Abstract
In recent years, impregnation of biomaterial (wood veneers) with eutectic phase change materials (PCM) has been investigated to increase the thermal capacity of bio-based materials, which significantly affects the thermal capacity, especially in building applications requiring low heat. In this study, four eutectic phase change materials were prepared using three different fatty acids and impregnated on two wood veneers (oak and ash). The structural characterization of the prepared eutectic mixtures was examined using FT-IR, while the morphological properties of the wood veneers were examined by scanning electron microscopy (SEM). The thermal performance was analyzed via differential scanning calorimetry (DSC) and their thermo-mechanical properties by dynamic mechanical analysis (DMA). Additionally, the thermal conductivity (k values) was determined. From the results obtained, it was possible to prepare eutectic mixtures with melting temperatures around 30°C with heat capacities up to 225.5 J/g. It was also generally determined that eutectic PCM, prepared from mixtures of lauric acid and palmitic acid, have lower thermal conductivity but show higher storage and loss modulus for the wood coatings leading to a balance between mechanical and thermal properties.
Introduction
With increasing world population in recent years, several issues are expected to meet the energy needs within a green environmental cycle. Today, the energy consumption associated with buildings represents about 40% of the total energy consumed around the world, especially in the structural engineering sector. This situation puts the development of the building construction sectors under serious threat. 1 Improving energy efficiency in buildings, reducing greenhouse gas (GHG) emissions and combating climate change are extremely important for energy storage technology. These technologies can be classified as mechanical, thermochemical, chemical, electrical and thermal energy. The most important storage technology is thermal energy storage and phase change materials (PCM) are used for this application.2–4
PCM have three important properties: latent heat storage (LHS), controllable phase transition temperature (PTT) and chemical stabilization. Their main feature is to stabilize the ambient temperature and the most effective solution to replace air conditioning. 5 By using these materials, the temperature variations are substantially reduced due to their high energy storage density and their ability to store/release thermal energy during a phase transition. These properties are highly interesting for energy management applications such as energy saving in buildings, photovoltaic solar energy use, air conditioning condenser heat recovery, solar energy storage, cryogenic cooling, and indoor temperature control systems.6–15
PCM are generally divided into organic and inorganic materials according to their components. The main materials are salt hydrates, paraffin-free organic compounds, fatty acids and their mixtures.16–19 Organic PCM, especially based on fatty acids, is the most preferred storage material in thermal energy storage systems including indoor coatings and solar heating/cooling and textiles integrated onto fabrics due to its excellent heat transfer rates and thermodynamic effects such as latent heat of fusion, supercooling, thermal and chemical stability, small volume change and wide melting point range.20–23
Using a single fatty acid leads to a constant phase/fixed transition temperature (intrinsic property), but preparing eutectic mixtures using binary or ternary aliphatic acids is known to expand the phase transition temperature value and range of possible fatty acids.24,25 Eutectic mixture is the mixing of at least two substances in an amount that must have the lowest melting point. In addition, the components forming the mixture crystallize simultaneously.
In the literature, information on thermal heat storage properties and application areas investigated the effect of eutectic mixtures compositions. In the study of Tunçbilek et al., 26 the melting temperature of the eutectic mixture based on 69% wt. lauric acid and 31% wt. palmitic acid was calculated as 35.2°C with a latent heat as 166.3 J/g. These values indicate that this material shows good thermal performances. Nazir et al. 27 compared eutectic fatty acid mixtures prepared in different ratios according to the Schrader equation and thermophysical properties. The phase transition temperature of eutectic fatty acid mixture (stearic acid/palmitic acid/lauric acid) with a weight ratio of 15.6/24.4/60 was determined as 35.1°C with a latent heat storage of 183.8 kJ/kg. The results showed that the mixture is suitable for solar water heating and building energy saving systems.
Wood, which is the most common biomaterial known as an organic building material in nature, is frequently preferred in engineering applications due to its superior technological properties, sustainability, and renewability. 28 Due to its porous structure, it can effectively absorb PCM and is very suitable for high heat storage technology. Especially in cold weather conditions, the PCM absorbed into a building material melts when heated and prevents air entry from the outside. As the amount of heat (energy supply) decreases at night, the heat is released back from the PCM in the building material, and this is how the building’s internal heat is actively used. While minimizing the use of solar energy with PCM addition in buildings, a more homogeneous temperature distribution inside the building is obtained which can optimize the energy costs for both heating and cooling. 29
In the literature, some research has been done on the use of wood and PCM. Myristic alcohol, 29 fatty acid, 30 eutectic mixture of lauric acid, myristic acid and capric acid,31,32 eutectic mixtures of capric acid and stearic acid, 33 paraffin34,35 were used in studies. Can 36 used the emulsification-solvent evaporation method to impregnate poplar wood by encapsulating palmitic acid in ethyl cellulose. As a result, the thermal conductivity of wood impregnated with PCM was found to be 40% higher than the control sample. Grzybek et al., 37 impregnated solid wood with paraffinic PCM to increase its heat capacity. According to the results of DSC analysis, they found that the heat capacity increased onefold compared to the control sample. Nazari et al. 38 impregnated Scots pine and beech massive trees with encapsulated biobased eutectic phase change materials and examined the thermal properties of the wood. The results showed that bio-PCM material increased the thermal capacity and thermal conductivity of wood for building applications.
In this study, the phase-changing materials (lauric acid, stearic acid, and palmitic acid) are combined with four different concentrations to be impregnated into two biomaterials (oak and ash wood) in the form of veneers. The morphological properties, structural analysis, thermal stability, dynamic mechanical properties and thermal conductivity of the prepared functionalized biomaterials are characterized.
Experimental
Materials
Lauric acid (LA) (C12H24O2, Tm = 43.2°C, Mw = 200.32 g/mol), stearic acid (SA) (C18H36O2, Tm = 67.8°C, Mw = 284.47 g/mol, and palmitic acid (PA) (C16H32O2, Tm = 62.2°C, Mw = 256.42 g/mol) were selected as the base materials to prepare the phase change materials. These PCMs were purchased from Sigma-Aldrich. Oak wood (Quercus petraea, density = 0.615 g/cm3) and ash (Fraxinus Excelsior L., density = 0.556 g/cm3) veneers were selected from boards cut (30 mm × 30 mm × 0.75 mm) entirely from sapwood. The wood veneers were supplied by a local firm.
Preparation of the eutectic PCM
Formulations of the eutectic mixtures.

PCM impregnation into biomaterial
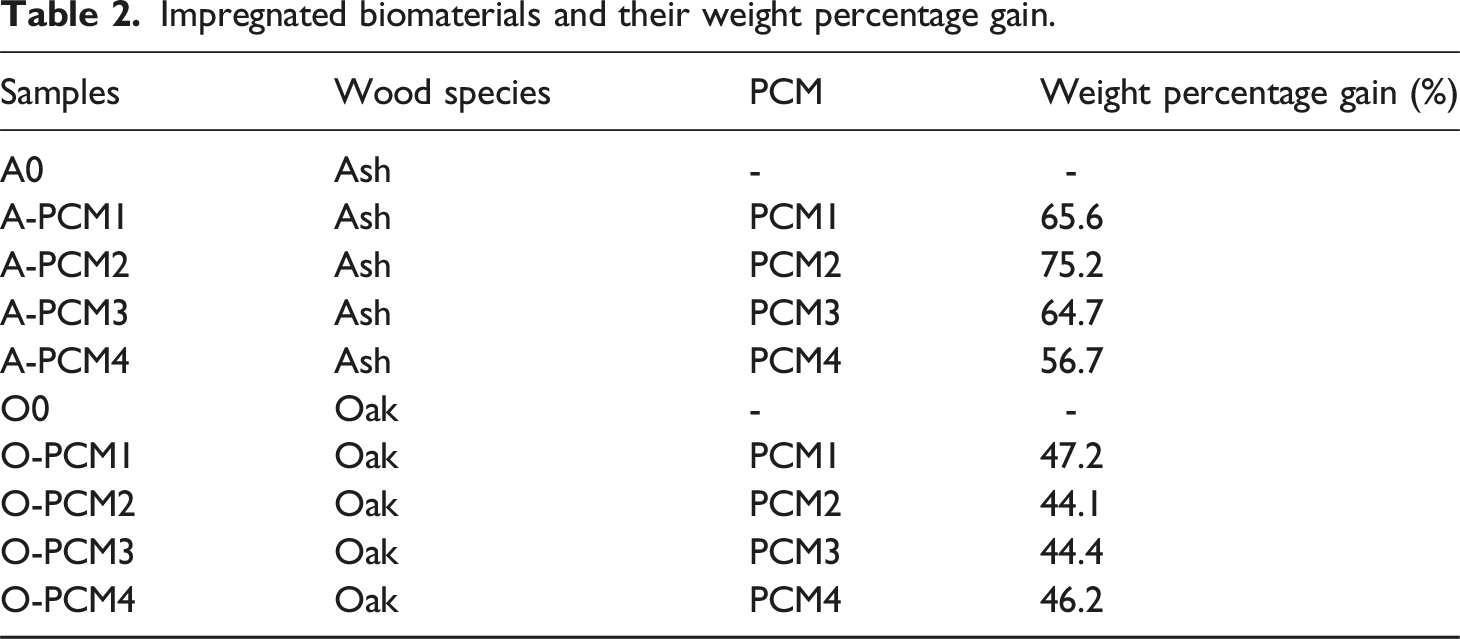
Impregnated biomaterials and their weight percentage gain.
Characterization of eutectic PCM and biomaterial
The morphological structure of each sample was determined using SEM (Sigma 300, ZEISS, Jena, Germany). Before analysis, the cross-sectional surfaces of the samples were coated with platinum. The chemical structure of eutectic PCM and impregnated biomaterial were determined by FTIR (Thermo Scientific, USA). The spectra were obtained with 32 scans for a wavelength range of 500–4000 cm−1 and a resolution of 4 cm− 1. The thermal properties were determined by DSC with a Pyris 1 (Netzsch DSC 404C Perkin–Elmer, USA). Measurements were carried out in an argon environment by heating and cooling in the temperature range of 0–90°C and at a temperature rate of 5°C/min. The dynamic mechanical analysis (DMA) tests were carried out on a DMA SA 3 (TA instruments, USA). The measurements were performed using a film tension mode with a strain of 0.1% and a frequency of 1 Hz for an appropriate temperature range depending on the blend composition, but the heating rate was fixed at 5°C/min.
Thermal conductivity (k) was determined by a home-made thermal conductivity analyzer according to ASTM E1225 using the method of Dou and Rodrigue. 39 The thickness (L) of the wood veneers (30 × 30 mm2) was measured using a digital caliper (Mastercraft, Canada). Each sample was placed between thin aluminum sheets (low thermal resistance). The upper plate temperature was fixed at 33°C and the lower plate temperature at 13°C, giving a temperature difference of 20°C with an average of 23°C. The temperatures were controlled with water-cooled Pelletier plates (Model K20, Haake, Germany) and measured with thermistances (TC-720, TE-Technology, USA), while the equilibrium heat flux (Q) was obtained by a PHFS-01 heat flux sensor (Flux Teq LLC, USA). The data reported represented an average of three replicates with standard deviations. An independent sample t test was used to determine whether there was a significant difference between two different wood species in thermal conductivity measurements.
Results and discussion
Morphological properties of impregnated biomaterial
Figure 1 shows images (different magnifications) of samples impregnated with eutectic phase change materials of both wood species. Eutectic PCM extensively filled cracks within the wood cladding and were impregnated into the lumen and the wood cell wall. This shows that the prepared eutectic mixture had good fluidity and compatibility with wood material. No obvious interfacial gap was observed between the eutectic PCM and the wood cell wall. These SEM images reveal that eutectic PCM were successfully located in wood cavities and homogeneously distributed. When images at higher magnification (x5000) are examined, the eutectic phase change materials are evenly distributed within the pores of both types of wood. As a result, a layered porous structure prevents the leakage of PCM during the impregnation process. SEM images of impregnated wood (A: ash wood veneer; O: oak wood veneer).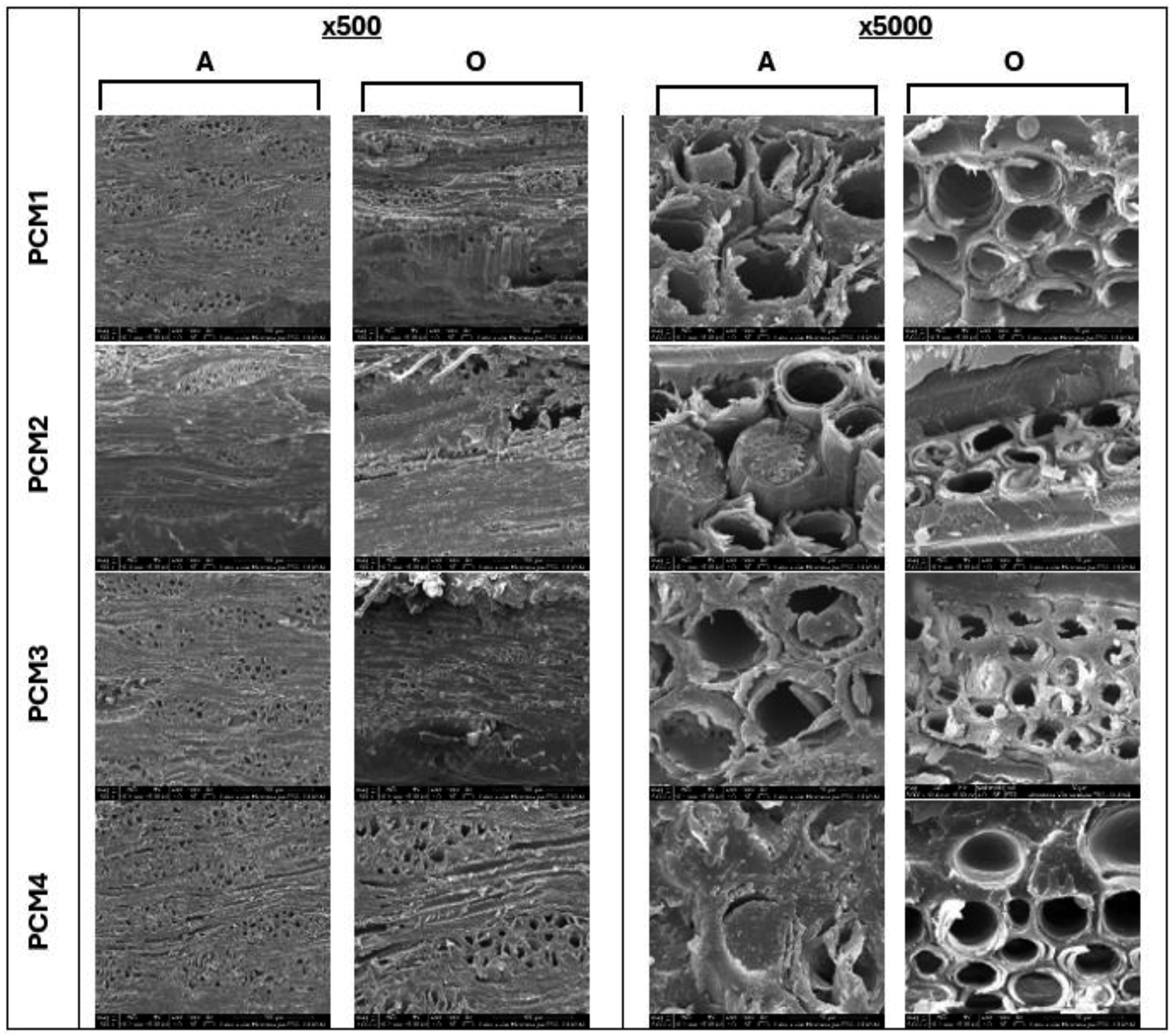
Structural characterization of eutectic PCM
The chemical structure of the prepared eutectic PCM composite was characterized by FTIR spectroscopy (Figure 2). In the LA-PA-SA spectra, the peaks at 2914 cm−1 and 2846 cm−1 represent the stretching vibration of the -CH3 and -CH2 group of LA-SA-PA, respectively. The peak at 1697 cm−1 is the characteristic absorption peak for the stretching vibration of C = O. The peak at 1465 cm−1 is the -CH- bending peak, while 935 cm−1 and 726 cm−1 correspond to rocking vibration and bending, which are characteristics of the aliphatic chain of LA-PA-SA, respectively. Similar results were also reported in the literature for other PCM samples.40,41 FTIR spectra of the impregnated wood.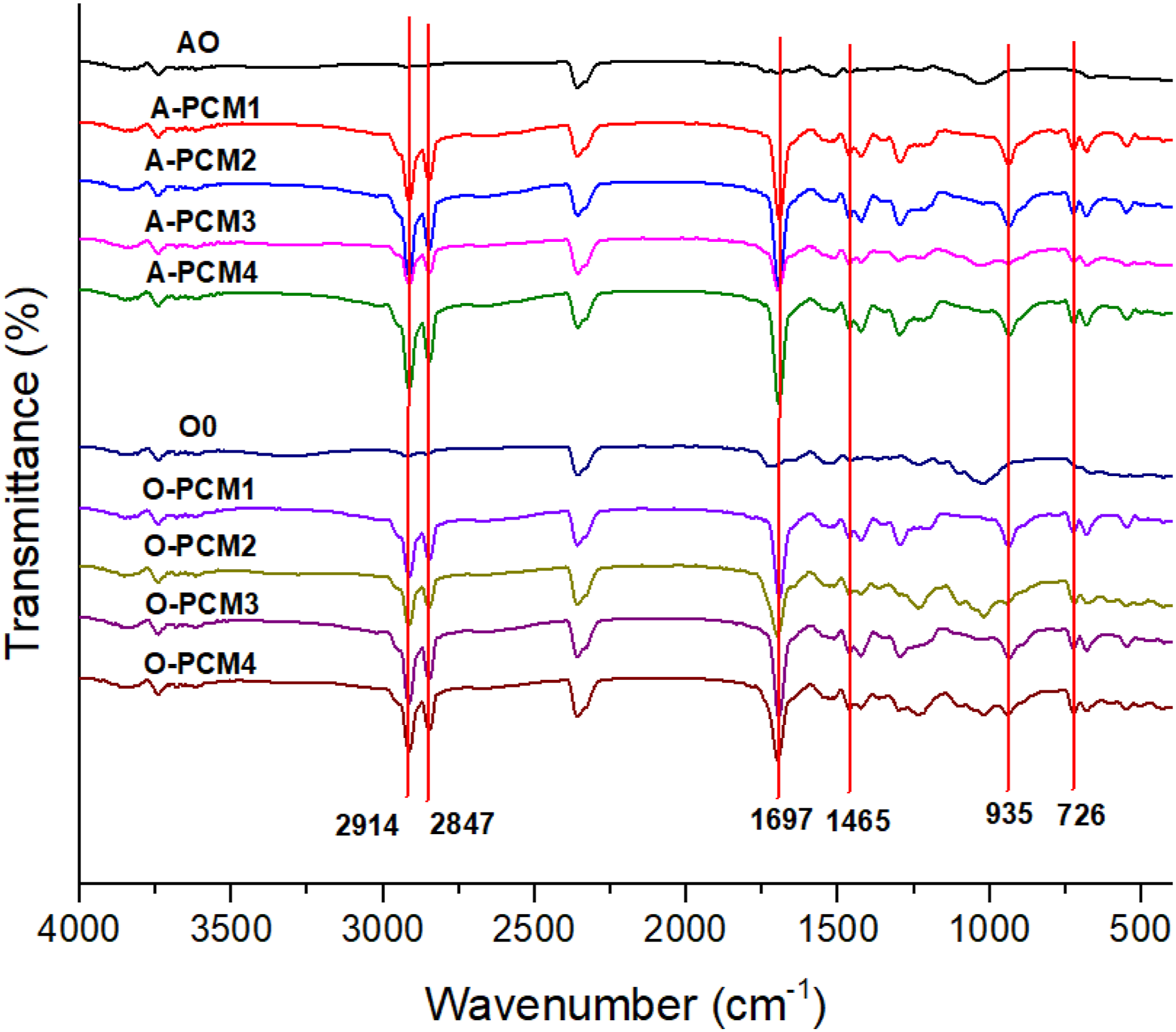
The FTIR spectra of wood veneers and PCM impregnated biomaterials are shown in Figure 3. While the characteristic peaks of PCM are not seen in the FTIR spectra of wooden coatings, they are seen in the FTIR spectra of all impregnated biomaterial coatings. These results prove that PCM were successfully impregnated into the grain coating. FTIR spectra of the eutectic PCM.
Thermal properties of eutectic PCM
To reduce the phase change temperature of fatty acids, using an eutectic mixture is a good method for low temperature building applications and Figure 4. presents typical DSC curves of the PCM produced. Zhang et al.
40
measured the melting temperature of LA, PA, and SA as 44°C, 62°C and 68.5°C respectively, but these phase change temperatures are too high to be used in building energy efficiency applications. This is why LA:SA:PA eutectic phase change materials with different mass ratios (PCM1; PCM2; PCM3 and PCM4) were prepared. DSC thermograms of the eutectic PCM.
Thermal properties of the eutectic PCM prepared.

Thermo-mechanical properties of impregnated wood
The storage modulus (E′) of impregnated biomaterial with different PCM was determined by dynamic mechanical analysis (DMA) by applying tensile tests. The storage modulus is used to indicate the strength of viscoelastic materials. The storage modulus curves of biomaterials impregnated with different PCM are shown in Figure 5, and the data obtained from the curves are shown in Table 4. It is observed that the rigidity of PCM decreases with increasing temperature and their energy storage tendency decreases due to chemical degradation. For ash wood, the best storage modulus is for A-PCM1 (11592 MPa at 25°C), while the lowest storage modulus is for A-PCM4 (6218 MPa at 25°C). For oak wood, the best storage modulus is for O-PCM3 (13937 MPa at 25°C), while the lowest storage modulus is for O-PCM4 (8448 MPa at 25°C). The modulus differences in the control samples of both wood veneers are lower than the modulus differences in the PCM impregnated biomaterial. The reason for this is that the mechanical strength of PCMs is lower than wooden materials. With increasing temperatures, PCMs undergo deformation, and the modules gradually decrease. As a result, it is clearly seen that the storage modules of biomaterials using eutectic mixtures prepared from three different PCM materials are significantly reduced. Storage modulus curves of impregnated biomaterial with different PCM. Storage modulus (E′, MPa) of impregnated biomaterial with different PCM.

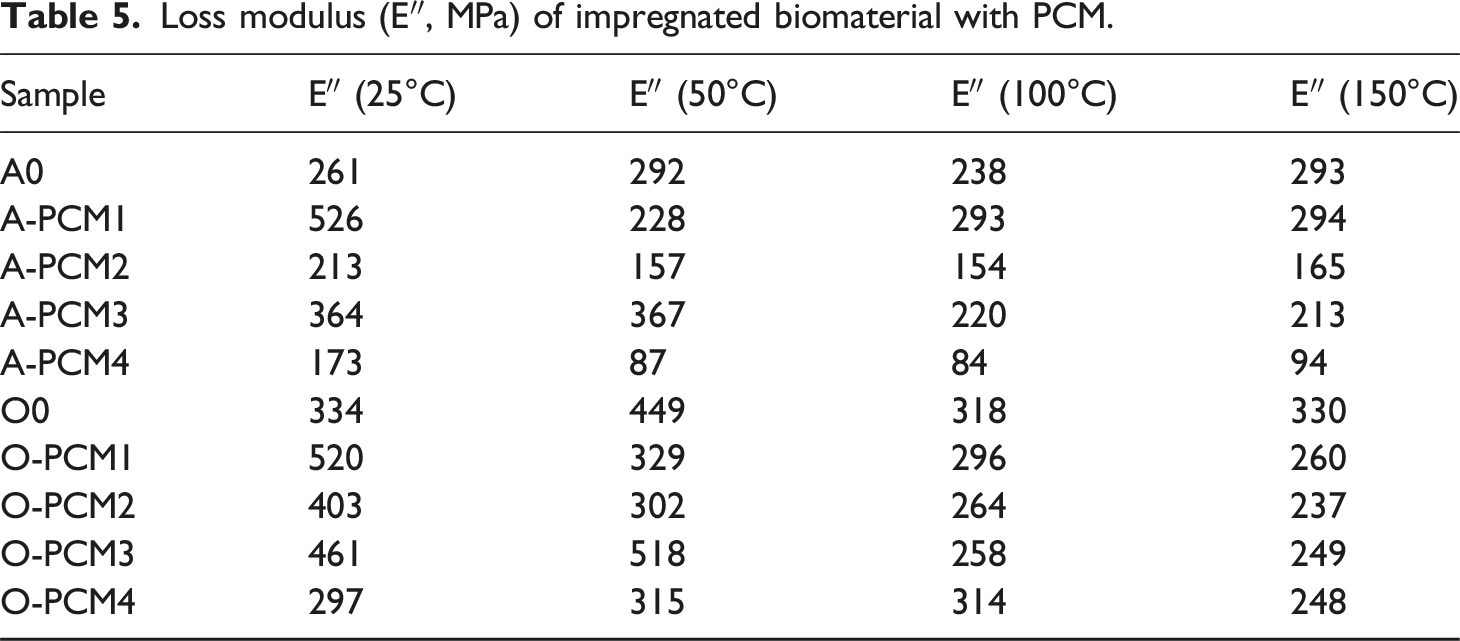
The loss modulus curves of biomaterials impregnated with different PCM are shown in Figure 6, and the data obtained from the curves are shown in Table 5. It is seen that the viscous behavior (E″) decreases with increasing temperature from 25°C to 40°C. For ash wood, the highest loss modulus is for A-PCM1 (526 MPa at 25°C), while the lowest loss modulus is for A-PCM4 (173 MPa at 25°C). For oak wood, the highest loss modulus is for O-PCM1 (520 MPa at 25°C), while the lowest loss modulus is for O-PCM4 (297 MPa at 25°C). As in the storage modules, the lowest loss modules are seen in the biomaterials where the eutectic mixture prepared with the mixture of triple PCM ratios is used. Loss modulus curves of impregnated biomaterial with PCM. Loss modulus (E″, MPa) of impregnated biomaterial with PCM.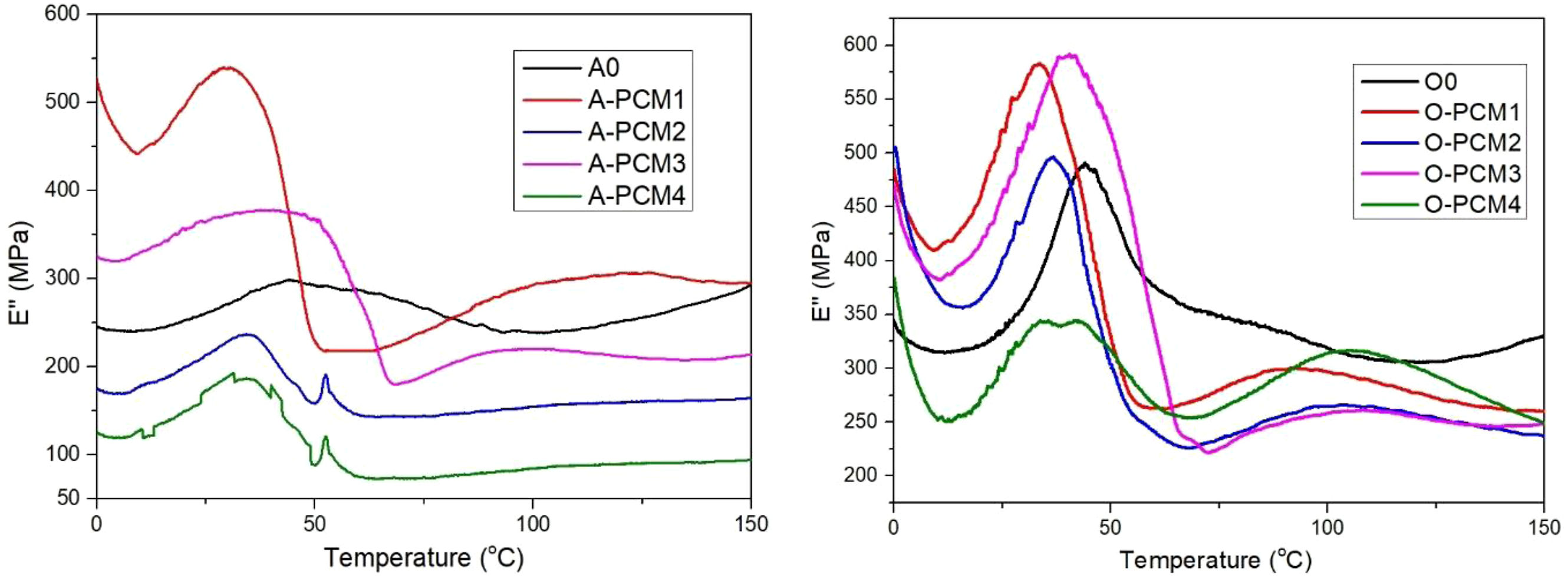
Thermal conductivity of impregnated biomaterial
Figure 7 reports the thermal conductivity of wood with eutectic PCM. In general, the thermal conductivity of woods without PCM is higher than those containing PCM. Since PCM have heat storage capacity, they store heat and prevent heat conduction. For ash wood, A-PCM4 has a slightly higher thermal conductivity compared to the control sample (A0). On the other hand, lower thermal conductivity values were measured for oak species compared to their control (O0). This is because solid wood is an amorphous polymer, and it is known that amorphous polymers mainly rely on the vibration and transformation of atoms in the cell wall to transfer heat to the next atom during heat transfer. Because the heat transfer of amorphous polymers is random and slow, it causes the thermal conductivity of wood to decrease.
42
On the other hand, Li et al.
43
used PEG as an energy storage polymer in delignified poplar wood using a high-temperature immersion method. According to the results obtained, they stated that the thermal conductivity of phase change impregnated (PEG-W) wood increased by 190% compared to the unimpregnated control sample. The t test results of thermal conductivity values are shown in Table 6. All materials except the A-PCM2 and A-PCM4 sample belonging to the ash wood species are statistically significant different from sample A0 as p < 0.05. All materials belonging to the oak wood species are statistically significant different from sample O0 as p < 0.05. Thermal conductivity of wood with eutectic PCM. T test results of thermal conductivity values.

Conclusion
In this study, four different eutectic phase change materials were prepared using three different fatty acids (lauric acid, stearic acid and palmitic acid) and impregnated in two different biomaterials (ash and oak wood). The SEM images showed that the PCM were successfully impregnated into the biomaterials and filled all the pores leading to homogeneous samples. Structural characterization of the PCM was carried out via FTIR which confirmed that they were successfully impregnated with biomaterial. The DSC results showed that the lowest melting point was 30.4°C for PCM4, while the highest latent heat capacity was 225.5 J/g for PCM2. In addition, it was generally determined by DMA that PCM1-based wood coating produced the highest and PCM-4 based wood coating produced the lowest storage modulus and loss modulus at 25°C. From the thermal conductivity results, it was determined that PCM reduced the thermal conductivity of wood coatings due to their heat storage properties. While the thermal conductivity of A0 was 0.151 W/m.K, the value for A-PCM1 was 0.108 W/m.K (28% less). On the other hand, the thermal conductivity of O0 was determined as 0.172 W/m.K, while O-PCM1 was determined as 0.105 W/m.K (39% less).
Footnotes
Acknowledgments
The authors gratefully acknowledge the Scientific and Technological Research Council of Turkey (TUBITAK-2219/BIDEB, 2022) for a visiting researcher scholarship to Dr Mustafa Zor to perform a post-doctoral stay Laval University (Quebec, CANADA).
Declaration of conflicting interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work was supported by Turkiye Bilimsel ve Teknolojik Arastirma Kurumu; 1059B192201030.
