Abstract
Background:
Anti-programmed cell death protein-1 immunotherapy has been approved as a new treatment option for advanced hepatocellular carcinoma (HCC) based on the promising results of several studies.
Methods:
This retrospective study included 71 patients with advanced HCC treated with anti-programmed cell death protein-1 immunotherapy between June 1, 2017 and September 30, 2020 at the First Affiliated Hospital of Anhui Medical University. Responses to pulmonary metastases were evaluated.
Results:
The median follow-up duration was 7.73 months (95% confidence interval (CI), 4.48-10.98). Of 71 patients, the overall response rate (ORR) and disease control rate (DCR) were 32% (23/71) and 73% (52/71), respectively. The median progression-free survival (PFS) and overall survival (OS) were 4.90 (95% CI, 2.71-7.09) and 20.23 (95% CI, 6.87-33.59) months, respectively. Forty-two patients had HCC pulmonary metastases, whereas 29 did not have pulmonary metastasis. No significant differences were observed in the ORR (38% [16/42] vs. 24% [7/29], P = 0.22) and DCR (74% [31/42] vs. 72% [21/29], P = 0.90) between groups. In patients with pulmonary metastases, the median disease control duration of pulmonary lesions was significantly longer than extrapulmonary lesions (Not Reached vs. 12.37 months, P = 0.048). Pulmonary metastases were not associated with an increased incidence of adverse events (67% vs. 62%, P = 0.69).
Conclusions:
Anti-programmed cell death protein-1 immunotherapy showed promising efficacy and safety in patients with advanced HCC, with good responses observed in pulmonary metastases. The mechanism underlying the differences in responses between pulmonary and extrapulmonary metastases requires further investigation.
Introduction
Hepatocellular carcinoma (HCC) accounts for 90% of all cancers in the liver and has become the third leading cause of cancer-related mortality worldwide. 1,2 In China, more than 78% of HCCs are caused by chronic viral infections, most commonly due to hepatitis B virus (HBV). 3,4 The intrinsic immune tolerance of liver leads to the “immune evasion” of HCCs through multiple pathways. Specifically, the programmed cell death protein-1 (PD-1)/programmed cell death ligand-1 (PD-L1) pathway mediates immunosuppression through the negative regulation of CD8+ T cell activity. 5 Therefore, the anti-PD-1 antibodies, pembrolizumab, nivolumab, and camrelizumab, have displayed antitumor activity with good safety in patients with advanced HCC in clinical trials 6 -8 as well as in real-world settings. Furthermore, immunotherapy in combination with targeted anti-angiogenesis therapy for advanced HCC has shown efficacy in preliminary findings from several clinical trials. 9,10
The recurrence or metastasis of advanced HCC generally involves intrahepatic and extrahepatic lesions. The lung is the most common extrahepatic site of metastasis in advanced HCCs. 11 Studies have confirmed that the immune microenvironment in the liver is different to that in other organs. Based on previous clinical work, we observed organ-specific responses to immunotherapy, particularly in the lung. The aim of the present study was to evaluate the clinical outcomes, including efficacy and adverse events of anti-PD-1 immunotherapy in patients with advanced HCC and compare the efficacy of immunotherapy between pulmonary and extrapulmonary metastases.
Methods
Study Design and Patients
This retrospective study included patients with advanced HCC who received anti-PD-1 immunotherapy at the First Affiliated Hospital of Anhui Medical University between June 1, 2017 and September 30, 2020.All data were retrospectively collected and included patient demographics, laboratory results, and radiological information. The efficacy of anti-PD-1 immunotherapy in patients with or without pulmonary metastases was compared and analyzed, and the efficacy of of immunotherapy in lung lesions in patients with pulmonary metastases was evaluated separately. This study was approved by the Medical Ethics Committee of the First Affiliated Hospital of Anhui Medical University (PJ 2020-15-35). Considering the retrospective nature of this work, the requirement for informed consent was waived for individual participants as per the committee standards.
The inclusion criteria were as follows: age of at least 18 years; histologically or clinically confirmed advanced primary HCC according to the Guidelines for the Diagnosis and Treatment of Primary Liver Cancer in China 12 ; Eastern Cooperative Oncology Group performance score, 0-2; Child-Pugh class, A or B; HBV DNA load, normal range (<1000 copies/ml or <500 IU/ml); monotherapy with at least 2 doses of anti-PD-1 antibodies (pembrolizumab, nivolumab, camrelizumab, sintilimab, or tislelizumab) or combination therapy of anti-PD-1 antibodies with anti-angiogenesis drugs(sorafenib, lenvatinib, or apatinib).
The exclusion criteria were as follows: other concomitant malignant tumors; insufficient information on the antitumor efficacy of anti-PD-1 immunotherapy, such as lack of data on follow-up imaging, loss to follow-up (the minimum follow-up duration was 1.5 months), or death before response assessment; previously or concurrently administered immunotherapy, including those targeting PD-L1, PD-L2, or cytotoxic T lymphocyte antigen (CTLA-4); concurrent administration of chemotherapy or local treatment, including radiofrequency ablation, microwave ablation, and transcatheter arterial chemoembolization (TACE).
Study Outcomes
Efficacy outcomes included the following: 1) overall response rate (ORR) and disease control rate (DCR) as evaluated by study investigators or attending clinicians using the Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1 13 ; 2) response rate of pulmonary metastases with assessment of individual pulmonary lesions according to RESIST 1.1; 3) progression-free survival (PFS) measured from the date of first dose to the date of radiographic or clinical evidence of disease progression, overall survival (OS) measured from the date of first dose to the date of death from any cause; and 4) disease control duration of pulmonary and extrapulmonary metastases measured from the date of first dose to the date of radiographic evidence of progression of pulmonary and extrapulmonary lesions.
Safety outcomes were the rates of all immune-related adverse events (irAEs), those leading to the interruption and discontinuation of treatment, and those requiring steroids, which were graded according to the National Cancer Institute Common Terminology Criteria for Adverse Events [CTCAE] version 4.03.
Statistical Analysis
Baseline demographics, treatment characteristics, and irAEs were analyzed using descriptive statistics. PFS, OS, and disease control duration were estimated using the Kaplan-Meier method and compared by the log-rank test. The analysis for ORR was based on best overall response and compared by the chi-square test. All statistical analyses were performed using IBM SPSS Statistics version 20 (Chicago, IL, USA) and GraphPad Prism version 8. A P value of < 0.05 was considered to indicate statistical significance.
Results
Patients
During the study period, 106 patients receiving anti-PD-1 immunotherapy either as monotherapy or combination therapy as first- or second-line treatment of advanced HCC in the First Affiliated Hospital of Anhui Medical University were screened. The final analyses included 71 patients fulfilling the inclusion and exclusion criteria (Figure 1). All patients had a history of HBV infection. Forty-two patients had pulmonary metastases, whereas 29 did not have lung metastasis. The baseline demographic and clinical characteristics of all patients and the 2 groups are summarized (Table 1).
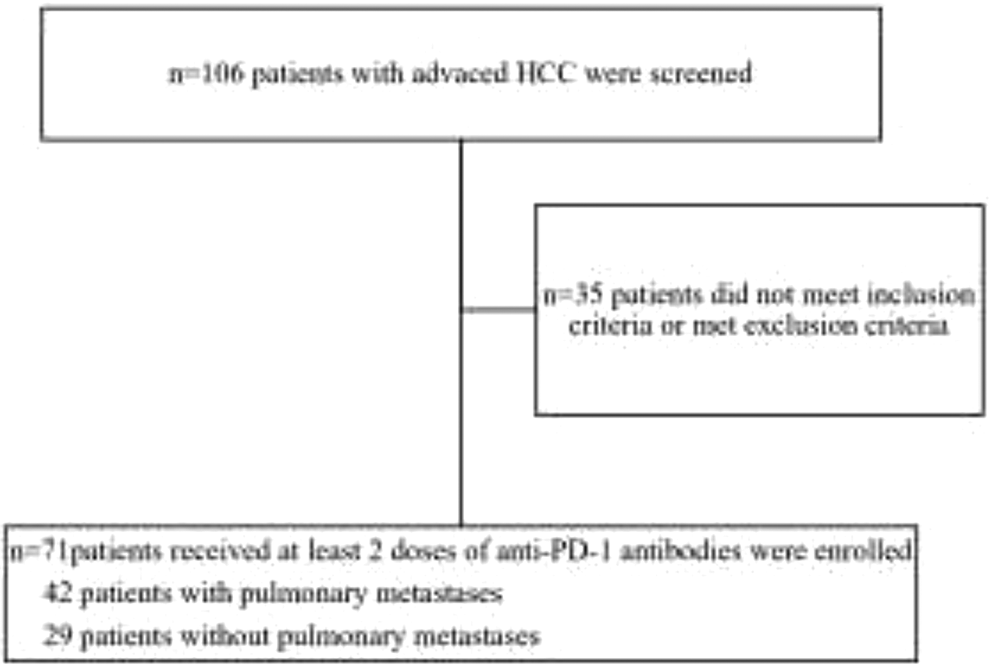
Inclusion and exclusion criteria flowchart.
Baseline Demographic and Clinical Characteristics.
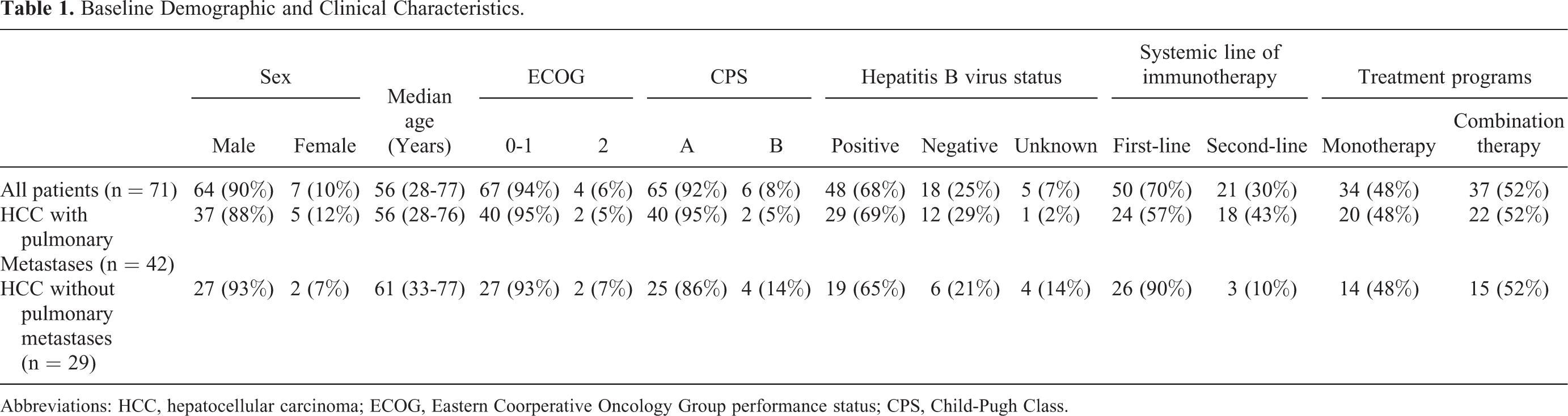
Abbreviations: HCC, hepatocellular carcinoma; ECOG, Eastern Coorperative Oncology Group performance status; CPS, Child-Pugh Class.
The anti-PD-1 antibodies used as systemic first- or second-line immunotherapy included pembrolizumab, nivolumab, camrelizumab, sintilimab, and tislelizumab in 16 (22%), 3 (4%), 36 (51%), 7 (10%), and 9 (13%) patients, respectively. Combination therapy with anti-angiogenesis drugs administered in 37 (52%) patients included sorafenib, lenvatinib, and apatinib in 7 (19%), 12 (32%), and 18 (49%) patients, respectively.
The reasons for treatment discontinuation were disease progression, adverse events, patient decision, liver transplantation, and complications leading to death in 19 (27%), 8 (11%), 7 (10%), 1 (1%), and 2 (3%) patients, respectively. At the data cut-off date, 34 (48%) patients were still on immunotherapy and 17 (24%) patients were maintaining response.
Efficacy
All 71 patients underwent at least one follow-up radiographic examination and were evaluated for treatment efficacy. The overall response and the response rate of pulmonary metastases are shown in Table 2. There were no significant differences in the ORR (38% vs. 24%, P = 0.22) and DCR (74% vs. 72%, P = 0.90) between patients with and without pulmonary metastases of HCC. In 6 patients with only pulmonary metastases, the ORR and DCR were 50% and 83%, respectively. The median PFS and OS of these 6 patients were 15.17 (95%CI, 0.00-34.15) months and Not Reached (NR), respectively.
Tumor Responses and Survival Data.a
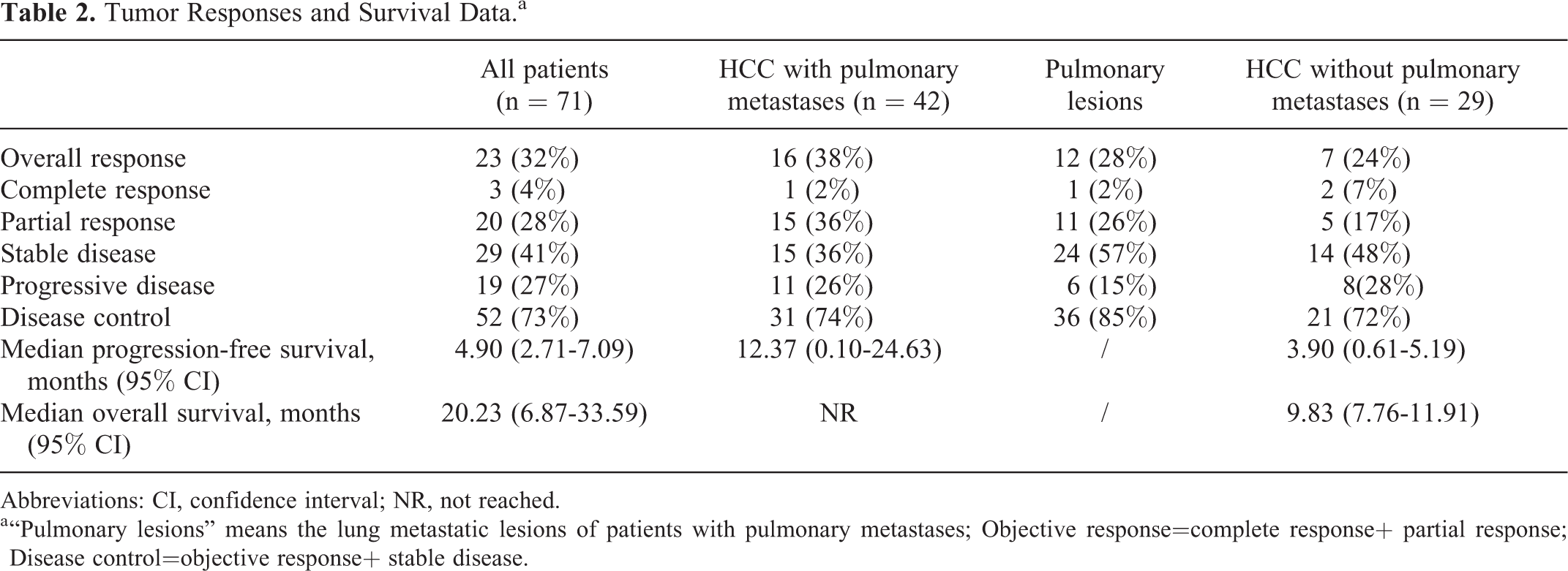
Abbreviations: CI, confidence interval; NR, not reached.
a “Pulmonary lesions” means the lung metastatic lesions of patients with pulmonary metastases; Objective response=complete response+ partial response; Disease control=objective response+ stable disease.
The median follow-up duration of the overall cohort was 7.73 (95% CI, 4.48-10.98) months. The median PFS and OS of the study cohort was 4.90 (2.71-7.09) and 20.23 (6.87-33.59) (Figures 2 and 3), respectively (Table 2). In patients with pulmonary metastases, the median disease control duration of pulmonary and extrapulmonary lesions was NR vs. 12.37 (95% CI, 0.00-24.85) months, respectively (P = 0.048) (Figure 4).

Kaplan-Meier curve showing progression-free survival in all patients.

Kaplan-Meier curve showing overall survival in all patients.

Kaplan-Meier curve showing disease control duration of lung lesions and extrapulmonary lesions in patients with pulmonary metastases.
Safety
The irAEs observed in all patients are shown (Table 3). At least one irAE occurred in 46 (65%) of the 71 participants; irAEs were graded 3-4 in 13 (18%) patients and severe in 10 (14%) patients. The incidence of irAEs did not differ between patients with and without pulmonary metastases (67% vs. 62%, P = 0.69). Two patients who developed immune pneumonia were without pulmonary metastases.
Adverse Events.

Abbreviations: irAEs, immune-related Adverse Events; RCCEP, reactive cutaneous capillary endothelial proliferation.
During the follow-up period, the causes of death were disease progression, obstructive jaundice, and spontaneous peritonitis in 15 (21%), 1 (1%), and 1 (1%) patient, respectively. Importantly, no patient died because of an irAE.
Discussion
Several anti-PD-1 antibodies are currently available for clinical use in China. All anti-PD-1 antibodies used in the present study are either recommended by the Guidelines for the Diagnosis and Treatment of Primary Liver Cancer in China 12 or effective in clinical trials. We demonstrated that different anti-PD-1 antibodies showed promising efficacy and manageable toxicity in patients with advanced HCC. The incidence of irAEs was similar to that observed in previous reports. In our study, one patient developed immune hepatitis, immune cystitis, and urinary tract infection successively, then recovered after steroid and symptomatic treatment, and is still responding to immunotherapy.
We found that the median PFS (12.37 vs. 3.90 months, P = 0.049) and OS (NR vs. 9.83 months, P = 0.26) of patients with pulmonary metastases were longer than those of patients without pulmonary metastases. Given the difference in follow-up times (median follow-up time 10.07 vs. 5.13 months, P = 0.002) between groups, this difference may not be meaningful. But we separately analyzed the response of lung lesions and extrapulmonary lesions in patients with pulmonary metastases and observed significant differences. The median disease control duration was significantly longer for pulmonary lesions than for extrapulmonary lesions (NR vs. 12.37 months, P = 0.048). A previous study found that apatinib, an anti-angiogenesis drug, was particularly effective for advanced HCC with pulmonary metastases. 14 One potential mechanism underlying the efficacy of apatinib is the normalization of abnormal tumor vasculature, which potentially transforms the tumor microenvironment and activates hepatic and pulmonary immune systems, thereby preventing lung metastases from the liver and reducing the size and number of metastatic pulmonary lesions. 15 -17 In our study, 22 patients with pulmonary metastases received anti-PD-1 immunotherapy in combination with anti-angiogenesis drugs. On the other hand, several studies have demonstrated that heterogeneous tumor immune microenvironments of various organs and different gene expression profiles of multiple tumor types may affect tumor growth and responses to anticancer treatments. 18 For example, studies have found that several characteristics of the immune environment, such as the density of CD8+ T cells and PD-L1 expression, were different in paired primary and metastatic renal cell carcinoma and lung cancer specimens. 19 -21 Lu et al retrospectively analyzed the data of 75 patients with advanced HCC who received ICIs and found that tumor responses varied considerably depending on the tumor site, specifically in lung metastatic lesions (ORRs of hepatic tumors, lung, lymph node, and other intra-abdominal metastases were 22.4%, 41.2%, 26.3%, and 38.9%, respectively). The study found that of the 16 patients with evaluable tumors in the liver and lungs at baseline, 8 had disease control in the lungs while PD in the liver, and none experienced disease control in the liver while PD in the lungs (P = 0.005). The study hypothesized that it may be related to the imbalance of tumor burden among different organs. 21 Another retrospective study published recently also described organ-specific responses (OSR) to first-line lenvatinib plus anti-PD-1 antibodies in 60 patients with unresectable HCC. The data showed that the OSR of macrovascular tumor thrombi was 54.5% and the OSR of extrahepatic lung metastases (37.5%) was higher than that of intrahepatic tumors (32.8%) and lymph node metastases (33.3%). 22 However, information on the immunological characteristics of metastatic HCC lesions in different organs is lacking and warrants further investigation.
Although this was a retrospective study and lacked a control group, its main strength was the inclusion of real-world data on patients with advanced HCC receiving anti-PD-1 immunotherapy. Future studies will include the evaluation of biopsy specimens from pulmonary metastases of patient with advanced HCC to elucidate the potential mechanisms underlying the differences observed in the response of pulmonary lesions to anti-PD-1 immunotherapy. Additionally, future investigations should increase participant numbers in studies and extend the follow-up period to balance the follow-up time and determine the efficacy and safety of anti-PD-1 immunotherapy.
Footnotes
Abbreviations
CI, confidence interval; CTCAE, National Cancer Institute Common Terminology Criteria for Adverse Events; CTLA-4, cytotoxic T lymphocyte antigen; DCR, disease control rate; HBV, hepatitis B virus; HCC, hepatocellular carcinoma; irAEs, immune-related adverse events; NR, Not Reached; PD-1, programmed cell death protein-1 (PD-1); PD-L1/PD-L2, programmed cell death ligand-1/ligand-2; PFS, progression-free survival; ORR, overall response rate; OS, overall survival (OS); RECIST, Response Evaluation Criteria in Solid Tumors; TACE, transcatheter arterial chemoembolization.
Authors’ Note
Jie Da, MD, and Yingying Du, MD, contributed equally to this work. The present study was approved by the Committee on Medical Ethics of the First Affiliated Hospital of Anhui Medical University (PJ 2020-15-35). Given the retrospective nature of this work, the requirement for informed consent was exempted for the individual participants included in the study in accordance with the standards of the Ethics Committee of the First Affiliated Hospital of Anhui Medical University.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
