Abstract
Hepatocellular carcinoma is the fourth leading cause of cancer-related death worldwide. Depending on the extent of disease and competing comorbidities for mortality, multiple liver-directed therapy options exist for the treatment of hepatocellular carcinoma. Advancements in radiation oncology have led to the emergence of stereotactic body radiation therapy as a promising liver-directed therapy, which delivers high doses of radiation with a steep dose gradient to maximize local tumor control and minimize radiation-induced treatment toxicity. In this study, we review the current clinical data as well as the unresolved issues and controversies regarding stereotactic body radiation therapy for hepatocellular carcinoma: (1) Is there a radiation dose–response relationship with hepatocellular carcinoma? (2) What are the optimal dosimetric predictors of radiation-induced liver disease, and do they differ for patients with varying liver function? (3) How do we assess treatment response on imaging? (4) How does stereotactic body radiation therapy compare to other liver-directed therapy modalities, including proton beam therapy? Based on the current literature discussed, this review highlights future possible research and clinical directions.
Keywords
Introduction
Hepatocellular carcinoma (HCC) is the third most common gastrointestinal malignancy and the sixth deadliest solid malignancy with increasing incidence in the United States. 1 Worldwide, HCC is the fourth leading cause of cancer-related death. 2 The majority of patients with HCC have underlying chronic liver disease caused by viral hepatitis and/or nonalcoholic/alcoholic fatty liver disease. For many patients, their chronic liver disease presents a competing comorbidity risk for mortality.
Surgery remains the gold standard for curative treatment and includes either a partial hepatectomy or liver transplantation. Transplantation is associated with an 84% 2-year overall survival (OS) 3 ; however, only 15% to 30% are candidates due to tumor extent and underlying liver dysfunction. 4 For these patients, other liver-directed therapies (LDTs) are evaluated in a multidisciplinary setting with various treatment intents, such as bridge-to-transplant, definitive/curative treatment, and/or palliation. Most patients with nonmetastatic HCC receive one or more of the following LDT over the course of their treatment: radiofrequency ablation (RFA), transarterial chemoembolization (TACE), transarterial radioembolization (TARE), and external beam radiotherapy (RT). Until recently, RT was used cautiously due to the narrow therapeutic window when balancing tumor control against radiation-induced liver disease (RILD). 5
However, technological advancements in radiation oncology over the past 10 to 15 years have enabled the development of stereotactic body RT (SBRT), which delivers highly conformal dose distributions with a rapid dose drop off that offers the ability to spare large portions of the liver while simultaneously allowing for dose escalation with ablative potential within the tumor. Stereotactic body RT presents an alternative and/or combined modality to use with other LDTs. In 2000, the Barcelona Conference (BCLC) outlined the optimal treatment algorithms for primary liver cancer without inclusion of RT, because it was only until afterward that RT emerged as a promising treatment for HCC. 4 However, SBRT is now included in the most recent version of the National Comprehensive Cancer Center Network guidelines for primary liver cancer under the indication for unresectable disease or medically inoperable patients. 6
Despite the mounting worldwide evidence of SBRT as an acceptable LDT option for patients with HCC, several important issues remain unresolved. In addition to reviewing the current clinical SBRT data, we review unresolved issues and controversies regarding the use of SBRT for HCC, specifically: (1) Is there a radiation dose–response relationship with HCC? (2) Dosimetric predictors of hepatotoxicity: do Child-Pugh B patients have differing constraints? (3) How do we assess treatment response on imaging? (4) How does SBRT compare to other LDT modalities, including proton beam therapy (PBT)?
Overview of Clinical SBRT Data
Prospective clinical trials of liver SBRT have demonstrated high rates of local control (LC), typically defined as no progression of disease per RECIST criteria, ranging from 87% to 100% at 1 to 3 years (Table 1).
7
-9
Local control may decrease slightly to 82% to 100% for patients with worse baseline liver function such as Child-Turcotte-Pugh (CP)-B patients, which may be related to lower tumor dose delivered in order to decrease the risk of RILD.
7,9
-13
Evidence for the safety and efficacy of SBRT for HCC is the strongest in patients with well-compensated baseline cirrhosis (eg, CP-A). In 2006, Mendez-Romero
Summary of Prospective Studies of SBRT for HCC.a

Abbreviations: CP, Child-Pugh; f/u, follow-up; n, number of patients; fx, fraction; G, grade; GI, gastrointestinal; GTV, gross tumor volume; HCC, hepatocellular carcinoma; LC, local control; LDT, liver-directed therapy; NR, not reported; OS, overall survival; PVTT, portal vein tumor thrombosis; SBRT, stereotactic body radiation therapy.
a At 1 year unless otherwise stated.
Culleton
Cardenes and colleagues at Indiana University analyzed the results of a phase I/II trial of SBRT for HCC restricted to CP-A (n = 38) and CP-B7 patients (n = 21) with lesions treated to a median total dose of 48 Gy in 3 fractions (range: 36-48) and 40 Gy in 5 fractions, respectively.
9
One-year LC remained excellent at 91% in CP-A patients with a modest decrease to 82% in CP-B7 patients. Consistent with the results from Princess Margaret demonstrating increased toxicity in CP-B patients, Indiana University reported rates of grade 3+ liver toxicity of 11% versus 38% in CP-A and -B patients, respectively. Similarly, Scorsetti
Besides demonstrating the prognostic significance of the CP score, these studies showed that PVTT, multinodular disease, and high serum AFP portend poor prognosis. Since these are often considered relative or absolute contraindications for other non-SBRT LDT, this makes direct comparisons between SBRT and other LDT challenging. Additionally, many patients undergoing SBRT were previously treated with other LDTs (range: 44-100%), which may confound local control, intrahepatic control, and hepatotoxicity reporting. 7, 11, 13
The presence of PVTT, which often prohibits the feasibility of other LDTs, has been analyzed specifically in the context of SBRT.
14
-16
Kang
Recently, Yoon
A limitation of SBRT for HCC is the lack of long-term prospective data beyond 2 years. At a median follow-up of 28.7 months, Kwon
Radiation Dose Response and the Role of Dose Escalation
In the era of rapid technological advancement, the technical ability to escalate dose may be outstripping our knowledge of whether dose escalation is beneficial in SBRT for HCC. Our understanding of the nature of the dose response for HCC is based on tumor control probability (TCP) models, expert opinion, and heterogeneous clinical case series reports. Understanding the dose response curve, if one exists, provides clinicians the ability to appropriately escalate dose to provide a defined amount of incremental tumor control. This escalation often comes at the price of increased risk of toxicity, which in the case of hepatic irradiation can be fatal. 20 For example, if the dose response is sigmoidal, small changes in dose in the middle dose range of the curve may result in large increases in TCP. However, if the dose increase is entertained at the upper or lower flatter portions of the curve, large dose escalations may provide minimal improvement in TCP at the price of an increasing risk of hepatotoxicity.
Dose response models and literature review
As the optimal dose is unknown and varied dose data are relatively limited in this heterogeneous patient group, models were developed to clarify whether a dose–response relationship exists for HCC. Lausch
In contrast to these models, recently published TCP model developed with multi-institutional data for 394 patients with HCC by Ohri
Most current models that guide our dose selection are derived from single institutions and small data sets. Does the broader literature confirm the conclusions? The earliest data suggesting a dose response comes from hepatic irradiation case series trials, where a wide range of doses were used within a dose escalation maximally tolerated dose (MTD) protocol or in radiobiologically guided studies.
We performed a systematic literature review of trials with LC data at 2 years, with limited dose ranges to allow for an appropriate link with outcome and of sufficient size to avoid individual patients from skewing the data (defined as >50 patients). The trials in this review included terms “hepatocellular and radiation” and “liver and SBRT” in a PubMed search performed on January 19, 2018. Equivalent dose if given in 2 Gy fractions and BED, assuming an α–β ratio of 10, were calculated for all trials to qualitatively identify any dose response. Based on this review, these models fit well with a review of the literature, where an ordering of published data by local control relative to dose suggests that 2-year LC rates of 90% can be achieved with common dose regimens such as 40 to 48 Gy in 3 fractions and 35 to 40 Gy in 5 fractions (Table 2). Given the importance of CP score, many institutions recommend higher fractionation schemes for CP-B7+ patients. The EQD2 range for these regimens is between 50 Gy and 78 Gy. One important caveat to this comparison and the Ohri TCP model 23 is that this method of trial selection results in a collection of trials with an arguably narrow range of doses and apply only to relatively small lesions. Trials with a wider range of doses and tumor sizes, such as the trial by the Princess Margaret, 7 have more varied outcomes that differ from the majority of included trials.
Summary of HCC Radiotherapy Studies in Order of Local Control at 2 Years.a
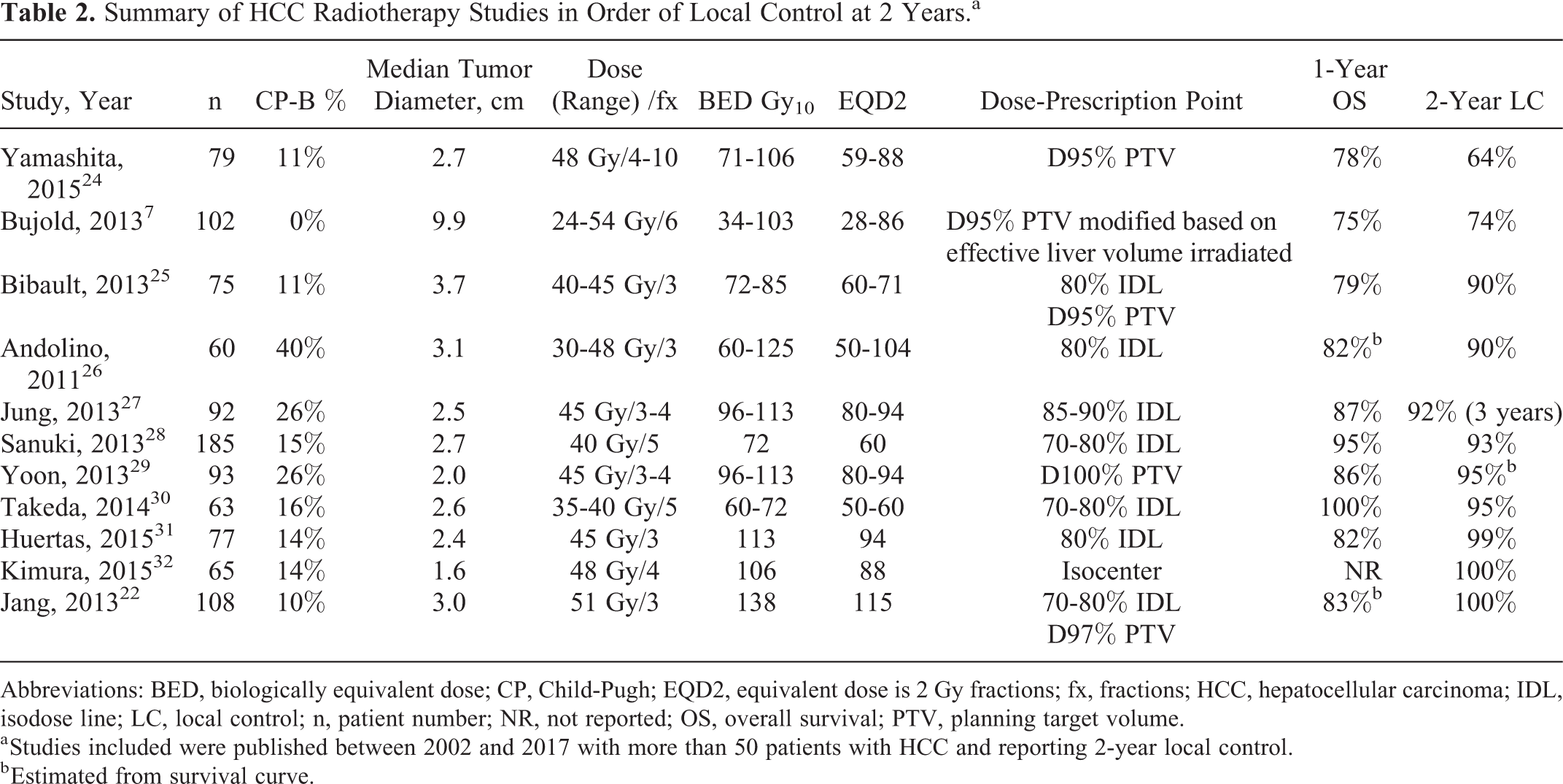
Abbreviations: BED, biologically equivalent dose; CP, Child-Pugh; EQD2, equivalent dose is 2 Gy fractions; fx, fractions; HCC, hepatocellular carcinoma; IDL, isodose line; LC, local control; n, patient number; NR, not reported; OS, overall survival; PTV, planning target volume.
a Studies included were published between 2002 and 2017 with more than 50 patients with HCC and reporting 2-year local control.
b Estimated from survival curve.
Dose escalation and LC in the context of survival and toxicity
Even if a significant dose–response relationship existed for LC, another critical issue to consider is whether LC with SBRT is associated with improved OS. In a 2013 prospective report on 102 patients with HCC (CP-A 100%) series by Princess Margaret, Bujold
In a similar trial in North American patients, the Indiana University group also used 3 fractions in a formal dose escalation protocol.
8
Starting at 36 Gy in 3 fractions, this group increased dose in 2 Gy per fraction steps and found that 48 Gy in 3 fractions (BED = 125 Gy10, EQD2 = 104 Gy) resulted in excellent results (2-year LC rate of 90%) and minimal toxicity in CP-A patients. Compared to the Korean data by Jang
Patients with HCC often have underlying cirrhosis or have tumors located near important organs at risk such as gastroduodenal or hepatobiliary structures. This may limit the ability to dose escalate in a large portion of patients. The CP-B patients and/or those with low platelets have less dose–volume dependency and may be harmed by increasing radiation dose. Data from Indiana University, among others, have demonstrated an increased risk of toxicity for patients with hepatic insufficiency as measured by the CP score. 8 The Indiana University group went further and was able to determine an MTD of 40 Gy in 5 fractions (BED = 72 Gy10, EQD2 = 60 Gy) for CP-B patients, and a CP score greater than B7 was associated with an increased risk of RILD. Therefore, the question of dose response beyond 60 Gy EQD2 in this situation may be moot, as no further dose escalations are possible in this higher risk population. Given that patients in CP-A group were able to tolerate up to 48 Gy in 3 fractions (BED = 124 Gy10, EQD2 = 104 Gy) and achieve good results, should techniques such as PBT be considered to provide more normal liver sparing and a higher chance of dose escalation in patients with hepatic insufficiency? Perhaps, this may not be a useful strategy, as regression data from LRCP suggested that CP-B patients do not benefit from dose escalation. 34 In contrast, in that same study of 195 patients, dose escalation was found to provide a significant improvement in OS for CP-A patients.
Limitations and caveats
There are limitations to this investigation of whether a dose response or at least a threshold dose exists for HCC. As noted in Table 2 and in other reviews, 35 the published literature may have a range of doses that might reflect a narrow portion of the dose–response curve. Despite the ability to achieve higher doses of significantly over 100 Gy EQD2 in certain patients, clinicians are rightly reluctant to give these doses routinely. Therefore, there are fewer high dose data points, which limit the ability to model the dose–response curve. 21,22 Furthermore, outcomes may be driven by factors other than dose alone. Nomograms and multivariable models 34 demonstrate that liver function and tumor size are much more significant in determining survival compared to an increase in radiation dose. This is consistent with the Japanese data from Yamashita who retrospectively studied 79 patients with HCC from 7 Japanese institutions. 24 They found no difference in LC with doses above or below 100 Gy10. Their 2-year LC rate was 75% with a median BED of 106 Gy10. However, there was a statistically important difference in outcome when comparing lesions above and below 3 cm in maximum diameter (LC 64% vs 85%). Size is a predominant factor in outcome. The dose response noted above may simply reflect the size variation in trials and the ability to give a higher dose to smaller lesions (Table 2).
The clinical benefit of radiation, particularly OS, may be driven less by the tumor–dose control interaction and more by the underlying fitness of the patient and hepatic insufficiency, especially in CP-B/C patients. Therefore, these patients treated with a lower BED may not display a typical dose–response relationship seen in other tumors and patient groups due to the fragile nature of their underlying cirrhosis and competing risk factors for mortality, which could lead to erroneous dose–response relationship in some studies. 36 Despite the knowledge that dose is correlated with LC, and LC is correlated with OS, 22,37 only a minority of patients derive this survival benefit. This highlights the need for better patient selection methods, such as nomograms 34,38 and/or biomarkers. 39
Conclusion: Is there a dose response for HCC?
Hepatocellular carcinoma is a relatively radiosensitive tumor in contrast to the relatively poor clinical prognosis of the patient group. In terms of LC and OS, the collective data suggest a threshold of 30 Gy EQD2 below which the impact of radiation is muted. Between approximately 53 and 84 Gy EQD2, the LC rates increase from 50% to 90%. Beyond 84 Gy, the degree of incremental LC improvement decreases while, depending on the anatomy, there is a continued incremental risk of toxicity. However, there is evidence that escalating the dose further may lead to a survival benefit in some patients based on correlation data from nonrandomized studies. Current dose recommendations such as 40 to 45 Gy in 3 fractions or 40 to 50 Gy in 5 fractions, which are within the 53 to 84 Gy EQD2 range, have been demonstrated to be safe and provide consistently high LC rates. Additional work is required to determine an optimal dose regimen for different subgroups.
Dosimetric Predictors of Hepatotoxicity: All for One and One for All?
Historically, conventional definitive fractionated radiation therapy to the liver resulted in a high incidence of “classical“ RILD (cRILD), defined as hepatomegaly, anicteric ascites, and alkaline phosphatase elevated disproportionately compared to other hepatic enzymes.
40
Despite the technological advancements in radiation therapy with SBRT and proton RT,
39
3% to 44% of patients remain at risk of developing “nonclassical” RILD (ncRILD), which can be fatal in up to 5% to 13%.
7,9,10,20,41
-45
Various empiric end points have been used to describe ncRILD, including changes in CP score
44,46,47
and Radiation Therapy Oncology Group (RTOG)/Common Terminology Criteria for Adverse Events (CTCAE) grade 2 to 4 laboratory abnormalities.
9,43,48
The large variation in the reported rates of ncRILD suggests the need for a standardized consensus on reporting of ncRILD end points.
44
One such end point may be CP score increase of 2 or greater (CP + 2). Chapman
At present, most of the data for dosimetric predictors of ncRILD are derived from patients with well-compensated liver function. In 2010, Quantitative Analysis of Normal Tissue Effects in the Clinic reported recommendations regarding the importance of mean normal liver dose of <18 Gy and <6 Gy for a 6-fraction SBRT regimen in patients with primary liver cancer with baseline CP-A and CP-B function, respectively. 39,49 These early recommendations were largely based on a collection of studies using conventional or hyperfractionation studies for the treatment of primary liver cancer and/or metastatic disease to the liver 50 as well as the early SBRT experience from Princess Margaret 51,52 and University of Colorado. 53 The University of Colorado phase 1 clinical trial of SBRT for liver metastases was the first to describe the importance of the liver volume spared, that is, the “critical volume model,” (eg, critical minimum volume of 700 cc of liver should be uninjured by SBRT by receiving <15 Gy) a concept akin to surgical sparing of the future liver remnant. 53 This critical volume of liver spared concept has also been applied to patients with HCC.
Recent studies have reported significant heterogeneity in the types and cutoffs of dosimetric parameters correlated with RILD (Table 3). Several possible reasons why such heterogeneity exists include (1) limited sample size and RILD events in cohorts limiting statistical power, (2) variations in reporting of RILD outcomes, (3) the subjective nature of baseline liver assessments with traditional metrics (eg, CP-score), 54 and (4) dosimetric models are based on anatomic computerized tomography (CT) imaging and do not incorporate regional functional heterogeneity within the liver. The last point is particularly relevant as many patients with HCC referred for SBRT have significant regional heterogeneity, partially due to prior LDT. 55 -57 Future studies would benefit from consensus reporting of toxicity outcomes and validation of objective parameters to evaluate liver function in the setting of RT. There is a need for validated global and spatial functional liver parameters that can be used for guiding precision RT treatment planning.
Summary of the Dosimetric Constraints of the Uninvolved Liver for Minimizing Risk of Radiation-Induced Liver Disease (RILD).

Abbreviations: CP, Child-Pugh; CP + 2, CP score increase of 2 or greater; CP + class, CP class progression; fx, fractionation; G3-4 HE, grade 3-4 hepatic enzyme elevation; V<X Gy, absolute normal liver volume (cc) spared from at least X Gy.
Velec
These studies were predominantly composed of well-compensated CP-A patients. Whether applying these dosimetric parameters to patients with more severe baseline liver disease (CP-B/C) results in the same risk of RILD is uncertain. Indiana University reported toxicity data from their phase I to II trial of SBRT for HCC in CP-A (n = 38) and -B (n = 21) patients.
9
Due to increased toxicity observed in patients with CP > B7 in the phase I trial (3 events of classical RILD),
8
the phase II study included only patients with CP ≤ B7 and tailored dosimetric constraints to baseline CP score. For a treatment regimen of 48 Gy in 3 fractions, CP-A patients were required to either limit the dose to 33% of the uninvolved liver (D33%) ≤ 10 Gy and/or maintain the liver volume receiving < 7 Gy to ≥500 cc. For a more conservative treatment regimen of 40 Gy in 5 fractions, CP-B7 patients had to meet constraints of D33% ≤ 18 Gy and/or > 500 cc receiving < 12 Gy. Dosimetric correlates were identified for grade 3 to 4 hepatic enzyme toxicity observed in 10.5% and 38.8% of CP-A and CP-B patients, respectively. For CP-A patients, there was a trend toward importance of the dose to 500 cc (
Caution when treating CP-B/C patients (CP-B7 69%) with SBRT was further supported by Princess Margaret’s prospective trial of 29 CP-B/C patients treated with a mean dose of 30 Gy in 6 fractions. 10 Sixty-three percent of these patients experienced ncRILD as determined by CP + 2 within 3 months. For this study, the planning liver dosimetric parameters were based on the biological NTCP model with the goal of <5% and preferably less than <1% in fragile patients 59 ; however, there were no significant predictive dosimetric factors found on UVA.
Conclusion: Dosimetric predictors of hepatotoxicity—all for one and one for all?
The limited data available regarding CP-B patients consistently suggest they are at heightened risk of RILD, and dosimetric parameters ought to be more conservative than for CP-A patients. The optimal liver dose constraints for CP-B+ patients, however, remain unclear at this time and highlight the need for additional research in this patient subgroup.
Image Response Assessment
The Liver Imaging Reporting and Data System (LI-RADS), initially launched in 2011 and recently updated in 2017, standardized the terminology and classification of liver lesions. 60,61 The following characteristic imaging features of HCC are considered sensitive and specific enough to diagnose HCC independently (LI-RADS category 5) without requiring pathologic confirmation: mass-like, arterial-phase hyperenhancement, with either portal venous phase hypoenhancement and/or increase in size by ≥5 mm by 6 months, or definite tumor within the lumen of the hepatic veins.
The LI-RADS v2017 introduced 4 additional criteria specific for treatment response (LR-TR): (1) LR-TR nonevaluable due to poor image quality; (2) LR-TR viable for treated lesions with residual enhancing nodular, mass-like, or thick irregular tissue in or along the margin of the treated lesion with any of the following: arterial phase hyperenhancement, washout, or enhancement similar to pretreatment enhancement; (3) LR-TR nonviable for lesions without enhancement or a treatment-specific expected enhancement pattern; and (4) LR-TR equivocal for lesions with atypical enhancement pattern not meeting the other criteria. 61 The LR-TR is based on the modified Response Evaluation Criteria in Solid Tumors (mRECIST), and European Association for the Study of Liver Diseases (EASL) which define response assessment based on the volume of residual arterial hyperenhancement as a surrogate for tumor necrosis. 4,62 -64
Historically, RECIST v1.1 criteria relied on changes in tumor size for posttreatment assessment which failed to detect nearly all complete responders (CRs) and underestimated the number of partial responders (PRs). 62,65 Response Evaluation Criteria in Solid Tumors v1.1 demonstrated poor concordance to newer response criteria, such as mRECIST and EASL, which define response based on the volume of residual arterial hyperenhancement as a surrogate for tumor necrosis. 4,62,63 In mRECIST criteria, 100% disappearance of arterial phase hyperenhancement is considered a complete response (CR), while 30% decrease is considered a partial response (PR), and progressive disease is defined as 20% increase. 64 If none of these changes are present, then the tumor is considered to be stable. The mRECIST, EASL, LR-TR criteria may be accurate for ablative, embolic, or systemic therapies, but application of these criteria to lesions treated with SBRT is unclear. 66 Figure 1 demonstrates a characteristic CP-A5 patient with HCC treated with SBRT and the associated treatment response assessment over time.

Characteristic arterial phase T1 MR imaging for a Child-Pugh A5 patient with HCC (arrow) treated with SBRT to 50 Gy in 5 fractions are shown: pre-SBRT (A), 6-weeks post-SBRT (B), and 4-months post-SBRT (C). Below each MR image is a correlative schematic to demonstrate either the corresponding LI-RADS diagnostic category (D), or treatment response assessment criteria of LI-RADS treatment response (LI-TR) and the modified RECIST criteria (mRECIST) (E-F). HCC denotes hepatocellular carcinoma; LI-RADS, Liver Imaging Reporting and Data System; LI-TR, Liver Imaging Treatment Response; RECIST, Response Evaluation Criteria in Solid Tumors; SBRT, stereotactic body radiation therapy.
Although the majority of HCC SBRT clinical trials continue to employ the mRECIST criteria for response assessment, there is an unmet need for a standardized response assessment system designed to address the unique imaging changes observed in HCC after SBRT.
28
Mendiratta-Lala
Other reports have described similar findings suggesting the importance of timing with regard to imaging response assessment using multiphase CT or MRI scans (Table 4). Sanuki
Summary of Key Studies Evaluating Imaging Treatment Response After SBRT with Multiphasic CT and/or MRI.

Abbreviations: APHE, arterial phase hyperenhancement; CP, Child-Pugh; CR, complete response; n, patient number; CT, computerized tomography; EASL, European Association for the Study of Liver Diseases; MRI, magnetic resonance imaging; NR, not reported; OLT, orthotopic liver transplantation; PR, partial response; κ, weighted κ statistic to analyze concordance; RECIST, Response Evaluation Criteria in Solid Tumors; mRECIST; modified Response Evaluation Criteria in Solid Tumors; SBRT, stereotactic body radiation therapy.
a Imaging–pathology correlation.
Price
The imaging characteristics of HCC tumor response after SBRT is distinct compared to the changes seen in the surrounding uninvolved liver parenchyma. Regarding normal hepatic parenchyma changes, Park
Conclusion: Imaging response assessment
Collectively, these studies highlight the potential pitfalls of radiographic response assessment after SBRT. Common observations across studies include (1) optimal time for response assessment is at least 6 to 12 months after SBRT; (2) stability or decrease in lesion size is associated with successful local control; (3) arterial phase hyperenhancement may persist despite pathologic CR 67 ; and (4) washout on delayed phases may persist after SBRT. If the RECIST, mRECIST, EASL, or LI-RADS TR v2017 criteria had been applied, many of these lesions would have been improperly categorized as treatment failures potentially leading to unnecessary additional therapies.
Comparison With Other Local Treatments
With so many LDT options, the question is often which LDT is optimal for patients ineligible for liver transplantation or surgical resection. Unfortunately, there currently is a paucity of head-to-head prospective randomized data, so answering this question often relies on synthesizing the results of various retrospective nonrandomized studies each with their particular weaknesses. Table 5 contains a brief summary of studies comparing SBRT alone or in combination with other modalities versus other LDT. The heterogeneity in patients, LDT techniques, and end points reported further challenges drawing definitive conclusions.
Summary of Key Clinical Data of SBRT Compared to Other Liver-Directed Therapies.

Abbreviations: BED, biological equivalent dose; CP, Child-Pugh; cTACE, complete TACE; curative, includes surgery, RFA, and percutaneous ethanol injection; DMFS, distant metastasis free survival; EQD2, equivalent dose in 2 Gy fractions; GI, gastrointestinal; HCC, hepatocellular carcinoma; iTACE, incomplete TACE; LDT, liver-directed therapy; LRFS, local recurrence free survival; MST, median survival time; n, patient number; NCDB, National Cancer Database; non-curative, includes TACE, sorafenib, or chemotherapy; NR, not reported; OS, overall survival; PFS, progression-free survival; PVT, portal vein thrombosis; RFA, radiofrequency ablation; SBRT, stereotactic body radiation therapy; TACE, transarterial chemoembolization; TAE, transarterial embolization; Tx, treatment; WHO, World Health Organization.
a Statistically significant.
Surgical resection for medically operable patients with small early-stage HCC remains first-line treatment. There are no prospective data comparing the efficacy of resection to SBRT. Su
Although no randomized evidence exists comparing RFA to SBRT in the treatment of early-stage HCC, there are several nonrandomized comparative cohort studies. In Japan, a single-center pilot trial of SBRT versus RFA was performed in 73 patients with solitary HCC with CP-A to B8 cirrhosis (≤3 cm for RFA and ≤5 cm for SBRT) treated to 60 Gy in 3 to 5 fractions (dose reduction based on tumor size).
82
Despite the SBRT cohort representing patients deemed unfit medically due to comorbidities, tumor location, and/or size >3 cm for RFA, there were no significant differences in 1-year local control (97% vs 97%) and OS (95% vs 100%) between SBRT and RFA, respectively. Wahl
For BCLC class B and C patients without PVTT, TACE is considered by many to be the standard of care treatment. A propensity score analysis compared TACE versus SBRT in patients with 1 to 2 tumors found superior 2-year LC with SBRT (91% vs 23%).
75
However, this large difference in LC did not translate into an OS benefit. Several studies have examined the potential role of combining SBRT with other LDTs. In a retrospective cohort study, Su
Transarterial chemoembolization, RFA, and SBRT have all been used as a bridge to transplantation. A retrospective cohort analysis testing TACE, RFA, and SBRT showed no survival difference between them in patients treated as bridge to transplantation at Princess Margaret.
81
Of note, most patients treated by RFA (88%) met the Milan criteria for transplant, indicating they had limited stage disease. However, only 24% and 36% of TACE and SBRT patients met these criteria, respectively. A retrospective study by Mannina
Besides TACE, RFA, and SBRT, TARE is another LDT option for BCLC stage B/C tumors. Transarterial radioembolization involves administering yttrium-90 (Y-90)-containing microspheres through arterial catheters to the hepatic arteries feeding the tumor, enabling high radiation dose in the tumor and minimal exposure to normal tissues. Two large phase III trials tested selective internal radiation therapy versus sorafenib. 83 Selective internal radiation therapy resulted in lower risk of progression in the liver and higher local response rates but no difference in OS.
Conclusion: Comparison with other local treatments
Most studies comparing 2 or 3 LDTs are retrospective and thus have low control of confounding variables. Even with matched analyses of large databases, the risk of imbalances between treatment groups is high. Despite many retrospective studies, the need for randomized trials to clarify the roles of SBRT, TACE, and RFA in HCC remains. A randomized phase III trial of sorafenib alone versus SBRT followed by sorafenib is currently ongoing (RTOG 1112, NCT01730937).
Comparison With PBT
The low radiation tolerance of cirrhotic liver tissue can limit the utilization of photon-based radiation therapy in treatment of HCC. 51 Due to the risk of RILD or deterioration in CP score, which in some cases may be fatal, 7 only a small number of patients with relatively few small tumors may be eligible for SBRT. Data exploring the critical volume model discussed earlier in this review demonstrate reduced risk of toxicity by limiting low-dose radiation to uninvolved liver as much as possible. However, due to the intrinsic physical properties of photon or X-ray-based SBRT, there are limits to how much the low-dose volume can be reduced. Alternatively, the physical properties of PBT allows substantial decrease in low to moderate dose to surrounding tissue without compromising high-dose coverage of tumor and may make some patients with HCC ideal candidates for PBT.
Delivering protons to hepatic targets is technically challenging due to liver motion with breathing. The proton treatment technique of pencil beam scanning (PBS) allows better high-dose conformity to the target compared to the older more ubiquitous passive scattering proton therapy (PSPT) technique, but PBS is potentially more susceptible to interplay effects of liver movement. Successfully accounting and/or controlling motion is mandatory to use PBS for liver tumors. Breath-hold, gating, and repainting technologies may be solutions, but they are dependent on real-time monitoring of target motion with equipment that is fully integrated with the treatment machine (“real-time target monitoring“). However, real-time target monitoring is not available at most proton facilities, as image-guided RT is relatively immature in PBT compared to SBRT. Thus, most published work with PBT for HCC uses PSPT and only few proton centers treat liver tumors.
In a phase I dose escalation study of unresectable HCC using moderately hypofractionated regimens of 60 to 72 Gy in 20 to 24 fractions, only 1 of the 27 patients had a 1-point increase in CP score, and no major hepatotoxicity was present. 87 There were no statistical differences in LC or OS between the dose schedule arms. Loma Linda and Massachusetts General Hospital published their phase II results showing favorable LC, PFS, and OS rates with 63 Gy or 58 to 67.5 Gy, both in 15 fractions. 88,89 Local control rates were as high as 95% at 2 years. Overall survival rates was also high; in a selected group of patients treated with PBT and subsequent liver transplant, 70% survived at 3 years. An interim analysis of a randomized trial testing PBT versus TACE in 69 patients with inoperable HCC from Loma Linda revealed excellent control rates of 88% and PFS and OS of 48 and 58%, respectively, among PBT-treated patients at 2 years. 90 There was a nonsignificant trend for improved LC and PFS in patients treated with PBT.
A meta-analysis of 73 cohorts from 70 observational studies showed heavy-ion therapy and SBRT resulted in similar survival which was superior to that of conventional photon RT for HCC. 91 Heavy-ion therapy resulted in the lowest risk of complications (6% vs 10% overall acute toxicity and 3% vs 6% late toxicity, heavy ion therapy vs SBRT, respectively). However, these results are based on a comparison of very heterogeneous patient populations and treatment techniques, including substantially different total dose (median dose: 69.3 GyE charged particle therapy vs 37 Gy SBRT vs 50.9 Gy conventional photon RT).
Conclusion: Comparison with PBT
Where photon therapy of HCC is clearly limited in terms of volume and target numbers, PBT may allow for treatment of more advanced cases of HCC. Kim
Summary
Hepatocellular carcinoma is a common cause of cancer-related death worldwide. Treating HCC is challenging and complex because of the natural history of the disease itself and the prevalence of advanced comorbidities seen in patients with HCC. Patients who are ineligible for liver transplant or partial hepatectomy have many different LDT options. Choosing an option that maximizes clinical benefits and limits risk of toxicity is essential. Stereotactic body radiation therapy has emerged as an effective LDT for properly selected patients with HCC having excellent rates of LC and minimal treatment associated morbidity, especially in those with well-compensated liver function (eg, CP-A). Technological advancements in SBRT and PBT have facilitated dose escalation, despite the inherent radiosensitivity of normal hepatic parenchyma. However, whether further dose escalation is beneficial (eg, beyond 84 Gy EQD2) remains controversial. Further study is necessary to fully characterize and understand the HCC dose–response curve. One key limitation for dose escalation is the need for validated dosimetric parameters predictive of RILD. To achieve this, objective global and spatial metrics of baseline liver function that have been validated in SBRT-treated patients are warranted, in addition to a standardized consensus regarding reporting hepatotoxicity endpoints. Current available data suggest patients with CP-B7+ cirrhosis ought to have more conservative dosimetric constraints because they are at elevated risk of RILD. A need remains for validated consensus criteria to improve accuracy of post-SBRT imaging-based response assessment to minimize the risk of over-treatment. Lastly, despite the challenges with comparing SBRT to other treatment modalities in the absence of randomized data, SBRT appears to be an effective LDT for local control with a safe toxicity profile in well-selected patients, and further work is ongoing regarding the role of SBRT in the setting of combined modality treatments. Ongoing phase III clinical trials will address the questions of SBRT versus PBT (NRG-GI003, NCT03186898) and sorafenib with/or without SBRT (RTOG 1112, NCT01730937).
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Michael I. Lock has received fees as a consultant or research funding from Accuray Incorporated, 3 M Canada, and Abbvie Corporation.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
