Abstract
Purpose:
To assess the efficacy and safety of stereotactic body radiation therapy using an abdominal compression technique and modified fractionation regimen (5-10 fractions) in patients with small-sized hepatocellular carcinoma.
Methods:
A total of 101 patients with small-sized hepatocellular carcinoma treated with stereotactic body radiation therapy using an abdominal compression technique and modified fractionation regimen were registered between June 2011 and June 2019 in our hospital. A total dose of 48 to 60 Gy was applied over 5 to 14 consecutive days. Liver motion was controlled by abdominal compression, and a helical intensity-modified radiation therapy-based stereotactic body radiation therapy administrated in tomotherapy platform.
Results:
The median follow-up period was 23.2 months (range: 4.1-99.2 months). Complete response and partial response were observed in 63 (62.4%) patients and in 24 (23.8%) patients, respectively. At the time of our analysis, the 1-, 3-, and 5-year local control rates after stereotactic body radiation therapy were 96.1%, 89.0%, and 89.0%, respectively. However, logistic regression analysis revealed no correlation between the biologically effective dose and 3-year local control rates. The 1-, 3-, and 5-year overall survival rates were 96.9%, 69.0%, and 64.3%, respectively. For patients who were treatment-naive, the 1-, 3-, and 5-year overall survival were 96.3%, 82.0%, and 82.0%, respectively. No patients experienced classic radiation-induced liver disease or nonclassic radiation-induced liver disease after stereotactic body radiation therapy completion.
Conclusions:
When using an abdominal compression technique and modified fractionation regimen (5-10 fractions) based on helical intensity-modified radiation therapy, stereotactic body radiation therapy led to a lower toxicity and comparative rate of local control and overall survival for patients who with small-sized hepatocellular carcinoma.
Keywords
Introduction
Hepatocellular carcinoma (HCC) is one of the most common malignancies in the world, especially in China, which accounts for half of the global incidence. 1,2 Surgical resection and radiofrequency ablation (RFA) are considered as the standard treatments for patients with early-stage HCC, according to the guidelines. 3,4 In recent years, stereotactic body radiation therapy (SBRT), as a new technique, has developed rapidly as a safe and effective treatment option for patients with small-sized HCC. It has a favorable local control rate and survival outcome, 5 -9 despite being reserved for cases inappropriate for standard modalities.
Stereotactic body radiation therapy is a type of radiotherapy technique that delivers high-dose radiation to tumors and sharp dose gradients to spare normal tissues, fewer needed fractions and sharp dose gradients spare normal tissues. 5 However, it is common for these patients to exhibit liver cirrhosis or close proximity of the tumor to the gastrointestinal tract. Therefore, modified fractionation regimen with increased limited fractions may decrease the risk of liver injury and gastrointestinal injury. On the other hand, techniques for managing respiration-related liver motion, such as active breathing control (ABC), tumor tracking, and gating, are complex and time-consuming. Furthermore, some of these techniques require good cooperation from patients.
In this study, we used a relatively simple technique based on helical intensity-modified radiation therapy (IMRT), including abdominal compression (AC) for immobilization, 4-dimensional computed tomography (4D-CT) for simulation, modified fractionation regimen for treatment plan, and on-board mega-voltage CT (MVCT) for daily image guidance.
Methods and Materials
Patients
Patients with small-sized HCC who received SBRT were reviewed from June 2011 to June 2019, and this study was approved by the institutional review board of the Ethics Committee of Zhongshan Hospital, Fudan University.
The diagnosis of HCC was according to the guideline proposed by the Chinese Liver Cancer Association. 10 Clinical diagnosis should be established by characteristic findings, α-fetoprotein level, and history of hepatitis or liver cirrhosis. The inclusion criteria for participants in our study were ECOG score: 0 to 2, age ≥18 years, volume of normal liver: >700 cm2, Child-Pugh class of A or B, and tumor ≥1 cm from the luminal gastrointestinal (GI) tract. Patients with lymph node metastasis, distant metastasis, or double primary malignancy were excluded from the study.
Stereotactic Body Radiation Therapy Treatment
Before treatment, patients were trained to maintain shallow breathing with respiratory exercise. Patients were immobilized using a customized vacuum body mold in the supine position, while the abdomen was compressed using the Body Pro-Lok system. Abdominal compression techniques (Body Pro-Lok system) were used as part of a fixed position to minimize the movement of the liver. The AC was applied to the subxiphoid area based on the patient’s maximum tolerability, as previously described. 11 Patients underwent 4D-CT scans with slice thickness of 3 mm (Siemens Somatom Sensation; Siemens Healthineers Corporation). The primary tumor in the enhanced CT was defined as the gross tumor volume (GTV). The internal target volume (ITV) was generated after including the extension of GTV on the 4D-CT scanning. The planning target volume (PTV) was created as the ITV plus a radial margin of 3 mm. Stereotactic body radiation therapy was administered using a Helical Tomotherapy (HT) Hi-Art Treatment System (Accuray). Patients received a total dose of 48 to 60 Gray (gy) in 5 to 10 fractions, 5 times a week. Patients underwent on-board MVCT daily for image guidance.
Follow-Up
All patients were followed up using CT or MRI 6 to 8 weeks after the completion of SBRT and every 3 months routinely thereafter. Follow-up time was defined from the start date of SBRT. Overall survival (OS) was calculated as the time from the first treatment to death due to any cause. Local control was defined as freedom from local disease progression according to Modified Response Criteria in Solid Tumors guidelines. 12 Toxicity was evaluated by the Common Terminology Criteria for Adverse Events, version 4.0 (CTCAE v 4.0). Radiation-induced liver disease (RILD) is defined as anicteric ascites and elevation of alkaline phosphatase levels to at least twice above the pretreatment values in the absence of progression (classic) or elevation of transaminases to at least 5 times above the upper normal limit or pretreatment level within 3 months after the SBRT (nonclassic).
Statistics Analysis
Data analysis was performed using SPSS Statistics 23.0 (IBM Corp). Descriptive statistics were summarized as the median ± standard deviation. Local control rate and OS were estimated using the Kaplan-Meier method. Variables significant in univariate analysis were further analyzed in multivariate analysis to evaluate for independent prognostic significance using the Cox regression model with hazards ratios (HRs) and 95% CIs.
Results
Patient Characteristics
From November June 2011 and June 2019, 101 patients with small-sized HCC underwent SBRT in our hospital. The median follow-up duration was 23.2 months (range: 4.1-99.2 months). Baseline characteristics are summarized in Table 1. There were 84 male patients and 17 female patients. The median age was 60.0 years (range: 22-98 years), and the median maximum tumor size was 2.6 cm (range: 0.8-6.8 cm). The Child-Pugh index was CPA in 99 patients and CPB in 3 patients. The median dose of SBRT was 48 Gy and ranged from 48 to 60 Gy. When converted into the biologically effective dose (BED10), the median dose was 86.4 Gy and ranged from 75 to 119 Gy. Patients most commonly received 6 fractions of SBRT, with a total dose of 48 or 54 Gy (73/101, 71.3%).
Patient Baseline Demographics.a

Abbreviations; AFP, α-fetoprotein; BED10, biologically effective dose; SBRT, stereotactic body radiation therapy; RFA, radiofrequency ablation.
a Group A: Patients who were suitable for surgery but refused; Group B: Surgery or RFA was not feasible for technical or medical reasons, such tumor location or a poor liver function, old-aged patients; Group C: SBRT was used as salvage treatment for intrahepatic tumor recurrence after surgical resection or RFA; Group D: SBRT as an adjuvant treatment for intrahepatic tumors with incomplete iodized oil retention, or residual cancer after surgical resection or RFA.
Next, we classified patients into 4 categories, depending on their indications (Table 1). Group A (n = 11): Patients were naive to treatment and were suitable for surgery or RFA but refused either intervention. Group B (n = 17): Patients were naive to treatment and were not suitable for surgery or RFA due to technical or medical reasons, such as tumor location, poor liver function, or old age. Group C (n = 31): SBRT was used as salvage treatment for intrahepatic tumor recurrence after surgical resection or RFA. Group D (n = 42): SBRT was used as a consolidated treatment for intrahepatic tumors with incomplete iodized oil retention or residual cancer after surgical resection or RFA.
Radiologic Response and Local Control Rates After SBRT
The treatment responses were evaluated at the time of second follow-up (4.5 months) after completion of SBRT. Complete response, partial response, and stable disease were achieved in 63 (62.4%), 24 (23.8%), and 13 (12.9%) patients, respectively, and 1 (1.0%) patient was observed as progressive disease. At the time of our analysis, the 1-, 3-, and 5-year local control rates after SBRT were 96.1%, 92.1%, and 89.0%, respectively (Figure 1). Age, BED10, gender, tumor size, and tumor number were not correlated with 3-year local control rates according to logistic regression analysis (Table 2).

Local control rate in all patients. The 1-, 3-, and 5-year overall survival (OS) rates were 96.9%, 69.0%, and 64.3%, respectively.
Factors Associated With 3-Year Local Control in Patients With Hepatocellular Carcinoma Treated With Stereotactic Body Radiation Therapy by Logistic Analysis.

Abbreviations; AFP, α-fetoprotein; BED10, biologically effective dose; HR, hazard ratio.
Overall Survival Rates
Among all the evaluable patients, the 1-, 3-, and 5-year OS rates for all evaluable patients were 96.9%, 69.0%, and 64.3%, respectively (Figure 2A). Then we analyzed OS by subgroups mentioned above. The 1-, 3-, and 5-year OS for group A (n = 11) is 90.9%, 90.9%, and 90.9%, respectively. The 1-, 3-, and 5-year OS for group B (n = 17) is 100.0%, 71.4%, and 71.4%, respectively. The 1-, 3-, and 5-year OS for group C (n = 31) is 96.8%, 71.1%, and 59.6%, respectively. The 1-, 3-, and 5-year OS for group D (n = 42) is 97.6%, 56.7%, and 56.7%, respectively (Figure 2B). At last, when combined Group A with Group B as treatment-naive patients, the 1-, 3-, and 5-year OS were 96.3%, 82.0%, and 82.0%, respectively.
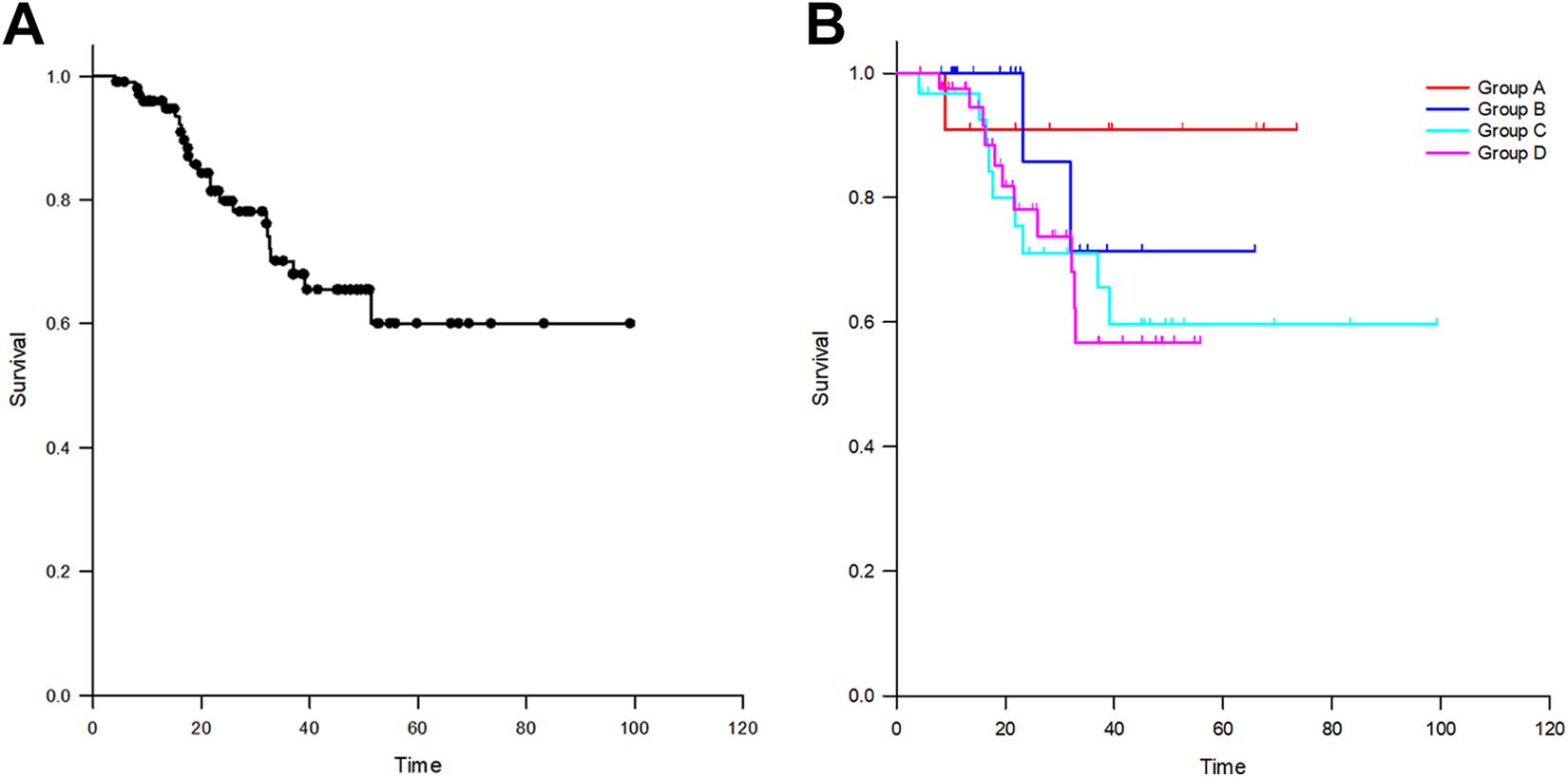
(A) Over survival outcome for all patients. (B) Overall survival by stratified into 4 groups.
Toxicity
All patients received the planned SBRT without any interruptions, and no patient experienced classic radiation-induced liver disease (classic RILD) or nonclassic RILD within 3 months after completion of SBRT. Grade 2 hepatic toxicities included elevated aspartate transaminase levels in one patient and hyperbilirubinemia in one patient. Grade 2 hematologic toxicities included leukocytopenia in 5 patients and thrombocytopenia in 3 patients. Two patients had a gastric ulcer, as confirmed by endoscopy. Treatment-related toxicities are summarized in Table 3.
Treatment-Related Toxicities After SBRT.

Abbreviation: SBRT, stereotactic body radiation therapy.
Discussion
Stereotactic body radiation therapy is an emerging radiotherapy treatment modality that is based on CT imaging and has enabled the delivery of ablative doses to tumors while sparing a sufficient portion of the normal liver. 13 High local control of SBRT may be comparable to RFA or surgery. 14 -16 Thus, SBRT could be considered as an alternative treatment. However, in patients with HCC, there are no specific consensus guidelines for SBRT, including determination of the optimal dose fractionation and methods for controlling respiration-induced organ motion and compromised hepatic function due to chronic viral hepatitis. 17 In previous studies, 5,18 3 to 5 fractions were most commonly used, and the adopted techniques for respiration control were the ABC, respiratory gating system, and real-time tracking system. 19 However, the current method for liver SBRT has some disadvantages. First, patients with HCC in close proximity to major vessels, the luminal tract, bile duct, or diaphragm are not suitable for surgery or RFA. In these patients, fewer fractions may increase the probability of gastrointestinal and bile duct injury. Second, the abovementioned techniques for respiration control are complex, require good cooperation from patients, thereby decreasing the efficiency of treatment. In this study, we used a relatively simple technique for SBRT of HCC, including AC for immobilization, 4D-CT for simulation, modified fractionation regimen for treatment plan, and on-board mega-voltage CT (MVCT) for daily image guidance. Our protocol was easy to use and potentially decreased toxicities by increasing the number of total fractions. The advantages of helical IMRT (tomotherapy) in the treatment of liver tumors include its ability to effectively treat multiple targets at the same time 20 and to improve tumor coverage. 21 The special design enabled tomotherapy to treat all tumor parts simultaneously, instead of intermittently, which is usually seen with conventional IMRT or Cyberknife. In contrast, the irradiation time was also less than that of other techniques, because correction was not necessary during irradiation in our protocol.
Abdominal compression is commonly used for reducing thoracic or abdominal tumor motion during radiation therapy. 22,23 As reported previously, 11 a compression plate on the subxiphoid area can minimize the magnitude of liver motion in 3 directions (X: 2.53 ± 0.93 mm; Y: 4.53 ± 1.16 mm; Z: 2.14 ± 0.92 mm). We adopted this technique in the current study. Although the slow acquisition times of MVCT may be disadvantageous, AC can improve the accuracy of image guidance by reducing breathing motion to <5 mm and liver-to-liver registration in the 3-dimensional view. 24
Different institutions use widely variable total doses in treating HCC with SBRT. However, unlike early-stage lung cancer, 25 there are few data to indicate whether similar dose–response relationships apply to hepatic SBRT. Sanuki et al 9 showed the possibility of an acceptable local control rate of 91% at 3 years for small and single HCCs using BEDs 59.5 Gy/72 Gy. Studies by Bujold et al 23 and Scorsetti et al 26 using BEDs lower than 100 Gy showed lower local control rates, although most patients in their studies had relatively large or multiple HCCs. Ohri et al 27 reported the results of a systematic quantitative review of published experiences with liver SBRT to determine whether there is a relationship between liver SBRT dosing and clinical outcomes. They found no evidence that local control is influenced by BED within the range of schedules used. In the current study, the 3-year local control rates after SBRT were 89.0%, and BED10 was not correlated with 3-year local control rates.
A meta-analysis conducted by Lee et al 28 showed LC was equivalent between SBRT and RFA for HCC, and the efficacy of SBRT might be better especially for tumors larger than 2 to 3 cm; however, RFA was associated with better survival. The discrepancy between LC and OS requires further investigation, as they are both local modalities showing equivalent efficacy. In fact, direct OS comparisons were difficult because of differences in patient information, tumor status, and treatment characteristics. Therefore, patients were stratified into 4 groups. In this study, patients in group A were all suitable for surgery or RFA. Previously reported 3-year OS rates for early-stage HCC treated by surgery and RFA were 75% to 90% and 67.2% to 71.4%, respectively. 28 -32 Given these comparable outcomes, SBRT constitutes a viable alternative if such therapies fail or cannot be applied.
In the current study, treatment-related toxicities were mild and transient. No patient experienced classic RILD or nonclassic RILD, and only 2 patients developed gastrointestinal ulcers. In contrast, some studies have reported relatively high complication rates, for examples, Scorsetti et al 26 reported grade 3 hepatic toxicity rates of 16.3%, and all reported cases involved the transient elevation of liver enzymes. In the studies by Kim et al 33 and Weiner et al, 34 grade 3 hematologic toxicity rates were 28% and 27%, respectively. Our lower rates may be due to the modified fractionation regimens and lower total doses used in the study.
The study has also limitations due to the relatively small number of patients and the retrospective design. In addition, although AC technique could be a good tool to reduce the liver dose and treatment time, AC cannot solve all the issues in the treatment of HCC, and the accuracy of image guidance by MVCT should be inferior to that of tracking technique.
Conclusion
In this study, helical IMRT-based SBRT using an AC technique and modified radiotherapy regimens for small-sized HCC provided a lower toxicity and comparative rate of local control and OS for patients with small-sized HCC. Our protocol was easy to use and potential benefits included shorter treatment set-up and delivery times.
Footnotes
Authors’ Note
Y-.X.C. and Y.Z. have contributed equally to this work. This study was approved by the Ethics Committee of Zhongshan Hospital, Fudan University (approval no. B2018-272). All patients provided written informed consent prior to enrollment in the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research was supported by grants from the National Natural Science Foundation of China (No. 8160110123), the National Key R&D Program of China (2017YFC0112100), and the special clinical research foundation of Zhongshan Hospital, Fudan University (2016ZSCL20).
