Abstract
Introduction:
Neoadjuvant chemoradiotherapy (nCRT) followed by operation has become the standard treatment for locally advanced rectal cancer (LARC). However, considering the possible toxicity and complications of radiochemotherapy, nCRT is seldom used for the elderly. The purpose of this study was to assess the safety and long-term effect of nCRT combined with TME in elderly patients with LARC.
Method:
Four-hundred-fourteen LARC patients were divided into 2 groups: 108 patients were in the elderly group (≥ 65 years old) and 306 patients were in the non-elderly group (<65 years old). The side effects, toxicity, complications, disease-free survival (DFS), and overall survival (OS) of all of the patients were assessed.
Results:
The data comprised 103 patients in the elderly group and 292 patients in the non-elderly group who completed nCRT sessions following operation. The treatment-completion rates of the elderly and non-elderly groups were 95.37% and 95.42%, respectively. Twenty-two patients developed radiotherapy complications (grade III) in the elderly group and 37 such cases developed in the non-elderly group. Diarrhea, skinulcer, and perianal pain were ranked as the top 3 most common complications. The incidence of infection, anastomotic leakage, and intestinal obstruction was 0.97% in the elderly group. The 5-year DFS and 5-year OS rate were 70.7% and 80.8% in the elderly group, 67.3% and 81.6% in the non-elderly group respectively.
Conclusions:
nCRT are safe and effective for elderly patients, and it does not increase the risk of postoperative complications for the elderly. Hence, nCRT should not be withheld based on age alone.
What Does This Paper Add to the Literature?
This study evaluated whether neoadjuvant chemoradiotherapy should be used in elderly patients with locally advanced rectal cancer safety. We have proved that elderly people (≥ 65 years) would not have more complications when they were underwent neoadjuvant treatment, and their DFS and OS are almost the same with non-elderly patients.
Introduction
The morbidity of rectal cancer is 13.67 per 100,000 in China, and most of the patients have been found to be in the advanced stages. 1 Several studies have shown that the median age for diagnosis of colorectal cancer is 69–72 years old, and about 60–70% of the patients are over 65 years old. 2 -4 Neoadjuvant chemoradiotherapy (nCRT) combined with total mesorectal excision (TME) has become the standard treatment for locally advanced rectal cancer (LARC). 5 Several large randomized controlled studies have shown that neoadjuvant therapy combined with TME for LARC patients can not only improve the local control rate of tumors, but also reduce the local-recurrence rate. 6 -11 However, in clinical practice, clinicians do not usually recommend neoadjuvant therapy for elderly LARC patients over 65 years old, and instead they recommend operation directly. 12 One of the reasons is that elderly patients often have other chronic diseases, such as diabetes, hypertension, and cardiovascular diseases. 13,14 In addition, considering the toxicity and side effects of nCRT, most physicians believe that this may lead to a poor tolerance of elderly patients to neoadjuvant therapy, and they may become unable to complete neoadjuvant therapy or incur delayed treatment. 15,16 Therefore, there are few studies on the safety and cancer outcomes of neoadjuvant therapy in elderly LARC patients. The lack of research evidence makes clinicians reluctant to choose neoadjuvant therapy for elderly LARC patients. The purpose of this retrospective study was to observe the safety and cancer outcomes of neoadjuvant therapy in elderly patients with LARC.
Patients and Methods
Patient Population
This study retrospectively analyzed 414 rectal cancer patients who received nCRT from August 1, 2010 to July 31, 2018 in the Cancer Hospital of the Chinese Academy of Medical Sciences. There were 278 males and 136 females aged 23 to 82 years. They were divided into the elderly group (≥ 65 years old) and non-elderly group (<65 years old). This grouping yielded 108 cases in the elderly group and 306 cases in the non-elderly group.
Inclusion and Exclusion Criteria
Inclusion criteria for patients were as follows: (i) All of the patients received endoscopy at admission and were diagnosed as having rectal adenocarcinoma by biopsy; (ii) All of the patients underwent chest, abdominal CT, and pelvic MR examination at admission and before operation. The imaging diagnosis was T3-4/N+.
Exclusion criteria for patients were as follows: (i) multiple primary colorectal cancer; (ii) suffering from or have suffered from malignant tumors in other parts of the body; (iii) patients undergoing short-range radiotherapy (a total of 25 Gy radiation in 5 fractions); (iv) patients who achieved clinical complete remission (cCR) after nCRT, and were treated with “watch and wait;” and (v) patients who did not undergo surgical treatment in our center after neoadjuvant therapy.
Treatment Protocol
In brief, a total dose of 45-50 Gy (1.8-2.0 Gy/fraction) to the whole pelvis in 25 fractions for 5 weeks, plus 5.4 to 9 Gy (1.8 Gy/fraction) to the tumor volume, with 6 to 15 MV energy photons. 17 There were 3 schemes of concurrent chemotherapy as follows: (i) capecitabine 1650 mg/m2 daily concurrent with radiotherapy; (ii) oxaliplatin combined with capecitabine (capecitabine 1650 mg/m2, oxaliplatin 130 mg/m2/w). All of the patients received TME surgery for at least 4 weeks after nCRT treatment. 18
Toxicity and Postoperative Complications
Evaluation of toxicity during nCRT was according to Common Terminology Criteria for Adverse Events Version No. 3 recommendations. 19 Postoperative complications and mortality were defined as those occurring within the 30 days following surgery.
Follow-Up
All of the patients received postoperative reviews every 3 months at our center or local hospital within 2 years, and every 6 months for 3 to 5 years after operation. The postoperative-review examinations included physical examination, peripheral blood tumor markers (e.g., CEA, CA19-9), fecal occult blood, chest and abdominal CT, pelvic CT or MRI, and whole-body PET-CT if necessary. Tumor metastasis or recurrence was defined by imaging results or histopathological diagnosis.
Statistical Analysis
SPSS software (IBM, v.19.0, IBM SPSS Statistics, IBM Corporation, Armonk, NY) was used to test all of the data. We used chi-square tests for counting data and 2 independent samples t tests for comparisons between 2 groups, which was in accordance with the homogeneity of normal distribution and variance. Non-parametric Mann-Whitney U tests were used to compare the measurement data between the 2 groups that were not in accordance with the homogeneity of normal distribution and variance. The data obtained have been expressed in the form of mean ± standard deviation, with significant differences between the 2 groups being established at P < 0.05. Disease-free survival (DFS) and overall survival (OS) were analyzed by the Kaplan-Meier method. OS was defined as the time between the date of surgery and the date of death from any cause or the last follow-up. A P-value of < 0.05 was considered to be statistically significant.
Results
Patient Characteristics
In this study, 414 patients were enrolled, but 19 patients dropped out during the treatment. A total of 395 cases completed the whole course of neoadjuvant therapy combined with TME surgery. The completion rate of nCRT was 95.37% (103/108) in the elderly group and 95.42% (292/306) in the non-elderly group. These patients and tumor characteristics are listed in Table 1. There were 77 (19.49%) of 395 patients achieved pCR after treatment, of which 23 (22.3%) patients in the elderly group and 54 (18.5%) patients in the non-elderly group. The pCR rate in the elderly group was significantly higher than that in the non-elderly group (F = 41.238, P < 0.001).
Demographic and Clinical Features for All Patients.
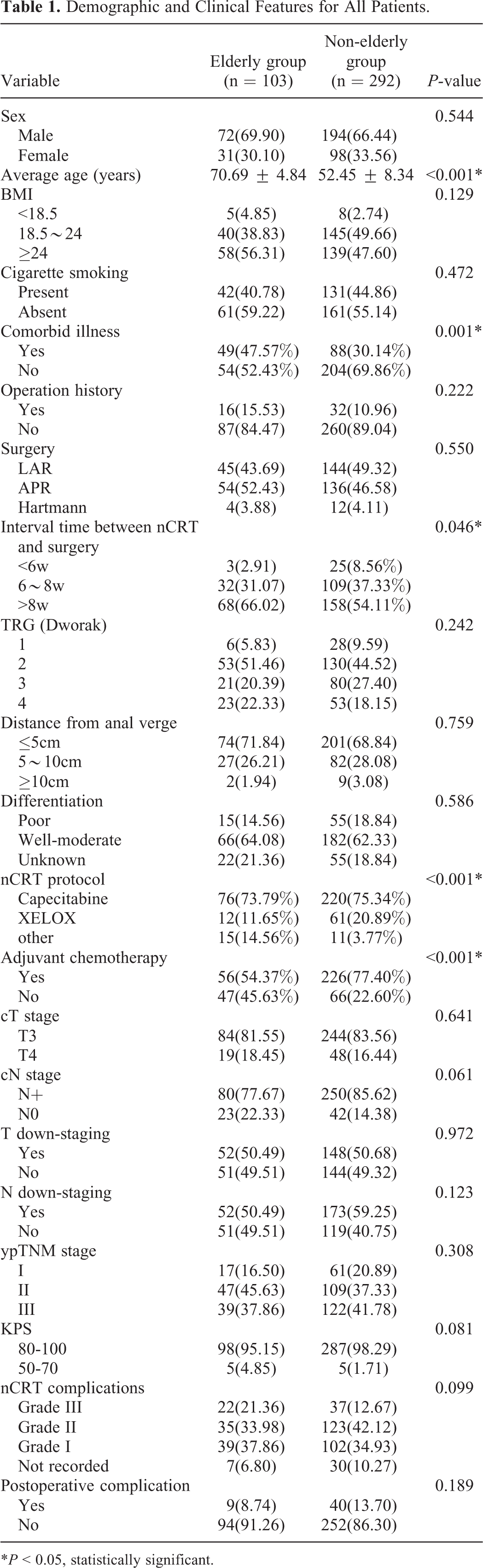
*P < 0.05, statistically significant.
Toxicity
No grade-IV or -V toxic reactions were found. There were 8 kinds of grade III toxic reactions in 60 patients, including 21 cases in the elderly group and 39 cases in the non-elderly group. In the elderly group, 10 patients had grade III diarrhea/constipation, 7 patients had grade III radiation dermatitis, 2 patients had grade III nausea/vomiting and abdominal discomfort, 1 patient had grade III hematochezia/anal distention and other local rectal reactions, 1 patient had myelosuppression and 1 patient had asthenia. Among them, 1 patient had grade III diarrhea and asthenia at the same time, 1 patient had grade III diarrhea and abdominal discomfort at the same time, 1 patient had grade III diarrhea and radiation dermatitis at the same time. In the non-elderly group, 15 patients had grade III radiation dermatitis, 14 patients had diarrhea/constipation, 6 patients had grade III rectal pain, 5 patients had grade III myelosuppression, 2 patients had grade III nausea/vomiting and 2 patients had abdominal discomfort. Among them, 4 patients had grade III diarrhea and radiation dermatitis at the same time. These acute toxicities are listed in Table 2.
Acute Toxicity in Rectal Cancer Patients Treated With Neoadjuvant Chemoradiotherapy.

*P < 0.05, statistically significant.
Postoperative Complications
A total of 49 patients had complications within 30 days after the operation, of which 40 cases were in the non-elderly group and 9 were in the elderly group. Among all of the complications, there were 19 cases of infection (perineal infection in 10 cases, pelvic infection in 5 cases, pulmonary and urinary-tract infection in 2 case, fever of unknown origin in 1 case, and systemic inflammatory response syndrome in 1 patient), 11 cases of leakage (anastomotic leakage in 9 cases, ureteral leakage in 1 case, small intestinal leakage in 1 case), and 11 cases of intestinal obstruction. Additionally, 5 patients had urinary retention, 3 patients discharged without removal of the drainage tube, 3 patients suffered from abdominal bleeding after operation, and 3 patients had necrosis or severe edema of the intestinal tube at stoma. One patient had poor self-control of defecation after operation. It is worth noting that the first 3 complications (infection, leakage, and intestinal obstruction) with the highest incidence rate did not show statistical differences between the 2 groups. The incidence of infection was 3.88% (4/103) in the elderly group and 4.79% (14/292) in the non-elderly group (P = 0.703). The incidence of intestinal obstruction was 0.97% (1/103) in the elderly group and 3.42% (10/292) in the non-elderly group (P = 0.193). The details are presented in Table 3.
Postoperative Complications in Rectal Cancer Patients Treated With Neoadjuvant Chemoradiotherapy.

Disease-Free Survival Rate and Overall Survival Rate
Next, 395 patients who completed nCRT combined with TME were followed up in terms of their progress. The 5-year DFS rate was 70.7% in the elderly group and 67.3% in the non-elderly group. The 5-year OS rates were 80.8% and 81.6% in the elderly group and non-elderly group, respectively. It was found that there was no significant difference in DFS (P = 0.837) and OS (P = 0.608) between the 2 groups (Figure 1).

Kaplan–Meier curves showed no significantly disease-free survival (A) and overall survival (B) difference on 2 subgroups in accordance with age.
Discussion
Preoperative neoadjuvant chemoradiotherapy combined with total mesorectal excision has become the standard method for the treatment of locally advanced rectal cancer. However, clinicians are reluctant to provide nCRT for elderly patients (≥ 65 years old) with LARC because of the possible toxicity and intolerance of radiotherapy and chemotherapy. In the past 2 years, only 16.92% of elderly patients have received nCRT. At present, there are few studies on the effect of nCRT on the elderly, and the age limit for elderly patients is inconsistent. 20 -22 In the absence of sufficient clinical evidence, clinicians also lack the courage to carry out nCRT for the elderly. Although nCRT cannot improve the OS rate of patients, it does improve the local-control rate of tumors, reduces the tumors stage, and improves the R0-resection rate. The significance of the present study is that it will add to the “marked paucity of published data in this area” to guide management of elderly patients. 23
The results showed no significant difference on the nCRT completion rate between elderly patients and non-elderly patients. Nearly half of the elderly patients were complicated with chronic diseases, but it did not affect the nCRT-completion rate in the elderly group. There were no grade-IV or -V complications in the elderly group during nCRT. Although about 1 in 5 patients in the elderly group had grade III complications and this data was slightly higher than that in the non-elderly group, the incidence of grade-II complications in the elderly group was slightly lower than that in the non-elderly group. Although the incidence of anorexia in the elderly group is significantly higher than that in the non-elderly group, they are all grade I-II, with mild symptoms, which can be relieved after symptomatic treatment. In addition, there was no significant difference in the incidence of various complications between the 2 groups (P > 0.05).This is similar to previous reports. 24 The results of our present study show that elderly patients tolerated neoadjuvant CRT in nearly equivalent terms to younger patients.
It is worth noting that the incidence of postoperative complications in the elderly group is slightly lower than that in the non-elderly group, but no statistical difference was found. Moreover, there was no statistical difference in the incidence of infection, leakage, and intestinal obstruction, which were the main complications after operation between the 2 groups. This suggests that elderly LARC patients can well tolerate nCRT and that their age will not increase the incidence of complications of neoadjuvant therapy and post-operative complications.
The present study found that there were no significant difference in 5-year DFS and OS rate between the elderly group and the non-elderly group respectively. This is inconsistent with some existing research results, which may be due to inconsistencies in the age requirements of the “elderly patients” included in various studies and it may also be due to the fact that the number of elderly patients included in various studies is very small and there may be an inherent bias. 22,25 -28 Interestingly, in this study, there was almost no significant difference in the general data and tumor characteristics between the 2 groups, but the proportion of elderly patients who did not receive adjuvant chemotherapy after surgery was almost twice that of non-elderly patients. The main reason might be that the patients were reluctant to accept postoperative chemotherapy subjectively. Even so, there was no difference in DFS and OS between the elderly group and the non-elderly group. In order to elucidate how postoperative chemotherapy impact on prognosis, which might lead to shortened the survival time in non-elderly group, we further analyzed the data and found that no significant difference on OS (χ2 = 3.009, P = 0.083) and DFS (χ2 = 0.120, P = 0.730) (Figure S1). While the OS in the elderly group has no concern with that whether they received adjuvant chemotherapy (χ2 = 0.989, P = 0.320), and the DFS in elderly patients receiving adjuvant chemotherapy was significantly lower than that of patients without adjuvant chemotherapy (χ2 = 6.082, P = 0.014) (Figure S2). So whether adjuvant chemotherapy after the operation is meaningful to elderly patients is an issue that merits further discussion.
Our study has several limitations. First, it was a single-center retrospective analysis, and the number of cases in the elderly group was relatively small. There were only 56 elderly people over 70 years old, while only 23 patients were over 75 years old. The reason may be due to clinical scruples about nCRT for elderly patients, which also illustrates the importance of relevant research from another perspective. Second, some high-risk patients and patients with multiple comorbidities tend to transfer to general hospitals after nCRT completion.
The results of our study indicate that nCRT is safe and effective for the elderly, and that it does not increase the postoperative complications of the elderly. Hence, nCRT should not be refused for patients based on age alone. To achieve more accurate insights, more trials focused on treating elderly patients with cancer should be performed.
Supplemental Material
Supplemental Material, Fig_S1_DFS - Safety and Long-Term Effect Assessment of Neoadjuvant Chemoradiotherapy for Elderly Patients With Locally Advanced Rectal Cancer: A CHN Single-Center Retrospective Study
Supplemental Material, Fig_S1_DFS for Safety and Long-Term Effect Assessment of Neoadjuvant Chemoradiotherapy for Elderly Patients With Locally Advanced Rectal Cancer: A CHN Single-Center Retrospective Study by Hengchang Liu, Chunxiang Li, Zhixun Zhao, Xu Guan, Ming Yang, Zheng Liu, Yuan Tang, Zheng Jiang and Xishan Wang in Technology in Cancer Research & Treatment
Supplemental Material
Supplemental Material, Fig_S1_OS - Safety and Long-Term Effect Assessment of Neoadjuvant Chemoradiotherapy for Elderly Patients With Locally Advanced Rectal Cancer: A CHN Single-Center Retrospective Study
Supplemental Material, Fig_S1_OS for Safety and Long-Term Effect Assessment of Neoadjuvant Chemoradiotherapy for Elderly Patients With Locally Advanced Rectal Cancer: A CHN Single-Center Retrospective Study by Hengchang Liu, Chunxiang Li, Zhixun Zhao, Xu Guan, Ming Yang, Zheng Liu, Yuan Tang, Zheng Jiang and Xishan Wang in Technology in Cancer Research & Treatment
Supplemental Material
Supplemental Material, Fig_S2A - Safety and Long-Term Effect Assessment of Neoadjuvant Chemoradiotherapy for Elderly Patients With Locally Advanced Rectal Cancer: A CHN Single-Center Retrospective Study
Supplemental Material, Fig_S2A for Safety and Long-Term Effect Assessment of Neoadjuvant Chemoradiotherapy for Elderly Patients With Locally Advanced Rectal Cancer: A CHN Single-Center Retrospective Study by Hengchang Liu, Chunxiang Li, Zhixun Zhao, Xu Guan, Ming Yang, Zheng Liu, Yuan Tang, Zheng Jiang and Xishan Wang in Technology in Cancer Research & Treatment
Supplemental Material
Supplemental Material, Fig_S2B - Safety and Long-Term Effect Assessment of Neoadjuvant Chemoradiotherapy for Elderly Patients With Locally Advanced Rectal Cancer: A CHN Single-Center Retrospective Study
Supplemental Material, Fig_S2B for Safety and Long-Term Effect Assessment of Neoadjuvant Chemoradiotherapy for Elderly Patients With Locally Advanced Rectal Cancer: A CHN Single-Center Retrospective Study by Hengchang Liu, Chunxiang Li, Zhixun Zhao, Xu Guan, Ming Yang, Zheng Liu, Yuan Tang, Zheng Jiang and Xishan Wang in Technology in Cancer Research & Treatment
Footnotes
Abbreviations
Authors’ Note
All authors were involved in the conception, design, analysis, data interpretation drafting and final approval of this manuscript. Our study was approved by Cancer Hospital, Chinese Academy of Medical Sciences; National GCP Center for Anticancer Drugs; The Independent Ethice Committee (approval no. NCC2016JZ-06). All patients were informed of the purpose of the study at follow-up and received oral consent.
Acknowledgments
The authors wish to thank all the patients enrolled in this study. This work was supported by National Program Project for Precision Medicine in National Research and Development Plan of China (2016YFC0905300), National Natural Science Foundation of China (81572930), National Key Research and Development Program of the Ministry of Science and Technology of China (2016YFC0905303, 2016YFC1303200), Beijing Science and Technology Program (D17110002617004), Non-profit Central Research Institute Fund of Chinese Academy of Medical Sciences(2018PT32012), CAMS Innovation Fund for Medical Sciences (CIFMS) (2016-I2M-1-001), Incentive Fund for Academic Leaders of Oncology Hospital, Chinese Academy of Medical Sciences (RC2016003), and Beijing Hope Run Special Fund from Cancer Foundation of China (LC2017A19).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
