Abstract
Background:
Triple-negative breast cancer encompasses heterogeneous subtypes. Neoadjuvant chemotherapy is ineffective against some triple-negative breast cancers, while others show a favorable prognosis despite chemoresistance.
Methods:
A total of 51 cases with stages I and II triple-negative breast cancer were analyzed; 34 triple-negative breast cancers treated with neoadjuvant chemotherapy were divided into “good responders” (n = 22), showing therapeutic effect G2b or G3 in surgical specimens, and “poor responders” with therapeutic effect G0, G1a, G1b, and G2a (n = 12). Neoadjuvant chemotherapy was spared in 17 cases (non-neoadjuvant chemotherapy group). Apocrine-type triple-negative breast cancer was defined as triple-negative breast cancer immunoreactive for both androgen receptor and forkhead-box protein A1. Triple-negative breast cancer other than apocrine-type (n = 16) and special types (myoepithelial, medullary, adenoid cystic, and spindle cell carcinomas, n = 6) was categorized as basal-like subtype (n = 29). Prognosis was evaluated in each category.
Results:
Neoadjuvant chemotherapy provoked significant effects against basal-like triple-negative breast cancer with high Ki-67 labeling (≧50%), and tumor-infiltrating lymphocytes predicted high chemosensitivity. Neoadjuvant chemotherapy was avoidable in triple-negative breast cancer of apocrine- and special types showing low (<50%) Ki-67 labeling. Ten (59%) lesions in the non-neoadjuvant chemotherapy group belonged to the apocrine-type. When clinical complete remission shown by contrast-enhanced magnetic resonance imaging was reached in the course of neoadjuvant chemotherapy against basal-like triple-negative breast cancer, the neoadjuvant chemotherapy period was shortened in 14 (64%) of 22 good responders. Disease-free and overall survival rates were excellent in all groups.
Conclusions:
The following 2 hypothetical proposals should be proven by large-scale clinical trials. Immunohistochemical recognition of apocrine-type triple-negative breast cancer with low Ki-67 labeling is important for avoiding ineffective/unnecessary neoadjuvant chemotherapy. By employing appropriate clinical imaging, period-shortening is achievable in basal-like triple-negative breast cancer with high Ki-67 labeling.
Keywords
Introduction
Breast cancer, the most common cancer of women worldwide, remains an important global health issue. 1 Center for Cancer Control Information Services of National Cancer Center, Tokyo, Japan, estimated 92 200 new cases of breast cancer in 2019 in Japan, with more than 15 000 deaths, and every 1 of 11 Japanese women suffers from breast cancer during the lifetime. 2
Over the decade, messenger RNA expression profiling has been applied to classifying invasive breast cancer into biologically and clinically distinct intrinsic subtypes. The basal-like subtype is found in a younger age group and at an advanced clinical stage and accompanies frequent
Triple-negative breast cancer accounts for approximately 15% of breast cancer. Because of the absence of molecular targeted therapy, cytotoxic chemotherapy should be chosen for TNBC of basal-like subtype, but the clinical outcome is poor, compared with other intrinsic subtypes. 10,11
It has been shown that neoadjuvant chemotherapy (NAC) is poorly effective in more than half of cases of TNBC, while some cases of TNBC show a favorable prognosis despite chemoresistance. 12 -14 In 2012, one of the authors (YT) defined apocrine-type breast cancer as the lesion immunohistochemically showing ER/PgR-negative and androgen receptor (AR)-positive phenotypes, estimating 44 (13.5%) of 325 invasive ductal carcinomas. 15 Classical apocrine appearance was histopathologically suggested only in half of these lesions. Overexpression rate of HER2 in the apocrine-type breast cancer was as high as 23 (52%) of 44. Importantly, a half of the AR-expressing apocrine-type lesions lacked the HER2 overexpression and were thus categorized in TNBC. When compared with genuine TNBC of basal-like subtype quadruple-negative for ER, PgR, AR, and HER2, the apocrine-type TNBC showed a lower histological grade and a lower Ki-67 labeling index. It has been indicated that patients with apocrine-type TNBC may have a better prognosis than those with basal-like TNBC, despite lower pathological complete response (pCR) rates after NAC. 16 -22
The molecular apocrine subgroup was first described on a transcriptomic analysis by Farmer and colleagues
23
in 2005 and confirmed thereafter by Doane and colleagues
24
and Guedj and colleagues.
25
The molecular apocrine subgroup is effectively defined as the ER/PgR-negative, AR-positive, and forkhead-box protein A1 (FOXA1)-positive tumor with AR pathway activation, and in 50% of cases, HER2 overexpressed. Immunohistochemical study of Tsutsumi
15
is fully in line with these descriptions. Lehmann and colleagues
26
specifically analyzed the TNBC subgroup by gene expression profiling in 2011 and described 6 subtypes, including luminal androgen receptor (LAR) subgroup, corresponding to the HER2-negative molecular apocrine breast cancer by Farmer
Tumor-infiltrating lymphocytes (TILs) are a predictive microscopic predictor of good responses of TNBC to NAC, and TILs-rich TNBC may thus represent TNBC of immunomodulatory intrinsic subtype. 30 -33 Androgen receptor-positive TNBC also occasionally accompanies the lymphoid stroma. 34
Forkhead-box protein A1, also called hepatocyte nuclear factor 3α, is a member of intranuclear transcription factors of the forkhead gene family lacking basic amino acids in the molecule. 35 In the breast and prostate, FOXA1 colocalizes with ER or AR in the nuclei, interacts with cis-regulatory regions in heterochromatin, and enhances interaction of the hormone receptor with chromatin. Forkhead-box protein A1, indispensable for normal development of the breast and prostate, 36,37 is also required for both ER- and AR-regulated transcription in hormone receptor-positive carcinoma of the breast and prostate. 38 -40
In the present retrospective study performed in a small clinic in Yokohama, Japan, we evaluated effects of NAC against stages I and II TNBC and clinical outcome of both the basal-like (quadruple-negative) type and apocrine-type by analyzing a total of 51 cases with TNBC, including 34 with NAC (NAC group) and 17 without NAC (non-NAC group). Apocrine-type TNBC was defined when the tumor cells expressed both AR and FOXA1 in the nuclei, as has been indicated by Robinson and colleagues 41 and Sasahara and colleagues. 42 We propose hypothetical schemes for the avoidance of chemotherapy for the apocrine-type TNBC with low Ki-67 labeling, as well as for period-shortening of NAC for basal-like TNBC with high Ki-67 labeling, in order for patients to relieve adverse drug reactions.
Material and Methods
Patients
In the period from 2011 to 2016, we experienced a total of 434 patients with operable primary breast cancer, including 48 patients with ductal carcinoma in situ; and 122 patients with operable invasive breast cancer, including 43 patients with TNBC, received NAC in Yokohama Breast and GI Clinic, Yokohama. The surgical procedure was performed in Kawasaki Municipal Ida Hospital, Nakahara-ku, Kawasaki; Saiseikai Yokohamashi Tobu Hospital, Tsurumi-ku, Yokohama; and 1 other hospital. The subtypes of the NAC group were judged by immunohistochemical findings. 7 -9,15 Of 43 TNBC lesions, 34 (79%) cases were categorized in TNBC in stages I and II. Seventeen patients with TNBC in stages I and II who had undergone breast surgery in Kawasaki Municipal Ida Hospital, Saiseikai Yokohamashi Tobu Hospital, and 2 other hospitals in the same period but without NAC were categorized in the non-NAC group.
A flow diagram of patients included in the present retrospective study is shown in Figure 1.
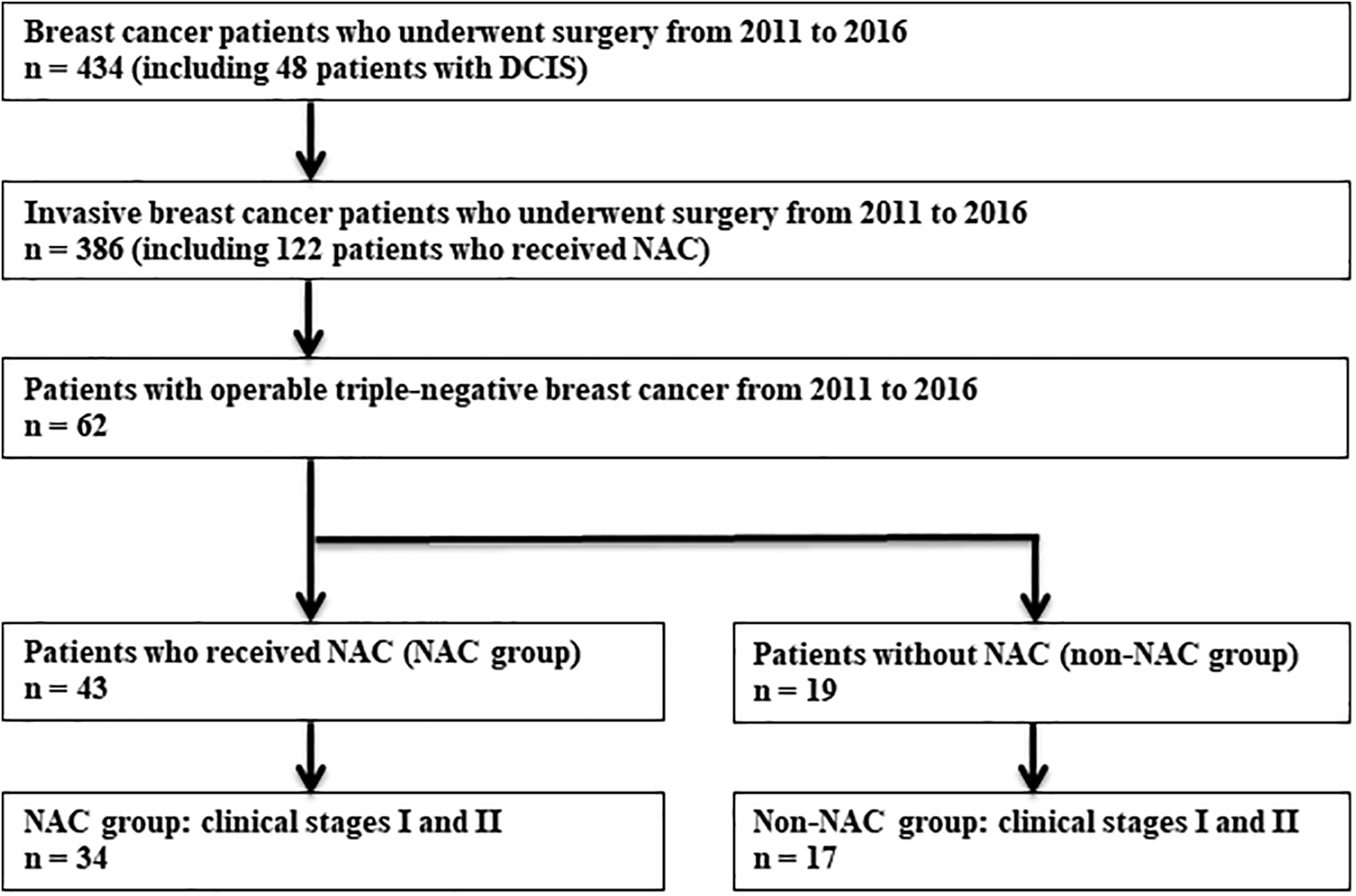
Flow diagram of patients included in the present retrospective study. DCIS indicates ductal carcinoma in situ.
Since the present study was performed solely using cases in a local clinic, the sample size was restricted. Therefore, hypothetical proposals are to be presented based on our observations of a limited number of cases.
Evaluation of Effects of NAC
As NAC, the anthracycline (A)-based regimen (epirubicin and cyclophosphamide [EC] with or without 5-fluorouracil, 3 weeks/cycle) followed by the taxane (T)-based regimen (weekly paclitaxel [PTX], triweekly docetaxel [DTX] or weekly nanoparticle albumin-bound paclitaxel [nabPTX]) was administered, in accordance with the National Comprehensive Cancer Network Guidelines 43 and the Guidelines for Breast Cancer in Japan. 44 The standardized cycle numbers were 4 cycles for A and 12 cycles for T, as well as 3 for A and 15 for T or 6 for A and 6 for T. Triweekly DTX was comparable with 3 cycles of weekly PTX or weekly nabPTX. When clinical complete remission (cCR) was revealed by contrast-enhanced magnetic resonance imaging (MRI) in the course of NAC, we progressed to surgical removal of the lesion. When the tumor was significantly regressed to 5 mm or less in size by ultrasound examination, the chelated nonionic gadolinium-enhanced MRI analysis was performed when necessary with an ad libitum fashion. We did not accept systematic MRI formulation for the NAC response. By palpation, the tumor commonly becomes unpalpable when the tumor size is shrunken to less than 10 mm. By ultrasound screening, we can recognize the tumor regression up to 5 mm in diameter (or 50 mm3 in volume), but the residue of viable tumor tissue becomes uncertain when the ultrasound shadow size is shrunken to less than 5 mm. The period of NAC was thus shorter than the standardized cycles, and we regarded such situation as period-shortening. As a surgical procedure, partial mastectomy (breast-conserving surgery) with postoperative whole breast irradiation or total mastectomy, including nipple-sparing mastectomy without reconstruction, was performed in all the cases. In one case in the good responder, radiotherapy was omitted after partial mastectomy, because of patient’s refusal. Four cases with BRCA mutations (categorized in the good responder) underwent total mastectomy with breast reconstruction.
Disease (relapse)-free survival (DFS) was defined as the interval between the date of biopsy confirmation of the primary cancer and the date at which relapse was confirmed or the date of the last follow-up, as of the end of March 2020. Overall survival (OS), an interval between the date of biopsy and the last follow-up or the date of death, was also evaluated. Based on the chemotherapeutic effect in the surgically resected samples, 34 TNBC lesions were divided into 2 groups: 22 lesions showing marked response to NAC, grades 2b or 3, were categorized in the good responder, while 12 lesions with less response, grades 2a, 1b, 1a, or 0, were categorized in the poor responder. The judgment was authorized by the General Rules for Clinical and Pathological Recording of Breast Cancer, version 18 (2018) by the Japanese Breast Cancer Society. 44
Adjuvant (postoperative) chemotherapy was performed in 3 cases of basal-like TNBC in the non-NAC group, instead of NAC. In 3 stage II cases of 8 basal-like TNBCs in the poor responder, additional adjuvant chemotherapy using EC, nabPTX, or cyclophosphamide/methotrexate/5-fluorouracil was supplemented. No adjuvant chemotherapy was given for the good responder. After 2017, adjuvant capecitabine administration became common post NAC against TNBC. 45 The present study was performed using cases in 2011 through 2016.
Histopathological and Immunohistochemical Evaluation
A total of 34 TNBC tissues prior to NAC were sampled by core needle biopsy, and breast-conserving surgery or total mastectomy was performed after NAC. Needle-biopsied TNBC lesions without NAC (n = 17), as well as the surgical specimens of the respective cases, were also examined. The tissues were fixed in 10% buffered formalin and routinely embedded in paraffin wax. The paraffin blocks were kept in the clinic or in the hospitals. Paraffin sections of 4 µm thickness were mounted onto 3-aminopropyltriethoxysilane-coated glass slides (Matsunami Glass Industry). Hematoxylin and eosin (H&E) staining was performed for evaluating histopathological features. Tumor-infiltrating lymphocytes were microscopically evaluated as immune cells (small lymphocytes and plasma cells) seen in or around the invasive cancer nests, according to the international guideline. 31 Tumor-infiltrating lymphocytes were judged positive when more than 50% of stromal area within the cancer tissue was occupied by the mononuclear cells. Lymphoid cells accumulating only at the invasion front or around the intraductal cancer lesion were not regarded as TILs.
The amino acid polymer technique (Simple Stain Max-PO, Nichirei Bioscience) was utilized for immunoperoxidase staining of ER, PgR, AR, HER2, p53 oncoprotein, Ki-67, epidermal growth factor receptor (EGFR or HER1), cytokeratin 5/6 (CK5/6), CK14, and gross cystic disease fluid protein-15 (GCDFP15). The antibodies and the soaking solution for heat-induced epitope retrieval by pressure pan heating for 10 minutes were described previously. 15 For demonstrating FOXA1, biotin-free catalyzed signal amplification-II modification provided by Agilent Technologies was employed. Rabbit antiserum available from Abcam was diluted at 1:4000, and the antigenicity was retrieved with 10 mM citrate buffer, pH 7. Positive signals were colored brown with diaminobenzidine and hydrogen peroxide, and the nuclei were briefly stained with Mayer hematoxylin.
Judging Criteria for the Effect of NAC
Chemotherapeutic effect was judged according to the General Rules for Clinical and Pathological Recording of Breast Cancer, version 18 (2018), edited by the Japanese Breast Cancer Society. 46 The effect on invasive lesions was microscopically evaluated. Grade 0: little change, grade 1a: mild effect, grade 1b: moderate effect, grade 2a: marked effect but with viable cancer cells, grade 2b: significant effect with only a few viable cancer cells, and grade 3: no viable cancer cells seen (pCR). In the Japanese guideline, the pCR status may contain viable in situ lesions. Clinical relevance has so far been reported by Japanese investigators. 47,48
Judging Criteria for Immunohistochemical Staining
The judging criteria were described previously. 15 The 10% criterion was utilized for the judgment of hormone receptors (ER, PgR, and AR). For the judgment of HER2, the 10% criterion (American Society of Clinical Oncology/College of American Pathologists, 2013) 49 was adopted, although in most cases the tumor cells showed uniform reactivity. Fluorescence in situ hybridization study for HER2 genome was added in case of 2+ immunohistochemical positivity. For FOXA1, diffuse nuclear staining was judged positive, while cytoplasmic reactivity of FOXA1 was not evaluated.
Dowsett and the International Ki67 in Breast Cancer Working Group emphasized the importance of assessing Ki-67 labeling in invading breast cancer cells. 50 In the present study, the mean (not hot spot) percentage of Ki-67 nuclear positivity was evaluated in invasive lesions in a stepwise way, such as 1%, 2%, 3%, 5%, 10%, 15%, 20%, 25%, 30%, 40%, 50%, 60%, 70%, 80%, and 90%, as reported previously. 15 Overexpression of p53 was regarded as positive when more than one-third of the nuclei of cancer cells were stained. Expression of EGFR, CK5/6, CK14, and GCDFP15 was judged positive when the plasma membrane or cytoplasm of more than 10% of cancer cells were stained.
Statistical Analysis
The chi-square test was utilized for comparing 2 or 3 opposing items. When the cell of the division display contained the number less than 10, 2-sided Fisher exact test was applied. Disease-free survival and OS were calculated by the Kaplan-Meier method, and the prognosis was compared by the log-rank test. Bonferroni correction was introduced when comparing 3 or more groups. Statistical software R version 3.5.1 was utilized for analysis. Statistical significance was defined as
Results
Clinicopathological Features of Patients
The age of the patients with NAC (n = 34) ranged from 34 to 71 years with the mean of 54.0 and the median 53.5, and the tumor size ranged from 6 to 31 mm with the mean of 18.3 and the median 18. The mean follow-up period of the NAC group was 70.1 months with the median 71, ranging from 30 to 113. The age of the patients of non-NAC group (n = 17) ranged from 37 to 87 years (mean: 61.3, median: 60), and the size of the tumors ranged from 6 to 40 mm (mean: 17.5, median: 18). The follow-up period ranged from 19 to 129 months (mean: 75.1, median 72).
Good Responders Versus Poor Responders, in Relation to Ki-67 Labeling Index
In all, 19 (56%) of 34 TNBC lesions showed pCR (pathological therapeutic effect grade 3). The rate of grade 2b plus grade 3 (pCR) was 22 (65%) of 34.
When the chemotherapeutic effect microscopically judged using the surgical specimens was categorized in grade 2b and grade 3, we categorized them in the good responder (n = 22). When the chemotherapeutic effect was in grades 2a, 1b, 1a, and 0, the lesions were included in the poor responder (n = 12).
The clinicopathological features of the patients in respective categories (good responders, poor responders, and non-NAC group), as well as those of histological subtypes (basal-like, apocrine-type, and others), are summarized in Table 1. Four good responders (basal-like 2, apocrine-type 1, and myoepithelial carcinoma 1) received total mastectomy, due to the association of
Summary of Clinicopathological Features.a
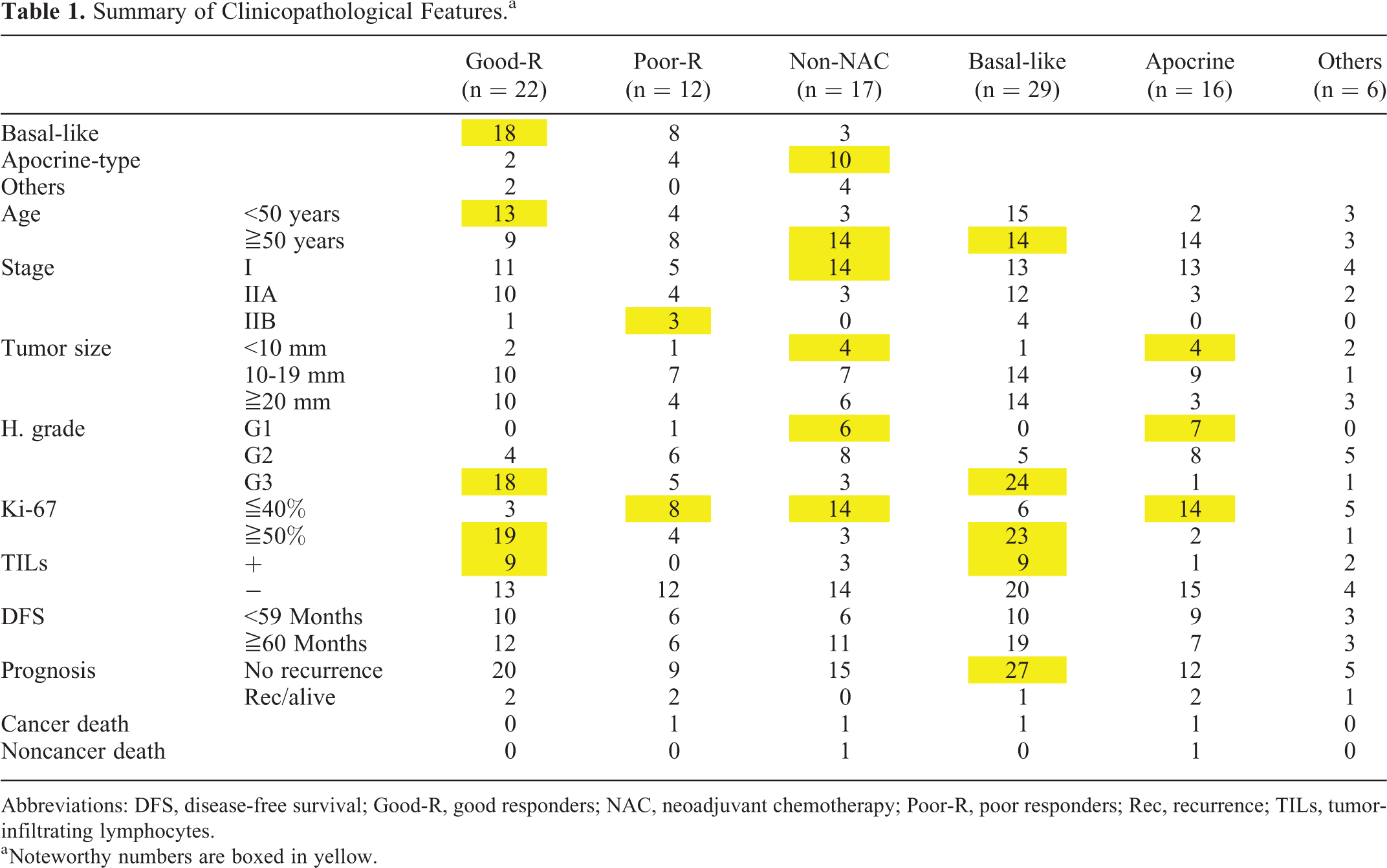
Abbreviations: DFS, disease-free survival; Good-R, good responders; NAC, neoadjuvant chemotherapy; Poor-R, poor responders; Rec, recurrence; TILs, tumor-infiltrating lymphocytes.
a Noteworthy numbers are boxed in yellow.
Importantly, the chemotherapeutic effect was closely correlated with Ki-67 labeling index. Namely, 19 (86%) of 22 good responders showed Ki-67 labeling 50% or more, whereas 4 (33%) of 12 poor responders were categorized in the high Ki-67 labeling group. The statistical significance (
Figure 2 illustrates a schematic relationship between the chemotherapeutic effect of NAC and Ki-67 labeling indices of TNBC (n = 34).

Schematic illustration of the relationship between the chemotherapeutic effect of NAC and Ki-67 labeling indices in TNBC (n = 34), including 22 good responders and 12 poor responders. TNBCs with high Ki-67 labeling (50% or more) showed a good response to NAC, while those of low Ki-67 labeling (<50%) were frequently included in the poor responder. Statistical significance (
Histopathological and Immunohistochemical Characterizations of Apocrine- and Basal-Like Phenotypes
The results of immunohistochemical findings are summarized in Table 2. Estrogen receptor and PgR were consistently negative in all the TNBC lesions. The HER2 expression was judged as negative or 1+, except for two 2+ lesions (one in the good responder and one in the poor responder). Fluorescence in situ hybridization study failed to identify HER2 gene amplification in these 2 lesions.
Summary of Immunohistochemical Features.a

Abbreviations: AR, androgen receptor; CK5/6, cytokeratin 5/6; EGFR, epidermal growth factor receptor; FOXA1, forkhead-box protein A1; GCDFP15, gross cystic disease fluid protein-15; Good-R, good responders; NAC, neoadjuvant chemotherapy; Poor-R, poor responders.
a Noteworthy numbers are boxed in yellow.
In the NAC group, AR was expressed in the nuclei in 6 (18%) of 34 TNBC lesions, and 8 (24%) showed diffuse nuclear expression of FOXA1. Forkhead-box protein A1 positivity without AR expression was seen in 1 basal-like lesion in the poor responder and 1 myoepithelial carcinoma in the good responder. In 6 lesions, both AR and FOXA1 were simultaneously expressed to be categorized as the apocrine-type. The apocrine-type lesions comprised 2 (9%) of 22 good responders, and 4 (33%) of 12 poor responders (
In the non-NAC group, the ratio of the apocrine-type was higher: 10 (59%) of 17 lesions were categorized in the apocrine-type (
Special types phenotypically resembling TNBC included myoepithelial carcinoma (n = 3), medullary carcinoma (n = 1), spindle cell carcinoma (n = 1), and adenoid cystic carcinoma (n = 1). One myoepithelial carcinoma and 1 medullary carcinoma belonged to the good responder. In the non-NAC group, 2 myoepithelial carcinomas, 1 adenoid cystic carcinoma, and 1 spindle cell carcinoma were included. All the special types, except for medullary carcinoma, showed low Ki-67 labeling indices.
The lesions other than the apocrine-type and the special types were regarded as the basal-like subtype. A total of 3 (1 basal-like and 2 special types) expressed FOXA1 without AR positivity, including 1 spindle cell carcinoma in the non-NAC group. The basal-like subtype represented 18 (82%) of 22 good responders, 8 (67%) of 12 poor responders, and 3 (18%) of 17 non-NAC group lesions (
Of note is that TILs positivity (lymphoid stroma) was seen in 10 (45%) of 22 good responders, including 9 (50%) of 18 TNBC lesions of basal-like subtype and 1 medullary carcinoma, while no lesions in the poor responder were associated with TILs (
Representative immunohistochemical features of TNBC of basal-like subtype and apocrine-type are illustrated in Figures 3 and 4, respectively.

Immunohistochemical features of TNBC of basal-like subtype (a representative lesion in the good responder; A, H&E; B, ER; C, AR; D, FOXA1; E, p53; F, EGFR; G, CK5/6; H, GCDFP15). Highly atypical quadruple-negative cancer cells (histological grade 3) accompany the lymphoid stroma. ER, AR, FOXA1, and GCDFP15 are not expressed, while p53, EGFR, and CK5/6 are positive in the nuclei, on the plasma membrane, and in the cytoplasm of the cancer cells, respectively. AR indicates androgen receptor; CK5/6, cytokeratin 5/6; EGFR, epidermal growth factor receptor; ER, estrogen receptor; FOXA1, forkhead-box protein A1; GCDFP15, gross cystic disease fluid protein-15; H&E, hematoxylin and eosin; TNBC, triple-negative breast cancer.

Immunohistochemical features of TNBC of apocrine-type (a representative lesion in the good responder. A, H&E; B, ER; C, AR; D, FOXA1; E, p53; F, EGFR; G, CK5/6; H, GCDFP15). Highly atypical ER-negative cancer cells (histological grade 3) express AR, CK5/6, and GCDFP15 focally and FOXA1, p53, and EGFR diffusely. AR indicates androgen receptor; CK5/6, cytokeratin 5/6; EGFR, epidermal growth factor receptor; ER, estrogen receptor; FOXA1, forkhead-box protein A1; GCDFP15, gross cystic disease fluid protein-15; H&E, hematoxylin and eosin; TNBC, triple-negative breast cancer.
When the microscopic features were reviewed, apocrine-type TNBC commonly showed plump amphophilic cytoplasm, while TNBC of basal-like type often revealed indistinguishable histopathological appearance. Classical apocrine features with plump eosinophilic cytoplasm were seen in one poor responder and in 6 lesions in the non-NAC group with histological grade 1. Figure 5 represents comparative features of H&E staining and Ki-67 labeling in basal-like and apocrine-type TNBCs (2 lesions each).

Histopathological appearance and Ki-67 immunostaining of TNBC of basal-like subtype and apocrine-type (upper panels: H&E, lower panels: Ki-67; A and E, a basal-like lesion in the good responder; B and F, another basal-like lesion in the good responder: C and G, an apocrine-type lesion in the good responder; D and H, another apocrine-type lesion in the non-NAC group). Triple-negative breast cancer of the basal-like subtype commonly reveals high histological grade (G3) and high Ki-67 labeling. Dense lymphoid stroma is evident in panel A. Triple-negative breast cancer of the apocrine type often reveals lower histological grade (G2 in panel C and G1 in panel D) and lower Ki-67 labeling. Typical apocrine appearance is seen in panel D. H&E indicates hematoxylin and eosin; NAC, neoadjuvant chemotherapy; TNBC, triple-negative breast cancer.
Expression of p53, EGFR, CK5/6, CK14, and GCDFP15
The results are summarized in Table 2. p53 was overexpressed in 17 (59%) of 29 basal-like lesions and 6 (38%) of 16 apocrine-type lesions (
Regarding representative immunohistochemical features, see Figures 3 and 4. Detailed data are shown in Supplementary Tables S1 to S3.
Evaluation of Prognosis and Avoidance of NAC
The good responder to NAC contained lesions with therapeutic effects G2b (3 lesions) and G3 (19 lesions). All the cases of the basal-like subtype (n = 18) were alive without recurrence, and 2 cases revealed local recurrence (apocrine-type with G2b effect and myoepithelial carcinoma with G3 effect). It is of note that the clinical outcome of cases with therapeutic effect G2b was comparable with cases with G3 effect.
A total of 26 patients with basal-like TNBC in the NAC group, 18 good responders and 8 poor responders, were followed up for a mean period of 71.4 months (median 70.5) ranging from 30 to 113. Two basal-like lesions in the poor responder with high Ki-67 labeling, adjuvant chemotherapy given, showed multifocal local recurrence at the 12th month and at the 38th month, and the former patient died at the 30th month. “All but 1 (96%)” should be reworded as “Twenty-five (96%) of 26”. Among 20 basal-like lesions with high Ki-67 labeling, 17 (85%) were categorized in the good responder and 3 (15%) in the poor responder. Among 6 basal-like lesions with low Ki-67 labeling, 1 responded to NAC (G2b), while the remaining 5 belonged to the poor responder. Of particular note is that no recurrence was recorded after surgery in these 6 cases: the follow-up period ranged from 39 to 101 months with the mean 73.7 and the median 80.5.
All 6 patients with apocrine-type lesions, 2 in the good responder and 4 in the poor responder, were alive after the mean follow-up period of 73.5 months (the median 72.5) ranging from 34 to 108. Two cases (1 in the good responder and 1 in the poor responder) showed local recurrence at the 12th month and at the 44th month, respectively. In the non-NAC group, 3 of 17 cases revealed the basal-like lesions with high Ki-67 labeling (1 with TILs), and no recurrence was recorded for 88, 99, and 119 months after adjuvant chemotherapy. Of 17 lesions, 10 (59%) in the non-NAC group belonged to the apocrine-type with low Ki-67 labeling, and 4 belonged to the special types. One apocrine-type lesion in stage I recurred at the 20th month and died after 78 months. In this case, the recurrent tumor in the axillary node showed subtype conversion to ER/PgR-negative, AR-positive but with overexpression of HER2. Cancer-unrelated death was recorded at the 19th month in another apocrine-type case aged 87 years.
In the past 5 years, we dared to avoid NAC in apocrine-type TNBC or special type carcinomas in stages I and II with low (<50%) Ki-67 labeling. These included 4 apocrine-type lesions, 1 spindle cell carcinoma, and 1 myoepithelial carcinoma in the non-NAC group. The tumor size ranged from 6 to 40 mm (mean 21.5 mm, median 20.5). In these 6 cases, no recurrence has been experienced, although the follow-up period still remains short, ranging from 32 to 60 months (mean: 43.8, median 38.5).
The prognosis of the patients with the good responder, poor responder, and non-NAC group was evaluated by Kaplan-Meier method. No significant difference was observed for OS (

Disease-free survival of cases with TNBC in stages I and II: comparison among the good responder (n = 22), the poor responder (n = 12), and the non-NAC group (n = 17). No statistical significance was observed among the groups (
Period-Shortening of NAC
When cCR was revealed by contrast-enhanced MRI, the period of NAC was able to be shortened. Among 22 good responders, period-shortening was achieved in 14 (64%) cases, as shown in Figure 7. In 2 cases in the good responder, pCR (therapeutic effect G3) was reached just after 4 and 3 cycles of anthracycline administration, respectively. It is noteworthy that basal-like TNBC with high Ki-67 labeling was particularly susceptible to anthracyclines. Two cases (apocrine-type TNBC and myoepithelial carcinoma) with BRCAness showed local recurrence after the shortened NAC and total mastectomy, but after local excision of the small-sized recurred tumor and radiotherapy, the patients remained disease free for 22 and 21 months after recurrence without additional chemotherapy.

The number of cycles of NAC and therapeutic effect against TNBC in stages I and II (n = 34). A, anthracycline-based regimen; T, taxane-based regimen. The number next to A and T means the repeated cycle number of the regimen. The areas on the thick red line (shadowed boxes) indicate standardized therapy zone of NAC. The areas indicated by the thick red arrow demonstrate cases with shortened NAC periods. Good responders are shown in red. In some poor responders, NAC was not completed because of poor responsiveness. NAC indicates neoadjuvant chemotherapy; TNBC, triple-negative breast cancer.
Discussion
Immunohistochemical Definition of Apocrine-type TNBC
We would like to emphasize the importance of immunohistochemical recognition of apocrine-type TNBC as triple-negative and AR-positive phenotype. As reported previously, the incidence of apocrine-type breast cancer is much higher than expected under H&E observation: 44 (13.5%) of 325 invasive ductal carcinomas. 15 It should be noted that histologically the apocrine-type breast cancer does not necessarily reveal typical apocrine appearance in H&E preparations, 15 and that apocrine carcinoma diagnosed under H&E preparations alone may occasionally express ER (not apocrine immunohistochemically). 51,52 Vranic and colleagues proposed a strict definition of apocrine carcinoma of the breast as ER/PgR-negative and AR-positive invasive ductal carcinoma, 53 in accordance with proposal of Tsutsumi. Overexpression rate of HER2 in the apocrine-type invasive breast cancer was as high as 23 (52%) of 44, and the remaining half of apocrine-type breast cancer was categorized in TNBC. Genuine TNBC of basal-like subtype was quadruple-negative for ER, PgR, AR, and HER2. Importantly, the apocrine-type TNBC showed lower histological grading and lower Ki-67 labeling. Expression of p53 and basal cell markers (EGFR, CK5/6, and CK14) was common among both basal-like and apocrine-type TNBC. 15 Frequent expression of EGFR and CK5/6 in TNBC has been reported repeatedly. 5,6,9 In our present series, the apocrine-type lesions showed significantly low expression of CK5/6 and CK14 and significantly high GCDFP15 positivity when compared with basal-like lesions.
In the present study, we adopted the strict definition of apocrine-type TNBC as TNBC immunoreactive for both AR and FOXA1 in the nuclei, as has been indicated previously. 41,42 Forkhead-box protein A1 is an intranuclear transcription factor colocalizing with ER or AR in the normal and neoplastic cells of the breast and prostate. 36,37 In fact, hormone receptor-positive cancer cells of the breast and prostate commonly express FOXA1 in the nuclei. 38 -40 Triple-negative breast cancer of basal-like subtype is featured by the lack of nuclear expression of FOXA1 (while the cytoplasmic positivity was common). In the present series, 1 basal-like lesion in the poor responder and 2 special type lesions (myoepithelial carcinoma in the good responder and spindle cell carcinoma in the non-NAC group) exceptionally expressed FOXA1 in the nuclei but without hormone receptor positivity.
Gene expression profiling studies have subclassified TNBCs into several intrinsic subtypes. 3 -7 The HER2-negative molecular apocrine subtype or LAR subtype was proven to be a distinct molecular subtype of TNBC. 23 -29 In the current study, AR was expressed in 6 (18%) of 34 TNBC lesions in the NAC group, and all of them showed distinct nuclear expression of FOXA1, categorized as the apocrine-type. By the molecular study, Lehmann and colleagues found the LAR subtype in 12% or 16% of TNBC. 26,27 Liu and colleagues reported the LAR subtype in 29 (18%) of 165 TNBC lesions. 29 It has been shown that molecular apocrine breast cancer was ER-negative and expressed AR and FOXA1, 41,54 while 10% of molecular apocrine breast cancer lacked FOXA1 expression. 54 Coexpression of AR and FOXA1 in apocrine-type TNBC has also been described by 3 independent research groups. 41,42,55 Nakashoji and colleagues reported that chemosensitive TNBC tended to show low expression rates of AR and FOXA1. 56 It is very likely that apocrine-type TNBC immunoreactive for both AR and FOXA1 represents the HER2-negative molecular apocrine or LAR subtype defined by the molecular study.
Expression of AR in TNBC
Androgen receptor is expressed in 53% to 90% of breast cancers.
15,57
-60
Hickey and colleagues suggested that AR signaling exerts an antiestrogenic, growth inhibitory influence in ER-positive breast cancer.
61
By the meta-analysis, AR expression in breast cancer predicted favorable DFS, and in ER-positive cases better OS was noted.
62
The complexities of AR signaling in breast cancer were recently reviewed by McNamara and colleagues.
63
Expression of AR in TNBC has also been studied extensively, and the AR positivity rate in TNBC ranged from 6.6% to 75%.
16
-22
Vera-Badillo and colleagues meta-analyzed 7693 cases in 19 researches, and reported AR was expressed in 31.8% of ER-negative breast cancer.
59
In the present study, the apocrine-type represented 31% (16/51) of the TNBC lesions analyzed. The rate of
Low aggressiveness of AR-positive TNBC has repeatedly been reported.
16
-22
However, some researchers described controversial findings: Lehmann-Che and colleagues suggested aggressiveness of molecular apocrine breast cancer,
54
and Choi and colleagues described decreased survival in cases with AR-positive TNBC.
65
Guiu and colleagues suggested a worse outcome of AR-positive/FOXA1-positive TNBC compared to other TNBC and a higher risk of the late recurrence.
55
Liu and colleagues found no significant correlation between AR expression and NAC effect in TNBC, and the prognosis was poor in AR-positive TNBC in stage III.
66
In contrast, the recent molecular analysis has indicated low aggressiveness of the LAR subtype.
26
-29
In our current study analyzing stages I and II cases, 3 (19%) of 16 apocrine-type lesions recurred and 1 patient died, while 2 (7%) of 29 basal-like lesions in the poor responder recurred and 1 died (
Therapeutic Effect of NAC in Relation to Ki-67 Labeling Index
It has been reported that NAC and adjuvant chemotherapy reveal comparable effects on operable breast cancer. 67 -69 Unlike adjuvant chemotherapy, NAC has an advantage to allow us an opportunity to observe tumor shrinkage by palpation or on image, enabling rapid assessment of clinical response. 70 Neoadjuvant chemotherapy shows favorable effects on downstaging of cancer, availability of conservative surgery, and increased DFS and OS. 12 -14,70,71 Neoadjuvant chemotherapy has induced pCR in roughly one-third of TNBC cases, 13,14 and in pCR-induced TNBC cases, excellent (90%) long-term survival was obtained. 12,72 Neoadjuvant chemotherapy was particularly effective in 52% of basal-like TNBC, while low effectiveness (10%) of NAC was seen in LAR-type TNBC. 73 Similar findings have been reported by the Japanese groups. 56,74,75 In the present study, TNBC of basal-like subtype in the good responder (n = 18) showed no recurrence after NAC, and apocrine-type TNBC definitely revealed the chemoresistance, as mentioned above. Of note is that 3 of 4 stage IIB cases of basal-like TNBC were categorized in the poor responder, suggesting the significance of staging of the lesion. The importance of the appropriate recognition of intrinsic subtypes in TNBC has repeatedly been emphasized. 76,77 Of particular note is that the prognosis of HER2-negative molecular apocrine or LAR-type TNBC was favorable despite chemoresistance. 72,76,77
In our current study, TNBC with NAC effect G2b and G3 was included in the good responder group. In fact, among 3 cases with G2b effect (a small volume of viable invasive cancer cells remaining after NAC), 1 apocrine-type lesion showed local recurrence without adjuvant chemotherapy. The data were as comparable as 19 cases with G3 effect: local recurrence was recorded in 1 myoepithelial carcinoma (
Tumor-infiltrating lymphocyte-positive TNBC (basal-like TNBC 8 and medullary carcinoma 1) was observed in the good responder and consistently showed high Ki-67 labeling. Tumor-infiltrating lymphocytes represented an excellent predictive marker for the responsiveness to NAC in TNBC, as was described previously. 30 -33
More than half cases in the non-NAC group (10/17 = 59%) belonged to the apocrine-type TNBC with low Ki-67 labeling. Of 16 apocrine-type TNBC lesions evaluated, 14 (88%) showed low Ki-67 labeling. In contrast, 23 (79%) of 29 basal-like TNBC lesions accompanied high Ki-67 labeling (
The apocrine-type lesions comprised 2 (9%) of 22 good responders, and 4 (33%) of 12 poor responders (
Avoidance and Period-Shortening of NAC
In the present series, the prognosis was excellent even in cases with TNBC of basal-like subtype. In the NAC group, local recurrence was recorded in 2 basal-like, 2 apocrine-type, and 1 myoepithelial carcinoma lesions. Cancer death was recorded in 2 cases: 1 basal-like TNBC in the poor responder (death after multifocal local recurrence and following systemic metastasis) and 1 apocrine-type TNBC in the non-NAC group causing patient’s death after phenotypic conversion into HER2-overexpressed type.
The non-NAC group was subdivided into 2 categories. In the recent 5 years, NAC was prospectively avoided for apocrine-type TNBC (n = 4) or special-type (spindle cell and myoepithelial) carcinomas (n = 2) with low (<50%) Ki-67 labeling in stages I and II. The follow-up period for these 6 cases remained short, ranging from 32 to 60 months (the mean 43.8, the median 38.5). The second category (n = 10, except for 1 case with cancer-unrelated death) with longer follow-up periods (mean 99.5, median 103, range: 67-129) included basal-like TNBC with high Ki-67 labeling and effective adjuvant chemotherapy (n = 3), and TNBC of apocrine-type (n = 5) or special type (n = 2) with low Ki-67 labeling. Of 16 cases, 15 (94%) in the non-NAC group, excluding 1 case with cancer-unrelated death in the aged, were alive without recurrence. These included all 6 cases in the first category and all 3 basal-like TNBC cases treated with adjuvant chemotherapy.
Our study included 3 myoepithelial carcinomas, 1 medullary carcinoma, 1 spindle cell carcinoma, and 1 adenoid cystic carcinoma expressing TNBC features. In addition to medullary carcinoma, 1 myoepithelial carcinoma responded well to NAC. No NAC was administered to the remaining cases. The importance of appropriate histopathological recognition of such special types with TNBC phenotype should be emphasized. 12 -14,56,73 -75
In addition to the avoidance of NAC against TNBC of apocrine- or special type with low Ki-67 labeling as described above, period-shortening of NAC was commonly achieved for basal-like TNBC cases with high Ki-67 labeling when we retrospectively analyzed the period of NAC in our clinical practice. In the course of NAC, cCR was effectively judged by contrast-enhanced MRI. 84 We introduced MRI evaluation in an ad libitum fashion when the lesion was shrunken to 5 mm or less by ultrasound screening. The standardized cycle numbers of NAC were 4 cycles for anthracycline (A)-based regimens and 12 cycles for taxane (T)-based regimens, as well as 3 for A and 15 for T or 6 for A and 6 for T. In our analysis, among 22 good responders, period-shortening (de-escalation of NAC) was achieved in 14 (64%) cases as an unintentional result. In 2 cases in the good responder, pCR was reached just after A regimens, and no T regimens followed. The period-shortening must be a significant advantage of NAC. In case of adjuvant chemotherapy, it is hard to shorten the period because of the paucity of appropriate barometers. The idea of “relative dose intensity” indicates that the maintenance of the total amount of drug given per cycle (“dose intensity”) at more than 85% of the standardized therapy is crucial to controlling cancers. 85,86 In our protocol, the dose intensity itself was maintained to achieve shortening of the dosing period.
Recent paradigm shift in breast cancer biology indicates changes of breast cancer management from radical mastectomy to precision (personalized) medicine. Both Halstedian and Fisherian hypotheses recommend certain treatment protocol to all patients with breast cancer. Along with “the spectrum theory” as reviewed by Özmen, 87 we should separate low-risk patients from high-risk patients to avoid overtreatment for low-risk patients. It is likely that certain percentage of low-risk patients accompanying good prognostic factors do not require systemic and/or radiation therapy. The same hypothesis may be appliable to patients with TNBC accompanying higher or lower risks, as we suggested in the present study.
It is of no doubt that the period-shortening of NAC should be imperious for the patients to relieve cytotoxic drug-induced serious adverse reactions, such as cardiac toxicity by anthracycline and peripheral neuropathy and dysgeusia by taxane. The patient-friendly chemotherapy will also contribute to health economics by saving medical expenses.
Therapeutic Strategy for Apocrine-Type TNBC
The effectiveness of antiandrogen therapy against apocrine-type breast cancer has been described repeatedly. 17,18,26,57,59,73,76,88,89 In Japan, however, this has not yet been authorized in the clinical practice. We cordially expect practical introduction of antiandrogen therapy against apocrine-type TNBC. Arce-Salinas and colleagues reported the usefulness of AR antagonist bicalutamide for the treatment of metastatic AR-positive TNBC. 90 Effectiveness of an AR blocker, enzalutamide, against AR-positive TNBC has been reported. 91 Hilborn and colleagues described beneficial tamoxifen response in ER-negative and AR-positive breast cancer. 92 Hormonal therapy strategy against apocrine-type TNBC should thus be reappraised. Recently, Bareche and colleagues identified frequent (75%) somatic mutations in the phosphatidylinositol 3-kinase (PI3K)-AKT (v-Akt murine thymoma viral oncogene) signaling pathway in the LAR subtype of TNBC and proposed possible use of a PI3K/AKT inhibitor as the molecular target therapy for AR-positive TNBC. 93 Appropriate recognition of this unique subtype of TNBC distinguished by adding immunostaining for AR and FOXA1 in the routine panel must again be emphasized.
Conclusive Remarks
We propose herein the following 2 major hypothetical schemes. Appropriate immunohistochemical recognition of apocrine-type TNBC with low Ki-67 labeling is critically important for avoiding ineffective and unnecessary NAC. Basal-like TNBC with high Ki-67 labeling is highly susceptible to NAC to achieve period-shortening (de-escalation of NAC). The choice of treatment in the present study was different from the current guidelines, but we sincerely hope that our approach can have a far-reaching guidance to the clinical practice. The present study belonged to a small-scale retrospective analysis, performed in a local clinic in Yokohama, Japan. Large-scale prospective clinical trials are requested to confirm our hypothetical proposals.
Supplemental Material
Supplemental_material - Avoidance and Period-Shortening of Neoadjuvant Chemotherapy Against Triple-Negative Breast Cancer in Stages I and II: Importance of Ki-67 Labeling Index and the Recognition of Apocrine-Type Lesions
Supplemental_material for Avoidance and Period-Shortening of Neoadjuvant Chemotherapy Against Triple-Negative Breast Cancer in Stages I and II: Importance of Ki-67 Labeling Index and the Recognition of Apocrine-Type Lesions by Koichi Kubouchi, Kyosuke Shimada, Takamichi Yokoe and Yutaka Tsutsumi in Technology in Cancer Research & Treatment
Footnotes
Authors’ Note
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. The research conformed to the provision of the Declarations of Helsinki in 1995. The study protocol was approved in 2016 by the Ethics Committee for Medical Research of Fujita Health University, Toyoake, Japan (#HM16-028), and Yokohama Breast and GI Clinic, Yokohama. Written informed consent was obtained from each patient after the approval of our study protocol. All the histopathological and immunostaining data in the signed histopathological diagnostic report by a pathologist (YT) were explained for the patient when the treatment plan was presented by attending physicians (KK and KS). Since our treatment plan was not necessarily in accordance with the internationally standardized therapeutic discipline, all patients were also verbally informed of the investigational nature of the present study, when they visited the outpatient clinic. The verbal informed consent was recorded in the clinical chart.
Acknowledgments
The authors cordially thank Toshihito Shinagawa, MD, Department of Diagnostic Pathology, Kawasaki Municipal Ida Hospital, Nakahara-ku, Kawasaki, Japan; and Tokuhiro Kimura, MD, Department of Diagnostic Pathology, Saiseikai Yokohamashi Tobu Hospital, Tsurumi-ku, Yokohama, Japan, for their kind cooperation in making histopathologic diagnosis of surgical specimens and in giving us suggestions on the biopsy diagnosis. The positive assistance by Shin Nishiya, MD, Department of Surgery, Saiseikai Yokohamashi Tobu Hospital, is also deeply acknowledged. Mr Takashi Suzuki, General Affairs Division, Yokohama Breast & GI Clinic, Kohoku-ku, Yokohama, Japan, considerably contributed to assembling case information. Part of the current study was presented at the 24th, 25th, and 26th Annual Meetings of the Japanese Breast Cancer Society in 2016 (Tokyo), 2017 (Fukuoka), and 2018 (Kyoto) at the 54th Annual Meeting of Japan Society of Clinical Oncology in 2016 (Yokohama), at the 2nd World Congress of Breast Cancer in 2016 (Phoenix, Arizona), at the 117th and 119th Annual Congress of Japan Surgical Society in 2017 (Yokohama) and 2019 (Osaka), and at the 106th Annual Meeting of the Japanese Society of Pathology in 2017 (Tokyo).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
Abbreviations
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
