteosarcoma cells expressing miR-NC (Figure 2A).

Overexpression of miR-100 inhibited osteosarcoma cell proliferation, migration, and invasion. A, CCK-8 assay shows the effect of miR-100 on cell proliferation of MG-63 and U-2OS. B and C, The effects of miR-100 on cell migration and invasion were measured by transwell assays using MG-63 and U-2OS cells stably expressing miR-NC or miR-100. Overexpression of MiR-100 decreased cell migration and invasion in MG-63 and U-2OS cells. Data represent mean ± standard deviation of 3 replicates. *Significant difference at P < .05; **significant difference at P < .01.
Since migration and invasion are key characteristics of malignant tumor, we next investigated the effects of miR-100 on migration and invasion. Restoration of miR-100 dramatically inhibited the normally strong migration and invasive capacity of osteosarcoma cells (Figure 2B and C). Thus, our results suggest that overexpression of miR-100 suppresses osteosarcoma cell proliferation, migration, and invasion.
MiR-100 Inhibits Phosphatidylinositol 3-kinase (P13)/AKT and Mitogen-activated protein kinases (MAPK)/ERK Pathways via Targeting Insulin-Like Growth Factor I Receptor
Insulin-like growth factor I receptor has been reported to be putative target of miR-100. Osteosarcoma cells were cotransfected with the wild-type or mutated-type IGFIR luciferase reporter vector together with miR-100 or miR-NC for 24 hours, and luciferase activities in those cells were measured. As shown in Figure 3A, luciferase activities were significantly reduced in those cells transfected with the wild sequence and miR-100 but not in the cells with the mutant sequence and miR-100. Then, Western blotting analysis was conducted to measure the levels of IGFIR protein, and we found that the expression of IGFIR protein was downregulated in miR-100-treated cells (Figure 3C). These results suggest that miR-100 directly targets IGFIR by binding its seed region to their 3′-untranslated regions in osteosarcoma cells. To further determine the correlation between miR-100 and IGFIR levels, we measured the levels of IGFIR proteins in osteosarcoma tissues. Spearman correlation analysis demonstrated that IGFIR levels in osteosarcoma samples were inversely correlated with miR-100 expression levels (Spearman correlation r = −0.5263; Figure 3B).

MiR-100 inhibits PI3K/AKT and MAPK/ERK pathways via targeting insulin-like growth factor I receptor (IGFIR). A, Luciferase assay on MG-63 cells, which were cotransfected with miR-100-negative control (miR-NC) or miR-100 and a luciferase reporter containing the full length of IGFIR 3′-untranslated region (3′-UTR; wild type [WT]) or a mutant (Mut) in which 4 nucleotides of the miR-100-binding site were mutated. Luciferase activities were measured 24 hours posttransfection. MiR-100 markedly suppressed luciferase activity in IGFIR 3′-UTR (WT) reporter constructs. The data are means ± standard deviation for separate transfections (n = 4). B, Spearman correlation analysis was used to determine the correlation between the expression levels of IGFIR and miR-100 in human osteosarcoma specimens. C, The expression levels of IGFIR, phosphorylated AKT (p-AKT), phosphorylated ERK1/2 (p-ERK1/2), and hypoxia-inducible factor 1α (HIF-1α) were decreased in cells with miR-100 overexpression by immunoblotting, whereas AKT and ERK1/2 protein levels were not changed. Overexpression of IGFIR restored miR-100–inhibited cellular protein levels of p-AKT, p-ERK1/2, and HIF-1α. D, Overexpression of IGFIR rescued vascular endothelial growth factor (VEGF) mRNA expression inhibited by miR-100. The VEGF mRNA level was normalized to that of glyceraldehyde 3-phosphate dehydrogenase (GAPDH). Data represent mean ± standard deviation of 3 replicates. * or # P < .05. *Significant difference compared to control; #significant difference compared to miR-100 treatment alone.
The PI3K/AKT and MAPK/ERK pathways act as major downstream of IGFIR signaling, and several downstream factors such as HIF-1α and VEGF have been linked to the PI3K/AKT and MAPK/ERK pathways. Cellular levels of p-AKT and p-ERK1/2 were significantly decreased in osteosarcoma cells stably expressing miR-100 compared with miR-NC, whereas no statistically significant reduction of AKT and ERK1/2 was detected (Figure 3C). Here, we observed that HIF-1α and VEGF levels in miR-100–expressing osteosarcoma cells were both downregulated (Figure 3C and D). Overexpression of IGFIR restored miR-100–inhibited cellular protein levels of p-AKT, p-ERK1/2, HIF-1α, and VEGF. These results suggest that miR-100 inhibits PI3K/AKT and MAPK/ERK pathways via targeting IGFIR.
Overexpression of miR-100 Increases Chemosensitivity of Osteosarcoma Cells to Cisplatin by Inhibiting Its Target IGFIR
Resistance to cisplatin treatment is one of the major causes for the failure of chemotherapy in treating osteosarcoma. Therefore, it is critical to discover new strategies to increase the effectiveness of cisplatin for therapeutic purposes. Our results showed that overexpression of miR-100 in osteosarcoma cells significantly increased chemosensitivity to treatment of cisplatin (Figure 4A). Furthermore, cell growth rate in the presence of cisplatin (5 μmol/L) was assayed by CCK-8 proliferation assay at different time points; interestingly, forced expression of IGFIR reversed miR-100–induced osteosarcoma cell chemosensitivity to cisplatin (Figure 4B). To further study whether miR-100 and its target IGFIR play a role in cell apoptosis in the presence of cisplatin treatment, fluorescence-activated cell sorting analysis was performed to detect cell apoptosis rates. The combination of miR-100 and cisplatin treatment significantly induced cell apoptosis, whereas forced expression of IGFIR partially abolished the effect induced by miR-100 plus cisplatin treatment (Figure 4C and Supplemental Figure 3). We also found that the activity of caspase-3, a key executor of cell apoptosis, was significantly upregulated upon treatment by miR-100 plus cisplatin compared with miR-100 or cisplatin treatment alone, whereas IGFIR overexpression attenuated the activation of caspase-3 induced by miR-100 plus cisplatin treatment (Figure 4D). These results indicated that miR-100 renders osteosarcoma cells more sensitive to cisplatin treatment, and miR-100 and cisplatin combination induced apoptotic effect through targeting IGFIR in osteosarcoma cells.
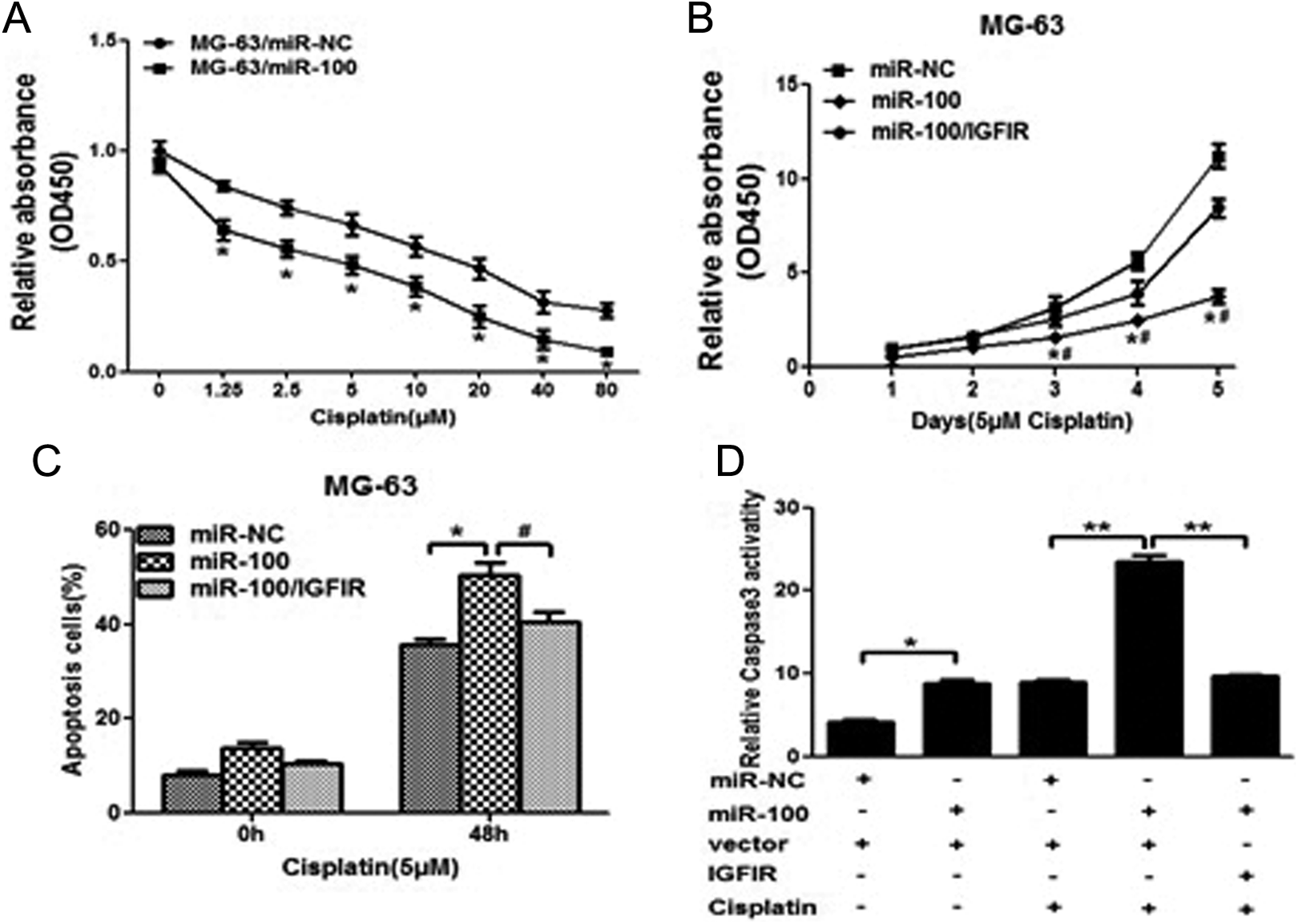
Overexpression of miR-100 increases chemosensitivity of osteosarcoma cells to cisplatin by inhibiting its target IGFIR. A, MG-63 cells stably expressing miR-100-negative control (miR-NC) or miR-100 were pretreated with various concentration of cisplatin for 48 hours and subjected to CCK8 assay. B, MG-63 cells stably expressing miR-NC, miR-100, or miR-100 forced expression of IGFIR were pretreated with 5 μmol/L of cisplatin for definite time points and subjected to CCK8 assay, and apoptosis analysis by flow cytometry (C). D, MG-63 cells stably expressing miR-NC or miR-100 were transfected with 2 mg pCMV6 vector or pCMV6–IGFIR plasmid and cultured with or without cisplatin. After 72 hours, the relative caspase-3 activities were determined. Data represent mean ± standard deviation of 3 replicates. * or # P < .05. **P < .01. *Significant difference compared to control; #Significant difference compared to miR-100 treatment alone.
Discussion
Dysregulation of various miRNA has been implicated in the development and progression of human cancers. 17,18 Maire et al found that 38 miRNAs differentially expressed were identified in 7 human osteosarcoma tissue samples compared with normal human osteoblasts. 19 Here, we identified that miR-100 was downregulated in osteosarcoma tissue samples. In combination with previous reports revealing the roles of miR-100 in some other types of cancer, 12 -15 we further confirmed that miR-100 may function as a tumor suppressor in osteosarcoma carcinogenesis and progression. Meanwhile, we further confirmed that IGFIR was a target of miR-100 consistent with previous studies. More importantly, for the first time, we showed that IGFIR was upregulated in osteosarcoma tissue samples and was inversely correlated with miR-100 levels. Thus, this study may provide new therapeutic strategies for osteosarcoma prevention and treatment.
Insulin-like growth factor I receptor is a tyrosine kinase receptor that is mainly activated by IGF1 and IGF2 in autocrine and paracrine manners. 20,21 Insulin-like growth factor I receptor activates multiple downstream signaling cascades, including PI3K/AKT and MAPK/ERK signaling pathways, which regulate cell proliferation, differentiation, and survival. 22 -27 Recent studies have identified some miRNAs posttranscriptionally regulating IGFIR, such as miR-145, miR-375, and miR-143, suggesting that miRNAs targeting IGFIR have an important role in carcinogenesis. 28,29 In this study, we confirmed that overexpression of miR-100 in osteosarcoma cells inhibited cell proliferation, migration, invasion, and decreased the levels of p-AKT and p-ERK1/2, HIF-1α, and VEGF. Meanwhile, cisplatin is still one of the most commonly used agents in chemotherapy due to the therapeutic advantages. However, resistance to this drug is also often observed, and therefore enhancing the sensitization of cancer cells to cisplatin-induced apoptosis became an important strategy for chemotherapy. A large body of evidence has indicated that IGFIR plays important roles in regulating tumor resistance to cisplatin. 30,31 Interestingly, we also found that forced overexpression of miR-100 promoted the effects of cisplatin in osteosarcoma cells, promoted apoptosis, and increased the activity of caspase-3. Thus, it is important that an miR-100 restoration approach may offer a new modulation strategy to overcome chemoresistance to cisplatin treatment in osteosarcoma.
In conclusion, our present investigation suggests that miR-100 functions as a tumor suppressor in osteosarcoma by negatively regulating IGFIR. In human osteosarcoma tissues, miR-100 levels are inversely related to the protein levels of IGFIR. MiR-100 impairs tumor cell growth, migration, and invasion through the PI3K/AKT and MAPK/ERK signaling pathways. Moreover, we demonstrate that miR-100 sensitizes cisplatin treatment in an IGFIR-dependent manner. Although miRNA-based therapeutics are still in the initial stages of development, our findings are encouraging and suggest that miR-100 could be a potential target for the treatment of osteosarcoma in future.
Conclusions
Our results indicated that miR-100 could exert tumor suppression function by inhibiting PI3K/AKT and MAPK/ERK signaling pathways via targeting IGFIR and could be used as a potential tumor suppressor for future novel therapeutic development.
Footnotes
Authors’ Note
Yang Liu and Shu-Tao Zhu contributed equally to this work. The study was conceived and designed by Yang Liu and Shu-Tao Zhu. The experiments were performed by Yang Liu, Shu-Tao Zhu, and Xiao Wang. The data were analyzed by Jun Deng, Wei-Hua Li, Peng Zhang, and Bing-Shan Liu. The paper was written by Yang Liu and Shu-Tao Zhu.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Abbreviations
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
