Abstract
Keywords
Introduction
Thyroid nodules are widespread in the general population, with a prevalence of approximately 70.0% in an adult undergoing ultrasonography, but only 5.0%–15.0% of nodules are malignant.[1–3] Therefore, it is important to differentiate benign from malignant nodules so as to prevent undertreatment or overtreatment. Ultrasound-guided fine-needle aspiration (FNA) biopsy is a reliable method with good sensitivity and specificity for a definitive benign or malignant diagnosis of thyroid nodules.[4,5] However, the limitations of FNA include nondiagnostic modality, indeterminate cytology, and false-negative and false-positive results, which are 10.0%–40.0% of all cytological results.[6,7] An indeterminate cytopathology report results in difficult clinical decisions. The options for management may include sonographic monitoring, repeat FNA, or surgical removal. To address this situation, molecular biomarkers are used to discriminate benign from malignant nodules and thus guide management.[8] The BRAF V600E mutation activates the classical thyroid tumorigenic Ras/Raf/MEK/ERK (MAPK) signaling pathway. It is the most common genetic alteration in papillary thyroid carcinoma (PTC) and has a high positive predictive value (PPV, 95.5%–100.0%) in PTC.[9–11] Besides its diagnostic value, many studies have shown the utility of BRAF V600E in prognosis prediction, as this mutation is associated with the aggressive clinicopathological features of PTC.[11]
Tumor tissue samples or cytology samples are considered as standard materials for genetic molecular testing. However, this test is often limited due to the insufficiency of cytology samples.[12] FNA washout precipitation specimens have become a promising alternative for molecular analysis. The detection of endocrine biochemical markers in FNA washout was widely used due to its high diagnostic efficiency, for example, FNA-tg, FNA-CT.[13,14] However, relevant reports on molecular variations among FNA washout precipitation specimens are still rare to date. Therefore, the goal of this study was to demonstrate the feasibility of the residual, routinely discarded FNA washout precipitation specimens for genetic analysis.
Materials and Methods
Patient Selection and Study Design
A total of 115 consecutive patients with thyroid nodules underwent FNA were enrolled from The Third Affiliated Hospital of Sun Yat-sen University from January 2020 to September 2020. Ultrasound-guided FNA specimens collected from all patients were divided into two parts including consecutive FNA biopsy specimens and discarded FNA washout precipitation (according to the preliminary experimental results, the FNA washout precipitation was a suitable specimen for examination compared to other alternative specimens, Supplementary Table1). In fact, BRAF V600E mutation was confirmed by FNA biopsy specimens soon after the FNA biopsy specimen were obtained for routing clinical diagnosis, with a result of 59 were BRAF V600E positive and 56 were negative. As a comparison test, the FNA washout precipitation were collected for detecting BRAF V600E mutation too. The clinicopathological data, including age, gender, nodule information, Thyroid imaging reporting and data system (TIRADS) scores and The Bethesda System for Reporting Thyroid Cytopathology (TBSRTC) scores were collected from the electronic medical records. This observational clinical study using real world data was approved by the institutional review board by the Third Affiliated Hospital of Sun Yat-sen University Ethics Committee with exemption of informed consent (approve number: [2020] 02-289-01), and all participants provided informed verbal consent.
Biopsy Formalin-Fixed Paraffin-Embedded Specimens
Thyroid FNA biopsy samples were centrifuged at 3500 rpm for 10 min in a 15-mL centrifuge tube with sample preservation solution. After the supernatant was discarded, 1 mL of buffer solution was added to clean the sediment. After centrifugation at 3500 rpm for 10 min again, the precipitate was wrapped in rice paper and put into an embedding box, followed by dehydration using a Leica dehydrator. The next day, the embedding box was taken out, and wax blocks were prepared.
FNA Washout Precipitation Specimens
After collecting FNA cytology samples, each needle was rinsed with 1.0 mL of saline (0.9% w/v NaCl). The wash solution from all needles was centrifuged at 2000rpm for 10 min to collect the residue. All the samples were stored at 4 °C until use, and the maximum storage time of all samples was less than 24 h.
DNA Extraction
DNA was extracted from the formalin-fixed paraffin-embedded (FFPE) samples and FNA washout precipitation specimens using an AmoyDx FFPE DNA Extraction Kit (Amoy Diagnostics Co., Ltd, Xiamen, China) following the manufacturer's protocols. A Nanodrop spectrophotometer (Thermo-Fisher Scientific, USA) was used to assess the concentration and purity of extracted DNA.
Mutation Screening of BRAF V600E
BRAF V600E mutations were tested using an AmoyDx BRAF Mutations Detection Kit (Amoy Diagnostics Co., Ltd, Xiamen, China) following the principle of the amplification refractory mutation system. Briefly, ARMS-PCR was carried out on a 7500 Real-Time PCR System (Applied Biosystems) following the manufacturer's protocol. The cycling conditions were as follows: 1 cycle of initial denaturation for 5 min at 95 °C (1 cycle), followed by 15 cycles at 95 °C for 25 s, 64 °C for 20 s, and 72 °C for 20 s, with a final step of annealing and elongation of 31 cycles at 93 °C for 25 s, 60 °C for 35 s, and 72 °C for 20 s. Threshold cycle (CT) values were recorded when the mixtures were amplified to a specific fluorescent level. All results were confirmed according to the criterion suggested by the manufacturer.
Statistical Analysis
The analysis was performed using SPSS 23.0 (IBM Inc., IL, USA). McNemar–Bowker test and kappa (κ) statistics were used to analyze and indicate the concordance of results generated by washout precipitation and FFPE samples. The relationship between gene mutations and clinicopathological characteristics was analyzed using the χ2 test or Fisher's exact test. The two-sided significance level was set at P < 0.05.
Results
Case Date and Clinical Features
Ultrasound-guided FNA was performed in 115 patients with thyroid nodules. The clinical characteristics of 115 patients are shown in
The clinical characteristics and BRAF mutation detected using FNA samples

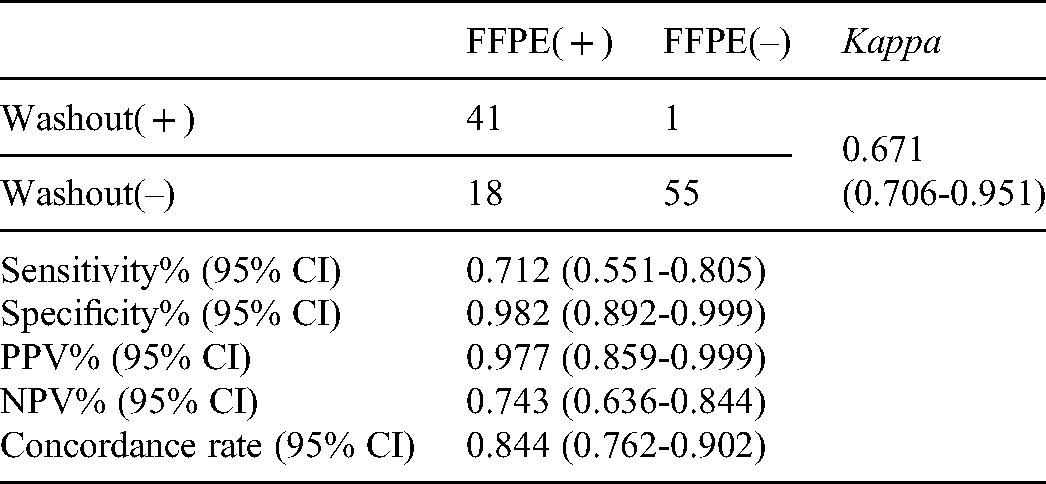
Concordance of BRAF V600E detected using FNA washout precipitation and biopsy FFPE specimens
A total of 115 samples were successfully analyzed using both FNA washout precipitation and biopsy FFPE specimens. The results of 96 patients were consistent between FNA washout precipitation and biopsy FFPE specimens, with 41 BRAF V600E positive and 55 BRAF V600E negative, achieving a concordance rate of 84.4% [95% confidence interval (CI): 0.762 to 0.902]. Further, 19 samples had inconsistent results, including 18 FFPE-positive/washout-negative specimens and 1 FFPE-negative/washout-positive specimen. As shown in
Performance evaluation of BRAF V600E detected using FNA biopsy FFPE and washout precipitation specimens
Comparison of BRAF V600E detected using biopsy FFPE and FNA washout precipitation specimens among patients with PTC
A total of 79 patients diagnosed as PTC by histopathology (including 2 TBSRTC II, 47 TBSRTC V, and 30 TBSRTC VI) were analyzed. The results showed that 74.7% (59 / 79) patients with PTC carried BRAF V600E mutation in the FNA biopsy FFPE specimens, while the rate detected in the FNA washout precipitation specimens was 54.4% (43/79). In addition, the results of BRAF V600E detected with FNA washout precipitation specimens were used to diagnose PTC in patients classified as TBSRTC V, and a 100.0% PPV was obtained (

Positive predictive value (PPV) of BRAF V600E detected using FFPE and FNA washout for PTC diagnosis.
Comparison of BRAF mutation detection in FNA biopsy PPFE or washout precipitation specimens from patients with PTC with or without capsule invasion

Without capsule invasion; bwith capsule invasion
Relationship between clinical characteristics and BRAF V600E status in patients with PTC
Furthermore, the relationship between BRAF V600E mutation status and clinical characteristics, including sex, age, lesion numbers, tumor size, capsule invasion, and lymph metastasis of patients with PTC, was analyzed. The results showed that the BRAF V600E mutation was significantly associated with tumor size (P = 0.030) and was more common in patients with a tumor diameter ≥10 mm (
Relationship between clinical characteristics and BRAF V600E mutation in PTC

Mut: Mutation; WT: Wild Type.
Discussion
The TBSRTC system (2017 version) is generally used as the diagnostic standard for thyroid cytopathology in China and the preferred method for benign and malignant screening of thyroid nodules.[15,16] However, TBSRTC III–V, which are uncertain diagnoses, pose significant clinical challenges because benign and malignant diagnoses directly determine the subsequent treatment. The BRAF V600E mutation is commonly found in PTC; in China, its mutation rate is up to 69.0%–85.3% in patients with PTC.[17–19] The BRAF V600E mutation is proposed as a diagnostic adjunctive tool in the differential diagnosis of indeterminate cytological findings and prognostic evaluation of thyroid nodules, which guide the optimal management.[20,21] Moreover, BRAF/RAS mutations and RET/PTC rearrangement are also used for the diagnosis of thyroid nodules.[4,8]
With the growing number of molecular biomarkers needed for the clinical management of patients with thyroid cancer, the judicious use of limited-volume specimens is critically needed. Thyroid FNA washout precipitation specimens have become a promising alternative for molecular analysis because they are of a less invasive nature and easy to perform. Giulia et al. evaluated the practical application of BRAF V600E mutation analysis in FNA washout precipitation specimens from thyroid nodule FNA; 20 nodules were positive for BRAF V600E, while 869 nodules were wild type.[22] However, direct head-to-head comparison studies for detecting BRAF V600E between FNA washout precipitation and biopsy FFPE specimens are scarce.
In this study, 115 samples were successfully analyzed using both FNA washout precipitation and biopsy FFPE specimens. The results showed that the BRAF V600E status detected in 96 FNA washout precipitation specimens were consistent with that detected in FNA biopsy FFPE specimens, including 41 BRAF V600E positive and 55 BRAF V600E negative, achieving a concordance rate of 84.4% (kappa = 0.671). The low proportion of tumor cells in washout specimen may contribute to the inconsistency of the 19 samples, including 18 FFPE ( + ) / washout (-) specimens and 1 FFPE (-) / washout ( + ) specimen. It is quite possible that the proportion of tumor cells in washout specimen is too low to get adequate positive BRAF V600E signal. In clinical practice, it is generally recommended that the content of tumor cells in the samples used for molecular detection should be above 20%. Unfortunately, due to the lack of sufficient FNA washout precipitation, we were unable to assess the percentage of tumor cells. Next, the comparative studies of proportion of tumor cells between different specimens will be considered in the future. For the FNA washout precipitation specimen with BRAF V600E positive, while the FNA biopsy FFPE specimen was negative. The histopathological diagnosis was papillary carcinoma. Therefore, it was highly suspected that the FNA washout precipitation specimens that detected the BRAF mutation originated from a malignant thyroid nodule.
In addition, a higher mutation frequency of BRAF V600E was observed in patients with higher TIADRS score and TBSRTC score. Some studies have shown the rates of malignancy in TBSRTC category I, III, and V nodules were 15.3%, 39.3%, and 96.8%, respectively, and BRAF V600E exhibited similar diagnostic performance to that of TIRADS in BSRTC III/V nodules, the combination of the two diagnostic approaches significantly enhanced the sensitivity, especially among high TBSRTC higher TIADRS score and a TBSRTC score.[23]
The BRAF V600E mutation status was shown to be positive in 90.9% (10/11) patients with capsule invasion using FNA washout precipitation specimens, which was higher than that detected in patients without capsule invasion using FNA washout precipitation specimens 44.4% (12/27). As previous studies have shown that BRAF alterations were significantly more common in PTCs with thyroid capsule invasion than in tumors that without thyroid capsule invasion.[24] Giovanni have proposed that Large subcapsular mPTCs (group A: tumors size ≥5 mm and distance from the thyroid capsule = 0 mm) represent the group with the most worrisome characteristics, including aggressive growth with distinctively invasive features, such as infiltrative tumor border, intraglandular tumor spread, presence of psammoma bodies into the surrounding parenchyma, vascular invasion, and lymph node metastases.[25] So, tumor cells were more readily available in the patients with capsule invasion. On the other hand, for the patients without capsule invasion, FNAB specimens tend to have a higher proportion of normal cells.
The results showed that FNA washout precipitation was a feasible specimen for BRAF V600E mutation detection. In addition, the BRAF V600E mutation was detected in thyroid FNA washout precipitation specimens, providing rapid genetic testing results for improving the accuracy of cytopathological diagnosis. Thus, the FNA washout precipitation specimen was an important supplement for the genetic detection of thyroid nodules. Among these 77 patients diagnosed as TBSRTC V and TBSRTC VI by cytopathology, 1 was found to be false negative using an FNA biopsy FFPE specimen, indicating that the FNA washout precipitation specimen, combined with the FNA biopsy FFPE specimen, could be used as an effective supplementary specimen to reduce the false-negative rate of BRAF gene detection in thyroid cancer.
This study had several limitations. First, the proportion of tumor cells in fresh FNA thyroid tissue samples was unknown. Fresh samples might have no or insufficient tumor cells, which did not reach the detection limit of the genetic test kit. Second, the number of patients included in the study was not sufficient. Third, only cytology FFPE samples were used for comparison due to the lack of tissue sample validation (histopathological results of 82 patients were obtained). Fourth, fewer genes were detected. Thus, further large-scale and multi-gene prospective investigations are warranted to solve some of these limitations.
Conclusion
115 samples were successfully tested using both FNA washout precipitation and biopsy FFPE specimens in this study. The results indicated that FNA washout precipitation specimens could be used as a supplementary sample type to shorten the genetic testing cycle on the basis of ensuring PPV. Furthermore, BRAF V600E mutations in FNA washout precipitation specimens showed 100.0% sensitivity and 100.0% specificity for diagnosing patients with PTC having thyroid capsule invasion.
Supplemental Material
sj-docx-1-tct-10.1177_15330338211057982 - Supplemental material for Fine-needle Aspiration Washout Precipitation Specimens: An Acceptable Supplement to Genetic Mutation Detection of Thyroid Nodules
Supplemental material, sj-docx-1-tct-10.1177_15330338211057982 for Fine-needle Aspiration Washout Precipitation Specimens: An Acceptable Supplement to Genetic Mutation Detection of Thyroid Nodules by Yongmei Cui, Xiangqi Huang, Jinrui Guo, Nana Zhang, Jing Liang, Yiwang Zhang, Yueting Liao and Dan He in Technology in Cancer Research & Treatment
Supplemental Material
sj-docx-2-tct-10.1177_15330338211057982 - Supplemental material for Fine-needle Aspiration Washout Precipitation Specimens: An Acceptable Supplement to Genetic Mutation Detection of Thyroid Nodules
Supplemental material, sj-docx-2-tct-10.1177_15330338211057982 for Fine-needle Aspiration Washout Precipitation Specimens: An Acceptable Supplement to Genetic Mutation Detection of Thyroid Nodules by Yongmei Cui, Xiangqi Huang, Jinrui Guo, Nana Zhang, Jing Liang, Yiwang Zhang, Yueting Liao and Dan He in Technology in Cancer Research & Treatment
Supplemental Material
sj-docx-3-tct-10.1177_15330338211057982 - Supplemental material for Fine-needle Aspiration Washout Precipitation Specimens: An Acceptable Supplement to Genetic Mutation Detection of Thyroid Nodules
Supplemental material, sj-docx-3-tct-10.1177_15330338211057982 for Fine-needle Aspiration Washout Precipitation Specimens: An Acceptable Supplement to Genetic Mutation Detection of Thyroid Nodules by Yongmei Cui, Xiangqi Huang, Jinrui Guo, Nana Zhang, Jing Liang, Yiwang Zhang, Yueting Liao and Dan He in Technology in Cancer Research & Treatment
Supplemental Material
sj-docx-4-tct-10.1177_15330338211057982 - Supplemental material for Fine-needle Aspiration Washout Precipitation Specimens: An Acceptable Supplement to Genetic Mutation Detection of Thyroid Nodules
Supplemental material, sj-docx-4-tct-10.1177_15330338211057982 for Fine-needle Aspiration Washout Precipitation Specimens: An Acceptable Supplement to Genetic Mutation Detection of Thyroid Nodules by Yongmei Cui, Xiangqi Huang, Jinrui Guo, Nana Zhang, Jing Liang, Yiwang Zhang, Yueting Liao and Dan He in Technology in Cancer Research & Treatment
Footnotes
Acknowledgments
We are extremely grateful to Dr Fei Yao and Yanfang Chen, Medical Research Department of Amoy Diagnostics Co., Ltd, for their support in this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
Dan He, Yueting Liao, Yongmei Cui, and Xiangqi Huang design the conception; Yongmei Cui, Xiangqi Huang collect and assemble data; Jinrui Guo, Nana Zhang and Jing Liang collect samples; and Nana Zhang, Yiwang Zhang are responsible for the cytopathological and histopathological diagnosis. Nana Zhang, Yongmei Cui, and Yueting Liao are the guarantors of this work. Yueting Liao and Dan He, had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (81702409,81902416), and the National Natural Science Youth Cultivation Foundation of The Third Affiliated Hospital of Sun Yat-sen University, and Foundation of Xiamen Municipal Bureau of Science and Technology (China, IDs: 3502Z20184038).
Ethical Statement
This observational clinical study was approved by the institutional review board by the Third Affiliated Hospital of Sun Yat-sen University Ethics Committee with exemption of informed consent (approve number: [2020] 02-289-01), and all participants provided informed verbal consent.
Supplemental Material
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
