Abstract
Background
For advanced hepatocellular carcinoma (HCC), sorafenib remains the established therapy. PD-1/PD-L1 inhibitors plus antiangiogenic drugs (PIAD) as a new therapeutic approach for advanced HCC is still a subject of clinical debate regarding whether they offer improved treatment outcomes. This study was conducted to compare the two treatments in terms of antitumor efficacy and safety.
Methods
Randomized controlled trials (RCTs) comparing PIAD and sorafenib for advanced HCC were retrieved from six databases. Survival (overall survival [OS] and progression-free survival [PFS]) were the main outcomes measured. Secondary endpoints included responses, adverse events (AEs), and effects on quality of life.
Results
Seven studies based on four RCTs (CARES-310, COSMIC-312, IMbrave150, and ORIENT-32) were included. The PIAD group exhibited better OS (hazard ratio [HR]: 0.69, 95% confidence interval [CI]: [0.53, 0.89], P = 0.005), and PFS (HR: 0.60, 95% CI: [0.53, 0.67], P < 0.00001). The survival advantages of OS and PFS were confirmed in almost all subgroups. The PIAD group exhibited higher OS rates at 6–18 months and PFS rates at 6–12 months. Additionally, the objective response rate, disease control rate, complete response, and partial response were higher in PIAD group. The PIAD group had a delayed decline in quality of life, physical functioning, and role functioning. However, the PIAD group experienced more grades 3–5 and serious AEs, along with treatment discontinuation, dose reduction, and dose interruption.
Conclusions
PIAD appears to be better than sorafenib for advanced HCC with better survival and responses. However, its higher rate of AEs requires cautious attention.
Introduction
In the past few decades, liver cancer has ranked among the top in both incidence and mortality rates. 1 Hepatocellular carcinoma (HCC) constitutes nearly 80% of the total cases. 2 For an extended period, sorafenib has been the standard treatment for advanced HCC. 3 However, over time, the effectiveness of sorafenib has increasingly failed to meet patient expectations. The advent of immunotherapy in the treatment of advanced liver cancer has introduced new hope for patients. 4 Nevertheless, the efficacy of immunotherapy alone remains suboptimal. 5 Recently, the use of PD-1/PD-L1 inhibitors combined with antiangiogenic drugs (PIAD) for treating advanced HCC has grown, yet its superiority in clinical outcomes is still debated.6–12
According to the NCCN guideline for hepatocellular carcinoma, both PIAD and sorafenib are recommended as first-line treatments for advanced HCC. 13 The IMbrave150 study reported that atezolizumab plus bevacizumab achieved better overall survival (OS) and progression-free survival (PFS) than sorafenib.9–11 Similar results were reported in the CARES-310 (camrelizumab plus rivoceranib) and ORIENT-32 (sintilimab plus bevacizumab) studies.6,12 However, the COSMIC-312 study found that atezolizumab plus cabozantinib achieved similar OS but had significantly higher rates of adverse events (AEs) compared to sorafenib.7,8
This meta-analysis aims to systematically review and synthesize data from phase 3 randomized controlled trials (RCTs) comparing PIAD with sorafenib, thereby establishing a strong basis for improving treatment protocols in advanced HCC.
Materials and Methods
The research adhered to PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guideline and was registered in PROSPERO (ID: CRD42024557133).
Search Strategy
The approach for searching included “PD-1/PD-L1 (details in Table S2)”, “Sorafenib”, “Hepatocellular carcinoma”, and “Randomized” as keywords. A comprehensive search for eligible RCTs was conducted across six databases (PubMed, ScienceDirect, Cochrane Library, Scopus, EMBASE, and Web of Science) from their inception until May 9, 2024 (Supplementary Table 1). In addition, reference lists of selected RCTs were also checked to locate additional qualifying studies.
Selection Criteria
Inclusion criteria (PICOS):
Participants (P): patients with advanced HCC. Intervention (I) and control (C): PIAD compared to sorafenib. Outcomes (O): survival, survival rate, responses, AEs and the impact on quality of life. Study design (S): phase 3 RCTs.
Exclusion criteria: animal experiment, meta-analysis, conference paper, and case report.
Data Extraction
Two researchers extracted data independently on study: study characteristics (such as geographic region and follow-up time), survival metrics (OS and PFS), survival rate (OS rate [OSR] and PFS rate [PFSR]), responses (objective response rate [ORR], complete response [CR], etc), AEs (total, grade 3-5, etc) and the impact on quality of life (physical functioning, role functioning, etc). Disagreements were resolved through careful review and dialogue.
Outcome Assessments
Subgroup analysis of OS and PFS were conducted based on age, sex, geographic location, race, ECOG PS, Barcelona Clinic Liver Cancer (BCLC) stage, Alpha-Fetoprotein (AFP), category, macrovascular invasion/extrahepatic spread at study entry, macrovascular invasion at study entry, extrahepatic spread at study entry, etiology, prior local therapy, PD-L1 expression, and PD1/PD-L1 type. Additionally, OSR and PFSR were also analyzed at 6–24 months. The responses were analyzed according to RECIST 1.1 14 and mRECIST. 15 The deterioration of quality of life free survival rate (DQL-FSR), deterioration of physical functioning free survival rate (PF-FSR), and deterioration of role functioning free survival rate (RF-FSR) were also analyzed at 6–24 months.
Quality Assessment
The quality of RCTs was measured using the Cochrane Risk of Bias Assessment tool 16 and the Jadad scale, a 5-point system that rates randomization, blinding, and patient inclusion, with a score ≥3 indicating high quality. 17 The quality of the results was assessed using the GRADE method (Grading of Recommendations, Assessment, Development, and Evaluation). 18
Statistical Analysis
The combined data were analyzed with RM 5.3 and STATA 12.0. Hazard ratio (HR) was employed to analyze survival variables. Risk ratio (RR) was used to analyze binary variables. We assessed heterogeneity through the use of I2 statistic and χ2 test. For I2 less than 50% or P greater than 0.1, signifying no significant heterogeneity, fixed-effects model was used. Alternatively, random-effects model was used. Statistical significance was established with P < 0.05. Funnel plots were visually inspected to evaluate publication bias.
Results
Search Results
A total of 2753 studies were identified through the initial database search. After removing 1051 duplicates, 1702 studies were screened based on titles and abstracts. Of these, 1566 studies were excluded due to irrelevance, leaving 136 full-text articles for further review. Ultimately, seven studies based on four RCTs (CARES-310, COSMIC-312, IMbrave150, and ORIENT-32) were included, with 1420 patients in the PIAD group and 844 patients in the sorafenib group (Figure 1).6–12 Table 1 presents a summary of the initial characteristics of the studies included. Three studies6–11 were conducted in global multicenter and one study 12 was conducted in China multicenter. All studies had a low risk of bias for random sequence generation, allocation concealment, blinding, incomplete outcome data and selective reporting. Overall, the quality of the included studies was deemed adequate for the purposes of this meta-analysis (Supplementary Figure 1). According to Jadad scale, all 4 studies were in high quality (Supplementary Table 2). According to the method of GRADE, all outcomes were within the medium to high spectrum (Supplementary Table 3).

Flow chart.
Baseline Characteristics of the Included Studies.
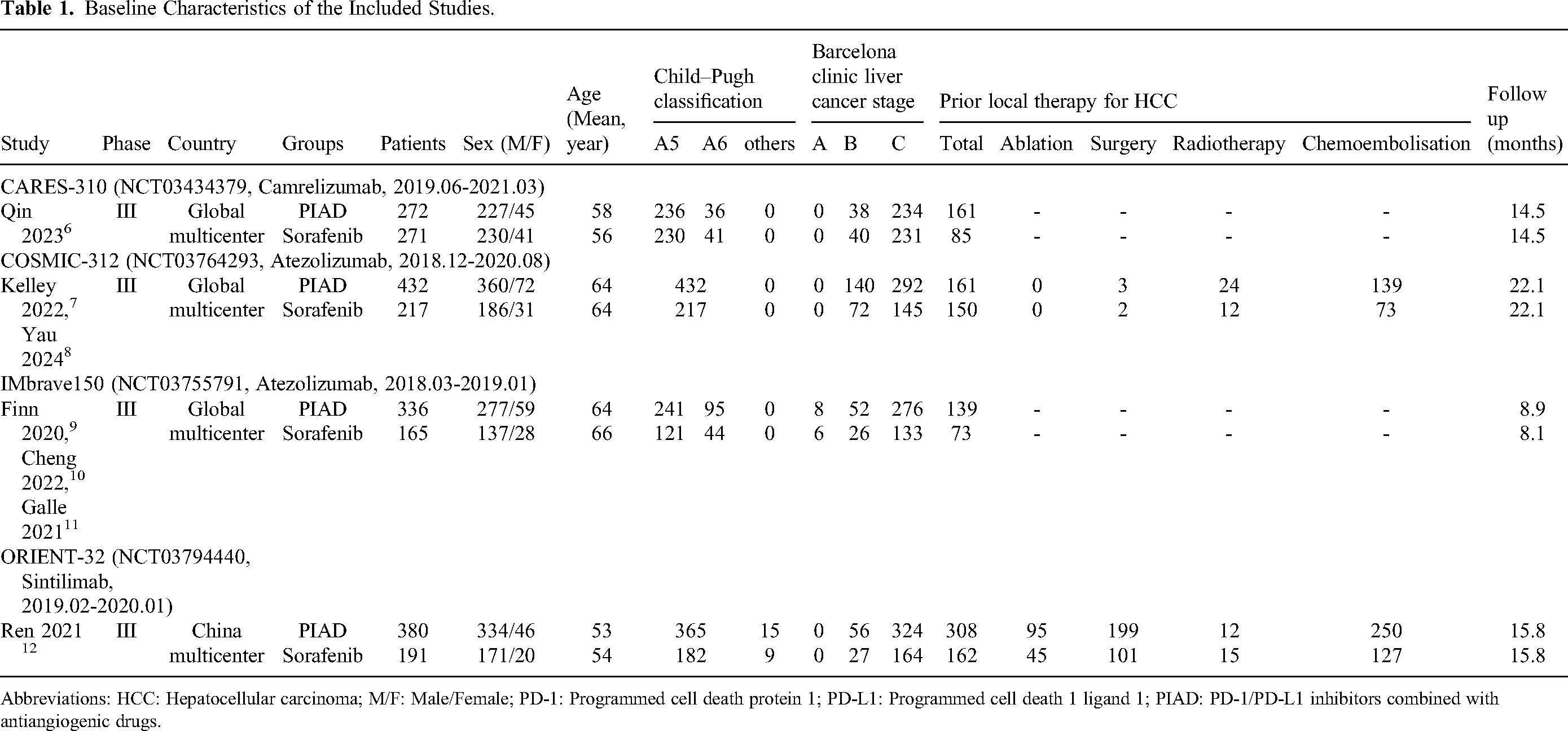
Abbreviations: HCC: Hepatocellular carcinoma; M/F: Male/Female; PD-1: Programmed cell death protein 1; PD-L1: Programmed cell death 1 ligand 1; PIAD: PD-1/PD-L1 inhibitors combined with antiangiogenic drugs.
Survival
The PIAD group exhibited improved OS (HR: 0.69, 95% confidence interval [CI]: [0.53, 0.89], P = 0.005) (Figure 2). The OSR at 6–18 months was significant higher in the PIAD group (Supplementary Figure 2). Nearly all subgroups showed a tendency for better OS in the PIAD group, except in the subgroups of race-white, extrahepatic spread at study entry-no, and etiology-nonviral (Table 2).

Forest plots of overall survival and progression-free survival associated with PIAD versus Sorafenib.
Subgroup Analysis of Overall Survival and Progression-Free Survival

Abbreviations: CI: Confidence interval; ECOG PS: Eastern Cooperative Oncology Group Performance Status; HR: Hazard ratio; PD-1: Programmed cell death protein 1; PD-L1: Programmed cell death 1 ligand 1; PIAD: PD-1/PD-L1 inhibitors combined with antiangiogenic drugs.
The PIAD group exhibited improved PFS (HR: 0.60, 95% CI: [0.53, 0.67], P < 0.00001) (Figure 2). The PFSR at 6–12 months was significant higher in the PIAD group (Supplementary Figure 3). All subgroups showed a tendency for better PFS in the PIAD group (Table 2).
Responses
According to RECIST 1.1, better ORR (RR: 3.40, 95% CI: [2.55, 4.53], P < 0.00001), disease control rate (DCR) (RR: 1.29, 95% CI: [1.21, 1.38], P < 0.00001), CR (RR: 6.94, 95% CI: [1.53, 31.40], P = 0.01), and partial response (PR) (RR: 3.24, 95% CI: [1.93, 5.41], P < 0.00001) were found in the PIAD group. Similar stable disease (SD) (RR: 1.05, 95% CI: [0.97, 1.14], P = 0.22) were found between the two groups (Figure 3).

Forest plots of responses according to RECIST 1.1 associated with PIAD versus Sorafenib.
According to mRECIST, better ORR (RR: 3.04, 95% CI: [2.35, 3.94], P < 0.00001), DCR (RR: 1.32, 95% CI: [1.22, 1.43], P < 0.00001), CR (RR: 4.86, 95% CI: [2.14, 11.03], P = 0.0002), and PR (RR: 2.80, 95% CI: [2.12, 3.69], P < 0.00001) were found in the PIAD group. Similar SD (RR: 0.96, 95% CI: [0.85, 1.07], P = 0.42) were found between the two groups (Supplementary Figure 4).
Quality of Life
Duration until decline in quality of life (HR: 0.76, 95% CI: [0.65, 0.89], P = 0.0008), physical functioning (HR: 0.65, 95% CI: [0.55, 0.77], P < 0.00001), and role functioning (HR: 0.72, 95% CI: [0.59, 0.89], P = 0.002) were all better in the PIAD group (Figure 4). The deterioration of PF-FSR at 6 month was higher in the PIAD group. The DQL-FSR 6–24 months (Supplementary Figure 5), PF-FSR 12–24 months (Supplementary Figure 6), and deterioration of role functioning free survival rate (RF-FSR) 6–24 months (Supplementary Figure 7) were similar between the two groups.

Forest plots of time to deterioration of quality of life, time to deterioration of physical functioning, and time to deterioration of role functioning associated with PIAD versus Sorafenib.
Safety
The rates of total AEs (RR: 1.04, 95% CI: [1.02, 1.05], P < 0.0001), grade 3–5 AEs (RR: 1.25, 95% CI: [1.12, 1.39], P < 0.0001), serious AEs (RR: 1.40, 95% CI: [1.21, 1.61], P < 0.00001), discontinuation due to AEs (RR: 2.35, 95% CI: [1.39, 3.98], P = 0.002), dose reduction due to AEs (RR: 1.48, 95% CI: [1.30, 1.70], P < 0.00001), dose interruption due to AEs (RR: 1.40, 95% CI: [1.13, 1.74], P = 0.002), grade 3–5 treatment-related AEs (TRAEs) (RR: 1.39, 95% CI: [1.04, 1.85], P = 0.03), and serious TRAEs (RR: 2.57, 95% CI: [1.56, 4.23], P = 0.0002) were higher in the PIAD group. The reduction due to AEs, and fatal TRAEs appeared to favor the sorafenib without statistical difference (Table 3).
Summary of Adverse Events
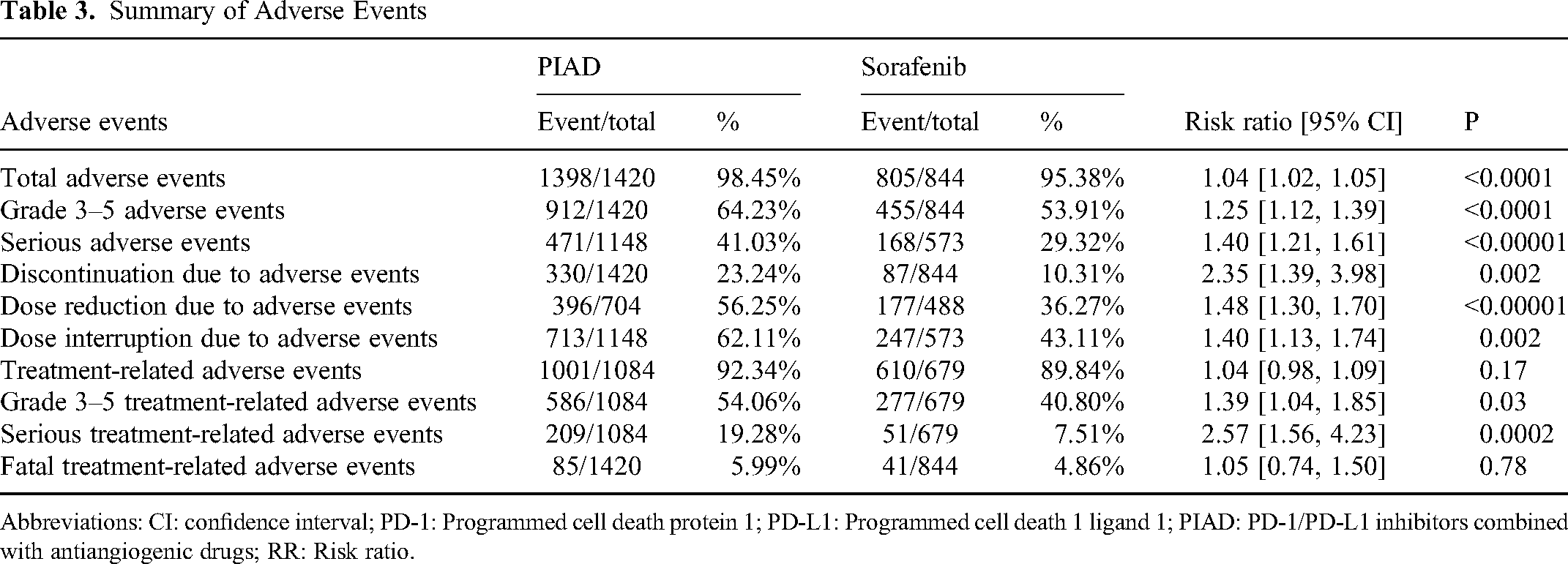
Abbreviations: CI: confidence interval; PD-1: Programmed cell death protein 1; PD-L1: Programmed cell death 1 ligand 1; PIAD: PD-1/PD-L1 inhibitors combined with antiangiogenic drugs; RR: Risk ratio.
In the analysis of any grade AEs, more proteinuria, hypertension, alanine aminotransferase increase, platelet count decrease, fatigue, hypothyroidism, pyrexia, pruritus, anemia, hypoalbuminemia, neutrophil count decrease, blood alkaline phosphatase increase, thrombocytopenia, headache, lipase increased, ascites, dyspnea, hyponatremia, epistaxis, amylase increase, and hyperglycemia were found in the PIAD group. The sorafenib group experienced higher incidences of palmar-plantar erythrodysesthesia syndrome, rash, and alopecia (Table 4, Supplementary Table 4).
Any Grade Adverse Events (Incidence Rate > 10% in the PIAD Group)

Abbreviations: CI: confidence interval; PD-1: Programmed cell death protein 1; PD-L1: Programmed cell death 1 ligand 1; PIAD: PD-1/PD-L1 inhibitors combined with antiangiogenic drugs; RR: Risk ratio.
In analyzing grade 3–5 AEs, more platelet count decrease, gamma-glutamyl transferase increase, proteinuria, neutrophil count decrease, hyponatremia, and lymphocyte count decrease were found in the PIAD group. More hypophosphatemia was found in the sorafenib group (Table 5, Supplementary Table 5).
Grade 3–5 Adverse Events (Incidence Rate > 2% in the PIAD Group)

Abbreviations: CI: confidence interval; PD-1: Programmed cell death protein 1; PD-L1: Programmed cell death 1 ligand 1; PIAD: PD-1/PD-L1 inhibitors combined with antiangiogenic drugs; RR: Risk ratio.
Sensitivity Analysis
Sensitivity analyses of OS, PFSR-6 m and grade 3–5 AEs were performed, demonstrating that excluding any single study had no impact on the credibility of the results (Supplementary Figure 8).
Publication Bias
Balanced funnel diagrams were observed regarding survival, response rates, impacts on quality of life, and safety summaries, indicating an acceptable level of publication bias (Figure 5).
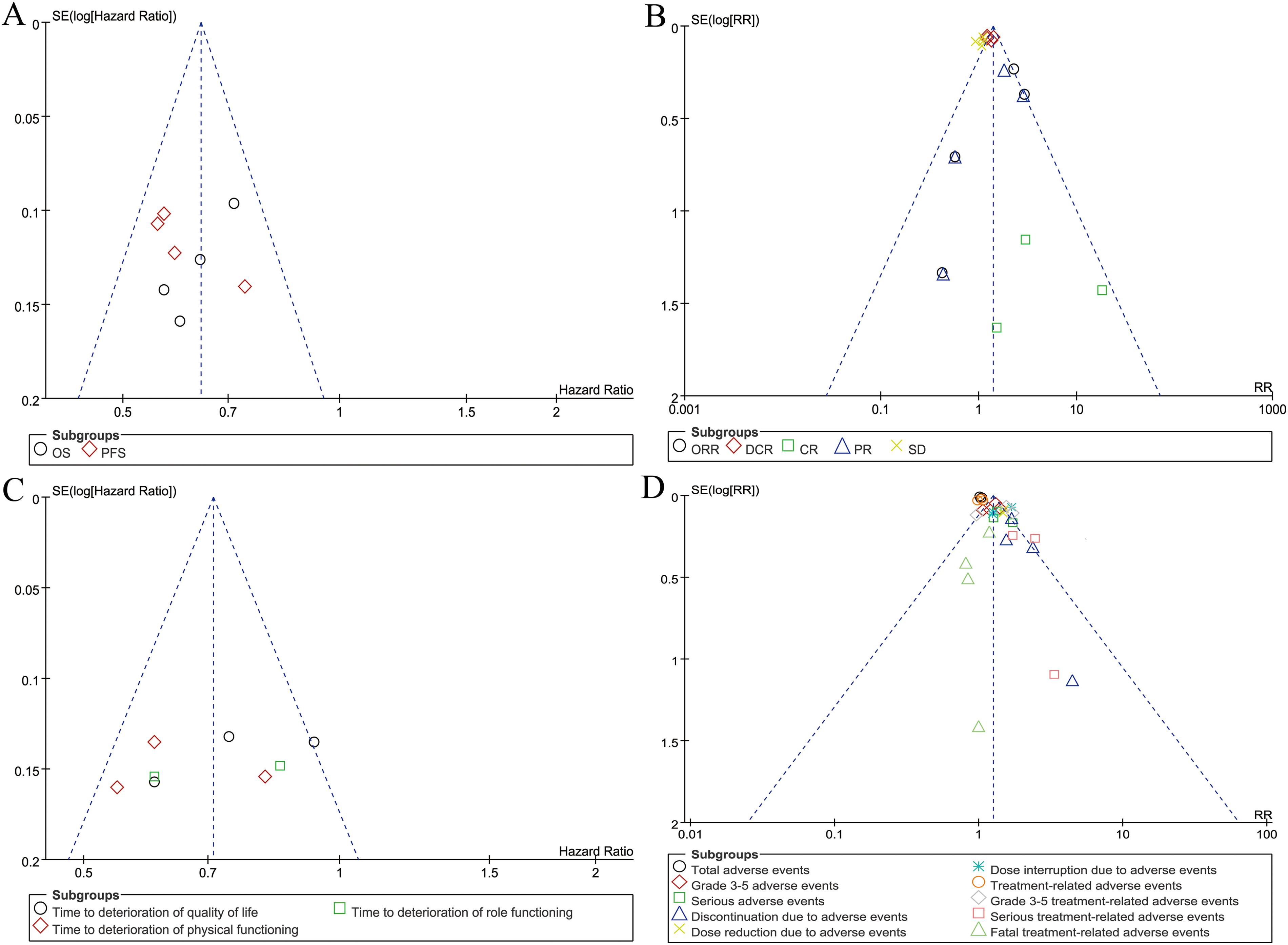
Funnel plots of survival (A), responses (B), the impact on quality of life (C), and safety summary (D).
Discussion
Sorafenib has served as the standard therapy for advanced HCC, a condition that ranks high in both incidence and mortality rates. 3 However, the effectiveness of Sorafenib has not met expectations. Previous meta-analyses have validated the clinical value of PD-1/PD-L1 inhibitors monotherapy in the treatment of advanced HCC. 19 Recently, PIAD has emerged as a new therapeutic option for advanced HCC, though whether it offers superior clinical outcomes remains a subject of ongoing debate.6–12 Our meta-analysis indicates that the PIAD group demonstrated better OS and PFS. The survival advantages of OS and PFS were confirmed across almost all subgroups. The OSR at 6–18 months and PFSR at 6–12 months were higher in the PIAD group. Additionally, ORR, DCR, CR, and PR were also higher in the PIAD group. The duration until decline in quality of life was notably extended in the PIAD group. However, there was a higher incidence of total AEs and grade III to V AEs in the PIAD group.
The superior survival outcomes observed in the PIAD group are a significant advancement in the treatment of advanced HCC. Notably, the PIAD group demonstrated better OS and PFS compared to the sorafenib group. This finding is consistent with the results from pivotal trials such as IMbrave150, which reported that patients treated with atezolizumab plus bevacizumab had a 42% decreased risk of death compared to those receiving sorafenib. 9 Similarly, the ORIENT-32 trial showed a significant OS benefit for sintilimab plus bevacizumab over sorafenib. 12 The improved efficacy of PIAD can be attributed to the complementary mechanisms of action of PD-1/PD-L1 inhibitors and antiangiogenic drugs therapy. PD-1/PD-L1 inhibitors rejuvenate the immune system's ability to identify and combat cancerous cells, while antiangiogenic drugs inhibit the formation of new blood vessels that tumors need for growth and metastasis. This dual approach not only targets the tumor cells directly but also disrupts the tumor microenvironment, making it less conducive to cancer progression.20,21 Subgroup analyses in our meta-analysis further reinforce the robustness of these findings. The survival benefits of PIAD over sorafenib were observed across various patient demographics and clinical characteristics, including age, sex, geographic region, and ECOG performance status.22,23 This suggests that PIAD could be a broadly applicable treatment option for advanced HCC. However, certain subgroups, such as patients with non-viral etiology or those without extrahepatic spread, did not show a significant survival advantage with PIAD. This indicates the need for personalized treatment approaches based on individual patient profiles to maximize therapeutic benefits.24,25
Despite the clear efficacy benefits, the use of PIAD is associated with a higher incidence of adverse events, which poses significant safety concerns. Patients receiving PIAD experienced more total AEs, and grade 3–5 AE compared to those treated with sorafenib. The most common severe AEs included proteinuria, hypertension, and neutropenia, which are consistent with the known AEs of both PD-1/PD-L1 inhibitors and antiangiogenic drugs.26,27 The increased incidence of AEs in the PIAD group necessitates careful patient monitoring and management. Regular assessment of renal function, blood pressure, and hematologic parameters is essential to promptly identify and manage AEs. Meanwhile, assessing liver functional reserve is crucial in guiding treatment choices for patients with HCC, especially those with prior treatments that may impair liver function. These treatments, while essential for controlling tumor growth, can reduce the liver's ability to tolerate further therapies. This makes the evaluation of liver functional reserve a vital step in determining the most appropriate treatment strategy, particularly in patients with underlying cirrhosis. 28 For instance, proteinuria, a common AE with antiangiogenic drugs therapy, requires regular urine tests and may necessitate dose adjustments or temporary discontinuation of treatment.29,30 Hypertension management may involve antihypertensive medications, and severe cases may require dose modifications or discontinuation of the antiangiogenic drugs drug. Neutropenia, observed with both PD-1/PD-L1 inhibitors and antiangiogenic drugs therapy, increases the risk of infections, necessitating vigilant monitoring and possibly the use of growth factors to mitigate the risk.7,31 The higher occurrence of grade III to V AEs in the PIAD group is particularly concerning, as these severe AEs can significantly impact patients’ quality of life and overall treatment adherence. For example, the IMbrave150 trial reported that the rate of grade 3–4 AEs was 56.5% in PIAD group VS 55.1% in sorafenib group. 9 While difference may seem marginal, the types and management of these AEs differ significantly. For instance, the PIAD group had higher rates of hypertension and proteinuria, which require specific management strategies not typically necessary with sorafenib.32,33
The findings from this meta-analysis suggest that while PIAD offers superior efficacy in terms of survival outcomes and tumor responses, its higher toxicity profile necessitates a cautious approach in clinical practice. Clinicians must weigh the benefits of improved survival and tumor control against the risks of severe AEs. This decision-making process should involve a thorough conversation with patients about the potential benefits and risks of PIAD, considering their overall health status, comorbid conditions, and preferences.34,35 To optimize the therapeutic use of PIAD in advanced HCC, future research should focus on several key areas. First, identifying biomarkers that can predict response and resistance to PIAD will help tailor treatments to individual patients, maximizing efficacy while minimizing unnecessary toxicity. Second, developing strategies to manage and mitigate AEs associated with PIAD is crucial. This includes refining dosing regimens, exploring combination therapies with lower toxicity profiles, and enhancing supportive care measures.36,37 Improvements in AE management over time led to prolonged treatment duration and improved OS in patients. This highlights the fact that, careful management can reduce the frequency of treatment discontinuation and optimize patient outcomes. This factor is particularly relevant when comparing sorafenib to newer therapies such as PIAD, which have shown superior efficacy but are associated with a higher incidence of serious AEs. 38 Third, recent clinical evidence has shown that TKI/ICI combinations offer enhanced anti-tumor efficacy and improved safety profiles compared to earlier treatments like sorafenib. These findings indicate that TKI/PD-1/PD-L1 inhibitors combinations could represent a future direction in the treatment of advanced HCC, warranting further studies to directly compare these regimens with PIAD. 39
Our study has certain limitations. Firstly, only English articles were included, causing language bias. Secondly, the inclusion of only 4 RCTs limits the clinical value. Thirdly, a lack of data prevented individual patient data meta-analysis, potentially reducing clinical value. Fourthly, In the PIAD group, the selection of PD-1/PD-L1 Inhibitory agents and antiangiogenic drugs varies, affecting the compatibility of data across studies.
Conclusion
PIAD appears to be better than sorafenib for advanced HCC, showing superior OS and PFS. The superior survival outcomes of PIAD are observed across various subgroups. However, the higher incidence of AEs associated with PIAD requires careful patient selection and management. Future research should focus on identifying predictive biomarkers and optimizing management strategies to enhance the therapeutic efficacy of PIAD for advanced HCC.
Supplemental Material
sj-docx-1-tct-10.1177_15330338241305700 - Supplemental material for PD-1/PD-L1 Inhibitors Plus Antiangiogenic Drugs Versus Sorafenib as the First Line Treatment for Advanced Hepatocellular Carcinoma: A Phase 3 RCTs Based Meta-Analysis
Supplemental material, sj-docx-1-tct-10.1177_15330338241305700 for PD-1/PD-L1 Inhibitors Plus Antiangiogenic Drugs Versus Sorafenib as the First Line Treatment for Advanced Hepatocellular Carcinoma: A Phase 3 RCTs Based Meta-Analysis by Jun Li, Chun Liao, Zhaohui Liu, Hu Xiong, Jing Cai and Tiande Liu in Technology in Cancer Research & Treatment
Footnotes
Abbreviations
Acknowledgements
The authors thank professor Wenxiong Zhang, MD (Department of Thoracic Surgery, The second affiliated hospital of Nanchang University) for his data collection and statistical advice.
Data Availability Statement
The data sets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval
This study is a meta-analysis of previously published data and did not involve direct participation of human subjects; therefore, ethical approval from an Institutional Review Board (IRB) was not required.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Training Program for Academic and technical leaders in major disciplines in Jiangxi Province (20212BCJL23063), Natural Science Foundation General Project of Jiangxi (20212BAB206051), and Jiangxi Provincial Health Department Project (202130375,202210610). The funding had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
