Abstract
Ensuring the complete removal of tumor tissue is the main challenge during resection operations. Recently, a technique of “indirect” contact laser surgery has been developed. In this study we assess the possibility of using such surgery for fluorescence image-guided tumor resection. Mouse colon adenocarcinoma CT-26 cells stably expressing the fluorescent protein mKate-2 was used as the tumor model. Resections of the tumor nodes were performed with either a scalpel blade, a laser scalpel with a bare tip, or a laser scalpel with a strongly absorbing coating on the fiber tip. Tumor-positive resection margins were detected using an IVIS Spectrum fluorescence imaging system. After tumor resection with the scalpel blade over half of the animals needed one additional resection to remove residual tumor cells. Animals in this group showed tumor recurrence within 7 days. Fluorescence imaging of the tumor bed, performed after resection to assess the presence of tumor cell clusters, was sufficiently effective only with a bloodless resection. The laser scalpels both with the bare tip and with the strongly absorbing coating on the tip provided such bloodless tumor resection in contact mode. Fewer animals required additional resections when the bare tipped scalpel was used and this also resulted in a reduction in tumor recurrence. After resections were carried out with the laser scalpel with the strongly absorbing coating on the tip, fluorescence was detected in the operative field and this led to undertaking additional resections, although subsequent investigation suggested that this was “false” fluorescence, resulting from the effects of the scalpel rather than the presence of residual tumor cells. The method of laser resection with a strongly absorbing coating on the tip therefore did not appear to demonstrate definite advantages over laser resection with a bare tip when removing tumors.
Introduction
The surgical treatment of solid tumors generally involves tumor excision including a wide margin in an attempt to ensure the complete removal of all the tumor cells. However, some tumor cells may remain in regions adjacent to the tumor removal area and form “tumor-positive resection margins.” It has been shown that local recurrence is increased in cases that involve a persistent positive margin compared to those where a negative surgical margin has been achieved. 1 Currently, several techniques (eg, frozen section 2 and touch imprint cytology 3 ) are being used to improve the intraoperative assessment of the tumor margins. However, they have some limitations (extend surgery time, highly operator-dependent) in their use. 4 Optical coherence tomography has also been used for high-accuracy real-time detection of tumor margins. 5,6 More often, however, tumor-specific near-infrared fluorescent agents have been used for intraoperative visualization of the excised tumor bed. 7 Near-infrared fluorescent light has many advantages, including high tissue penetration and low tissue autofluorescence, while not altering the surgical field. Many portable intraoperative fluorescence imaging devices for image-guided surgery are available. 8 Indocyanine green (emission at 820 nm), methylene blue (emission at 700 nm), and fluorescein (emission at 520 nm) are the fluorescent contrast agents currently registered by the Food and Drug Administration and European Medicines Agency (EMA) for clinical applications. 9 Furthermore, 5-aminolevulinic acid (5-ALA) may be used for fluorescence-diagnosis of tumor-positive margin. 10,11 The application of 5-ALA leads to an increased cellular accumulation of the highly fluorescent protoporphyrin IX in tumor cells.
The selection of an appropriate surgical technique can improve the accuracy of tumor resection. 12 On one hand, the technique must comply with the principles of ablastics. On the other hand, it must allow the operation to be performed sufficiently and quickly, while not restricting the possibility for further monitoring of the surgical field. Some laser surgery modes meet these requirements. Laser surgery may be classified by the laser light delivery mode: “free-beam mode” or “contact mode”. 13 In general terms, free-beam laser surgery provides for ablation or coagulation of the target tissues. However, this mode conflicts with the ablastic requirement due to its dissemination of the ablative tissue. In contact mode, the laser fiber tip can be used for resection by submerging it into the tissue, meaning that ablation is not the primary mechanism. The fiber tip moves apart the “softened” tissue 14 so that a cut is formed. In this case, the fiber tip is introduced directly into the tissue, that is, the action comes from inside the tissue. Tissue can soften due to the caramelization effect 13 or a porous structure can be created. 15 The thermally damaged layer, which occurs due to the action of the laser scalpel, 16 has optical parameters that are dependent on the contact laser resection technique (increased scattering, formation of carbonized tissue particles), and acts as an optical filter when using fluorescence-diagnostics to determine the tumor margins. With the emergence of new laser resection techniques, for example, “indirect” contact laser surgery, 17 this problem has become more important. The efficacy of cancer surgery can be improved by choosing an appropriate combination of surgical techniques and methods for detection of the true resection margins. 18,19 Recently, systems for the treatment of tumors that integrate detection and laser resection have been proposed. 6,20,21 Ex vivo experiments with a porcine brain demonstrated the effectiveness of the erbium:yttrium-aluminium-garnet (Er:YAG) laser ablation of a targeted area stained with protoporphyrin IX. It was shown that variation in the laser treatment parameters (eg, pulse frequency and power) enabled a variety of damage depths and widths to be achieved. 20 In vivo fluorescence-guided Nd:YAG laser ablation of residual tumor tissue in mice enhanced their recurrence-free survival when compared to untreated animals. 21 These theranostics systems used free-beam mode laser surgery for tumor ablation. Currently, there are no papers devoted to fluorescence-guided “indirect” contact laser surgery. Therefore, the aim of this study was to assess the possibility of using “indirect” contact laser surgery for fluorescence-image guided tumor resection.
Materials and methods
The study was performed in 45 BALB/c mice with subcutaneously inoculated tumors. To allow the fluorescence imaging of residual tumor cells in the surgical field, we used mouse colon adenocarcinoma (CT-26) cells stably expressing the fluorescent protein mKate-2 as a tumor model. This protein has a fluorescence emission peak at 633 nm, similar to that of protoporphyrin IX (636 nm). Tumor cells were inoculated subcutaneously into the left thigh at a concentration of 2.5 × 105 cells/100 μL of phosphate-buffered saline. The tumor was removed on the eighth day after inoculation, when the tumor node diameter had reached approximately 1 cm. Tumor sizes were measured mechanically. In addition, prior to surgery, palpation was used to determine the tumor depth. The mice were randomly separated into 3 groups (15 animals each) to receive resection. The tumor resection was performed either with a scalpel blade (first group); a laser scalpel with a bare tip (second group), or a laser scalpel with a strongly absorbing coating (SAC) applied onto the fiber tip (third group). Prior to operation, the mice were anesthetized with a mixture of Zoletil and Romethar. An IVIS Spectrum (Caliper, USA) fluorescence imaging system was used to detect the tumor boundaries before surgery as well as any residual tumor cells during each operation (Figure 1). This system enables the detection of autofluorescence with a high spatial resolution (down to 20 microns).

The flowchart of the study design.
An LSP-0.97/10 (IRE-Polyus, Russia) laser scalpel operating at a wavelength of 0.97 μm with an output power of 7 W was used to excise the tumor. A previous study had demonstrated the efficacy of this laser parameter for skin resection. 22 The laser light was delivered to the tissue via silica fiber 550 μm in diameter. A fiber tip cleared from the cladding layer was used for resection. Tumor resection was made using either the bare fiber tip, or using one with an SAC. The SAC was a mixture of graphite powder (5%-20%) with fractions less than 0.5 μm and silicone varnish (80%-95%) based on polymethylphenylsiloxane resin. In “indirect” laser surgery, the laser radiation heated the SAC on the optical fiber tip and did not modify the tissue directly. The heated fiber tip was used for tissue resection. In this technique, biological tissue heated by fiber tip softened and, under the mechanical action of the optical fiber, moved apart so that a cut was formed. 14,17 That is the principal difference of the laser resection from the laser ablation technique, which considers biotissue elimination.
The surgical procedure was carried out by a highly qualified surgeon and included the following steps. Prior detection of the lower tumor boundaries was made by palpation. The skin covering the tumor node was lifted back with a scalpel blade. Detection of the tumor boundaries was achieved using fluorescence visualization and the tumor node was excised with one of the instruments described above. During resection by both types of laser scalpel, the fiber was held in the surgeon’s hand in a similar manner to a pen. The output power of the laser scalpel was adjusted to the required 7 W. The fiber tip had direct contact with tissue and was moved along an excision line. This operation was repeated several times to increase the resection depth. Excision lines were located 1 mm outside the tumor boundaries indicated by fluorescence. Once the tumor node had been removed, the presence of fluorescence in the operative field was checked to reveal any residual tumor tissue. In the case of positive tumor margins, additional resection was carried out. After the resection, the skin flap was sutured and closed using medical glue. The animals were weekly monitored for 8 weeks. Subsequently, the mice were kept in standard vivarium conditions, but received an analgesic and an antibiotic in their drinking water. All animal studies were approved by the Ethical Committee of the Nizhny Novgorod State Medical Academy (Protocol No.14 of December 10, 2013).
Three mice from each group were killed immediately after various numbers of resection for histopathological analysis of any tumor positive margins to correlate with the fluorescence images. Local recurrences in the remaining mice were confirmed to be tumors by histopathological evaluation.
Results
This work demonstrates the first results from the development of an “indirect” laser surgery technique for the resection of tumors. A fluorescence imaging technique was used for detection of positive tumor margins, however, tumor resections made with a scalpel blade resulted in a pool of blood in the operative field. This presented a challenge for visual and fluorescence assessment of the presence of further tumor tissue. Therefore, the operative field was irrigated regularly with physiological saline to remove the blood. After the tumor resection, the presence of fluorescence in the bed of the excised tumor was assessed (Figure 2A and B). It was found that after use of the scalpel blade, fluorescence was present in 2 of the 12 mice (Table 1). Histopathological analysis revealed residual tumor tissue in the fluorescence positive margin (Figure 2C). Each of these animals received a single additional resection to completely remove any tumor-associated residual tissue showing a fluorescent signal.

Fluorescence images of tumor during resection using a scalpel blade: (A) before resection, (B) surgical field after resection, (C) H&E analysis of tumor positive margin after resection. T indicates tumor; M, muscle. Scale bar for fluorescent images is 1 mm, for H&E is 500 µm.
Features of Tumor Resection.

Abbreviation: SAC, strongly absorbing coating.
Tumor resection made using the laser scalpel with a bare tip was attended by strong carbonization of the tissue. This scalpel provided a relatively bloodless operative field. Visually, no tumor tissue was detected in the operative field after resection. However, 8 of the 12 mice exhibited a fluorescence signal in the tumor bed after the first resection (Figure 3A). Histopathological analysis showed tumor cells in the operative field (Figure 3B). Two additional resections were needed for total eradication of tumor tissue in all cases (Figure 3C and D; Table 1).
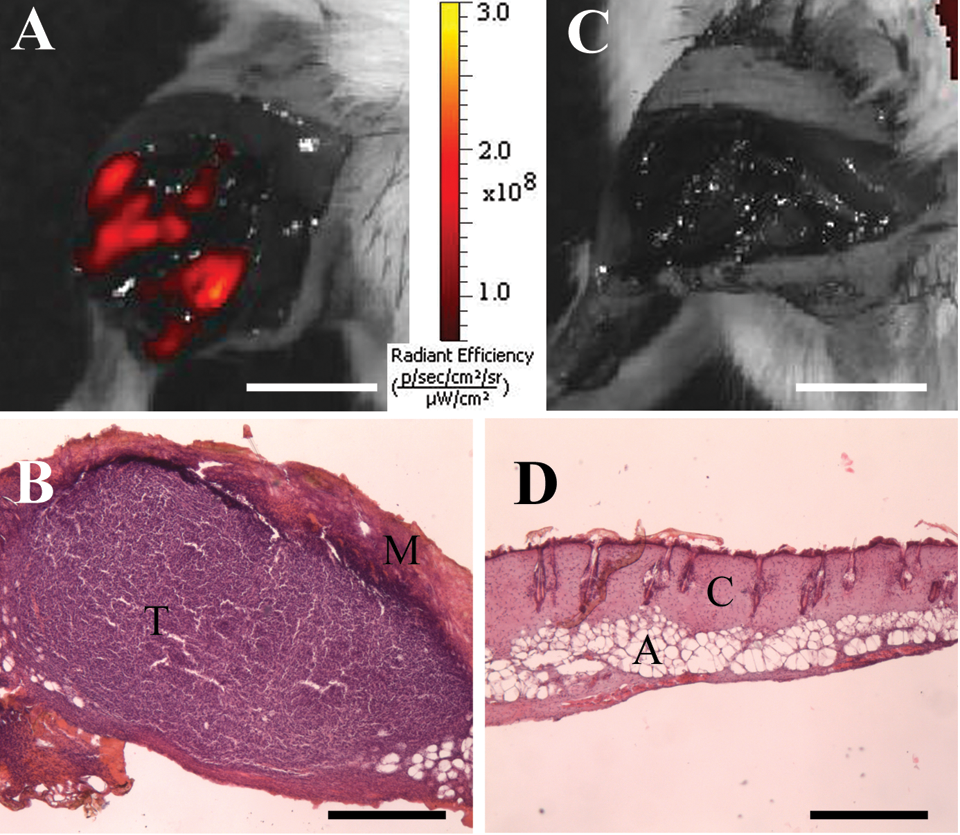
Fluorescence images of tumor during resection using the laser scalpel with a bare tip: (A) surgical field after first resection, (B) H&E analysis of tumor positive margin after the first resection, (C) surgical field after the second resection; (D) H&E analysis of the tumor positive margin after the second resection. T indicates tumor; M, carbonized muscle; C, coagulated connective tissue; A, adipose tissue. Scale bar for fluorescent images is 1 mm, for H&E is 500 µm.
Tissue carbonization was not very strong after resection performed using the laser scalpel with the SAC. It may be noted, that this laser scalpel provided good vessel coagulation, therefore the operative field was bloodless. However, fluorescence in the resection margin was detected in 4 of the 12 mice (Table 1). These animals received 4 additional resections for removing the fluorescence signal (Figure 4A and C). Histopathological analysis did not reveal any residual tumor tissue in the fluorescence-positive margin after the 4 additional resections (Figure 4B and D).

Fluorescence images of tumor during resection using the laser scalpel with SAC: (A) surgical field after the first resection, (B) H&E analysis of tumor positive margin after the first resection, (C) surgical field after the fourth resection; (D) H&E analysis of tumor positive margin after the fourth resection. T indicates tumor; M, muscle; C, carbonized tissue. Scale bar for fluorescent images is 1 mm, for H&E is 500 µm.
It was noted, that the location of the fluorescence signal correlated with the carbonized area in the operative field. To confirm this correlation, dissection of healthy muscle tissue was performed using both the laser scalpel types (Figure 5). A fluorescence signal was detected from the incisions in the case of the laser scalpel with SAC (Figure 5C), but not from those made with the bare tip (Figure 5A). Histopathological analysis revealed carbonization in this location (Figure 5D).

Fluorescence images (A and C) and corresponding H&E staining (B and D) of muscle excised by laser scalpel with a bare tip (A and D) and laser scalpel with an SAC (A and D). M indicates muscle; C, carbonized tissue. Scale bar for fluorescent images is 1 mm, for H&E is 200 µm.
The main indicator of the efficacy of tumor eradication is no recurrence for a long time. It was found that when the scalpel blade was used, recurrence occurred in 7 out of the 12 animals within 7 days of the resection (Table 2). Tumor recurrence appeared after 14 days in 2 animals from the “laser scalpel with a bare tip” group. Five of the 12 mice had tumor recurrence by 14 days after resection using the laser scalpel with an SAC. Local recurrences were confirmed to be tumors by histopathological evaluation. The remaining animals in each group had no recurrence within the 8 weeks of the investigation.
Appearance of Tumor Recurrence.

Abbreviation: SAC, strongly absorbing coating.
Discussion
In this article, the possibility of eradicating tumors using a laser scalpel with an SAC was evaluated in relation to 2 other methods. Fluorescence imaging was used to inspect for total elimination of the evident tumor tissue. In recent years, there have been numerous studies demonstrating the benefits of fluorescence image-guided surgery for the resection of tumors, and the overwhelming majority of these have utilized a specific contrast agent known as 5-ALA. 23,24 5-Aminolevulinic acid is the metabolic precursor of protoporphyrin IX in the biosynthetic pathway for heme. The natural heme feedback control can be bypassed by the administration of exogenous 5-ALA, resulting in an accumulation of protoporphyrin IX. Enhanced accumulation of 5-ALA within malignant cells is thought to occur as a result of their decreased levels of ferrochelatase (an enzyme that produces heme by the addition of iron) and selective uptake using the adenosine triphosphate-binding cassette transporter (ABCB6). 25 A cell line stably expressing the fluorescent protein mKate-2 was used for tumor modeling. The peak of fluorescence emission of mKate-2 is close to that of protoporphyrin IX.
In the “scalpel blade” group, a single additional resection for complete tumor eradication was performed in 2 animals. Resections made using a scalpel blade led to a pool of blood in the operative field. Whole blood has absorption maxima at 545 and 578 nm, as has been previously reported in the literature. 26 Thus, the presence of blood significantly hinders fluorescence imaging of any residual tumor cells. Wide-field visualization of 5-ALA-induced protoporphyrin IX contrast is effective at revealing regions of bulk tumor (deep red), but fluorescence intensities decay near the margins (lighter pink) and vanish entirely as the tumor cell density continues to decline. 27 The proportion of tumor recurrences in this group was higher compared to the other groups.
The laser scalpel with a bare tip provided good vessel coagulation. Thus, the operative field was clear of any blood preventing detection of the residual tumor cells. The proportion of tumor recurrences in this group was the smallest in comparison with the other groups. Furthermore, such tumor recurrence only appeared 14 days after tumor resection. Diode laser energy in the 0.97 μm range is only slightly absorbed by water. This is a key reason why such diode laser light can penetrate and scatter quite deeply into tissue. This can result in a substantial depth of thermal impact. 13 According to a previous report, resections made using a laser scalpel with a bare tip led to the formation of a wide region (nearly 300 μm) of thermally coagulated tissue. 22 The thermal response of pancreatic cancer cells has been investigated in vitro. Exposure to 50°C for 5 minutes resulted in a significant decline in cell viability (70%) on day 1 with a continued decline to 0% by day 7 following the exposure. 28 Probably, in this study, it was the thermal injury of residual tumor cells that led to the smallest number of tumor recurrences in the “bare fiber” group.
In the “laser scalpel with SAC” group, 4 additional resections were performed in 5 animals for complete eradication of the detectable tumor tissue. The laser scalpel with SAC provided good vessel coagulation and induced mild tissue carbonization. It should be noted that the number of animals with tumor recurrence was higher after resection with the laser scalpel with SAC than with the laser scalpel with a bare tip. The SAC on the fiber tip can absorb from 30% to 50% of the initial laser irradiation, as has been previously reported. 17 Therefore, the influence of the laser light on tissue including cancer cells was not as strong as with the bare fiber laser, and the thermal injury to the tissue was more mild. However, the carbonized area was capable of emitting fluorescence in the spectral range being used. A similar effect has been observed during contact laser resection in the work, 29 and was explained by the presence of diamond-like microparticles that fluoresce in the same region. In the research, 30 the fluorescence of porous carbon samples was investigated. The false fluorescence signal emitted by the carbonized layer causes difficulties for the practical use of fluorescence-guided contact laser resection. Thus, further optimization of the contact laser resection technique is needed.
It might be supposed that the “indirect” laser surgery is useful for treatment of several pathologies, in particular, malignant tumors due to efficient cutting capability combined with instantaneous coagulation of vessels in a wound, minimized intraoperative trauma, and absence of coarse scars in postoperative period. Morphological and functional features of tissue in ear, nose, and throat as well as trend to minimally invasive operation determine the main area of “indirect” laser surgery. Moreover, this technique may be employed for oral surgery.
Conclusion
Initial experimental results show that “indirect” contact laser surgery may be used for fluorescence-guided tumor resections. Fluorescence imaging of the tumor bed performed after resection to assess the presence of tumor cell clusters is sufficiently effective only with a bloodless resection. The comparative analysis of organ-preserving tumor resection techniques using a scalpel blade or the laser scalpels either with a bare tip or with an SAC reveals the advantage of the laser scalpels for fluorescence monitoring of the resection margin. However, false fluorescence detected in the operative field after use of the laser scalpel with an SAC is a possible limitation of this technology and can result in an indication of the need for additional resections. Furthermore, laser scalpels with either SACs or bare tips have no significant difference in tumor recurrence rate. In future studies, we plan to increase the number of animals used in order to reveal any statistically significant difference between a laser scalpel with an SAC and one with a bare tip. Moreover, a further tumor model will also be included in the research.
Footnotes
Abbreviations
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Russian Science Foundation, grant number 14-15-00840P.
