Abstract
Introduction
This study explored the clinical value and application of ultrasound contrast imaging technology in the American College of Radiology Thyroid Imaging Reporting and Data System (ACR TI-RADS) TR4 benign and malignant thyroid nodules.
Methods
We retrospectively analyzed data from the medical records of 40 patients who met the inclusion criteria between January 2020 and December 2023. Each patient was evaluated using the ACR TI-RADS classification and contrast-enhanced ultrasound (CEUS). The agreement between pathological outcomes and ultrasound indicators and the diagnostic value and significance of each parameter were assessed.
Results
The diameters did not differ between benign and malignant nodules (P = 0.324). Ring enhancement was closely associated with benign thyroid nodules, with a negative predictive value of 100%. Homogeneous enhancement and enhancement intensity showed good diagnostic value for pathological results, with an area under the curve (AUC) > 0.8. This parameter showed a high diagnostic value for serial and parallel combinations of homogeneous enhancement and enhancement intensity, with a sensitivity of 77.8% and specificity of 85.7% for the serial combination and 100% and 71.4%, respectively. for the parallel combination.
Conclusion
Among ACR TI-RADS TR4 nodules, diameter 1.0–1.5 cm was not significantly correlated with a benign or malignant nature. Nodules featuring ring enhancement with ring-enhancing features should be considered benign. Similarly, nodules showing no, homogeneous, or high enhancement with clear borders on CEUS imaging may be benign. However, nodules with uneven low enhancement or unclear borders may be malignant. Therefore, uneven and low enhancement on CEUS imaging may have a high diagnostic value for malignant nodules. Moreover, the combination of these features may have even higher specificity.
Keywords
Introduction
Thyroid nodules can be detected in most patients using routine imaging.1,2 More than 80% of these nodules are benign and do not require medical intervention if compression or thyroid dysfunction occurs. However, malignant nodules account for 5%–15% of cases and require immediate surgical treatment to prevent metastasis and further deterioration.3,4 Although the proportion of malignant thyroid nodules is low, the incidence of thyroid cancer ranks first among endocrine malignancies. 5 Thus, the determination of the benign or malignant status of thyroid nodules is critical for clinical diagnosis and treatment and rapid and accurate identification of meaningful nodules in the large population of thyroid nodules is urgently needed.
Ultrasound examination has the advantages of safety, convenience, and high repeatability and is currently the preferred method for the diagnosis of thyroid lesions.6,7 However, due to the phenomenon of “same image, different diseases; same disease, different images” in two-dimensional ultrasound images of thyroid nodules, the differential diagnosis of thyroid nodules remains challenging. 8 Contrast-enhanced ultrasound (CEUS) imaging is an important supplement to two-dimensional ultrasound examination that can detect low-speed blood flow and neovascularization within the lesion. The different microvascular perfusion and morphology inside thyroid nodules can help differentiate benign and malignant nodules.9,10
In 2017, The American College of Radiology (ACR) issued the Thyroid Imaging-Reporting and Data System (TI-RADS), 11 which further standardizes the evaluation criteria and establishes a risk stratification system for thyroid nodules. According to this classification, TI-RADS TR4 nodules have a malignancy risk of approximately 5%–20%. For TI-RADS TR4 nodules with a diameter ≥1.5 cm, fine-needle aspiration biopsy is recommended, while follow-up observation is recommended for nodules with diameters of 1.0–1.5 cm. Because TI-RADS TR4 nodules have a malignancy risk, a follow-up review of these nodules does not reduce patient anxiety and concerns. Thus, biopsies or surgical resection of these nodules are performed to reduce future anxieties.12,13 As the thyroid is an important endocrine organ in the human body, invasive examinations or excessive treatments adversely affect patient health. 14
Perspectives differ regarding the optimal management strategy for patients with type 4 thyroid nodules 1.0 −1.5 cm in diameter; namely, whether to implement a strategy of vigilant surveillance or to perform immediate biopsy or surgical intervention. 15 Sonographers maintain some reservations regarding proactive approaches given that the limited clinical evidence is available. To curtail the prevalence of unwarranted invasive diagnostic procedures and surgical operations in patients with thyroid nodules, this study retrospectively reviewed nodules 1.0–1.5 cm in diameter classified as ACR Thyroid Imaging Reporting and Data System (ACR TI-RADS) 4. By utilizing a combination of two-dimensional and CEUS, this study sought to identify a noninvasive, effective, and precise diagnostic strategy that is aligned with clinical practice.
Material and Methods
Patient Selection
We retrospectively examined a cohort of 40 patients (9 men and 31 women, mean age 45.2 ± 12.6 years [range 24-74 years]) and 42 thyroid nodules classified as ACR TI-RADS 4, with diameters of 1.0–1.5 cm (mean 1.14 ± 0.21 cm) detected by ultrasound within the medical record system of our institution between January 2020 and December 2023. Each nodule was confirmed through surgical pathology or fine-needle aspiration, and the clinical records of the enrolled patients were thoroughly reviewed with respect to their medical histories, clinical presentations, and physical examinations. All patient information was anonymized, and the reporting of this study conformed to Strengthening The Reporting of Observational studies in Epidemiology (STROBE) guidelines. 16 This retrospective study was conducted with the consent of the Medical Ethics Committee. All research protocols were approved by the Clinical Research Ethics Committee of Huizhou Central People's Hospital (approval dated May 20th, 2022, No. KYLL2022209) in compliance with the specified guidelines and all enrolled patients signed informed consent forms.
The exclusion criteria were patients: with a history of thyroid surgery or treatment for thyroid nodules, with incomplete imaging data or poor image quality not meeting the diagnostic requirements, who are allergic to contrast agents or in critical condition, and who are ineligible for fine needle aspiration and CEUS examination.
Instruments and Methods
CEUS was performed using a PHILIPS EPIQ 7 color Doppler instrument with nSIGHT system, and a pure-wave high-performance eL18-4 probe (4-18 MHz). Preoperatively, SonoVue contrast agent was dissolved in 5 mL of 0.9% NaCl. The final solution appeared milky white.
Before contrast examination, a routine two-dimensional ultrasound examination was performed with the patient in a supine position with a low pillow placed below the scapula to extend the neck, fully exposing the thyroid examination area. Multiple sections from each patient were scanned. Relevant information on the thyroid nodules was recorded according to the ACR TI-RADS scoring criteria, including the structure, internal echo, morphology, margin, and focal strong echo of the nodules in the lymph nodes in the neck area. Color Doppler Flow Imaging (CDFI) was used to examine the distribution of blood flow signals around the nodules for real-time monitoring. The 2017 ACR TI-RADS classification method was used to classify thyroid nodules into categories 1–5, with the following scoring criteria: Internal composition (cystic or almost cystic, sponge-like: 0 points; cystic-solid mixed: 1 point; solid or almost completely solid: 2 points); shape (aspect ratio <1:0 points; aspect ratio > 1:3 points); echo level (no echo: 0 points; high echo or isoechoic: 1 point; low echo: 2 points; very low echo: 3 points); margin (clear or blurred: 0 points; irregular or lobulated: 2 points; closely adherent to the capsule or invading beyond the capsule: 3 points); internal strong echo (no strong echo or strong echo with comet tail sign: 0 points; coarse calcification: 1 point; circular calcification or incomplete circular calcification: 2 points; punctate strong echo: 3 points). Finally, the scores for the five items were summed to obtain the total score. As the fifth item, internal strong echo has multiple options, the scores of corresponding internal strong echo conditions were also summed. Finally, the nodes were divided into five categories: TR 1–5 according to the total score (0, 2, 3, 4-6, and ≥7 points, respectively).
The present study included only nodules categorized as TR4 with dimensions of 1.0–1.5 cm. These nodules were subsequently examined using CEUS. The largest diameter section of the target nodule (usually the long-axis section of the thyroid) was selected and the target nodule and its adjacent normal thyroid tissue were fully displayed. Large calcifications inside the nodule were avoided as much as possible. The ultrasound machine was pre-set to CEUS mode, and the patients were instructed to reduce the amplitude of their breathing and to avoid coughing and swallowing. A dose of 2.4 mL (up to 4.8 mL to provide satisfactory contract) of pre-dissolved contrast agent was rapidly injected through the elbow vein (or brachial cephalic or great saphenous vein if the patient's elbow vein was too small), followed by immediate rapid injection of 5 mL of normal saline (0.9% NaCl). Contrast dynamic images were observed and recorded for at least 90 s, and the images were saved. If the image quality was not satisfactory during the examination, CEUS was repeated using a transverse section of the thyroid.
Image Analysis
The detailed characteristics of thyroid nodules using CEUS include enhancement uniformity, enhancement intensity, post-enhancement boundary clarity, and the presence of a halo sign. Regarding enhancement uniformity, uniform enhancement refers to a thyroid nodule showing relatively homogeneous enhancement with an even distribution of contrast agents, while non-uniform enhancement refers to a thyroid nodule with uneven contrast agent distribution, including mostly homogeneous enhancement with a small portion of non-enhancement or mostly non-enhancement with a small portion of enhancement. Regarding enhancement intensity, low enhancement refers to a nodule with lower enhancement than the surrounding normal thyroid tissue, equal enhancement refers to a nodule with no significant difference in enhancement compared to the surrounding normal thyroid tissue, high enhancement refers to a nodule with higher enhancement than the surrounding normal thyroid tissue, and no enhancement refers to a nodule with no contrast agent uptake throughout the entire contrast-enhanced period. Post-enhancement boundary considered whether the boundary between the enhanced nodule and surrounding normal thyroid tissue was clear or unclear. Halo sign was defined as circular, elliptical, or other complete halo-like enhancement structures present around the nodule. When a halo sign was present, the internal enhancement characteristics of the nodule, such as uniform enhancement, enhancement intensity, and lack of enhancement, were no longer considered.
Statistical Analysis
We screened and integrated the data to remove individual outliers (poor or suspicious values) caused by human error or machine instability during the experiment. Statistical analysis was performed using IBM SPSS Statistics for Windows, version 22.0. Participant comparisons were conducted using the kappa consistency test, and the independent sample t-test was used for quantitative data. P < 0.05 was considered statistically significant.
Results
General Information and Pathological Results
A total of 42 thyroid nodules were detected in 40 patients who met the inclusion criteria (38 cases were single nodules: 23 benign and 15 malignant; two cases had multiple nodules: one with two malignant nodules, and one with one benign and one malignant nodule). The patients included nine men and 31 women, with a male-to-female ratio of 5:19 in the benign group and 4:13 in the malignant group, which is consistent with previous studies. 17 The age of the patients ranged from 24 to 74 years (mean age 45.2 ± 12.6 years). The mean ages of patients with malignant and benign nodules differed significantly (38.44 ± 8.99 years vs 49.13 ± 13.62 years, P = 0.006). Receiver operator characteristic (ROC) curve analysis showed that age had a higher predictive value for pathological results (P < 0.05, area under the curve [AUC] = 0.738, cutoff value = 53). The pathological results showed that all 18 cases of malignant nodules were papillary thyroid carcinoma; among the 24 cases of benign nodules, 15 were benign follicular nodules, six were nodular goiters, one was a nodular goiter with small calcification, one was a toxic thyroid nodule with local adenoma formation, and one was a nodular goiter with eosinophilic adenoma formation.
Two-Dimensional Ultrasound Imaging Findings
In this study, two-dimensional ultrasound showed that the average diameters of nodules in the benign and malignant groups did not differ significantly (11.50 ± 1.61 mm vs 10.78 ± 2.29 mm, P = 0.324). The two-dimensional ultrasound imaging findings of benign nodules were as follows: among nodules with a score of 4, 12 showed solid or nearly completely solid echogenicity with low internal echogenicity, a longitudinal-to-transverse ratio of <1, and no internal calcification (Figure 1a). Among nodules with a score of 5, all showed solid or similar echogenicity, with a longitudinal-to-transverse ratio of <1, and coarse internal calcification. One nodule showed cystic-solid mixed echogenicity, with a solid portion showing similar echogenicity, a longitudinal-to-transverse ratio of <1, and incomplete peripheral ring calcification. Nodules with a score of 6 showed various ultrasound imaging findings, including one nodule with solid or similar echogenicity, a longitudinal-to-transverse ratio of <1, and multiple punctate strong internal echoes, and one nodule with nearly completely solid echogenicity, similar echogenicity internally, a longitudinal-to-transverse ratio >1, and no internal calcification.

A 30-year-old woman with multiple thyroid nodules that had persisted for >1 year. (a) and (c): Two nodules in the left lobe and isthmus of the thyroid. (a) Two-dimensional ultrasound showing a solid nodule in the lower pole of the left lobe, measuring approximately 12 × 10 × 7 mm³. It appears hypoechoic with smooth margins, an aspect ratio of <1, no calcification, and punctate blood flow signals. The American College of Radiology Thyroid Imaging Reporting and Data System (ACR TI-RADS) classification is TR4. (c) A cystic-solid mixed nodule measuring approximately 13 × 15 × 9 mm3 in the isthmus. The solid portion appears hypoechoic with smooth margins, an aspect ratio of <1, and incomplete peripheral ring-like calcification. Punctate linear blood flow signals are also observed. The ACR TI-RADS classification is TR4. Ultrasound imaging reveals uniform enhancement in (a), suggesting a benign nodule, while (c) shows uneven low enhancement, suggesting a malignant nodule. Postoperative pathology suggests that (a) is consistent with nodular goiter (b, hematoxylin and eosin ×40), while (c) is papillary thyroid carcinoma (d, hematoxylin and eosin ×100).
The two-dimensional ultrasound imaging findings of malignant nodules were as follows: among nodules with a score of 4, the ultrasound images were all solid or nearly completely solid, with low internal echogenicity, a longitudinal-to-transverse ratio of <1, clear or blurred margins, and no internal calcification (Figure 2a). Among nodules with a score of 5, three showed solid low echogenicity, a longitudinal-to-transverse ratio of <1, and coarse internal calcification; two nodules showed cystic-solid mixed echogenicity, with a solid portion showing similar echogenicity, a longitudinal-to-transverse ratio of <1, and multiple punctate strong internal echoes; and one nodule showed cystic-solid mixed echogenicity, with a solid portion showing similar echogenicity, a longitudinal-to-transverse ratio >1, and no internal calcification (Figure 3a). Only one nodule had a score of 6, and showed cystic-solid mixed echogenicity, with a solid portion showing low echogenicity, a longitudinal-to-transverse ratio of <1, and incomplete peripheral ring calcification (Figure 1c).
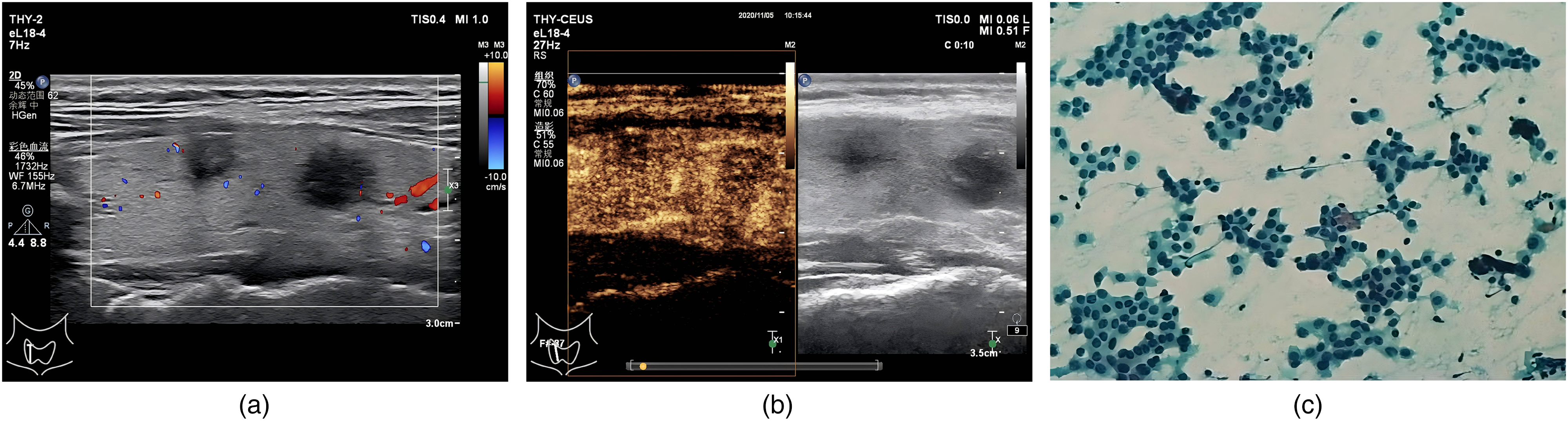
A 47-year-old woman with multiple nodules in the right lobe of the thyroid gland discovered during a physical examination >1 month previously. (a) Two-dimensional ultrasound examination shows two nodules in the right lobe of the thyroid gland located at the middle and lower Poles. The nodule in the middle pole of the right lobe was solid and hypoechoic, measuring approximately 10 × 8 × 6 mm³, with blurred margins, a longitudinal-to-transverse ratio <1, and no calcification or blood flow signal. The American College of Radiology Thyroid Imaging Reporting and Data System (ACR TI-RADS) classification is TR4. The nodule in the lower pole of the right lobe was solid and hypoechoic, measuring approximately 11 × 9 × 8 mm³, with blurred margins, a longitudinal-to-transverse ratio <1, and no calcification or blood flow signal. The ACR TI-RADS classification is also TR4. (b) Ultrasound contrast showing that both nodules exhibit unevenly low enhancement; thus showing potential malignancy. (c, papanicolaou ×100) Fine needle aspiration cytology showing that the pathological results of these two nodules as papillary thyroid carcinomas.

A 30-year-old woman was admitted to the hospital for thyroid nodules that had been present for >2 weeks. (a) Two-dimensional ultrasound showing a solid and cystic mixed nodule on the left lobe of the thyroid gland, measuring approximately 10 × 6 × 7 mm³. The solid part exhibits isoechoic echoes with blurred margins and a longitudinal-to-transverse ratio greater than 1. No obvious calcification was observed, and the blood flow signal was not abundant. According to the ACR of Rheumatology classification, it is considered a category 4 nodule. (b) Ultrasound contrast showing uneven enhancement of the nodule on the left lobe, suggesting potential malignancy. (c, hematoxylin and eosin staining ×20) Postoperative pathology indicates papillary thyroid carcinoma. The enhanced part of this nodule was the cystic part in two-dimensional ultrasound examination. Contrast ultrasound suggested that the cystic portion was hypoechoic. The TI-RADS classification of this nodule was corrected to TR5.
CEUS Imaging
We defined the early and late phases of contrast enhancement as the periods from the beginning of thyroid parenchymal enhancement to complete homogeneous enhancement and from the beginning of thyroid parenchymal attenuation to complete attenuation, respectively. Detailed information on CEUS enhancement patterns for 18 malignant and 24 benign nodules is shown in Table 1. Correlation analysis of various CEUS features and their pathological diagnostic values showed a 100% negative predictive value of rim enhancement (Table 2). However, owing to the different degrees of homogeneous enhancement within nodules with rim enhancement, the kappa value of homogeneous enhancement was relatively low. Therefore, a statistical analysis was performed after excluding rim enhancement (Tables 3 and Table 4). The ROC curve analysis showed that both homogeneous enhancement and enhancement intensity have good diagnostic value for pathological results, with an AUC >0.8 (Table 4). Therefore, we considered the joint evaluation of both factors for diagnosis and pathological results and performed a kappa consistency analysis between homogeneous enhancement and enhancement intensity. Serial connection refers to CEUS results of thyroid nodules simultaneously meeting the criteria of nonhomogeneous and low enhancement to indicate potentially malignant nodules. Parallel connection refers to CEUS results of nodules meeting either non-homogeneous or low enhancement criteria and an indication of potentially malignant nodules (Tables 5 and 6). The results of the ROC curve analysis showed the high diagnostic value of both serial and parallel connections (Tables 5 and 6). The sensitivity and specificity of the serial connection were 77.8% and 85.7%, respectively, and 100% and 71.4% for the parallel connection, respectively. These results showed that using uneven low enhancement as an ultrasound imaging manifestation for judging malignant thyroid nodules had a higher specificity and good diagnostic value.
Consistency of Findings Between Ultrasound Imaging and Pathological Examination (1).

Evaluation of the Diagnostic Value of Contrast-Enhanced Ultrasound Imaging Signs for Pathological Results (1).

Abbreviations: AUC, area under the curve; CI, confidence interval.
Consistency of Findings Between Ultrasound Imaging and Pathological Examination (2).

Evaluation of the Diagnostic Value of Contrast-Enhanced Ultrasound Imaging Signs for Pathological Results (2).

Abbreviations: AUC, area under the curve; CI, confidence interval.
Consistency of Findings among Homogeneous Enhancement, Series and Parallel Enhancement Patterns, and Pathological Results.

Diagnostic Value of Homogeneous Enhancement and Enhancement Patterns in Series and Parallel for Determining the Pathological Results.

Abbreviations: AUC, area under the curve; CI, confidence interval.
Discussion
Malignancy Rate of Thyroid TR4 Nodules
This study analyzed thyroid TR4 nodules with diameters of 1.0–1.5 cm. The malignancy rate of these thyroid nodules was approximately 40%, which is higher than the ACR rate of 5%–20% for TR4 nodules. Thyroid nodule CEUS technology is in the exploratory stage, and its diagnostic efficacy has not been widely recognized. In clinical practice, when suspicious nodules are detected during a two-dimensional ultrasound examination, doctors often recommend direct biopsy instead of CEUS for a definitive diagnosis. Additionally, the sample size in this study was relatively small owing to the impact of the coronavirus disease 2019 (COVID-19) pandemic, which may have led to an experimental bias in the results.
The ACR guideline recommends biopsy for TR4 nodules >1.5 cm; however, the nodules included in the present study had diameters ranging from 1.0–1.5 cm, which is smaller than the recommended biopsy criteria, which may also explain the inconsistent results. The TI-RADS score of thyroid nodules is based on the subjective judgment of the ultrasound physician on the two-dimensional images and lacks relatively stable absolute quantitative indicators and technical means, which may lead to differences in results across studies. For example, in the scoring of echo level, no echo was scored as 0, high echo or isoecho was scored as 1, low echo was scored as 2, and hypoechoic was scored as 3. In some cases, clinicians may judge hypoechoic as a low echo or no echo, thereby classifying nodules that might have been classified into five categories into four categories, thus increasing the malignancy rate of TR4 nodules. This study aimed to conduct a more comprehensive and systematic analysis of thyroid nodules through CEUS diagnostic techniques to obtain more scientific and objective diagnoses of thyroid nodules.
Related studies have shown that 75.5%–87.3% of the pathological types of thyroid cancer are papillary thyroid carcinomas, with an age of onset ranging from 10–88 years, (average 41.3 years). 18 The proportion of women aged 30–40 years has increased significantly, consistent with our findings. In this study, the average age of patients with pathologically malignant thyroid nodules was significantly lower than that of patients with pathologically benign thyroid nodules (P = 0.006). ROC curve analysis showed that age had a high predictive value for pathological results, with a corresponding cutoff value of 53 years. Therefore, the probability of malignancy may be higher in patients aged ≤53 years with TR4 nodules. Because the number of cases in this study was small and the age range of the patients was 24–74 years, these results may not fully coincide with actual clinical findings. Therefore, a cut-off of 53 years may have some bias. However, the existence of this intergroup difference also indicates the need to consider age in patients with TR4 nodules in clinical decision-making. Future diagnostic and treatment studies with more participants may provide a more accurate age threshold for malignancy risk.
Significance of CEUS Features and Circular Enhancement of TR4 Benign Nodules
The primary pathological types of benign thyroid nodules are adenomas and nodular goiters. Adenoma nodules primarily shows rapid enhancement compared with the surrounding thyroid tissue, with homogeneous, high, and circular enhancement with clear borders19–21; the nodular goiters primarily shows circular enhancement, equal to or low enhancement with clear borders.22–24 Bo zhang 25 and others reported that 83.02% of benign thyroid nodules show rim enhancement. The specificity, positive predictive value, and accuracy of rim enhancement in diagnosing benign thyroid nodules were 94.12%, 93.62%, and 88.46%, respectively, consistent with the results of this study. As the proliferation and atrophy of the internal tissues of nodular goiters alternate, the blood supply characteristics of nodular goiter lesions differ across pathological periods. 26 Therefore, the imaging features are diverse; however, overall, its blood supply characteristics are similar to those of normal thyroid tissues. Therefore, the main CEUS features of nodular goiters include homogeneous enhancement, iso-enhancement or hyperenhancement, and clear borders. From a histopathological perspective, rim enhancement in thyroid adenomas on CEUS occurs due to the rich rim vascular network around the adenoma nodule. Nodular goiters can also exhibit the characteristics of a rim vascular network; therefore, rim enhancement on CEUS may also be observed in nodular goiter lesions. In this study, seven cases diagnosed with nodular goiter by surgical pathology also showed various enhancement patterns on CEUS. Among them, three cases showed rim enhancement, two showed homogeneous iso-enhancement, one showed homogeneous hyperenhancement, and one showed no enhancement. Therefore, the main CEUS features of benign thyroid nodules include homogeneous enhancement, iso-enhancement or hyper-enhancement, clear borders, and rim enhancement. A nodule showing rim enhancement can be directly considered a benign nodule without considering the internal enhancement pattern.
CEUS Features of Malignant TR4 Thyroid Nodules
Due to their infiltrative growth characteristics, malignant nodules are prone to capsule invasion, resulting in irregular and ill-defined enhancement patterns.27,28 The application of CEUS in thyroid lesions remains underdeveloped, and considerable controversy persists regarding the CEUS features of benign and malignant nodules. 29 Another study showed that heterogeneous enhancement was the most prominent contrast-enhanced feature of thyroid cancer, with a specificity, positive predictive value, and accuracy of 92.45%, 91.84%, and 90.38%, respectively. 30 In this study, among the 18 malignant nodules, 14 showed irregular low enhancement with ill-defined borders, three showed irregular iso-enhancement with ill-defined borders, and one showed homogeneous low enhancement with ill-defined borders. The kappa consistency test results showed good consistency between the CEUS features and pathological results. Heterogeneous enhancement, low enhancement, and ill-defined post-enhancement borders indicated malignant thyroid nodules, whereas homogeneous enhancement, no enhancement, iso-enhancement, and high enhancement with clear post-enhancement borders indicated benign thyroid nodules. The differences were significant, with high consistency between homogeneous enhancement, enhancement intensity, and pathological results. Heterogeneous enhancement as a CEUS feature of malignant thyroid nodules had a sensitivity of 94.4% and specificity of 71.4%, whereas low enhancement had a sensitivity of 83.3% and specificity of 85.7%. CEUS imaging of malignant thyroid nodules showed uneven enhancement, which may be related to twisted internal blood vessels and uneven microvessel distribution. However, similar pathological changes may occur in nodular goiters, resulting in low specificity.
Bartolotta et al 31 reported that the enhancement pattern after contrast-enhanced imaging of thyroid nodules was related to nodule size, in which malignant nodules <1 cm, 1–2 cm, and >2 cm showed an avascular type, a small amount of punctate enhancement, and diffuse enhancement, respectively, consistent with our results. A small amount of punctate enhancement on CEUS imaging may correspond to an uneven low enhancement. The difference lies in the descriptive language used. Uneven enhancement on CEUS mainly occurs due to more necrotic blood vessels, the presence of cancer emboli, and the dysfunction of tumor blood vessels. The pathological basis of the low enhancement revealed that the early cancer lesion had fewer and thinner neovascularizations and the undeveloped vascular bed, resulting in reduced contrast agent perfusion.
We also compared the maximum diameter of benign and malignant nodules to investigate whether the ACR's recommendation of a minimum puncture diameter of 1.5 cm for TR4 nodules is reasonable. However, the nodule diameter did not differ significantly between benign and malignant nodules with diameters of 1.0–1.5 cm. Therefore, the ACR guideline recommendation for performing a biopsy for TI-RADS TR4 nodules with diameters >1.5 cm is reasonable. However, in clinical practice, for TI-RADS TR4 nodules with diameters 1.0–1.5 cm, we recommend performing color Doppler and CEUS follow-up examinations respectively, if the age at first discovery is >53 years and ≤53 years. Finally, subsequent diagnostic and treatment plans can be decided based on the CEUS results. Therefore, we also considered whether conducting CEUS studies on thyroid nodules grouped by size in the future development of thyroid nodule ultrasound imaging could provide more powerful evidence of imaging features to assist in the diagnosis of benign and malignant thyroid nodules.
Study Limitations
Our results demonstrated no significant correlation between ACR TI-RADS TR4 nodules 1.0–1.5 cm in diameter and their benign or malignant nature; however, the younger the age of onset, the higher the probability of malignancy. A nodule with ring-enhancing features is more likely to be benign and the contrast-enhanced appearance within the nodule does not require further evaluation. Similarly, nodules showing no enhancement or uniform or high enhancement with clear borders on contrast ultrasound imaging are also likely to be benign. However, nodules with uneven low enhancement or unclear borders are likely to be malignant. Both uneven enhancement and low enhancement on CEUS imaging have high diagnostic value for malignant nodules; however, the combination of the two demonstrated higher specificity.
This is a cross-sectional study, which was calculated based on the sample size formula:
Conclusions
The advantages of ultrasonography, including real-time imaging, non-invasive procedures, low cost, high resolution, and no radiation, make it an indispensable tool for clinical thyroid examination. Multimodal ultrasound combined with examination technology integrates the blood supply and enhancement features of nodules with two-dimensional structural characteristics. This approach provides doctors with more comprehensive diagnostic information, thereby allowing for a more accurate qualitative diagnosis of nodules.
However, the present study has several limitations, including the small number of participants, which require future exploration. However, our study provides sufficient power and clinical evidence for future analyses, which is of significance for the diagnosis of malignant or benign nodules. In addition, our findings provide novel and individualized value for future studies and clinical applications.
Footnotes
Abbreviations
Acknowledgements
I would like to acknowledge the contributions of those who supported me in my research. First, I wish to express my sincere thanks to my mentor, Dr Shengkai Li, and my teacher, Dr Hong Wen, for their invaluable guidance and patience throughout my study. Their expertise has greatly influenced my research direction. I also appreciate the help from my research team members, Mei Huang, Xumin Xie, and Zhijun Yi, for their collaboration and assistance in data collection and analysis. Thanks are also due to the Technology Plan Projects in the Medical and Health Field in Huizhou City Foundation for their financial support, which made this research possible. Finally, I am forever grateful to my family and friends for their encouragement and belief in me throughout this challenging yet rewarding journey.
Contributions
(I) Conception and design: X Yuan, S Li; (II) Administrative support: H Wen; (III) Provision of study materials or patients: X Yuan, H Wen, X Xie, Z Yi; (IV) Collection and assembly of data: X Yuan, H Wen, S Li; (V) Data analysis and interpretation: X Yuan, M Huang, S Li; (VI) Manuscript writing: All authors; (VII) Final approval of manuscript: All authors.
Data Availability
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Disclaimers
The authors of this manuscript declare no relationships with any companies, whose products or services may be related to the subject matter of the article.
Ethical Disclosure
The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. This retrospective study was conducted with the consent of the Medical Ethics Committee. All research protocols were approved by the Clinical Research Ethics Committee of Huizhou Central People's Hospital (No. kyll2022209) in compliance with the specified guidelines and all enrolled patients signed the informed consent forms.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study received funding from the Technology Plan Projects in the Medical and Health Field in Huizhou City, China (No. 2022CZ010093).
