Abstract
External beam radiation therapy (EBRT) has increasingly been utilized in the treatment of hepatocellular carcinoma (HCC) due to technological advances with positive clinical outcomes. Innovations in EBRT include improved image guidance, motion management, treatment planning, and highly conformal techniques such as intensity-modulated radiation therapy (IMRT) and stereotactic body radiation therapy (SBRT). Moreover, proton beam therapy (PBT) and magnetic resonance image-guided radiation therapy (MRgRT) have expanded the capabilities of EBRT. PBT offers the advantage of minimizing low- and moderate-dose radiation to the surrounding normal tissue, thereby preserving uninvolved liver and allowing for dose escalation. MRgRT provides the advantage of improved soft tissue delineation compared to computerized tomography (CT) guidance. Additionally, MRgRT with online adaptive therapy is particularly useful for addressing motion not otherwise managed and reducing high-dose radiation to the normal tissue such as the stomach and bowel. PBT and online adaptive MRgRT are emerging technological advancements in EBRT that may provide a significant clinical benefit for patients with HCC.
Keywords
Introduction
Liver cancer was the seventh most common cancer worldwide and the third leading cause of cancer-related death in 2020. 1 The high incidence of new diagnoses and death rates are the impetus for further research and innovation. Of all primary liver cancers, hepatocellular carcinoma (HCC) is the most prevalent, comprising about 75% to 85% of cases. 1 Standard local therapies include surgical resection, transarterial chemoembolization (TACE) or radioembolization, radiofrequency ablation (RFA), ethanol injection, and external beam radiation therapy (EBRT). The focus of this paper will be on EBRT, which offers a noninvasive modality for HCC.
Historically, radiation therapy (RT) for the treatment of HCC was limited to palliation since the safe delivery of definitive doses was constrained by suboptimal image guidance, inadequate motion management, and absence of highly conformal dose delivery.2,3 More recently, image-guided RT using computed tomography (CT) and magnetic resonance imaging (MRI), improved motion management, and highly conformal techniques such as intensity-modulated radiation therapy (IMRT) and stereotactic body radiation therapy (SBRT) have made definitive irradiation of HCC more feasible.2,4,5 CT-guided RT may either be accomplished with cone beam computed tomography (CBCT) or CT-on-rails.6,7 On-board CT or MRI improves daily treatment position by allowing one to visualize the patient's liver contour near the tumor target volume. If CT-guided RT is not available at certain proton centers, fiducial markers may be used along with image-guided kilovoltage orthogonal imaging to assist with daily localization. Motion management may include the use of breath-hold techniques, which involves holding one's breath in the end-exhale (preferred) or end-inhale phase for short intervals while radiation is being delivered, abdominal compression to reduce the internal margin of the internal target volume (ITV), or gating, which involves tracking movement during the respiratory cycle and only delivering radiation when the target is in the treatment field. The robotically guided radiosurgery system tracks the liver target volume in a near real-time fashion using implanted fiducial markers. Motion management is determined for each case prior to treatment, usually before or at the time of simulation, which typically involves obtaining a 4D CT for treatment planning and to assess motion. IMRT and SBRT both employ inverse planning, which involves computer-generated optimized treatment plans based on contours and physician-directed goals. SBRT further allows for therapeutic dose escalation to provide increased local control (LC) while minimizing dose to the surrounding normal tissue through steep dose fall-off outside of the target.
Several prospective and retrospective studies of SBRT showed excellent LC (74%-100%) and overall survival (OS) rates (53%-100%) at 1 to 2 years (yrs) with acceptable rates of high-grade toxicity (4%-13%) for the properly selected patient.8–20 Although more randomized data are warranted, prospective and retrospective studies have shown heterogeneous results when comparing SBRT to non-radiotherapeutic modalities (eg, TACE, RFA, or resection) and suggest SBRT may be preferred in certain patient populations.21–33 Still, modern radiation techniques have limitations. For example, SBRT is not always feasible, such as in the treatment of large tumors or patients with relatively poor liver function (eg, Child–Pugh [CP] B8 or greater), which may have a risk of radiation-induced liver disease (RILD) of up to 63% and 1-year (yr) OS rate as low as 32%.3,8,34–38 RILD can consist of nonmalignant ascites, hepatomegaly, and elevated alkaline phosphatase per the classical definition, or more commonly per the non-classic definition, greater than 5-fold increase in transaminases or worsening of Child–Pugh score by ≥2 and not attributable to disease progression.15,39 Limiting low-dose radiation to uninvolved cirrhotic liver is fundamental considering RILD may be associated with doses as low as 2.5 gray (Gy) to 10 Gy.8,40 Low-dose spillage is impossible to avoid with SBRT. In addition, CT imaging may be limited in its ability to delineate soft tissue and differentiate tumor from normal tissue (eg, liver or bowel). The purpose of this mini review is to discuss both proton beam therapy (PBT) and MR-guided radiation therapy (MRgRT) as ways of addressing the aforementioned issues and providing more targeted irradiation in the liver compared to conventional photon-based EBRT.
A PubMed literature review was performed on August 21, 2023. The PBT literature search included the terms “radiation,” “proton,” “hepatocellular,” and “liver.” A separate literature search was performed on the same date to identify MRgRT articles for HCC patients. The MRgRT literature search included the terms “radiation,” “MR,” “hepatocellular,” and “liver.” Inclusion criteria included prospective trials and well-designed retrospective studies to guide recommendations. Prospective PBT trials for HCC with greater than 20 patients and local control data at 2 yrs were summarized in Table 1. Given that MRgRT is a newer treatment modality, all prospective studies with HCC patients were included in Table 1. Discussion topics with limited research were supplemented with authors’ expert commentary based on clinical experience.
Summary of Select Prospective PBT and MRgRT Studies for Patients with HCC. All PBT Studies Utilized Passive Scattering

Abbreviations: n, number; pts, patients; CP, Child–Pugh; PV, portal vein; PVT, PV thrombosis; LDT, liver-directed therapy; RT, radiation therapy; Fx, fractions; OS, overall survival; LC, local control; BCLC, Barcelona Clinic Liver Cancer; AJCC, American Joint Committee on Cancer; Med, median; cm, centimeter; CGE, cobalt-gray equivalent; yr, year; G, grade; bili, bilirubin; CPCP, Child–Pugh class progression; RILD, radiation-induced liver disease; NR, not reported; MF, mean follow-up; GI, gastrointestinal; GIS, GI structure; m, months; HBE, hepatobiliary enzyme; Gy, gray.
Proton Beam Therapy (PBT) for HCC
History and Advantages of PBT
PBT was first used for cancer therapy in 1954 and for the treatment of HCC in the 1980s.57,58 Intensity-modulated proton therapy (IMPT) using pencil beam scanning (PBS) was clinically introduced in 1996. 59 Since then, more proton centers have opened, in part due to a lower initial cost option utilizing a compact proton accelerator and single-room gantry which was first used to treat patients in 2013. 60 PBT offers a significant advantage over photon-based RT: rather than depositing dose in an exponentially decreasing manner, which results in low-to-moderate doses of radiation beyond the tumor target, protons deposit the majority of their dose in a finite range, termed the Bragg peak, within the area of the tumor target. There is a sharp dose fall-off just past the target, thereby sparing normal tissue of irradiation.57,61 Technical advancements in the delivery of PBT include PBS which allows for IMPT. Prior to development of PBS, PBT was delivered with scattering techniques, which involved scattering a narrow proton beam over a larger area to cover a target volume. PBS involves magnetically scanning a proton beam across a 3D target. Compared to passive scattering, PBS provides better target conformality, particularly proximal to the target, which is advantageous for treating large, irregularly shaped tumors and sparing organs-at-risk (OARs).62–64 While PBS-IMPT may be more susceptible to changes in the tissues of the beam path than scattering PBT or photon-based IMRT, these uncertainties can be adequately addressed with motion management, image guidance, beam angle selection, and 4D optimization with treatment plan robustness testing.63,65–70
PBT offers the potential to widen the therapeutic window by allowing definitive doses of radiation to be delivered to HCC tumors while limiting exposure to critical OARs, notably the uninvolved liver (defined as liver minus gross tumor volume), and thereby reducing the risk of RILD.71–73 There is an increased risk of RILD when the mean liver dose exceeds 31 Gy, and even 2.5 to 10 Gy may increase the risk in the cirrhotic liver.8,40,74 Compared to photon-based RT of HCC, PBT provides the same target coverage as photon-based EBRT while significantly reducing the dose to surrounding normal structures. For example, Kim et al compared PBT and IMRT plans for patients with HCC and found that PBT reduced both the mean dose to the remaining normal liver and volume of normal liver exposed to 5 to 25 Gy by more than 50%. 75 PBT also significantly reduced the dose to the stomach and spinal cord. 75 Additionally, Wang et al showed PBT provided similar target coverage to IMRT and significantly reduced the radiation exposure to the normal liver, stomach, bowel, heart, and spinal cord. The radiation dose to the kidneys was also reduced but not statistically significant. 76 For visual representation,Figure 1 depicts a PBT and an IMRT plan, for a patient with HCC, which show similar target coverage and better normal tissue sparing with PBT compared to IMRT.
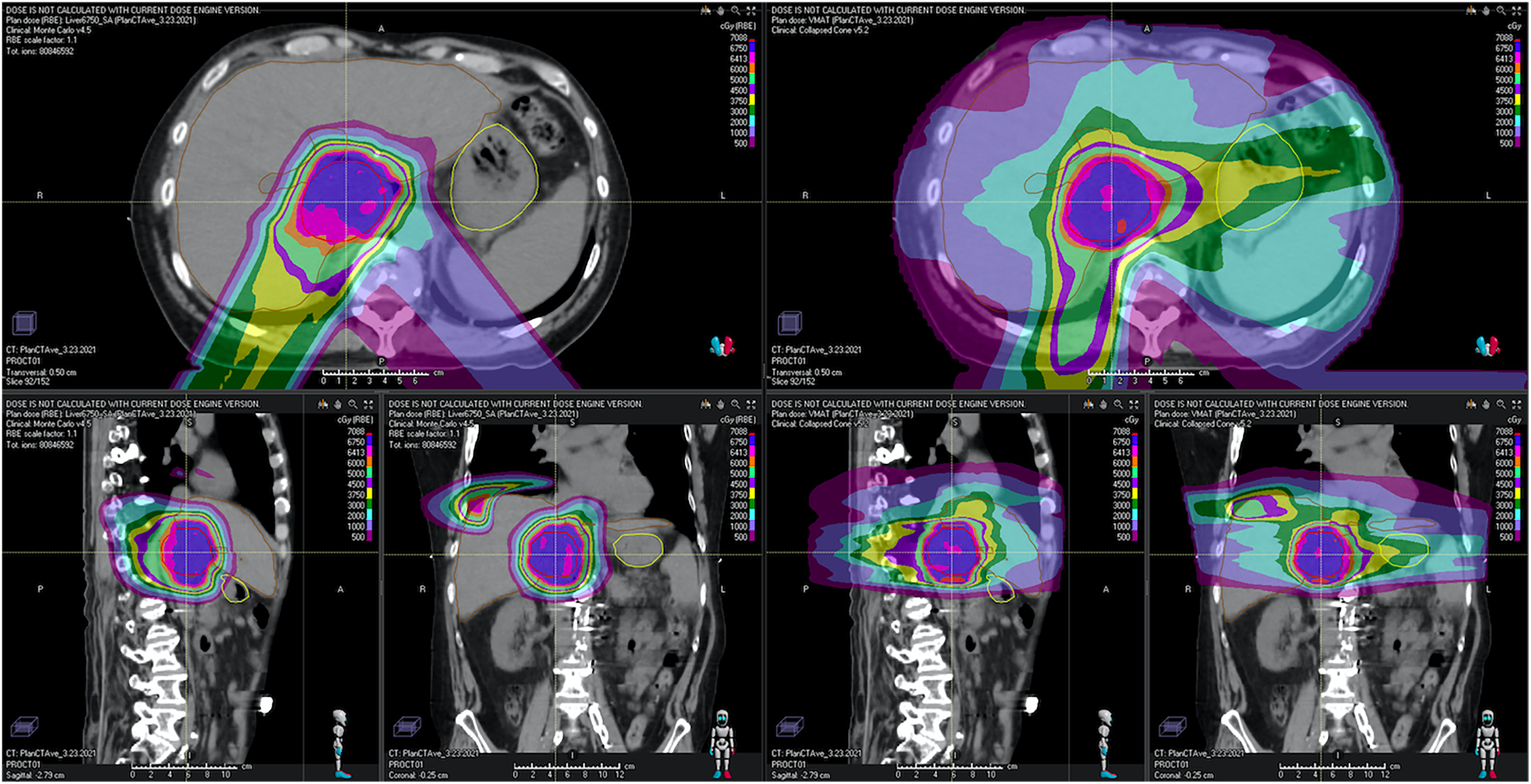
A 68-yr-old male with multifocal BCLC stage B HCC in segment 8 (1.7 cm), 1 (large, 5 cm), and 2 (1.5 cm) in the setting of hepatitis C (untreated) and alcohol-related CP-A5 cirrhosis. He was prescribed definitive PBT of 67.50 CGE in 15 fractions to all 3 lesions. This figure demonstrates significantly less normal tissue exposure to radiation (≤50 CGE) with PBT (axial, sagittal, and coronal views labeled A) compared to IMRT (axial, sagittal, and coronal views labeled B). The liver minus gross tumor volume is depicted in brown, and the same isodose line color schema is used for both plans.
Clinical Outcomes of PBT for HCC
Multiple retrospective and prospective studies of PBT for HCC have assessed if the dosimetric advantages of PBT translate to a benefit for patients and reported excellent clinical outcomes.41–53,77–82 Our PBT literature review yielded 12 prospective studies each with more than 20 patients and at least local control data at 2 yrs, which have further strengthened the rationale to consider PBT for HCC patients (Table 1). PBT achieved 5-yr LC rates of 80% to 94% and 5-yr OS rates of 39% to 70% depending on patient and tumor characteristics.48–50 The safety of PBT is well established in regard to hepatotoxicity with RILD rates of 3% to 11%.
40
Additionally, a retrospective study by Sanford and colleagues showed improved 2-yr OS (59.1% vs 28.6%) in favor of PBT compared to photon-based EBRT and suggested that this may be due to the lower risk of RILD (odds ratio 0.26;
Clinical Applications of PBT for HCC
The clinical benefit when PBT is utilized over photon-based EBRT for HCC patients has yet to be completely resolved. Some patients may benefit more from PBT than others. Ghandi et al developed a model that suggested protons would be more advantageous than SBRT either for central or dome tumors greater than 3 cm in size or those greater than 5 cm regardless of location in the liver. 85 Another study by Toramatsu et al suggested that tumors greater than 6.3 cm in size have a higher risk of RILD (94.5% vs 6.2%) when treated with IMRT rather than IMPT. 62 Even for tumors greater than 10 cm, which is larger than the typical tumor size represented in SBRT studies, Sugahara et al showed PBT had 2-yr LC of 87% and no late grade ≥3 treatment-related toxicity. 48 In addition to patients with large tumors, patients with CP-B–C cirrhosis, who often have very limited local therapy options due to the risk of liver toxicity, were observed to have better survival with PBT (1-yr OS of 53%) than what would be expected with photon-based SBRT (1-yr OS of 32%) or sorafenib (1-yr OS of 25%).35,81,86 Notably, the rate of RILD in patients with severe cirrhosis was 63% following SBRT, while no patients experienced RILD with PBT.37,81 A consensus report from a radiation oncology panel of experts suggested clinical settings where it may be appropriate to consider PBT: small uninvolved liver volume, high tumor-to-liver ratio, less compensated liver function (eg, CP-B and CP-C), and prior history of liver irradiation. 87 The results of the ongoing NRG Oncology GI-003 phase III randomized trial (NCT03186898) of IMRT versus PBT will provide level I evidence on the role of PBT in treating HCC.
Limitations of PBT for HCC
From a technical standpoint, the advantages of PBT may also be viewed as a double-edged sword. Because protons deposit the majority of their dose in a finite range, PBT may be more susceptible to various uncertainties; one has to be more cognizant of motion effects, daily positioning, and various density interfaces (eg, between air, soft tissue, and bone) that may affect the fidelity of the PBT plan, resulting in potential underdosing of tumor targets and/or overdosing of normal organs. 73 However, these uncertainties may be addressed with improved motion management and image guidance, both of which were discussed in the introduction of this article, as well as optimal beam angle selection, treatment plan robustness testing, and interval quality assurance scans.63,65–68 While development of a compact proton accelerator and single-room gantry has decreased the initial capital needed to open proton centers, another limitation of PBT is it is more expensive and, thus, not as widely available as photon-based EBRT.
Further Directions of PBT for HCC
PBS-IMPT provides an opportunity to deliver conformal PBT with a heterogeneous dose distribution. Utilizing PBS-IMPT, a simultaneous integrated boost (SIB) and protection (SIP) technique may be more feasible than with photon-based IMRT and allow for dose escalation to the part of the tumor with lower-dose levels near critical OARs.78,88 Kim et al reported results of a PBT-IMPT SIB approach in patients with advanced HCC with tumor vascular thrombosis (TVT): 2-yr local progression-free survival of 88%, OS of 51%, and no grade ≥3 toxicity. 78 Higher biologic equivalent dose was associated with better TVT response, which was an independent prognostic factor for improved OS. 78 Schaub et al reported results of a PBS-IMPT SIB/SIP technique in a population of patients with 20% CP-B or greater, nearly 50% BCLC stage C disease, 70% multifocal disease, and a median tumor size of 12.7 cm. 89 At a median follow-up of 18 months, crude LC was 94% (1-yr actuarial LC was 83%) with no isolated local failures or death from RILD. 89 This highlights a potential approach to safely allow for dose escalation when treating tumors in the proximity of critical OARs (Figure 2).

Example of a PBS-IMPT dose-painting plan for a patient with a large central HCC in close proximity to the esophagus treated to a total dose of 60 Gy in 15 fractions with dose reductions at the OAR dose limitations.
As previously mentioned for IMPT, treatment uncertainties can be addressed with various techniques. One technique currently used in photon-based RT but still in development for PBT is online adaptive therapy, which involves modifying the treatment plan to account for interfraction changes in the patient and/or tumor anatomy while the patient remains in the treatment machine. 90 Challenges involved with implementation include automated contouring, plan quality assurance, calculation times, and increased workload. 91 Yet, when online adaptive PBT is possible, radiation dose to normal tissue could be further reduced.
Another technique possible with PBT is FLASH RT, which is ultrahigh dose-rate (≥40 Gy/s) radiation initially demonstrated with electrons to potentially further limit normal tissue toxicity without compromising tumor control. 92 Similarly, several preclinical studies have shown normal tissue sparing with proton-based FLASH RT.93–96 Linear accelerators (linacs) can currently only deliver FLASH radiation using electrons and not photons due to limitations in beam current, thus limiting linac-based FLASH RT to superficial targets. Since PBT already involves accelerating protons with a cyclotron or synchrotron, PBT offers a unique ability to treat deep structures (eg, HCC) with FLASH RT. In fact, the feasibility of treating HCC patients with FLASH PBT is already being investigated. 97 Moreover, the first clinical trial involving FLASH PBT was recently published demonstrating feasibility in patients with bone metastases. Further investigation is expected to assess feasibility, normal tissue sparing, and effectiveness of FLASH PBT.
Magnetic Resonance Image-Guided Radiation Therapy (MRgRT) for HCC
History and Advantages of MRgRT
MRI has recently become available for image-guided photon-based EBRT, although not yet with PBT. 98 Proof of concept of the first MRI linear accelerator (MRI-linac) was achieved in 2009, and the first patients were treated in 2017.99–102 MRI inherently provides superior soft tissue resolution compared to CT, which has benefits in target volume and organ-at-risk segmentation for treatment planning as well as image registration on the day of treatment. Visualization is further enhanced by administrating gadoxetate disodium contrast about 20 to 30 min prior to simulation and each fraction. Active monitoring through continuous cine-MRI during treatment and soft tissue delineation with MRgRT render online adaptive replanning possible.
The potential benefits of MRgRT for liver tumors extend beyond online adaptive replanning. Due to improved visualization and real-time imaging with MRgRT, there is no need for fiducial marker placement. 103 In addition, unlike other RT modalities, MRI simulation and MRgRT for liver tumors can be performed without immobilization devices. 103 Moreover, a smaller uncertainty margin (0.3-0.5 cm) may be considered for the planning target volume (PTV), thereby leading to less irradiation of the normal tissue. 101 If breath hold or gating (available on MRIdian Linac) is used, an ITV is not needed, further leading to a smaller treatment volume. Van Sörnsen et al showed adequate target coverage could be achieved with breath-hold and real-time gated MRgRT, with only a 0.3 cm PTV expansion and without the use of an ITV, in the treatment of thoracic and abdominal tumors. 101 MR-linacs have increased versatility compared to conventional linacs since MRI simulation imaging can be acquired on the treatment machine, or departments may consider acquiring a MRI simulator, which could make more time available on MR-linacs for treatment, including online adaptive therapy. For visual representation,Figure 3 shows a case of online adaptive therapy for HCC that was near a critical organ such as the bowel.

A 55-yr-old male with 2.5 cm solitary HCC in the left lobe of the liver near the duodenum. He was treated with SBRT in breath hold on a 0.35 T MRI-linac prescribed to 50 Gy in 5 fractions. This figure demonstrates that while the original plan was able to satisfy all normal organ constraints using the anatomy from time of simulation (image on left), it was predicted that the original plan would have violated the duodenum constraint of V33 < 0.5 cc if used on the treatment day anatomy (center image), and this was resolved using online adaptive plan reoptimization while still delivering ablative dose to most of the target (image on right).
Clinical Outcomes of MRgRT for HCC
The feasibility and effectiveness of treating liver tumors with MRgRT are based on several single-arm prospective trials and retrospective studies since there have been no randomized control trials to date involving MRgRT for liver tumors.54–56,104–107 Specific to HCC patients, prospective clinical trials by Weykamp et al and Van Dams et al as well as a retrospective study by Feldman et al showed no grade ≥3 toxicity with MRgRT.54,55,106 A multicenter retrospective study by Rosenberg et al showed 100% crude LC rate at a median follow-up of 21.2 months for HCC patients treated with MRgRT. 107 A retrospective study by Boldrini et al showed a 90% crude LC rate without grade ≥3 toxicity at a median follow-up of 6.5 months in HCC patients treated with MRgRT. 105 The majority of patients in the summarized studies were treated with MRgRT without online adaptive therapy. A retrospective study by Chin et al found that online adaptive MRgRT could significantly improve PTV coverage while respecting OAR constraints, particularly when HCC was near the stomach or bowel. 108 A prospective clinical trial by Henke et al suggested the use of online adaptive therapy may improve outcomes when treating abdominal tumors with MRgRT. 56 Of all liver treatment fractions, 66% were adapted either because the initial plan would have violated an OAR constraint (36% of fractions) or there was an opportunity for dose escalation (30% of fractions). 56 Henke et al showed overall improved target coverage and normal tissue sparing could be achieved using online adaptive MRgRT, resulting in 100% crude LC of liver tumors and no grade ≥3 toxicity at a median follow-up of 15 months. 56 Results from additional prospective clinical trials (NCT04242342 and NCT04682847) involving photon-based adaptive MRgRT for primary liver cancers are anticipated.
Clinical Applications of MRgRT for HCC
While MRgRT is currently being studied in a broad variety of disease sites, it is especially helpful in the abdomen due to intra- and interfraction movement and proximity of OARs.56,109 Notably, the studies summarized in the clinical outcome section of MRgRT all involved treating patients without the use of an ITV expansion, invasive fiducial placement, or external respiratory device (eg, abdominal compression or spirometry). Therefore, MRgRT may be beneficial when these motion management techniques are not possible. Additionally, examples of OARs that may be near the liver tumors include the stomach and bowel (eg, duodenum), which have a risk of developing an ulcer or perforation if exposed to a high dose of radiation with SBRT. 110 However, SBRT delivered with online adaptive MRgRT may allow high doses of radiation to be more safely delivered even with nearby OARs. While the following study did not analyze HCC, the results are relevant since pancreatic tumors are also susceptible to motion and may be adjacent to the duodenum. Rudra et al showed online adaptive MRgRT for patients with inoperable pancreatic cancer allowed for dose escalation, which was associated with improved 2-yr OS and 2-yr freedom from local failure, without increase in grade ≥3 toxicity compared to standard dose radiation. 111 Also, as previously mentioned, the prospective trial of online adaptive MRgRT for abdominal tumors by Henke et al showed improved target coverage with the ability to dose-escalate and meet OAR constraints. 56 In summary, patients with significant motion, not otherwise addressed, or tumors near dose-limiting structures, may benefit from online adaptive MRgRT since MR-linacs can provide better soft tissue visualization compared to CT, real-time imaging during treatment, adaptive replanning at the time of each fraction, and gated treatment.54,56,101,103,105–107
Limitations of MRgRT for HCC
Currently, data on the clinical outcomes and toxicities of MRgRT in HCC patients are limited, and more studies are needed to determine its role and benefit over non-MRgRT modalities. Similar to PBT, cost and availability are limitations, but these may improve with further research and innovation. Standardized quality assurance protocols will need to be established as MRgRT becomes more common. 112 Notably, MRgRT currently is limited to machines that generate photon radiation and, therefore, still possesses the physical limitations of photons. Despite the advantages of improved target delineation, tracking, and adaptive therapy, MRgRT will still have limitations upon dose escalation in HCC patients, particularly those with more severe cirrhosis, due to the low-to-moderate dose spillage of photons in the uninvolved liver. Perhaps as technical challenges are overcome, the coalescence of PBT and MRgRT may potentially be on the horizon.98,113–116
Further Directions of MRgRT for HCC
Development of auto-contouring programs is a current area of interest as manual contouring can be laborious and time-consuming for clinicians. Improved soft tissue delineation with MRI, compared to CT, may improve anatomic segmentation. Liang et al showed feasibility and accuracy of fully automated contouring of normal structures (eg, liver, kidneys, and spinal cord) on scans obtained with a MR-linac. 117 Auto-contouring may shorten the overall treatment planning time as well as the time on the treatment table needed for online adaptive replanning.
Future technical advancements with novel imaging acquisition sequences, particularly on the MR-linac, may allow for functional adaptive radiotherapy planning while undergoing treatment. Attempts at toxicity mitigation exploring different noninvasive functional imaging modalities (eg, dynamic contrast-enhanced [DCE] CT or MRI, Gd_EOB-DTPA MRI, [18Fgalactose]-PET/CT, mebrofenin, or sulfur colloid SPECT/CT) are already underway.118–126 Functional imaging provides a quantitative objective measure of liver function as well as spatial distribution of the functional liver tissue to allow for preferential sparing of the most active liver parenchyma during radiotherapy planning, which may not always be apparent on conventional imaging with a CT or MRI. Furthermore, one could analyze images for predictors of response or toxicity. For example, Eccles et al showed that larger increases in the apparent diffusion coefficients on MRI correlated with higher doses and likelihood of response in liver tumors. 127 Since MRI scans of patients are obtained on treatment with a MR-linac, there is the possibility of biological image-guided adaptive radiotherapy (BIGART) as described by Van Houdt et al. 128 Due to its more recent development, future research opportunities for MRgRT are numerous. In the more immediate future, results from the prospective clinical trials (NCT04242342 and NCT04682847) of photon-based adaptive MRgRT for primary liver cancers are anticipated.
Conclusion
PBT and online adaptive MRgRT are emerging technologies in radiation oncology that help solidify our armamentarium against HCC by conferring the ability to deliver safer, more targeted definitive irradiation of liver tumors. These promising technologies offer specific and distinct advantages. The benefit of PBT is driven by the need to mitigate effects on radiosensitive organs: PBT reduces the integral dose of radiation to surrounding normal organs and the cirrhotic liver (due to the Bragg peak), thereby reducing the risk of RILD and potentially allowing safer dose escalation. The benefit of MRgRT with online adaptive therapy is driven by the need to address anatomical and tumor changes as well as normal organ motion: MRgRT with online adaptive therapy reduces the volume receiving prescription dose (due to smaller PTV margins and lack of ITV if using gating), thereby allowing definitive irradiation of tumors even when located adjacent to critical OARs (eg, stomach or bowel) or when there is a significant motion not otherwise managed. Both PBT and MRgRT improve the potential to personalize RT for patients with HCC. One can reference the ASTRO guidelines on primary liver cancers for recommended dose prescriptions and OAR constraints. 129 Table 2 summarizes clinical scenarios when either PBT or photon-based MRgRT may be preferred over conventional photon-based modalities. While these advanced therapies are limited by cost and accessibility, further innovation and research may address both of these issues. Future research should analyze the efficacy and side effect profile of treating HCC with PBT or photon-based MRgRT compared to conventional treatment modalities, ideally in a randomized control fashion.
Clinical Decision-Making Tool with Various Scenarios for When PBT or Photon-Based MRgRT May Be Preferred Compared to Conventional Photon-Based EBRT

Footnotes
Abbreviations
Acknowledgments
Declaration of Conflicting Interests
MDC reports honoraria, research funding, and advisory board role for ViewRay, board of directors and disease site committee chair role for Proton Collaborative Group, and honoraria from IBA. SSL reports research funding from the Kuni foundation, travel support from JASTRO, and leadership roles as Assistant Councilor and Chair of CARROS Nominating Committee for the ACR and Member of the Board of Directors and Medical Director of Distinction in Practice in Stereotactic Radiotherapy Program for the Radiosurgery Society. The author(s) declare no other potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics for Animal and Human Studies
Not applicable.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Correction (March 2024):
Article updated to correct caption of Figure 1.
