Abstract
Purpose:
Vascular invasion (VI) is associated with recurrence and is an indicator of poor prognosis in gastric cancer (GC). Pre-operative identification of VI may guide the selection of the optimal surgical approach and assess the requirement for neoadjuvant therapy.
Methods:
A total of 271 patients were retrospectively collected and randomly allocated into the training and validation datasets. The least absolute shrinkage and selection operator regression model was used to select potentially relevant features, and multivariable logistic regression analysis was used to develop the nomogram.
Results:
The nomogram consisted of pre-operative serum complement C3 levels, duration of symptoms, pre-operative computed tomography stage, abdominal distension and undifferentiated carcinoma. The nomogram provided good calibration for both the training and the validation set, with area under the curve values of 0.792 and 0.774. Decision curve analysis revealed that the nomogram was clinically useful.
Conclusion:
The present study constructed a nomogram for the pre-operative prediction of VI in patients with GC. The nomogram may aid the identification of high-risk patients and aid the optimization of pre-operative decision-making.
Keywords
Introduction
Gastric cancer (GC) is the fifth most common type of cancer and the third leading cause of cancer-associated deaths. It is estimated that there will be 27,600 new GC cases and 11,010 GC mortalities worldwide in 2020. 1 Radical surgery combined with regional lymph nodes dissection is the standard treatment for patients with stage I-III GC. However, 20-30% of patients suffer from local or distant recurrence and succumb to the disease following curative resection. 2,3 In order to improve the prognosis of GC, the application of multimodal therapy including chemotherapy, radiotherapy and surgery has been widely recognized. 4 Among them, neoadjuvant chemotherapy is an important supplement to surgical treatment. Preoperative application of neoadjuvant chemotherapy in advanced GC can effectively control tumor progression, promote tumor volume reduction or clinical stage reduction, enhance the success rate of the surgery and improve the overall survival of patients. 5,6 Despite advances in treatment techniques, postoperative recurrence is still a major problem for patients with GC. Therefore, the identification of patients with GC with a high risk of recurrence and poor prognosis may optimize the management of these patients and allow individualized treatment.
Vascular invasion (VI) occurs when tumor cells invade blood and/or lymphatic vessels near the tumor. 7 VI occurs in the submucosa and subserosa in the form of complete or partial tumor thrombus and vascular wall infiltration. 8 VI is a critical step in tumor cell dissemination and metastasis. It is reported that VI is associated with lymph node metastasis, advanced T stage and poor prognosis. 9 -12 In stage II GC, vascular invasion may serve as an additional independent prognostic predictor and can be used to identify patients with poor prognosis. 13 However, despite its strong prognostic value, VI status can only be assessed post-operatively. The identification of VI pre-operatively may improve pre-treatment decision-making, such as assessing the need for neoadjuvant therapy and the adequacy of surgical resection. While it has been reported that the expression of mucin 4 (MUC4) cell surface associated and the diffuse expression of human ring finger (hRFI) homologous to inhibitor of apoptosis protein related to VI in GC, 14,15 these biomarkers have limited use in clinical practice.
Nomograms have gained popularity in oncology and medicine and can be used to generate individual probabilities of clinical events by integrating different prognostic and deterministic variables. 16 In the nomogram, the prediction factors are quantified in the form of scores, and the probability of patient outcome events is calculated through the conversion function between the total score of each prediction factor and the probability of outcome events. 12,17,18 Nomograms are predictive models that have been widely applied in the field of cancer prognosis prediction, including GC. 19 -23 For example, nomogram for predicting disease-specific survival and recurrence in GC have been developed. 24,25 However, most of these nomograms were based on postoperative parameters, which can not guild neoadjuvant chemotherapy. To the best of our knowledge, a nomogram that can be used for the pre-operative prediction of VI in GC has not yet been described.
The aim of the present study was to develop and validate a nomogram for predicting the presence of VI based on pre-operative clinical features of patients with GC. To the best of our knowledge, the nomogram presented in the present study is the first that can be used for individual pre-operative prediction of VI in patients with GC. Therefore, this nomogram may aid clinicians make better clinical decision and select optimal treatment strategies.
Methods
Patient Selection Criteria
A total of 271 patients with GC who received surgical treatment in the Guangxi Medical University Cancer Hospital between October 2013 and May 2018 were retrospectively collected in the present study. The inclusion criteria were as follows: (i) Patients whose pathological diagnosis was gastric adenocarcinoma; (ii) patients who received resection of the primary tumor; (iii) a post-operative pathological report that suggested the presence of VI; and (iv) patients whose medical history and auxiliary examination results were complete and included computed tomography (CT), electronic gastroscopy and blood tests. The exclusion criteria were as follows: (i) Patients who received any pre-operative therapy (including radiotherapy, chemotherapy or chemoradiotherapy); and (ii) patients with other types of tumor in the same period. The protocol of this retrospective study was approved by the Ethics and Human Subject Committee of Guangxi Medical University Cancer Hospital (approval no. LW2019041), and all experiments and methods met the standards of the relevant guidelines and regulations. 26
The relevant demographic and pre-operative clinical features were collected. These features were divided into 8 types, including general information, symptom, previous history, family history, personal history, blood test results, imaging examination and pathological examination. General information, which were based on the patients’ identity cards, were extracted from the hospital database. Previous, personal and family history were extracted from the hospital admissions records, which were verified by a senior physician. Blood test results were collected from tests performed on the first admission. Imaging examination was based on patients’ pre-operative CT scans, which were read by 2 senior radiologists. Pathological examination included patients’ pre-operative endoscopic biopsies and the presence of VI on the post-operative pathological report. The 271 patients were randomly divided into the training and validation datasets at a ratio of 7:3 using computer-generated random numbers. The training dataset included 191 cases and the validation dataset included 80 cases. 27 The T and N stages were identified in accordance with the eighth Edition of the American Joint Committee on Cancer Staging Manual.
Feature Selection
The least absolute shrinkage and selection operator (LASSO) is a penalized regression method that estimates the regression coefficients by maximizing the log-likelihood function, while limiting the sum of the absolute values of the regression coefficients. The regression coefficients estimated by LASSO are sparse, and many components are exactly zero. Therefore, LASSO automatically deletes unnecessary covariates. 28,29 LASSO logistic regression algorithm is advantageous in high dimension regression. 30 LASSO was used to identify the predictive features in the training dataset, as previously described. 18 All the categorical variables were converted into dummy variables and the absence or presence of VI was considered to be a dependent variable. 10-fold cross validation was used to confirm the suitable tuning parameter (λ) for LASSO logistic regression. The most significant features selected by LASSO were subsequently used to construct the logistic regression model.
Constructing the Nomogram and Performance Assessment
The nomogram was constructed based on features with P-values less than 0.1 in the multivariate regression analysis. The goodness of fit between the observed and predicted values was evaluated by a calibration curve and Spiegelhalter’s Z-test. 31 A calibration curve that would perfectly fit the 45° reference line was plotted. The receiver operating characteristic (ROC) curve and the area under the curve (AUC) were used to evaluate the predictive discrimination of the nomogram. 32 An AUC of 1.0 was considered to indicate that the nomogram had perfect discrimination ability.
Validation of the Nomogram
The validation dataset was used to evaluate the performance of the nomogram. A predicted value was calculated for every patient in the validation dataset on the basis of the formula constructed using the training dataset. The ROC and AUC were used to assess the predictive discrimination ability of the nomogram in the validation dataset. The calibration curve and Spiegelhalter’s Z-test were used to evaluate the goodness of fit of the nomogram in the validation dataset. Additionally, 10-fold cross-validation was performed and mean-square error was calculated to assess the quality of the nomogram using the boot package (https://CRAN.R-project.org/package=boot) in R (Version: 3.4.0; https://cran.r-project.org/bin/windows/base/old/3.4.0/).
Clinical Use
The clinical significance of the nomogram was analyzed using decision curve analysis (DCA) by quantifying the net benefits at different threshold probabilities in the combined training and validation datasets, as previously described. 33 The treat-none scheme assumed no patient had a disease and the treat-all-patients scheme assumed all patients had a disease. 27
Statistical Analysis
The statistical analyses were performed using R statistical software version 3.4.0. LASSO logistic regression analysis was performed using the glmnet package (Version: 2.0-18; https://CRAN.R-project.org/package=glmnet). The rms package (Version: 5.1-3.1; https://CRAN.R-project.org/package=rms) was used to perform logistic regression analysis, to construct the nomogram and to plot the calibration curve. Spiegelhalter’s Z-test was performed using the val.prob function in the rms package. The DCA was performed using the dca.R function. 33 A P value less than 0.05 was considered to indicate a statistically significant difference.
Results
Clinical Characteristics
A total of 271 patients with complete clinical information were randomly divided into training dataset (n = 191) and the validation dataset (n = 80). The present study included 173 male patients and 98 female patients. The mean age of the patients was 56.20 years. The majority of the patients had stage II-III GC (206/271 cases; 76.02%) based on pre-operative CT scans. Additionally, stage IV patients were also enrolled in this study (7/271 cases; 2.58%). Although the determination of VI by the nomogram could not change the treatment strategies of stage IV patients, it could predict the prognosis of stage IV patients and guide clinicians to carry out further close follow-up and observation. Approximately half of the patents had VI confirmed by post-operative pathological examination (123/271; 45.39%). Patient demographical and clinical features are presented in Table 1. There were no significant differences in age, gender, pre-operative serum complement C3 levels, duration of symptoms, stage, abdominal distension and undifferentiated carcinoma between patients in the training and validation datasets (all P > 0.05).
Patient Background Characteristics.

Feature Selection
LASSO logistic regression analysis was used to construct a prediction model in the training dataset. The absence or presence of VI was a dependent variable and the remaining clinical features were independent variable in the LASSO logistic regression analysis. Using cross-validation, the most appropriate tuning parameter λ for LASSO logistic regression analysis was determined to be 0.060 when the binomial deviance was the smallest (Figure 1A). The 5 features with non-zero coefficients in the tuning parameter, including pre-operative serum complement C3 levels, duration of symptoms, stage (based on CT), abdominal distension and undifferentiated carcinoma, were selected and used to construct the regression model (Figure 1B).

Feature selection by LASSO logistic regression. (A) Tuning parameter (λ) selection in the LASSO logistic regression used 10-fold-cross-validation via minimum criteria. The binomial deviance is plotted versus log (λ). The black vertical lines are plotted at the optimal λ based on the minimum criteria and 1 standard error for the minimum criteria. (B) LASSO coefficient profiles for clinical features. Each coefficient profile plot was produced against the log (λ) sequence.
Performance Assessment of the Nomogram
The results of feature selection were used to construct a multivariate logistic regression model incorporating the 5 selected features using the training dataset. Features with P-values less than 0.1 in the multivariate regression analysis were used for nomogram construction. As shown in Table 2, among the 5 features incorporated in the multivariate logistic regression model, duration of symptoms (P = 0.024), abdominal distension (0.038), stage II (P = 6.78×10-7) and stage III (P = 0.0081) were independent risk factors of VI in GC. Patients symptoms lasting <1 month had a 0.44-fold risk of developing VI. Patients with abdominal distension had a 2.30-fold risk of developing VI. Stage was also an independent predictor for VI in GC. While pre-operative serum complement C3 levels and undifferentiated carcinoma were not independent predictors for VI (both of them have a marginally significant P-value), these 2 parameters were still included in the predictive model due to the scope of the present study, which was to develop a nomogram for pre-operative prediction of VI, rather than analyzing risk factors of VI.
Multivariable Logistic Regression Analysis of the Selected Clinical Features in the Training Set.

Abbreviations: CT, computed tomography; CI, confidence interval.
A scale was added at the top of the nomogram and was used to represent the points allocated to each variable. Each patient received a total number of points that could be converted into the prediction of the statistical probability of the presence of VI using the scale at the bottom of the nomogram (Figure 2). For example, a patient with symptoms persisting for >1 month (25 points), a serum C3 level of 0.6 g/l (52 points), stage III GC (77 points), abdominal distension (24 points) and non-undifferentiated carcinoma (0 points) had a total of 178 points and a VI probability of ∼55%.The calibration curve of the nomogram suggested that there was a good concordance between the statistically predicted probability of GC VI and the actual probability in the training dataset. The Spiegelhalter’s Z-test yielded a P-value of 0.92, which indicated that there was no significant departure from a perfect fit (Figure 3A). The ROC curve indicated that the nomogram provided good discrimination, with an AUC of 0.792 in the training dataset (Figure 3B).

Nomogram for predicting vascular invasion prior to surgery in gastric cancer (GC). Five points are allocated for preoperative serum complement C3 levels, duration of symptoms, preoperative computed tomography (CT) stage, abdominal distension and undifferentiated carcinoma. Draw a line on the corresponding value to the “points” line. Calculate the sum of these 3 points and getting the mark on the “Total points,” which is related to possibility of vascular invasion.
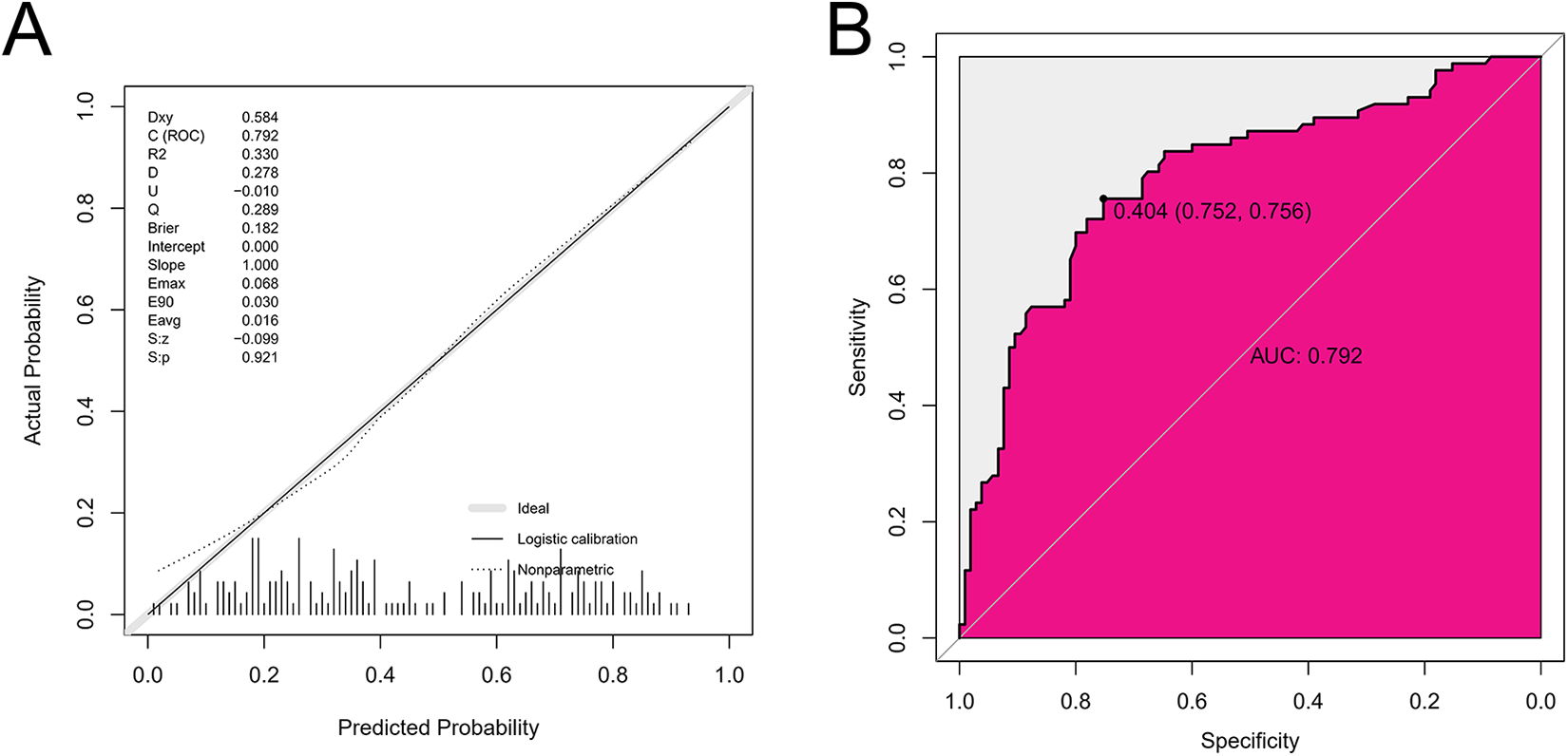
Performance evaluation for the nomogram in training dataset. (A) The x-axis is the nomogram-predicted probability of vascular invasion and the y-axis is the actual rate of vascular invasion. The reference line is 45° and indicates perfect calibration. (B) The ROC curves for the nomogram in training dataset.
Validation of the Nomogram
The nomogram was validated using the validation dataset by calculating the individual possibility of VI. The calibration curve revealed good concordance between the predicted and actual probabilities in the validation dataset (P = 0.50; Figure 4A). In addition, the nomogram provided an AUC of 0.77 in the validation set, which suggested a good discrimination ability (Figure 4B). Additionally, 10-fold cross-validation was performed 10 times to assess the nomogram. As shown in (Figure 4C), the mean-squares errors ranged between 0.255 and 0.263, indicating that the nomogram was robust.

Performance evaluation for the nomogram in validation dataset. (A) The x-axis is the nomogram-predicted probability of vascular invasion and the y-axis is the actual rate of vascular invasion. The reference line is 45° and indicates perfect calibration. (B) The ROC curves for the nomogram in validation dataset. (C) The 10-fold cross-validation of the nomogram. Shown are the mean-square errors in 10 times 10-fold cross-validation.
Clinical Utility of the Nomogram
DCA was performed to examine the clinical utility of the nomogram. As shown in Figure 5, when the probability of GC VI generated by the nomogram ranged between 25 and 80%, the application of the nomogram to predict the risk of VI added more benefit than both the treat-all-patients scheme and the treat-none scheme. For example, with a probability of VI of 45%, the nomogram added a net benefit of 21.7% compared with the other 2 schemes. This suggested that the nomogram developed in the present study had a high clinical utility.
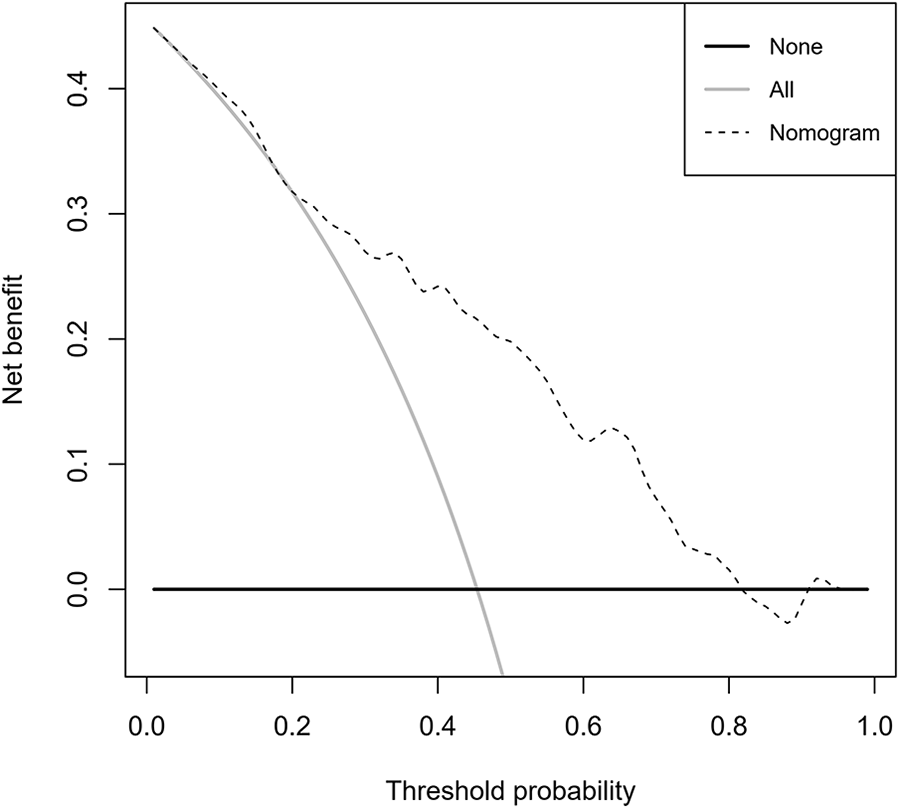
DCA curve of the nomogram. The threshold possibility was indicated by nomogram the represented by the dotted line. The gray line corresponds to the treat-all-patients and the black one represents the treat-none scheme.
Discussion
The present study constructed and validated a novel nomogram using data collected in clinical practice that could be used to predict pre-operative VI in patients with GC. The nomogram provided favorable discrimination and calibration values and may enhance the pre-operative management of patients with GC.
Recently, the importance of VI in GC has increasingly been recognized, but the specific molecular mechanism of VI has not been clearly explored. Several studies have found that MUC4 is associated with VI in a variety of cancers. 34 -36 Tamura et al. 14 reported that MUC4 expression was associated with VI. Another study showed that inhibiting the expression of MUC4 could reduce the invasive ability of GC cells. 37 Furthermore, the expression of MUC4 is also associated with GC recurrence and metastasis. In addition, the expression of hRFI in GC plays an important role in VI. Sasaki et al. 15 found that the diffuse expression of hRFI was significantly related to VI of GC, and patients with diffuse expression of hRFI were more likely to develop liver metastasis. Although some studies have been conducted, the molecular mechanism of VI has not been fully revealed. Therefore, it is a great challenge to find molecular markers that can accurately predict VI in GC and apply it to the clinic. Moreover, preoperative identification of VI requires endoscopic sampling and biopsy, which is invasive and causes harm to GC patients. Considering this, the nomogram for predicting VI created in this study is noninvasive, which can avoid the physical injury caused by biopsy to GC patients and facilitate clinicians to apply prediction of VI.
VI is a risk factor for the recurrence of GC. Evidence suggests that VI is associated with lymph node metastasis, advanced T stage, recurrence and poor prognosis. In order to reduce recurrence and extend disease-free survival, GC patients with T3-4/lymph node-positive generally receive adjuvant chemotherapy following curative surgery. 38 However, ∼20-60% patients who undergo radical surgical tumor dissection and receive adjuvant chemotherapy still experience recurrence or new occurrence of GC. Several reasons may account for treatment failure. Firstly, post-operative chemotherapy may be delayed for months after the initial diagnosis and cancer may progress during this period. 39 Secondly, surgery increases the production of growth factors and induces immunosuppression, which may facilitate the dissemination and metastasis of tumor cells. 40 Neoadjuvant chemotherapy has attracted increasing attention for its advantages in downstaging the primary tumor, facilitating complete surgical resection and treating systemic micrometastases. 41 Clinical trials have revealed that neoadjuvant chemotherapy improves overall survival time in patients with advanced GC. 42 -45 Hence, the National Comprehensive Cancer Network Guidelines recommend that patients with GC with T2 or higher stages receive neoadjuvant chemotherapy. 46 However, in Southeast Asia, neoadjuvant chemotherapy is recommended for patients with local advanced GC and no evidence of distant metastasis (T3/4, N+) or cIII. In China, patients with T2 GC who underwent radical surgical tumor resection had a 23% risk of recurrence. 47 Patients with a high risk of recurrence may benefit from neoadjuvant chemotherapy. 41 As VI may predict GC recurrence, a previous study suggested that VI should be included in risk stratification and selection criteria for patients entering novel adjuvant or neoadjuvant clinical trials. 48 However, VI status can only be assessed in the post-operative setting. In order to overcome this limitation, the present study developed a nomogram for the pre-operative identification of VI, which provides a novel strategy for identifying high-risk patients who may benefit from neoadjuvant chemotherapy.
In order to develop a pre-operative nomogram for VI prediction in patients with GC, 5 predictors, including pre-operative serum complement C3 levels, duration of symptoms, pre-operative stage, abdominal distension and undifferentiated carcinoma, were selected using LASSO logistic regression analysis, which is an efficient features selection statistical method for high dimensional data. 49 The discrimination and calibration were favorable in the training cohort (AUC of 0.795; and a P-value of Spiegelhalter’s Z-test of 0.81) and the validation cohort (AUC of 0.787; and a P-value of Spiegelhalter’s Z-test of 0.66). As favorable discrimination and calibration alone could not justify the clinical utility of the nomogram, DCA was performed. The DCA curve demonstrated that if the threshold probability of VI estimated by the nomogram was 25 and 80%, the nomogram provided greater benefit than either the treat-all-patients scheme or the treat-none scheme. Hence, the pre-operative nomogram could serve as a user-friendly predictor of VI in GC.
Among the 5 features selected in the present study, duration of symptoms, abdominal distension and CT stage was independent factors for VI in GC. VI is one of the earliest steps in tumor cell dissemination. 11 It was reported that ∼85% patients with N1-2 GC were VI-positive. 50 However, ∼30% of patients without lymph node metastasis were VI-positive. VI was significantly associated with T-status and N-status, 50 which was in line with the results obtained in the present study. CT is a non-invasive diagnostic test that is recommended as a standard examination in the diagnosis of GC. 51 The accuracy of CT for the evaluation of the T-stage and N-stage in GC is ∼80-89% and 63%, respectively. 52 Therefore, the use of CT to predict VI is convenient and accurate. The present study revealed that symptoms persisting <1 month conferred protection against VI. As VI is associated with TNM stage, 53 the probability of VI may increase with increasing duration of GC. The present study revealed that patients with abdominal distension had an increased risk of developing VI. However, the mechanisms underlying this remain unclear and future investigation is required.
The present study had several limitations. Firstly, the main limitation of this study is that the training set and validation set are from a single center, which can lead to potential selection bias. And an external validation with multi-center and larger samples may the optimal choice. Secondly, although a genome classifier is a promising predictor, no application of genome features has been considered. Thirdly, this study is a retrospective small sample clinical study. Further studies with large sample are needed to validate the results of this study.
Conclusions
To the best of our knowledge, the present study was the first to construct a nomogram that could be used as a pre-operative prediction tool for VI in patients with GC. The nomogram may provide novel strategy for the identification of high-risk patients and aid the optimization of pre-operative decision-making.
Supplemental Material
Supplemental Material, sj-jpg-1-tct-10.1177_15330338211004924 - A Novel Nomogram for Individually Predicting of Vascular Invasion in Gastric Cancer
Supplemental Material, sj-jpg-1-tct-10.1177_15330338211004924 for A Novel Nomogram for Individually Predicting of Vascular Invasion in Gastric Cancer by Yongsheng Meng, Xiaoliang Huang, Jungang Liu, Jianhong Chen, Zhaoting Bu, Guo Wu, Weishun Xie, Franco Jeen, Lingxu Huang, Chao Tian, Xianwei Mo and Weizhong Tang in Technology in Cancer Research & Treatment
Supplemental Material
Supplemental Material, sj-jpg-2-tct-10.1177_15330338211004924 - A Novel Nomogram for Individually Predicting of Vascular Invasion in Gastric Cancer
Supplemental Material, sj-jpg-2-tct-10.1177_15330338211004924 for A Novel Nomogram for Individually Predicting of Vascular Invasion in Gastric Cancer by Yongsheng Meng, Xiaoliang Huang, Jungang Liu, Jianhong Chen, Zhaoting Bu, Guo Wu, Weishun Xie, Franco Jeen, Lingxu Huang, Chao Tian, Xianwei Mo and Weizhong Tang in Technology in Cancer Research & Treatment
Supplemental Material
Supplemental Material, sj-jpg-3-tct-10.1177_15330338211004924 - A Novel Nomogram for Individually Predicting of Vascular Invasion in Gastric Cancer
Supplemental Material, sj-jpg-3-tct-10.1177_15330338211004924 for A Novel Nomogram for Individually Predicting of Vascular Invasion in Gastric Cancer by Yongsheng Meng, Xiaoliang Huang, Jungang Liu, Jianhong Chen, Zhaoting Bu, Guo Wu, Weishun Xie, Franco Jeen, Lingxu Huang, Chao Tian, Xianwei Mo and Weizhong Tang in Technology in Cancer Research & Treatment
Footnotes
Authors’ Note
Yongsheng Meng, Xiaoliang Huang, and Jungang Liu contributed equally to this work. The protocol of this retrospective study was approved by the Ethics and Human Subject Committee of Guangxi Medical University Cancer Hospital (approval no. LW2019041), and all experiments and methods met the standards of the relevant guidelines and regulations. Written informed consents were obtained from patients.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the 2019 Guangxi University High-Level Innovation Team and the Project of Outstanding Scholars Program, and Guangxi Science and Technology Project (2019AC03004); Guangxi Clinical Research Center for Colorectal Cancer (Guike: AD19245197); Research Basic Ability Improvement Project for Guangxi Young College Teachers (2021KY0087); Innovation Project of Guangxi Graduate Education. (YCSW2021133).
Supplemental Material
Supplemental material for this article is available online.
Abbreviations
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
