Abstract
Background:
Colorectal cancer is one of the leading causes of cancer-related death worldwide, but its mechanism has not been clarified clearly. Microfibrial-associated glycoprotein 2 is mainly located in extracellular matrix, and its role in colorectal cancer is obscure.
Methods:
Immunohistochemical staining and quantitative real-time polymerase chain reaction were used to compare the expression level of microfibrial-associated glycoprotein 2 in colorectal cancer tissues and adjacent tissues. Western blot was used to detect the expression of microfibrial-associated glycoprotein 2 in colorectal cancer cell lines and normal colonic epithelium cell line. Kaplan-Meier analysis and χ2 test were applied to evaluate the potential of microfibrial-associated glycoprotein 2 to function as cancer biomarker. Lentiviral transduction was used to induce microfibrial-associated glycoprotein 2 overexpression in HCT116 cells and NCM460 cells, followed by detecting cell proliferation, migration, and invasion. Quantitative real-time polymerase chain reaction was used to investigate the changes in downstream genes after microfibrial-associated glycoprotein 2 overexpression. Luciferase assay was conducted to validate whether miR-200b-3p can directly target microfibrial-associated glycoprotein 2.
Results:
We validated that microfibrial-associated glycoprotein 2 was upregulated in colorectal cancer samples and cells. We also demonstrated its upregulation was associated with several clinicopathologic features such as Dukes stage (P = .048), differentiation status (P = .034), and local lymphatic metastasis (P = .036) of patients with colorectal cancer, and its high expression indicated shorter overall survival of the patients. Microfibrial-associated glycoprotein 2 overexpression remarkably promoted cell proliferation and metastasis via regulating the downstream genes of Notch, including hes family bHLH transcription factor 1 (HES1), Slug, Snail, matrix metalloproteinase 2, matrix metalloproteinase 9, and Kruppel-like factor 4. We also identified miR-200b-3p as a posttranscriptional regulator of microfibrial-associated glycoprotein 2, which partly explain the high expression mechanism of microfibrial-associated glycoprotein 2 in cancer tissues.
Introduction
Extracellular matrix (ECM) provides both the structure and the signals that modulate biological behavior of cells, and recent studies have established the importance of the remodeling of ECM in cancer progression. 1 Microfibrial-associated glycoprotein 2 (MAGP2), also called microfibrillar-associated protein 5 (MFAP5), is a 25-kD protein mainly located in ECM. 2,3 It participates in bone growth, cardiovascular development, alveolar elastogenesis, and other biological process. 4 -6 Its N-terminal contains an Arg-Gly-Asp sequence (RGD) domain, cross-linked with elastic fibers in ECM and modulating the structure and functions of it, and its C terminal can activate Jagged1, which is an important ligand for Notch-1. 2,3 It is considered as a crucial regulator for cell adhesion and motility and participates in signal transduction (including Notch signaling) between many kinds of cells and their ECM. 2,4,5 In cancer biology, MAGP2 was reported to be negatively correlated with prognosis of patients with ovarian cancer. 7 Its aberrant expression or activation was proven to be involved in the tumorigenesis and progression of many other cancers such as tongue cancer, cervical cancer, breast cancer, and cholangiocellular carcinoma. 8 -11 Microfibrial-associated glycoprotein 2 secreted by cancer cells was demonstrated to promote cancer cell proliferation, motility, chemoresistance, and angiogenesis. 10
Colorectal cancer (CRC) is one of the most common cancers in Western countries. 12 In some rapidly developing countries including China, CRC also becomes a serious threat to human health. 13 In recent years, mounting studies have unveiled a complex genetic network involved in CRC initiation and progression. However, the mechanism of CRC tumorigenesis has not been clearly clarified. Extracellular matrix has been demonstrated to be a modulator for CRC cells proliferation, metastasis, and chemoresistance; and Notch signaling is also oncogenic in CRC. 14 -17 However, the expression and function of MAGP2 in CRC have not been investigated. In this study, we aimed to explore the role of MAGP2 in CRC. We demonstrated MAGP2 was significantly upregulated in CRC tissues compared to adjacent tissue, and the increase in it promoted malignant phenotypes of CRC cells including proliferation, migration, and invasion. Microfibrial-associated glycoprotein 2 can regulate the downstream genes of Notch, including hes family bHLH transcription factor 1 (HES1), Slug, Snail, matrix metalloproteinase (MMP) 2, MMP9, and Kruppel-like factor 4 (KLF4). We also proved that its aberrant expression in CRC is partly due to the decrease in miR-200b-3p. Our results implied that MAGP2 could be a potential biomarker and therapy target for this disease.
Methods and Materials
Cell Culture, Culture Conditions, and Antibodies
SW-60, Lovo, HCT-116, NCM460, and HEK293 cells were grown and routinely maintained in Dulbecco modified Eagle medium (DMEM) supplemented with 10% fetal bovine serum (FBS), 100 U/mL penicillin, and 100 mg/mL streptomycin. Cells were cultured at 37°Cin 5% CO2 and 95% humidified air. For establishment of stable MAGP2 overexpressing cells, overexpressing plasmid was constructed by Inovogen, China; then, the plasmid was packed by a lentivirus system (Inovogen, China). After removing the medium of the cells, the lentiviral supernatant was added (with 6 μg/mL polybrene). After 1 day, the supernatant was removed, and the cells were screened for 2 weeks using 10 μg/mL puromycin (Sigma-Aldrich, Shanghai, China). Anti-MAGP2 antibodies were purchased from Abcam (ab171737; Shanghai, China); anti-β-actin antibodies were purchased from Affinity Biosciences (AF7018; Guangzhou, China).
Patients and Immunohistochemical Staining
Our study was approved by the ethics review board of Affiliated Tumor Hospital of Guangxi Medical University (approval no. GMUTH-2016-0135). The need for patients’ written informed consent was waived by the ethics review board of our hospital, and no personal information of the patients was used in this study. All CRC samples and paired adjacent tissues used in immunohistochemical (IHC) and real-time polymerase chain reaction (RT-PCR) were retrieved from the Department of Pathology of our hospital. Deparaffinized sections were treated with 3% H2O2 and subjected to antigen retrieval by citric acid (pH 6.0). After overnight incubation with primary antibody at 4°C, sections were incubated for 30 minutes at room temperature with secondary antibody and incubated for 1 minute with diaminobenzidine. Then, the sections were lightly counterstained with hematoxylin. The expression level of MAGP2 was assessed by 2 pathologists. To validate our results of IHC and conduct survival analysis, we also refer to the results of The Human Protein Atlas (http://www.proteinatlas.org/).
Western Blot
Protein samples were equally loaded on 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis, electrophoresed, and transferred onto nitrocellulose membrane (Millipore, Darmstadt, Germany). After blocking with 5% nonfat milk in Tris-buffered saline with Tween 20 (TBST), the membranes were incubated with antibodies for MFAP5 and β-actin diluted at 1:1000 overnight at 4°C. After washing, the membranes were incubated with a 1:5000 dilution of horseradish peroxidase-labeled secondary antibody in TBST for 1 hour at room temperature. Then the signals were detected.
RNA Isolation and Quantitative Real-Time PCR Analysis
Total RNA was isolated from cell lines or tissues with TRIzol reagents (Invitrogen, California) according to the manufacturer’s instructions. The RT-PCR was performed to quantify mature microRNA (miRNA) expression with the NCode miRNA quantitative RT-PCR (qRT-PCR) analysis (Invitrogen) or messenger RNA (mRNA) expression with SYBR Green PCR Master Mix (TaKaRa, Japan). The primers were listed in supplementary material.
Cell Counting Kit-8 Assay
Cells of different groups were seeded in 96-well plates at a same density. The assessment was conducted every 24 hours. In brief, for every assessment, 10 µL Cell Counting Kit-8 (CCK-8; Dojindo, Japan) was added to each well and cultured at 37°C and 5% CO2 for 1 hour. Then, the absorbance was measured with the multifunctional microplate reader at 490 nm.
Migration and Invasion Assays
Cell migration assay was conducted using 8-mm pore size Transwell chambers (Corning, Shanghai, China). The lower chamber was filled with DMEM containing 10% FBS. Cells were suspended in serum-free DMEM and plated into the upper chamber. Then, the chambers were cultivated in 5% CO2 at 37°Cfor 2 days. After that, the cells in the upper chamber were removed with cotton swabs and the bottom surface of the polycarbonate membranes was counted visually using 0.1% Crystal violet dye and a light microscope. The invasion assay was same except that matrigel (Clontech, Mountain View, California) was used in the transwell chambers (Corning, Shanghai, China).
Luciferase Assay
Cells were seeded into 12-well plates 1 day before transfection. After 24 hours of culturing, the cells were cotransfected with 200 ng of psiCHECK-2 plasmids and 50 nmol/L of miR-200b-3p (or scramble miRNA). After 2 days, luciferase activities were measured with Dual-Luciferase Reporter Assay system (Promega, Madison, Wisconsin). Renilla luciferase activity was normalized to firefly luciferase activity.
Statistical Analysis
The correlation between IHC score and the clinicopathologic features was analyzed by χ2 test. Averaged replicates of 3 independent experiments were used in cellular studies, and results were statistically analyzed using the 2-tailed, unpaired Student t test. Results were expressed as mean values with 95% confidence intervals. Error bars represented standard error. P < .05 was considered statistically significant.
Results
MAGP2 is located in ECM and upregulated in CRC tissue
First of all, we examined MAGP2 expression by immunohistochemistry in 60 pairs of CRC tissues and their corresponding adjacent tissues. We demonstrated that MAGP2 was mainly localized in ECM (Figure 1A), which is consistent with the results from The Human Protein Atlas and previous reports focusing on other tumors. The percentage of samples strongly positive in CRC tissues is 81.7%(49/60), while there is no adjacent tissue with strongly positive stain (Figure 1B). Furthermore, MAGP2 expression of 10 randomly selected CRC tissues paired with adjacent tissues was investigated by qRT-PCR. We demonstrated that 9 of the 10 CRC tissues exhibited significantly upregulation of MAGP2 on mRNA level (Figure 1C). Additionally, we examined the expression level of MAGP2 in different CRC cell lines and normal colonic epithelium cell, namely, SW-60, Lovo, HCT-116, and NCM460. Western blot showed that MAGP2 protein expression was significantly higher in all CRC cell lines compared to immortalized colonic epithelium cell NCM460 (Figure 1D). From this, we concluded that after malignant transformation, cancer cells might obtain the ability to express and secrete MAGP2 into ECM.
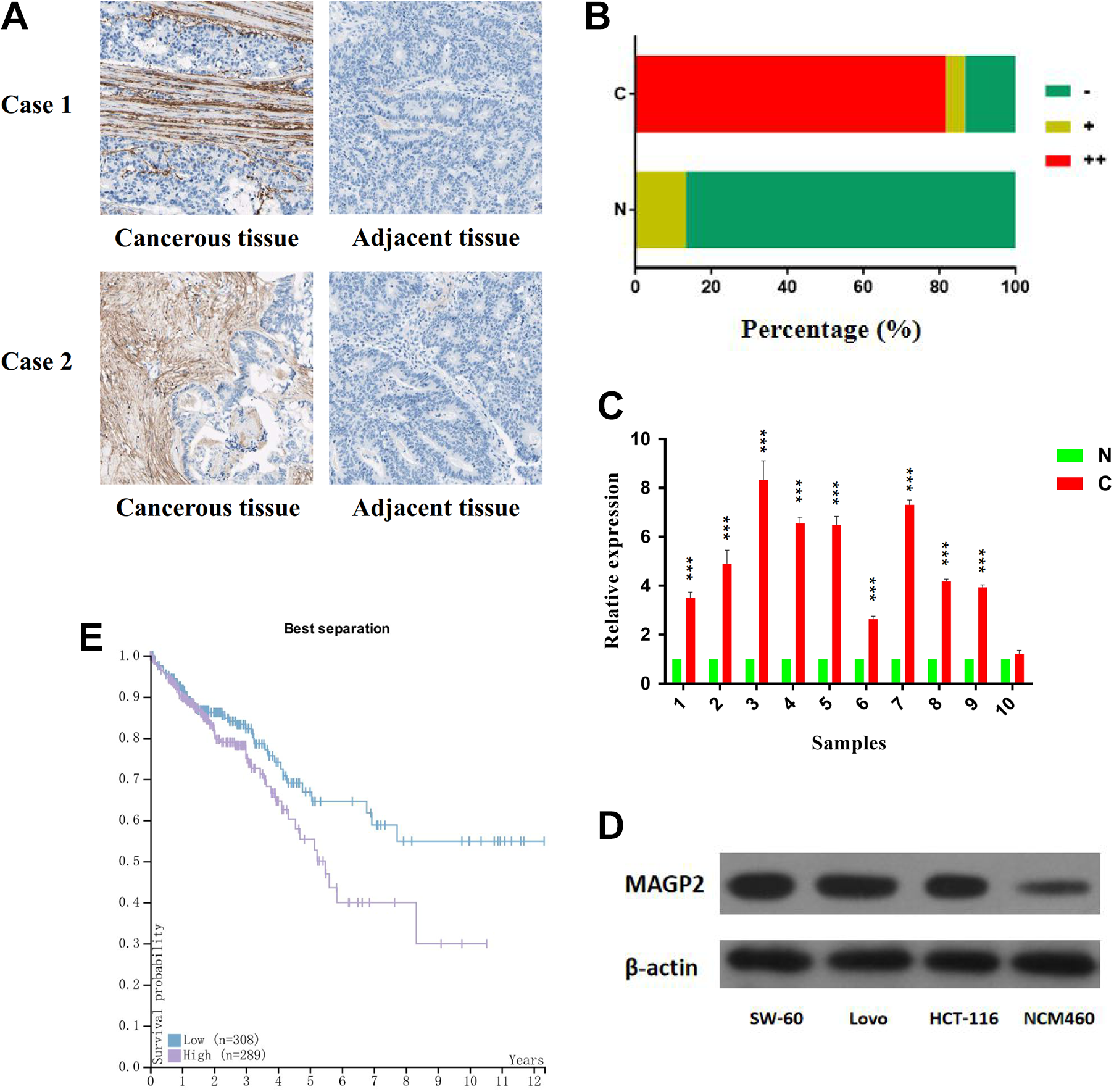
The expression level of MAGP2 is increased in CRC. A, Two representative cases: MAGP2 expression level in CRC tissue and adjacent tissue. B, Distributions of MAGP2 staining grades (−, +, and ++) in CRC tissue and adjacent tissue (magnification, ×200).C, Lysates from paired tissues of CRC and adjacent tissue were analyzed by qRT-PCR for the detection of MAGP2 mRNA. β-Actin was used as loading control. D, Western blotting analysis of MAGP2 expression in SW-60, Lovo, HCT-116, and NCM460 cell lines. β-Actin was used as loading control. E, High expression of MAGP2 is associated with shorter survival time of patients with CRC. The bars represent the mean values of 3 independent tests (mean [SD]). *** represents P < .001. CRC indicates colorectal cancer; MAGP2, microfibrial-associated glycoprotein 2; mRNA, messenger RNA; SD, standard deviation; qRT-PCR, quantitative real-time polymerase chain reaction.
2. The expression levels of MAGP2 in patients having CRC correlate with several clinicopathologic characteristics and patients’ survival
In order to confirm the correlation between the expression level of MAGP2 and clinicopathologic factors in CRC, the 60 cancerous samples mentioned above were classified into 2 groups according to the immunohistochemistry results, and χ2 test was used. As shown in Table 1, higher MAGP2 expression was closely associated with Dukes stage (P = .048), differentiation status (P = .034), and local lymphatic metastasis (P = .036). These results indicated that MAGP2 could influence the growth and metastasis of tumor. Importantly, using The Cancer Genome Atlas data, Kaplan-Meier survival analysis showed that the patients having CRC with tumors displaying higher MAGP2 expression levels had significantly shorter overall survival (Figure 1E, P < .001). This further suggested that MAGP2 functioned as an oncogene in CRC and could represent a potential novel prognostic biomarker for CRC after surgery.
The Correlation Between MAGP2 Expression and Pathological Characteristics.

Abbreviation: MAGP2, microfibrial-associated glycoprotein 2.
a Pearson correction.
3. MAGP2 enhances proliferation and metastasis of CRC cells and normal colonic epithelium cells
Then we detected the cytological effect of MAGP2. Microfibrial-associated glycoprotein 2 was also stably overexpressed by lentivirus-mediated packed pLV-MAGP2 vector in CRC cell line HCT116 and normal colonic epithelium cell line NCM460. We carried out the Western blot to verify the overexpression efficiency (Figure 2A). First, we used CCK-8 assay to compare the cell proliferation rate of MAGP2 overexpression cells with the control groups. As shown in Figure 2B, the growth rates of 2 MAGP2 overexpression cell lines were upregulated versus those of the control groups, suggesting that high expression level of MAGP2 resulted in cellular events about enhancing proliferation signals. For the purpose of confirming the effect of MAGP2 on CRC metastasis, migration and invasion assays were also conducted in the 2 cell lines mentioned above. Through the transwell assays, we found that higher levels of MAGP2 expression strongly exhibited increased rate of migration and invasion after 48 hours, compared to the control group in both cell lines (Figure 2C).
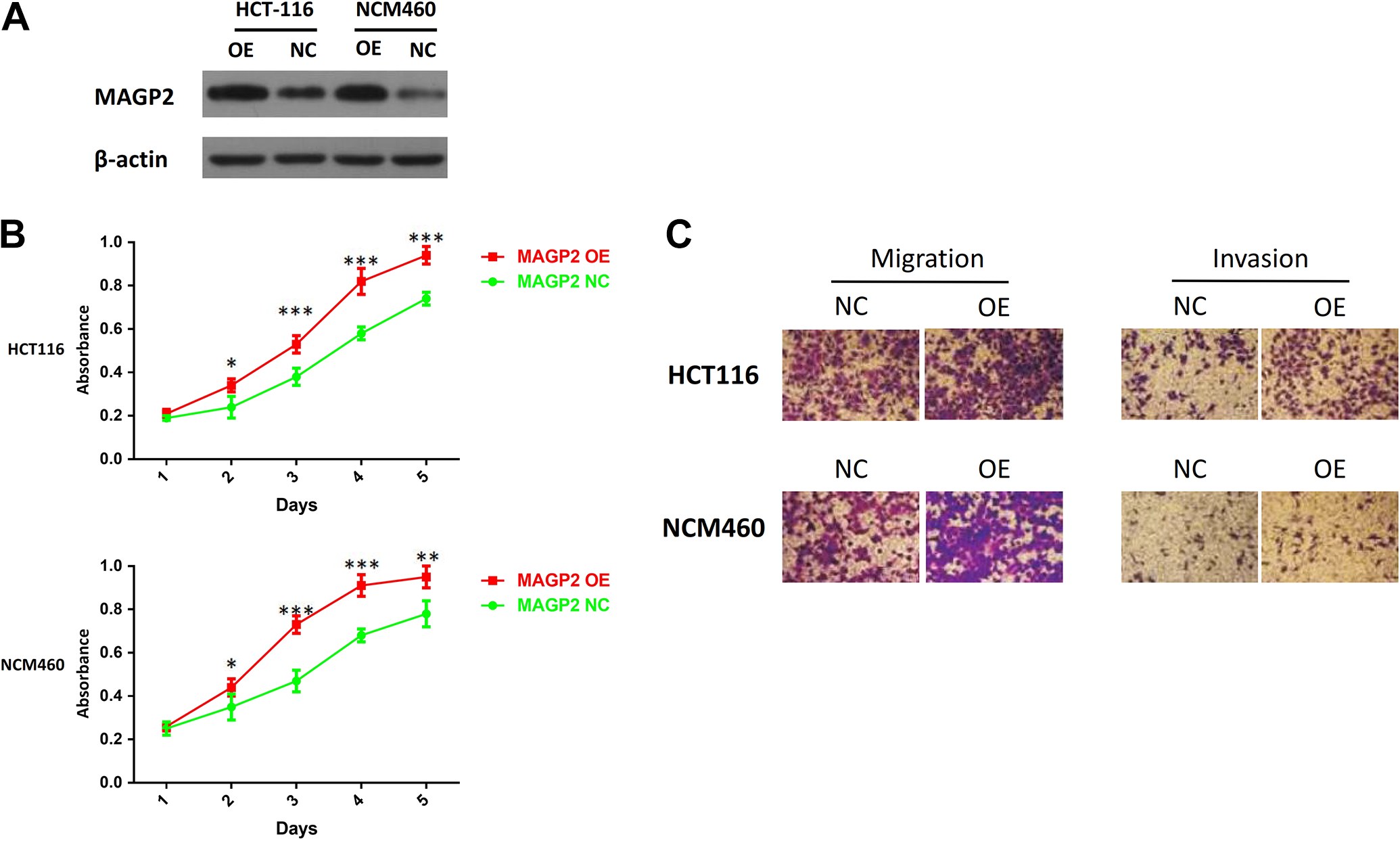
MAGP2 modulates proliferation and metastasis of CRC cells. A, Overexpression of MAGP2 in HCT116 cells (left) and NCM60 cells (right) was confirmed by Western blotting; β-actin was used as a loading control. B, Effect of MAGP2 overexpression on the proliferation of HCT116 and NCM60 cells was determined by CCK-8 assay. C, Effect of MAGP2 overexpression on the migration and invasion ability of HCT116 and NCM60 cells was determined by Transwell assay. The bars represent the mean values of 3 independent tests (mean [SD]). *, **, and *** represent P < .05, P < .01, and P < .001 respectively. CCK-8 indicates Cell Counting Kit-8; CRC, colorectal cancer; MAGP2, microfibrial-associated glycoprotein 2; SD, standard deviation.
4. MAGP2 activates the transcription of downstream genes of Notch pathway
It is reported that MAGP2 can control Notch signaling via interactions with RGD binding integrins in some kinds of cells. Notch pathway is a crucial cascade in cancer biology. So next we explored the possible relationship between MAGP2 and the downstream genes of Notch in CRC. By qRT-PCR, we assessed the effect of MAGP2 overexpression on the transcription of target genes of Notch-1 such as HES1, Slug, Snail, MMP2, MMP9, and KLF4. We found that overexpression of MAGP2 significantly increased the expression of HES1, Slug, Snail, MMP2, and MMP9 while reduced the expression of KLF4 when compared to the control group (Figure 3A). To imitate the function of microenvironment on cells, we also added ectogenic MAGP2 into the medium to observe the change in expression of these genes. The results were similar, which further validated that MAGP2 can modulate notch signaling in CRC (Figure 3B).

MAGP2 modulates Notch pathway in CRC. A, MAGP2 overexpression regulates the downstream genes of Notch, including HES1, Slug, Snail, MMP2, MMP9, and KLF4. B, Ectogenic MAGP2 regulates the downstream genes of Notch, including HES1, Slug, Snail, MMP2, MMP9, and KLF4. The bars represent the mean values of 3 independent tests (mean [SD]). *, **, and *** represent P < .05, P < .01, and P < .001, respectively. CRC indicates colorectal cancer; KLF4, Kruppel-like factor 4; MAGP2, microfibrial-associated glycoprotein 2; MMP, matrix metalloproteinase; SD, standard deviation.
5. MAGP2 is a direct target of tumor suppressor miR-200b-3p
Another issue remained to be solved, in not only CRC but also other cancers, is the high expression mechanism of MAGP2 in cancer tissues. MicroRNAs contribute importantly to gene expression regulation. It would be interesting to determine whether or not MAGP2 expression could be regulated by miRNAs in CRC. TargetScan database was searched computationally for potential microRNAs that are complementary to the 3′-untranslated region (3′-UTR) of MAGP2 (Figure 4A). The potential miRs includes miR-429, miR-200b-3p, and miR-200c-3p, which are all members of miR-200 family. After transfection with these 3 miRs (50 nmol/L), we, respectively, observed that the MAGP2 was significantly reduced on both mRNA levels in HCT-116 and Lovo cell lines (Figure 4B). Considering recent studies demonstrated that miR-429 was upregulated in CRC tissues, and only miR-200b-3p was proved to be downregulated in CRC tissues, we chose miR-200b-3p for further study. Our results showed that miR-200b-3p can also inhibit the expression of MAGP2 on protein level (Figure 4C). It was also found that miR-200b-3p can suppress reporter gene activity of MAGP2 in HEK293 cells, whereas mutated plasmid showed no change in the reporter gene activity (Figure 4D). Additionally, expressions of MAGP2 and miR-200b-3p were inversely correlated in the 30 CRC samples (Figure 4E). These data suggested that MAGP2 was a direct target of miR-200b-3p. Our results support the idea that MAGP2 expression is negatively regulated by miR-200b-3p in CRC, which partly explains the high expression mechanism of MAGP2 in cancer tissues.
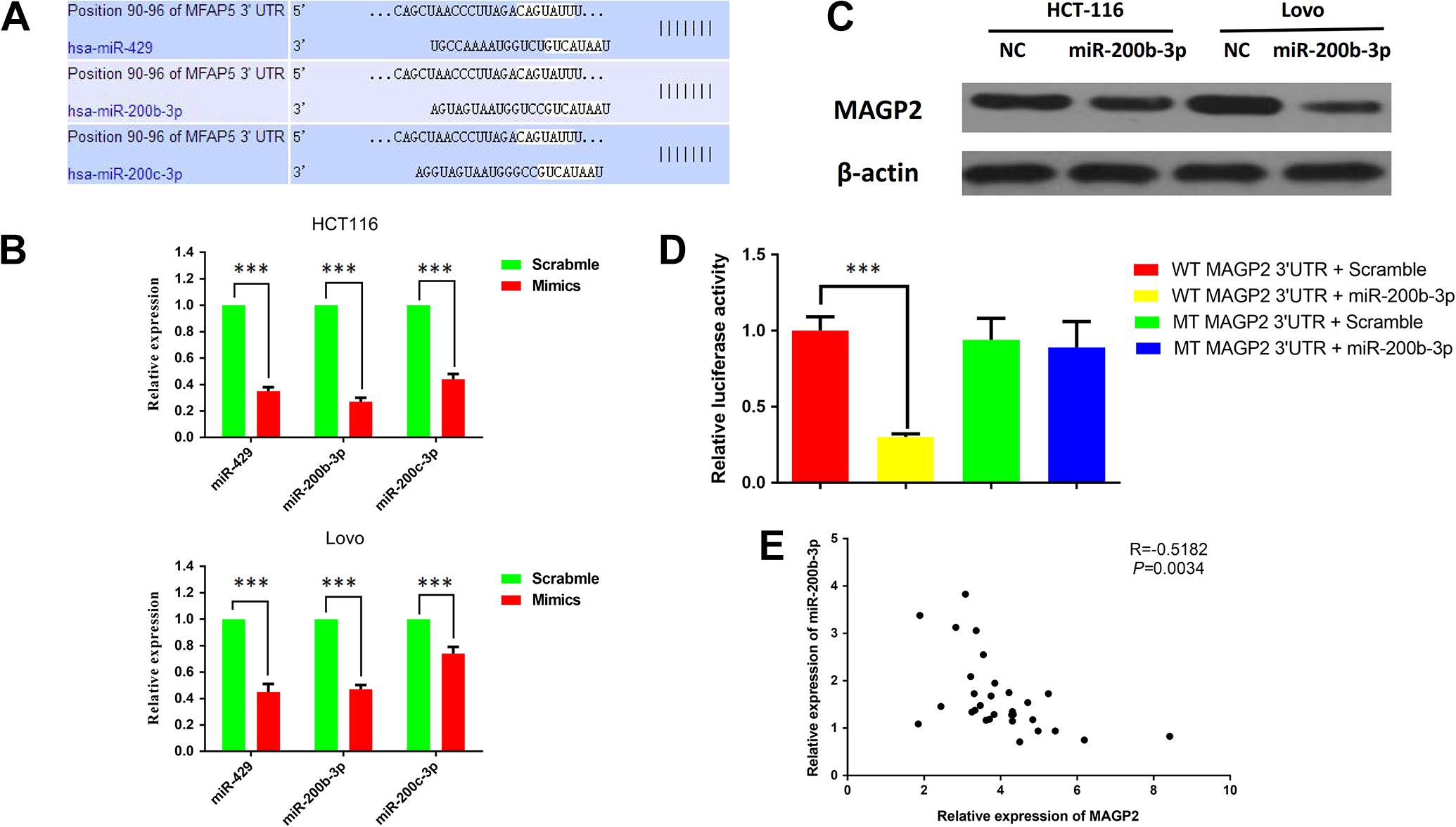
miR-200b-3p posttranscriptionally regulated MAGP2 expression by directly targeting its 3′-UTR. A, The target sites of miR-200 family members in 3′-UTR of MAGP2 are shown as a schematic representation. B, qRT-PCT showed the expression of MAGP2 mRNA in HCT116 and Lovo cells transfected with miR-429, miR-200b-3p, and miR-200c-3p. C, Western blotting assays showed the expression of MAGP2 protein in HCT116 and Lovo cells transfected with miR-200b-3p. D, Wild-type or mutant 3′-UTR constructs of MAGP2 were cloned into a psi-CHECK2 vector, respectively, and cotransfected with miR-200b-3p mimics in HEK293 cells. Renilla luciferase activities were normalized to firefly luciferase activities. E, The expression level of MAGP2 was negatively correlated with the expression level of miR-200B-3p in CRC samples. The bars represent the mean values of 3 independent tests (mean [SD]). *** represents P < .001. CRC indicates colorectal cancer; UTR, untranslated region; MAGP2, microfibrial-associated glycoprotein 2; mRNA, messenger RNA; SD, standard deviation; qRT-PCT, quantitative real-time polymerase chain reaction.
Discussion
Mutation or aberrant expression of MAGP2 is associated with many human diseases. Its mutation disrupts the function of vascular elastic fibers and is involved in the pathology of aortic aneurysms and dissections. 18 In adipose tissues, it modulates the remodeling of ECM and inflammation, taking part in the multiple metabolic diseases such as obesity. 19 In this study, we demonstrated that MAGP2 was significantly upregulated in ECM of CRC, and it was related to some pathological characteristics, and its high expression hinted worse prognosis. These indicated that MAGP2 may be used as a biomarker in CRC.
Microenvironment is a dynamic modulator for cell fate. In tumor biology, there are many information exchanges between tumor microenvironment and tumor cells. The microenvironment provides cancer cells with favorable factors, promoting their proliferation and metastasis. 20 One of the most abundant constituents of cancer microenvironment is ECM. On one hand, ECM regulates multiple concentrations of multiple molecules including ligands, growth factors, chemokines, and enzymes 21 ; on the other hand, the density and arrangement of protein components decide the physical properties of ECM such as structure and rigidness, and these properties greatly affect the potential of cancer growth and metastasis. 22 For example, matrix metalloproteinase (MMP) secreted by cancer cells will degrade surrounding matrix and promote cancer metastasis via formation of “micro-tracks” 23 crosslinking of ECM mediated by lysyl oxidase (LOX) results in increased tumor stiffness, and enhanced mechanical force will activate the integrin receptors and induce cytoskeleton rearrangement to endow the cancer cells with invasion ability 24 ; the urokinase receptor associated protein is able to reduce collagen content in ECM and promote the outgrowth of tumor. 23 Based on our results, we make a conclusion that the aberrant expression of MAGP2 may change the normal microenvironment of colorectal tissue, facilitating the progression of cancer.
It has been demonstrated that Notch signaling is dysregulated in CRC. Upregulation of Jagged-1, a Notch ligand, increases Notch-1 in CRC 25 ; furthermore, increased levels of Notch-1 is correlated with tumor grade and distant metastasis 26 ; Notch-1 can also activate oncogene HES1, which in turn suppresses KLF4, which is an inhibitor of the proliferation of colon cancer cells. 16 Notch can also interact with transcription factors Slug, Snail, and transforming growth factor-β (TGF-β), which are critical molecules for epithelial–mesenchymal transition (EMT). 16 In this study, we also proved that both endogenic and ectogenic MAGP2 can activate the transcription of the downstream genes of Notch-1, including HES1, Slug, and Snail, while suppress the expression of KLF4. Our results implies that MAGP2 can promote the progression of CRC, at least partly via Notch signaling. Microfibrial-associated glycoprotein 2 can also activate mitogen-activated protein kinase (MAPK) and AKT serine/threonine kinase (AKT) signaling in tongue cancer[8]; in breast cancer tissues and its downstream effectors include focal adhesion kinase (FAK), ERK, and so on. 10 In CRC tissue, MAGP2 may exert oncogenic functions via similar mechanism mentioned above. What’s more, MAGP2 probably activate integrin signaling in CRC, for its RGD domain can bind to integrin receptors. 2 These hypotheses remain to be proved in the following studies.
MiR-200 family of miRNAs plays an important role in tumor biology. 27 The miR-200 family includes 5 miRNA sequences: miR-200a, miR-200b, miR-200c, miR-141, and miR-429. 28 miR-200b-3p is reported to be significantly downregulated in CRC and it inhibits tumor metastasis and chemotherapeutic resistance of CRC via targeting peroxiredoxin 2. 28,29 In this study, we validated that miR-200b-3p can directly target the 3′-UTR region of MAGP2. Interestingly, it is reported that downregulation of the Notch pathway will increase the expression of miR-200 family members. 30 Collectively, these results imply that there may be a negative feedback among miR-200b-3p/MAGP2/Notch axis, and once one of them is dysregulated, then the whole pathway will be abnormally activated.
In this study, we found that MAGP2 protein is almost absent in normal colonic epithelium cell line NCM460, while is remarkably detected in cancer cell lines. So we think cancer cells can secrete MAGP2 into ECM via exosome or other ways. However, it is reported that MAGP2 can also be secreted from tumor-associated fibroblast (TAF). For example, in tongue squamous cell carcinoma, MAGP2 is enriched in TAF secretomes. 8 Recent studies have shown that TAF plays important roles in the progression and metastasis of CRC, 31,32 so an important issue remaining to be solved in the following study is that whether TAF in microenvironment of CRC is also producer of MAGP2.
In conclusion, we identified MAGP2 as an oncogene of CRC associated with clinical prognosis. We also demonstrated that the knockdown of MAGP2 could directly decrease cell proliferation and inhibit cell metastasis. It may function as a potential biomarker and therapeutic target for CRC.
Supplemental Material
Supplementary_Materials_(5) - MAGP2, a Component of Extracellular Matrix, Is Upregulated in Colorectal Cancer and Negatively Modulated by miR-200b-3p
Supplementary_Materials_(5) for MAGP2, a Component of Extracellular Matrix, Is Upregulated in Colorectal Cancer and Negatively Modulated by miR-200b-3p by Wei Feifei, Guo Hui, Zhao Ruiqiang, Jiang Qunxiang and Xie Yu’an in Technology in Cancer Research & Treatment
Footnotes
Authors’ Note
XYA and WFF conceived and designed the experiments. WFF, GH, ZRQ, and JQX performed the experiments. WFF performed statistical analysis. WFF and XYA wrote the paper. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statement
Our study was approved by the ethics review board of Affiliated Tumor Hospital of Guangxi Medical University.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from the National Natural Science Foundation of China (No. 81260320).
Supplemental Material
Supplemental material for this article is available online.
Abbreviations
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
