Abstract
Epithelial-mesenchymal transition (EMT) is a key step in tumor invasion and distant metastasis. Abundant evidence has documented that exosomes can mediate EMT of tumor cells and endow them with the ability of invasion and migration. However, there are few studies focusing on whether EMT can reverse the secretion of exosomes. In this study, 2 esophageal cancer cells (FLO-1 and SK-GT-4) were selected to compare the migration ability and EMT activation, and to further analyze the secretion ability of exosomes of the 2 cell lines. According to the results, inhibited activation of EMT in FLO-1 cells with relatively high migration ability could effectively reduce the secretion of exosomes. Besides, in SK-GT-4 cells, EMT activation induced by TGF-β could promote the secretion of exosomes. FLO-1 cell derived exosomes exhibited a paracrine effect of promoting the migration of SK-GT-4 cells, and the use of EMT inhibitors could weaken this ability. Furthermore, inhibition of EMT could change the relative content of some miRNAs in exosomes, with a particularly significant downregulation in the expression of miR-196-5p, miR-21-5p and miR-194-5p. Significantly, artificial transfection of the 3 miRNAs into exosomes by electroporation resulted in the recovery of migration-promoting effect of exosomes. Subsequent experiments further revealed that the effect of EMT on these miRNAs could be explained by the intracellular transcription level or the specific sorting mechanism of exosomes. To sum up, our study undoubtedly reveals that EMT has a regulatory effect on exosomes in the quantity and contents in esophageal cancer cells. Significantly, findings in our study provide experimental evidence for the interaction of EMT with the secretion and sorting pathway of exosomes, and also give a new direction for the further study of tumor metastasis.
Introduction
Epithelial-mesenchymal transition (EMT) is an extremely important basic procedure in the process of normal embryonic development, wound healing and malignant epithelial tumorigenesis. 1,2 It can alter epithelial cells morphology and promote the transition of these cells into motile mesenchymal cells. Consequently, these changes can damage cell-cell and cell-extracellular matrix junctions, so that cells can migrate to other parts of the body to participate in tissue reconstruction and repair consequently. Furthermore, EMT also plays a critical role in tumor cell migration/invasion and the progression of various cancers. It is a key step in tumor cell metastasis and a specific signaling pathway. EMT is characterized by the loss of cell adhesion, downregulated expression of epithelial markers such as E-cadherin and β-catenin and upregulated expression of interstitial markers such as N-cadherin and vimentin, with enhanced abilities of cell movement and invasion. 3 The progression of tumor EMT is attributed to the internal and external factors or both. Meanwhile, increasingly more attention has been paid to the role of miRNA in the intracellular and extracellular signaling pathways of EMT. 4 -6
MicroRNA (miRNA) is a kind of small non-coding endogenous RNA molecules which is about 22nt in length. miRNA can inhibit target mRNA translation or promote its degradation by binding 3′-untranslated region (3′-UTR) of target mRNA at post-transcriptional level, thus realizing negative regulation of target gene. Current research has found many miRNAs to be involved in the regulation of EMT. Most of the miRNAs that inhibit EMT share similarity of decreased expression in tumor cells, which leads to the occurrence of EMT. However, re-expression of such miRNA can inhibit this process by directly or indirectly inhibiting the expression of EMT related transcription factors such as ZEB, SNAIL, TWIST, SLUG, etc. 7 -10 Conversely, there must be some miRNAs that can promote EMT through other mechanisms. For instance, complementary sites exist between miR-9 and 3′-UTR of E-cadherin mRNA. In addition, stable transfection of miR-9 in human esophageal adenocarcinoma cell lines HKESC1 and KYSE410 resulted in a decrease in E-cadherin expression at both RNA and protein levels. 11 In liver cancer, miR-9 can promote EMT as well as invasion and metastasis of liver cancer cells by acting on KLF17. 12 Meanwhile, miR-21 inhibits the expression of PTEN and human sulfatase-1 (hSulf-1) in liver cancer cells, which leads to the activation of AKT/ERK pathway and the occurrence of EMT. 13 In addition, miR-21 can also promote the occurrence and progress of tumor EMT in cholangiocarcinoma and clear cell renal cell carcinoma. 14,15 miR-373 can down-regulate thioredoxin-interacting protein (TXNIP), which increases reactive oxygen species (ROS) production, activates the TWIST signal regulation axis of hypoxia inducible factor-1 (HIF-1) and thus induces EMT. 16 Moreover, in A549 and NCIH1650 cell lines, overexpression of miR-214 also promotes the development of EMT in lung adenocarcinoma cells. 17
Exosomes are nano-sized vesicles with lipid bilayer structure released by cells through efflux, with diameters ranging from 30 to 150 nm. Multiple cells have been discovered to possess the ability to secrete exosomes, such as B cells, epithelial cells, fibroblasts, a variety of tumor cells, etc. 18 Meanwhile, exosomes have been identified to carry a wide variety of proteins, lipids, nucleic acids and other active substances, which are obviously engaged into information exchange and material transfer among cells. It has been demonstrated that various proteins and miRNAs in exosomes participate in EMT process of tumor cells, exerting significant impact on the ability of tumor invasion and metastasis. 19 -21 The occurrence of EMT exhibits a strong association with exosomes, and the majority of the current studies focus on the promotion or inhibition of exosomes on EMT, revealing generous exosome/miRNA/EMT regulatory pathways. However, considering the presence of mutual influence, we are wondering whether the occurrence or inhibition of EMT will reverse the secretion of exosomes and the relative content of active substances. Accordingly, the present study was carried out with the purpose to explore the reverse regulation of exosomes by EMT in esophageal cancer cells.
Materials and Methods
Cell Culture and Reagents
Human esophageal adenocarcinoma cell lines (FLO-1 and SK-GT-4) were obtained from the Cell Resource Center, Peking Union Medical College. SK-GT-4 cell line was cultured in RPMI-1640 medium (Gibco, Grand Island, New York, USA) whereas Flo-1 was cultured in DMEM medium (Gibco) supplemented with 10% (v/v) fetal bovine serum (FBS; Invitrogen), 100 U/ml penicillin G, and 100 mg/ml streptomycin sulfate (Sigma-Aldrich) at 37°C with 5% CO2. EMT pathway related inhibitors: ML327 (C19H18N4O4, Med Chem Express, USA) and EMT inhibitor-1 (C12H12Cl2N2O2 S, Med Chem Express).
Exosomes Extraction
Cells were cultured in indicated medium supplemented with 10% exosome-depleted FBS for 48 h. Exosomes in the culture supernatant were prepared using the ExoQuick-TC kit (System Biosciences, Mountain View, CA, USA) as described by the manufacturer. In general, the medium was centrifuged for 15 min at 3000 g to remove apoptotic cells and cell debris. After adding 3.3 ml of the exosome-precipitating solution to each 10 ml of the culture supernatant, the cells were refrigerated overnight, and then the mixed liquid was centrifuged at 10000 g for 30 minutes, and the supernatant was discarded; the separated exosomes were suspended in PBS, stored at −80°C or used directly.
Exosome Quantification
Nanoparticle tracking analysis (NTA) was employed to check the prepared exosome samples. Briefly, exosomes re-suspended in 50 µL PBS were further diluted to 300-fold to achieve between 20 and 100 objects per frame and detected by NanoSight NS-300 (Malvern Panalytical, Malvern, UK). Each sample was measured in triplicate through the camera with an acquisition time of 30 s and the detection threshold was setting at 3. At least 200 completed were analyzed in each video. NTA 2.3 software was used to capture and analyze the result.
Scanning Electron Microscopy (SEM)
Exosome morphology was examined using Scanning Electron Microscopy. Pellets were fixed in 2% paraformaldehyde and 1% glutaraldehyde (Sigma). The sample was applied to a continuous carbon grid, washed in distilled H2O, then dehydrated, left to dry, and observed using SEM (Hitachi SU 6600) at 20 kV. Image J software was used for analysis.
miRNA Loading Into Exosomes by Electroporation
Briefly, exosome pellet was resuspended in PBS 0.1× and gently mixed with the appropriated µl of miRNA (10nM) in a final volume of 400 µl into 0.4 cm electroporation cuvettes. Exosomes were then electroporated using a Gene Pulser II Electroporator (Bio-Rad, Hercules, CA, USA), at 300 V and 25 μF of capacitance. Lastly, exosomes were incubated in ice for 30 min to allow the exosome membrane to be fully restored. To get rid of free nucleic acids, exosomes were diluted with cold PBS and isolated again by ultracentrifugation at 120,000 g for 90 min at 15°C.
Protein Preparation and Western Blot
Total exosomic and cellular protein was extracted using RIPA lysis buffer (Beyotime Institute of Biotechnology, Beijing, China). The protein concentrations of cell and exosome lysates were determined using BCA protein assay kit (Beyotime Institute of Biotechnology, Beijing, China). For Western blot analyses, the cellular and exosomic proteins (10 µg/well) were separated by electrophoresis in 10% sodium dodecylsulfate-polyacrylamide gel electrophoresis, transferred to polyvinylidene difluoride membrane (Amersham, Buckinghamshire, UK). The membrane was blocked with 5% skimmed milk in TBS-T (10 mM TrisCl, pH 8.0, 150 mM NaCl, 0.5% Tween 20) at 4°C overnight, rinsed 3 times (10 min/time) with TBS-T, followed by 3 h incubation at room temperature with the specific primary antibodies, followed by 1 h incubation with the second antibody. ECL developer (Thermo Scientific) was added and exposure imaging was performed by Odyssey gel imaging system. The specific antibodies to E-cadherin (ab1416), N-cadherin (ab76011), Vimentin (ab92547), Snail (ab216347), CD81 (ab109201), HSP70 (ab181606), TSG101 (ab125011), Calnexin (ab133615), were purchased from Abcam (Cambridge, UK). β-actin antibody (#4970) was acquired from Cell Signaling Technology (Danvers, MA, USA).
miRNA Sequencing
Exosomes were resuspended in 500 µL of Trizol (Invitrogen, CA, U.S) for high-throughput miRNA sequencing (Gx-health Biotech, Beijing, China). Briefly, total RNA was extracted and the A260/A280 ratio measured using a NanoDrop ND-1000 (Thermo Fisher Scientific, Waltham, MA, USA). All samples had an A260/A280 ratio >1.8. The miRNA sequencing library was established and the quality assessed using an Agilent 2100 Bioanalyzer (Agilent Technologies, Santa Clara, CA). The library was denatured and the single-stranded DNA molecules captured on Illumina flow cells. DNA was then amplified in situ as clusters and amplified for 50 cycles on an Illumina HiSeq sequencer according to the manufacturer’s instructions.
Quantitative RT-PCR (qRT-PCR)
Total RNA was extracted using TRIzol reagent (Invitrogen). Reverse transcription of cDNA was carried out using a RevertAid First Strand cDNA Synthesis Kit (Thermo Scientific, CA, U.S.). Pri-miRNA-specific PCR amplification was performed with Power SYBR Green Master Mix on ABI 7500 Real-Time PCR System (Applied Biosystems, Warrington, UK). Relative pri-miRNAs expression was calculated with the 2−ΔΔCt method after normalization to GAPDH expression.
An All-in-One miRNA qRT-PCR Detection Kit (GeneCopoeia, MD, U.S.) was used to measure miRNA expression. Briefly, total RNA was treated with poly A polymerase to add poly-A tails to the 3′-end of miRNAs. M-MLV RTase, with a universal adaptor PCR primer, was used to reverse transcribe miRNA tailed poly-A. Then, real-time qPCR was carried out using SYBR Green to measure microRNA expression. Intracellular miRNAs and pre-miRNAs were normalized against U6 snRNA. Cel-mir-39 was used as the exogenous control for miRNAs in exosomes. Primers for RT-qPCR sequences are listed in Supplementary Table S1.
Migration Assays
Transwell plates (BD Biosciences) were used to analyze cellular migration ability. Cells in serum-free medium were added to the upper chamber, and medium with 20% FBS was applied to the lower chamber. After 48 h, residual cells were removed from the upper chamber. Cells were then fixed with methanol, and stained with with 4′, 6-diamidino-2-phenylindole (DAPI) (Thermo Fisher Science). After washing with PBS, the number of cells passing through the membrane was counted under an inverted microscope (IX71, Olympus, Japan). Three random microscopic fields were selected for statistical analysis.
CCK-8 Assay
Cell proliferation capacity was measured using the CCK8 assay. The digested cells, 3 × 103 cells/well, were seeded into the 96-well plate. Incubate in a 5% CO2 incubator at 37°C for various time points, discard the original culture medium, add 100 µl of the mixture of CCK8 reagent (DoJinDo Laboratories, Japan) and medium at the ratio of 1:9, and incubate in the incubator for additional 1 hour. The absorbance values of 450 nm wavelength were determined by enzyme labeling instrument (Multimode Reader, PerkinElmer, USA).
Statistical Analysis
All data were processed by SPSS21.0 software and expressed as mean ± SD, and Shapiro-Wilk (W) normality test was carried out. For non-normally distributed data, the nonparametric Wilcoxon signed-rank test was used to evaluate the statistical differences between groups. When the data were close to normal distribution, one-way ANOVA was used to compare multiple sets of data, and LSD method was used to analyze the multiple comparisons of means. P < 0.05 was considered to be statistically significant.
Results
Comparison of Migration Ability Between Different Esophageal Cancer Cells
Two esophageal cancer cells, FLO-1 and SK-GT-4, were selected firstly for subsequent experiment. Cell migration test showed that the migration ability of FLO-1 cells was significantly higher than that of SK-GT-4 cells (Figure 1A and B). Considering the intimate association of EMT pathway with tumor invasion and metastasis, our experiment was continued to compare the relative expression levels of EMT related markers in the 2 cells. Corresponding results revealed no significant difference in the expression of epithelial-like cell marker E-cadherin between FLO-1 and SK-GT-4 cells, but the relative expression levels of mesenchymal-like cell markers N-cadherin, Vimentin and Snail were significantly higher in FLO-1 cells than those in SK-GT-4 cells (Figure 1C).
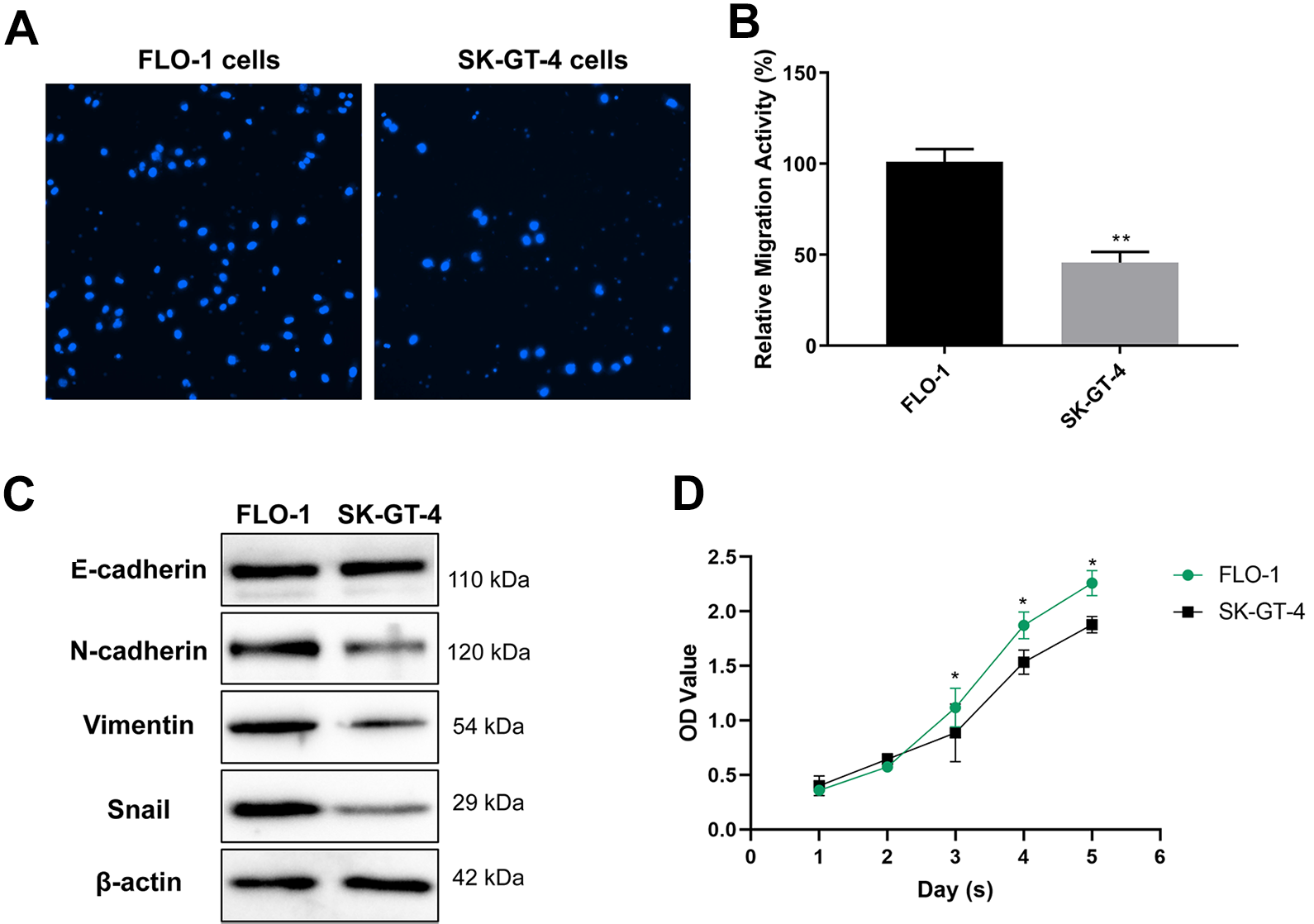
Analysis of differences in migration ability of 2 types of esophageal carcinoma cells (FLO-1 and SK-GT-4). A, Cell migration ability was tested by the transwell cell migration assay (magnification, 200×). B, The quantifications of cell migration were presented by the histogram. C, Western blot of EMT markers and MMP9 in FLO-1 and SK-GT-4 cells. D, CCK-8 was used to detect cell viability at different time points. Data are the means ± SD of 3 independent experiments. *P < 0.05 and **P < 0.01 versus control.
Correlation Between EMT and Exosome Secretion of Esophageal Cancer Cells
Subsequent experiment focused on the exosome secretion characteristics of the 2 cells. Exosomes derived from the 2 cells had typical exosomes marker proteins (HSP70, CD81 and TSG101), yet with the absence of endoplasmic reticulum marker Calnexin (Figure 2A). Meanwhile, according to the results of electron microscopy, no obvious difference was found in the morphology of exosomes derived from the 2 cells (Figure 2B). Interestingly, there was a significant difference in the number of exosomes derived from FLO-1 and SK-GT-4 cells. Specifically, the number of exosomes secreted by FLO-1 cells was much higher than that by SK-GT-4 cells in the same period of time (Figure 2C). We hence proposed a hypothesis that if the secretion capacity of exosomes associated with EMT activity.

Exosome characterization of 2 laryngeal carcinoma cells (FLO-1 and SK-GT-4). A, Expression of the exosome markers confirmed by Western blot. B, Representative SEM micrographs of exosomes (Exo) purified from the indicated cells. C, Exosome concentrations were quantified by NTA. The relative exosome concentration was normalized to the cell number. **P < 0.01.
In FLO-1 cells, with the use of 2 EMT pathway related inhibitors (ML327 and EMT inhibitor-1), there were effective reduction of the expression of EMT marker proteins N-cadherin, Vimentin and Snail, while increase of the relative content of E-cadherin (Figure 3A). Meanwhile, the migration ability of FLO-1 cells was also significantly downregulated by ML327 and EMT inhibitor-1 (Figure 3B and C). Moreover, the use of EMT inhibitors led to an obvious decrease in the number of exosomes secreted by FLO-1 cells (Figure 3D). In contrast, the classical TGF-β induced EMT activation scheme was applied in SK-GT-4 cells. Consequently, it was observed that the expression of E-cadherin was downregulated, and N-cadherin, Vimentin and Snail were upregulated accordingly following induction by using TGF-β (Figure 4A), accompanied by enhanced cell migration (Figure 4B and C). Besides, the number of exosomes secreted by SK-GT-4 after TGF-β induction was also significantly higher than that of the control group (Figure 4D). Collectively, all of these results support a positive correlation between EMT pathway and the number of exosomes secreted in esophageal cancer cells.

EMT inhibitor reduced exosome secretion in FLO-1 cells. A, Western blot detection of the relative expression of EMT marker proteins following FLO-1 cell treatment with ML327 (5 μM) and EMT inhibitor-1 (5 μM) for 24 h. B, Transwell cell migration test of FLO-1 cell migration ability in each group (magnification, 200×). C, Histogram of the relative migration ability of cells in each group. D, NTA detection of the secretion concentration of exosomes in FLO-1 cells of each group. The relative exosome concentration was normalized to the cell number. *P < 0.05 and **P < 0.01 versus control.

TGF-β induced EMT activation promoted exosome secretion of SK-GT-4 cells. A, Western blot detection of the relative expression of EMT marker proteins following SK-GT-4 cell treatment with TGF-β (5 ng/ml) for 24 h. B, Transwell migration test of SK-GT-4 cell migration ability after TGF-β induction (Magnification, 200×). C, Histogram for the quantification of the relative migration ability of cells in each group. D, NTA detection of the secretion concentration of exosomes in SK-GT-4 cells of each group. The relative exosome concentration was normalized to the cell number. **P < 0.01.
Impact of EMT on the Function and Contents of Exosomes
It is well-known that exosomes generally possess the characteristics of the derived cells and can act on the receiving cells in the form of paracrine. In our experiment, SK-GT-4 cells were treated with exosomes derived from FLO-1 cells with relatively high migration ability. The results showed that the FLO-1 exosomes could improve the migration ability of SK-GT-4 cells (Figure 5A and B). However, the exosomes derived from FLO-1 cells treated with EMT inhibitors displayed obviously weakened ability to promote cell migration (Figure 5A and B). In the next step, the exosomes derived from FLO-1 cells were extracted before and after EMT inhibitor treatment. The subsequent high-throughput sequencing results showed that the miRNAs in the 2 exosomes were not completely identical, with evident change in the relative content of many miRNAs (Table 1, Supplementary Table S2). After that, quantitative PCR verified the most obvious differences in the relative content of miR-196-5p, miR-21-5p and miR-194-5p, yet with significant decrease in exosomes derived from FLO-1 cells treated with EMT inhibitor (Figure 6A). The 3 miRNAs were then loaded into the exosomes of FLO-1 cells treated with ML327 by electroporation in vitro (Figure 6B). It could effectively increase the relative content of the 3 miRNAs in SK-GT-4 cells (Figure 6C), reactivate EMT pathway (Figure 6E), and partially restore the migration promoting ability of exosomes (Figure 6D). On the basis of the above, it suggested that the changes of EMT pathway not only altered the secretion quantity of exosomes, but also affect the contents of exosomes, thus affecting the paracrine effect of exosomes.

The migration promoting effect of FLO-1 cell derived exosomes on SK-GT-4 cells. A, The exosomes from normal and ML327 treated FLO-1 cells were collected and co-cultured with SK-GT-4 cells in the 24-well plate at the concentration of 50 μg/mL for 24 h. The migration ability of SK-GT-4 cells in each group was detected by Transwell migration test (Magnification, 200×). The control group was treated with PBS (PBS group). B, Histogram for the quantification of the relative migration ability of cells in each group. **P < 0.01.
Expression of MiRNAs in Exosomes Derived From FLO-1 Cells With or Without ML327 Treating.a

a Upregulated/downregulated, in the ML327 treated exosomes as compared to the control exosomes. Only the top 14 miRNAs with the most significant differences are listed (log2 ≥ 2 or ≤ −5).

The regulatory effect of EMT on the paracrine effect of promoting migration by changing the relative content of miRNA in exosomes derived from FLO-1 cells. A, Quantitative PCR verification of the differential expression of miRNAs in exosomes derived from FLO-1 cells treated with ML327. B, Transfection of miR-196-5p, miR-21-5p and miR-194-5p into the exosomes derived from FLO-1 cells treated with ML327 by electroporation to prepare exosomes rich in the 3 miRNAs. Quantitative PCR was used to detect the relative content of the 3 miRNAs in exosomes. C, Detection of the relative contents of the 3 miRNAs after co-incubation of exosomes in each group with SK-GT-4 cells for 6 hours. D, Western blot detection of the relative expression of EMT marker proteins after co-incubation of exosomes in each group with SK-GT-4 cells for 24 hours. E-F, Transwell migration assay detection of the migration ability of SK-GT-4 cells after the treatment of exosomes (magnification, 200×). Exo, exosomes derived from FLO-1 cells; ML327-Exo, exosomes derived from FLO-1 cells treated with ML327; ML327-Exo-ET, exosomes rich in miR-196-5p, miR-21-5p and miR-194-5p prepared by electroporation from ML327-Exo’; ET, electroporation. Comparisons among groups were determined by one-way ANOVA with Tukey post-hoc analysis. *P < 0.05 and **P < 0.01.
The Regulatory Effect of EMT on Exosome Output of miRNA at Multiple Levels
Why does inhibition of EMT pathway change the relative content of miRNA in exosomes derived from FLO-1 cells? Our experiment intended to address the issue in cells fundamentally. After cell treatment with ML327, the relative contents of the initial transcription product pri-miRNA, precursor miRNA (pre-miRNA) and mature miRNA corresponding to miR-196-5p, miR-21-5p and miR-194-5p in cells were detected by quantitative PCR assay. According to the results, only pri-miR-21, pre-miR-21 and mature miR-21 were detected to be downregulated in ML327 treated cells (Figure 7A-C), suggesting that EMT pathway can regulate the transcription level of miRNA-21. However, there was no significant difference in pri-miRNA and pre-miRNA corresponding to miR-196-5p and miR-194-5p (Figure 7A, B), but with an upregulation in the relative content of mature miRNA in ML327 treated cells (Figure 7C). These results suggest that the inhibition of EMT pathway in FLO-1 cells may result in the inhibited exosome release of miR-196-5p and miR-194-5p. Subsequently, cells were treated with ML327 and Actinomycin D was used to inhibit gene transcription to exclude the difference of transcription level. No obvious difference was detected in the relative contents of pri-miRNA and pre-mRNA corresponding to miR-21 (Figure 7D and E). While the amount of mature miR-21 increased after 4 hours of ML327 treatment (Figure 7F), but decreased in the corresponding exosomes (Figure 7G). In other words, following the exclusion of the impact of transcription level, inhibited EMT pathway resulted in the suppression of exosome release of miR-21-5p as well. More interestingly, when C. elegans cel-miR-73 was transferred into ML327 or DMSO treated FLO-1 cells, no significant difference was observed in the relative content of cel-miR-73 between ML327 treated cells and DMSO treated control cells for 4 hours (Figure 7H). Similarly, there was no obvious difference in the relative content of cel-miR-73 in its exosomes from that in the control exosomes (Figure 7I). Collectively, the aforementioned results undoubtedly reveal that EMT pathway can affect the emission of miRNA in exosomes selectively.
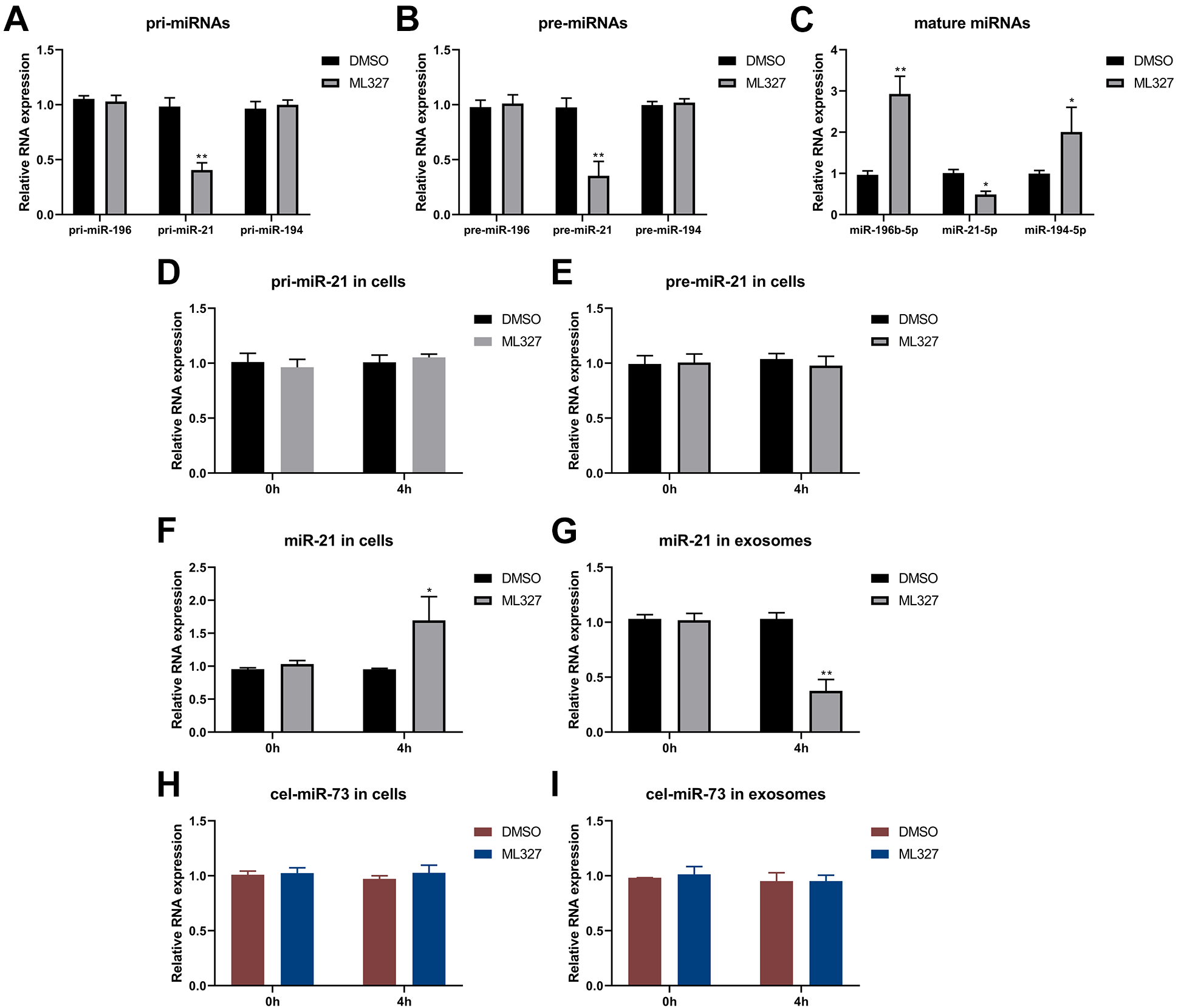
The regulatory effect of EMT on exosome output of miRNA at multiple levels in FLO-1 cells. A-C, Quantitative PCR detection of the relative content of pri-miRNA, pre-miRNA and mature miRNA corresponding to miR-196-5p, miR-21-5p and miR-194-5p after FLO-1 cell treatment with ML327. D-E, Quantitative PCR detection of the relative content of pri-miR-21 and pre-miR-21 in cells of the ML327 treatment group and DMSO control group following FLO-1 cell treatment with Actinomycin D (10 mg/L). F-G, Quantitative PCR detection of the relative content of miR-21-5p in cells and exosomes of the ML327 treatment group and DMSO control group following FLO-1 cell treatment with Actinomycin D. H-I, Quantitative PCR detection of the content difference of cel-miR-73 in cells and exosomes of the ML327 treatment group and DMSO control group following pre-transfection of cel-miR-73 (20nM) into FLO-1 cells for 4 hours. *P < 0.05 and **P < 0.01.
In addition, exosome biogenesis inhibitors GW4869 or Dimethyl amiloride (DMA) significantly decreased the amount of exosome secretion in FLO-1 cells (Figure 8A). The combination of ML327 did not enhance the inhibitory effect of GW4869 or DMA. However, interestingly, the addition of TGF-β could partially counteract the effect of the exosome biogenesis inhibitors (Figure 8A). GW4869 is a potent neutral sphingomyelinases inhibitor, 22,23 and DMA has the ability to inhibit H+/Na+ (NHE 1, 2 and 3) and the Na+/Ca2+ channels. 24 Therefore, these results suggest that the regulation of EMT on exosome secretion may be closely related to these pathways. However, GW4869 or DMA could not change the relative content of miR-196-5p miR-21-5p and miR-194-5p in exosomes derived from FLO-1 cells (Figure 8B). In other words, the inhibitor mainly acted on the number of exosomes, while EMT regulates exosome secretion in a complex multi-level way.

Functional analysis of exosome biogenesis inhibitors in FLO-1 cells. A, FLO-1 cells were treated with exosome biogenesis inhibitors GW4869 (10 μM) or DMA (10 μg/ mL), supplemented with TGF-β (5 ng/ mL) or ML327 (5 μM). Exosome concentration of the cell culture supernatant was confirmed by NTA. Relative exosome concentration was normalized to the cell number. B, Quantitative PCR was used to detect the relative content of the 3 miRNAs in exosomes derived from FLO-1 cells treated with GW4869 (10 μM) or DMA (10 μg/ mL) for 24 h. Comparisons among groups were determined by one-way ANOVA with Tukey post-hoc analysis. *P < 0.05 and **P < 0.01.
Discussion
Exosomes are a part of extracellular vesicles (EVS), which also include microvesicles and apoptotic bodies. Microvesicles and apoptotic bodies are produced directly by the budding of the cell membrane. Differently, the formation of exosomes is as follows: after invagination of cell membrane, endosomes are formed through a series of complicated procedures. Endosomes then limit the formation of multiple invagination, resulting in budding inward to form intraluminal vesicle (ILV), and form multivesicular bodies (MVBs), which consequently fuse with the plasma membrane and are secreted outside the cell. 25 Epithelial cells transform from polar epithelioid cells to mesenchymal-like cells during EMT. At present, it is still unclear with respect to the issue of whether the proposed depolarization will have an impact on the fusion of MVBs with plasma membrane, and then affect the secretion of exosomes. In this study, 2 esophageal cancer cell lines of FLO-1 and SK-GT-4 with different migration ability were selected for subsequent exploration (Figure 1). Consequently, it was found that FLO-1 cells with high migration ability had a higher quantity of exosomes simultaneously. While the number of exosomes decreased in FLO-1 cells when EMT inhibitors were used to weaken EMT pathway. Similarly, TGF-β was used to induce the activation of EMT pathway and it resulted in increased number of exosomes released from SK-GT-4 cells. These results undoubtedly support that the activation of EMT pathway may have a positive correlation with the ability of exosomes release in esophageal cancer cells.
It is generally believed at present that exosomes are mainly formed by endosome sorting complex required for transport (ESCRT) dependent and ESCRT independent pathways. 26 During the formation of exosomes, ESCRT recruits ubiquitinated proteins to plasma membrane, which in turn promotes MVB to release its intracellular ILV into the extracellular domain. ESCRT contains 4 different complexes and related accessory proteins. To be specific, ESCRT-0 can recognize and recruit most of the proteins, while ESCRT-Ⅰ and ESCRT-Ⅱ induce bud formation, and ESCRT-Ⅲ is responsible for promoting vesicle separation. 27 In addition, accessory proteins (VTA1, ALIX, VPS4, etc.), VPS4 in particular, promote the continuous separation and rebinding of ESCRT. 28 Clearly, these substances play their respective roles in the formation of exosomes. Furthermore, cells can also generate ILV and MVB by ESCRT independent mechanism, including transmembrane 4 superfamily molecules or heat-shock protein (HSP). Trajkovic et al for the first time confirmed that the formation of exosomes was independent of the mechanism of ESCRT. When ESCRT was knocked down in their experimental model, it was found that exosomes containing proteolipid protein (PLP) could form normally. 29 Kosaka et al applied nSMase inhibitor GW4869 to HEK293 cells, which could reduce the secretion of PLP exosomes, independent of ESCRT but dependent on the synthesis of ceramide. 30 Furthermore, Miller et al observed that the changes of ESCRT components can affect the output of TGF-β signal, alter the sensitivity of cells to ligands in microenvironment, and hence drive EMT. 31 In accordance with the above interpretation, there may be an interaction between ESCRT pathway and EMT, which, however, does not indicate that EMT can regulate the secretion of exosomes through ESCRT. With respect to the above, it is still unclear whether EMT affects the ESCRT dependent or independent pathway of exosome formation, which is worthy of further study.
Our study also indicated that EMT could change not only the number of exosomes, but also their contents. Inhibition of EMT in FLO-1 cells lead to the alteration of the relative content of many miRNAs in exosomes. Generally, EMT pathway involves multiple important transcription factors, including ZEB, SNAI1, TWIST, SLUG, etc. These transcription factors are regulated targetedly by related miRNAs, and regulate the transcription of related miRNAs simultaneously, resulting in a high possibility of forming a feedback regulatory loop. For example, miR-200/ZEB is a classical dual negative regulatory feedback loop. ZEB1/2 can induce EMT by inhibiting various epithelial genes. While miR-200 can target and inhibit ZEB1/2 to promote epithelial differentiation. On the other hand, the common promoter region of miR-200 family members contains highly conserved ZEB binding sites through which ZEB inhibits the transcription of miR-200 family. 32 Similarly, there is a negative feedback loop between miR-203 and SNAI1. 33 In our study, it was found that the inhibition of EMT pathway led to the downregulation of miR-21 at transcription level, suggesting a positive regulation of EMT on miR-21. While the process of EMT of SK-GT-4 cells was promoted through miR-21 in exosomes derived from FLO-1 cells. All the above results suggest a dual positive feedback pathway between EMT and miR-21. However, it shall be noted that not all miRNA differences in exosomes are from the transcriptional regulation of EMT. Findings in our study revealed that EMT displayed none obvious impact on the transcription levels of miR-196-5p and miR-194-5p, suggesting that there may be other pathways for EMT to regulate the relative content of miRNA in exosomes, not only at the cellular transcription level. And interestingly enough, the inhibition of EMT or not exerted no influence in exosome secretion of C. elegans cel-miR-73 transfected into cells artificially. There are many issues to be addressed currently, for example, whether the exosomes of esophageal cancer cells are specific for miRNA sorting, genus-species specific or sequence-specific (if any), and how EMT affects these sorting mechanisms? The preliminary results of our study are insufficient to clearly explain the specific relationship between EMT and miRNA release in exosomes, which requires to be clarified in future.
To sum up, the present study confirms that EMT can regulate exosomes in esophageal cancer cells, which involves the changes of the quantity and contents of exosomes. Findings in our study provide experimental evidence for the interaction of EMT with exosomes secretion and sorting pathway, and also offer a new direction for the research of tumor metastasis in future.
Supplemental Material
Supplemental Material, sj-pdf-1-tct-10.1177_15330338211033077 - EMT Participates in the Regulation of Exosomes Secretion and Function in Esophageal Cancer Cells
Supplemental Material, sj-pdf-1-tct-10.1177_15330338211033077 for EMT Participates in the Regulation of Exosomes Secretion and Function in Esophageal Cancer Cells by Chuangui Chen, Zhao Ma and Hongjing Jiang in Technology in Cancer Research & Treatment
Supplemental Material
Supplemental Material, sj-pdf-2-tct-10.1177_15330338211033077 - EMT Participates in the Regulation of Exosomes Secretion and Function in Esophageal Cancer Cells
Supplemental Material, sj-pdf-2-tct-10.1177_15330338211033077 for EMT Participates in the Regulation of Exosomes Secretion and Function in Esophageal Cancer Cells by Chuangui Chen, Zhao Ma and Hongjing Jiang in Technology in Cancer Research & Treatment
Footnotes
Abbreviations
Authors’ Note
Hongjing Jiang and Chuangui Chen conceived and designed the experiments. Chuangui Chen analyzed and interpreted the results of the experiments, Chuangui Chen and Zhao Ma performed the experiments. All data generated or analyzed during this study are included in this published article. Written informed consents were obtained from all the participants. No animal or human experiments were used in this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Key Clinical Specialist Construction Programs of China (No. 2013-544).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
