Abstract
To address the growing demand for efficient energy conversion and storage materials in wearable personal thermal management (PTM), this study proposes a cooperative strategy of “molecular interface engineering + multi-scale structural design” to develop a flexible composite phase change material (PU-rG/Cu/C-PW) with a triple-network enhanced architecture. Using polyurethane foam (PU) as a scaffold, a uniform coating of reduced graphene oxide (rGO), copper nanowires (CuNWs), and carbon nanotubes (CNTs) was constructed via a cyclic impregnation-gradient reduction process, forming a “hard core-soft shell” porous micro/nano thermal conduction network. Paraffin wax (PW) was then stably encapsulated into the framework through vacuum melt infiltration. The resulting composite exhibits outstanding comprehensive properties: the mechanical strength of 128.44 kPa, the energy storage density of 159.81 J·g−1, and the thermal conductivity of 0.702 W·m−1 K−1, which is 331% higher than that of PW. The material maintains high flexibility and thermal reliability over 100 melting-solidification cycles. Moreover, under the conditions of 1.5 kW·m−2 simulated solar radiation and 10 V voltage, it demonstrated outstanding photothermal and electrothermal conversion efficiencies, reaching as high as 87.8% and 80.4% respectively. and has excellent multi-source drive thermal energy conversion capabilities. This work provides a novel material solution to overcome the long-standing challenges of leakage, low thermal conductivity, and poor mechanical strength in phase change materials (PCM), demonstrating great potential for applications in wearable electronics, solar energy utilization, and multi-source driven thermal management.
Highlights
• A novel multi-functional flexible composite phase change material coated with a three-dimensional porous micro/nano structure of “hard core-soft shell” was fabricated through “molecular interface engineering + multi-scale structure design”. • Thermal conductivity of the PU-rG/Cu/C-PW was 331% higher than that of the PW. The reinforcement can significantly improve the solar/electro-thermal conversion and leakage-proof capacity of PU-PW. • PU-rG/Cu/C mechanical strength reaches 128.44 kPa, and the cycling stability is presented by the stress-strain curves of PU-rG/Cu/C at 90% strain after 10 loading and unloading cycles. It’s can spring back quickly without plastic deformation. • The enthalpy value of PU-rG/Cu/C-PW was 156.98 J·g−1 after 100 melting/solidification cycles, and it maintained the same latent heat of 1st, with excellent thermal reliability.
Introduction
Against the backdrop of the global energy structure transformation, the development of renewable energy and the efficient utilization of green energy have become core issues in addressing population growth, carbon emission constraints, and the depletion of fossil fuels.1–3 As one of the most promising energy storage methods, thermal energy storage (TES) technology, as a key means to enhance energy recovery and utilization efficiency, converts solar energy, electrical energy and other energy into thermal energy through heat storage materials for storage and release, thereby regulating energy conversion, transportation and resolving the contradiction of mismatch between energy supply and demand in time and space. 4 Phase change materials (PCM) have become ideal carriers for TES systems due to their high energy storage density, nearly constant phase change temperature and low cost advantages, and have shown great potential in the field of solar thermal storage.5,6 Although solid-liquid PCM such as paraffin (PW) and fatty acids have excellent energy storage properties, their practical applications are still limited by material failure caused by liquid phase leakage,7,8 heat transfer lag due to low intrinsic thermal conductivity, 9 and poor mechanical properties that are difficult to meet the dynamic deformation requirements of wearable devices. 10 These deficiencies severely restrict the application of PCM in emerging fields such as battery thermal management, 11 solar energy utilization and photothermal conversion, 12 building energy conservation, 13 and flexible wearable electronics. 14
To suppress leakage, the three-dimensional porous skeleton encapsulation strategy is widely adopted, such as Three-dimensional (3D) porous matrix adsorption,15,16 electrospinning, 17 graphene aerogel,18,19 and metal foam.20,21 Among them, polyurethane (PU) foam, due to its unique interpenetrating network structure, high elasticity and processability, has become an ideal matrix for flexible composite phase change materials (FCPCM).22–24 Experiments show that PU-based composites have a stable phase transition temperature and good mechanical flexibility, and can be made into different shapes as required.25–29 However, pure PU has the drawbacks of poor thermal conductivity 30 and lack of photo-thermal conversion capability, 31 making it difficult to meet the requirements of solar drive systems.
In recent years, surface nanoengineering has become an effective way to enhance the comprehensive performance of materials. graphene oxide (GO) is used to fix PCM through hydrogen bonding and capillary interaction due to its high specific surface area, rich surface functional groups and excellent photothermal properties.32–35 However, single GO modification has the problems of high contact thermal resistance 36 and structural collapse after reduction. The latest research indicates that constructing a multi-component nano-hybrid system can achieve synergistic performance enhancement. One-dimensional (1D) copper nanowires (CuNWs) can establish fast heat conduction channels,37,38 carbon nanotubes (CNTs) enhance photo-thermal conversion efficiency through phonon transport,39,40 2D rGO nanosheets provide mechanical support and adsorption sites.33,34,41 This “one-dimensional + two-dimensional” multi-element system is expected to break through performance bottlenecks through multi-scale heat transfer mechanisms, while achieving high thermal conductivity, structural stability and photothermal synergy. However, existing research still has deficiencies in the controllable assembly of multiple nanomaterials on the surface of PU, the mechanism of synergistic action, and the balance of composite materials among multiple goals such as flexibility, energy storage, and safety. Especially for dynamic and complex scenarios like wearable thermal management, there is still a lack of systematic material design and performance optimization strategies. Therefore, developing an integrated strategy that can actively construct a solid interface and achieve the coordinated construction of stable encapsulation of nano-fillers and efficient thermal conduction networks has become the key to promoting the development of high-performance CPCM, and this is precisely the missing link in current research.
It is worth further noting that the potential biological toxicity and raw material cost issues of nanomaterials (CuNWs and CNTs) may also affect their practical application prospects. Therefore, in material design, it is necessary to balance performance improvement and safety sustainability. For instance, this can be achieved by optimizing the loading capacity of nanomaterials, constructing stable packaging structures to reduce leakage risks, and developing low-content, high-efficiency nano-hybrid systems to control costs.
In response to the above challenges, this study proposes a collaborative strategy of “molecular interface engineering + multi-scale structure design”. Through a new process of cyclic impregnation-gradient reduction, the uniform loading of rGO/CuNWs/CNTs on the PU skeleton is achieved, and a 3D porous micro/nano thermal conduction network structure of “hard core-soft shell” (PU-rG/Cu/C) is constructed. The CuNWs bridging rGO nanosheets was utilized to reduce the interinterface thermal resistance. The interlaced structure of CNTs inhibited the stacking of rGO and maintained a stable porous framework. The vacuum melt permeation method was introduced to fill PW into the PU-rG/Cu/C porous skeleton to construct the PU-rG/Cu/C-PW composite phase change material. This strategy achieves compound collaborative enhancement of performance, thereby realizing light/electrical-thermal conversion and storage in thermal management applications. Combining various characteristics of PU-rG/Cu/C-PW, a simple, environmentally friendly and integrated portable PU-rG/Cu/C-PW module was designed, which is applied to the independent temperature control capability of wearable thermal management and is conducive to improving personal thermal comfort. This provides a theoretical basis and technical reference for the design of the new generation of flexible energy storage and thermal management materials.
Experiment
Materials
PW (pathological grade) with a melting point of 55°C, graphite (99.95%, 325 mesh), NH4Cl (PT grade >99.7%), CuCl (AR grade 97%), and oleylamine (90%) were provided by China Aladdin Reagent (Shanghai) Co., Ltd. PU with a void density of 50PPI (98% porosity) from Yuanshengxing Electronic Materials Factory, Suzhou, China. Tri (hydroxymethyl) aminomethane (biotech grade 99.9%), dopamine hydrochloride (98%), single-wall carbon nanotubes (CNTs, >90%), and glycerol (AR grade 99%) were purchased from McLean Company (Shanghai, China). Acetone and anhydrous ethanol (AR grade) were purchased from China Tianjin Yongsheng Fine Chemical Co., Ltd. Ethylene glycol (AR grade), H2SO4 (∼98%), NaNO3 (AR grade), KMnO4, and H2O2 (AR grade 30%) were provided by Sinopharm Chemical Reagent Co., Ltd. N-hexane (>95%) and isopropyl alcohol (>99.7%) were purchased from Tianjin Guangfu Technology Co., Ltd.
Pretreatment of PU
Commercial PU was repeatedly ultrasonic-washed with acetone, anhydrous ethanol, and deionized water three times to remove the oil on the surface. Then, it was immersed in a mixed solution of tri (hydroxymethyl) aminomethane (0.446 g), dopamine hydrochloride (0.258 g), and deionized water (100 mL) for 24 h.
Synthesis of CuNWs
CuNWs were synthesized using the salt-assisted polyol method. Measure 100 mL ethylene glycol solvent, add 0.054 g of NH4Cl and 0.6 g of CuCl in turn, stirred for 20 min to form a light green solution; slowly add 8.4 mL of oleamine to the solution, stir vigorously with a magnetic agitator until the solution turns dark blue, and pour it into a 200 mL reaction kettle and heated it at 180°C for 10 h. After heating, the solution in the reactor was poured into an Isopropanol (IPA) mixture (n-hexane: isopropanol = 3:1) in the centrifugal pipe for washing and centrifuging. The solution was discharged, and only CuNWs in the centrifugal pipe were retained. Repeated the wash with the IPA mixture three times, and the CuNWs were dispersed into isopropyl alcohol.
Preparation of PU-GO/Cu/C
GO was prepared by mild hummers and dispersed into isopropanol to form a 2.0 mg·mL−1 GO suspension. This concentration, after previous optimization, can ensure the stability of dispersion while providing sufficient functional group loading sites for subsequent compounding. 42 And 100 g CuNWs were dispersed into the above suspension at a dispersed mass concentration of 5 g·mL−1. Then, 33.94 g CNTs were added to form a composite mixed solution, this dosage was determined through pre-experiments to achieve effective bridging and interweaving between GO sheet layers, and to synergically enhance the mechanical and thermal conductivity of the composite skeleton. And a well-mixed GO/Cu/C mixed solution was obtained in an ultrasonic bath for 40 min. The pre-treated PU was immersed in the mixed solution for 30 min, repeated dipping. The impregnated PU-GO/Cu/C was then vacuum-dried at 85°C for 2 h. The impregnation and drying were repeated 3 times to ensure that the composite solution was uniformly introduced into the PU frame to obtain GO/Cu/C coated PU frame composite (PU-GO/Cu/C).
Preparation of PU-rG/Cu/C-PW
PU-rG/Cu/C was prepared by vacuum impregnation. The obtained PU was put into a 100 mL polytetrafluoroethylene pressure vessel, and the GO was reduced to rGO by vacuum reduction at 200°C for 30 min in the presence of glycerol. PW was then placed on PU composite material and in an oven at 75°C for 1 h, the PU-rG/Cu/C-PW composites were prepared, removed with tweezers and naturally cooled to room temperature for curing. Finally, the sample was polished with sandpaper to remove excess PW on the surface of the composite material to obtain PU-rG/Cu/C-PW. The preparation process diagram is shown in Figure 1. PU-rG/Cu/C-PW preparation flow chart.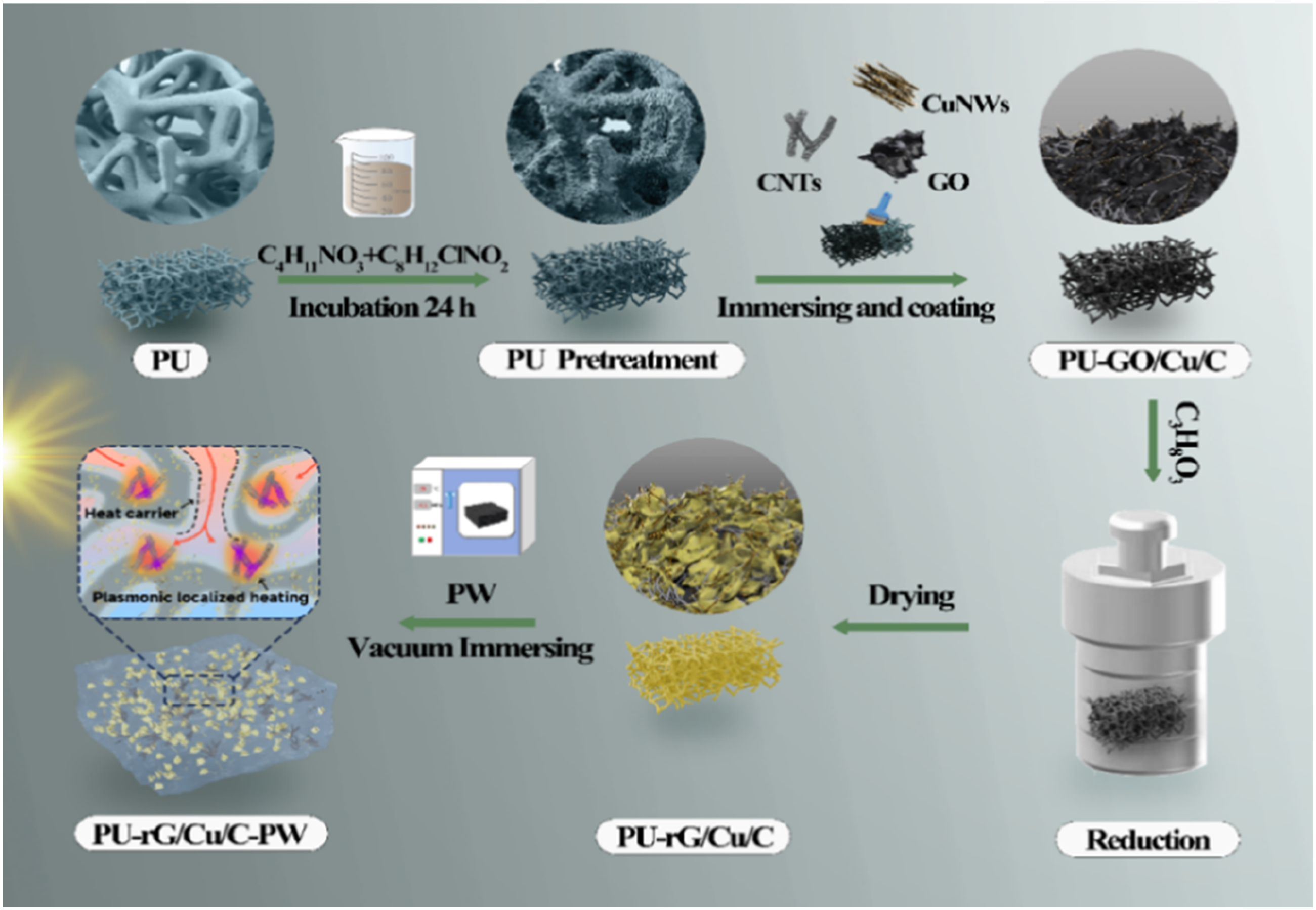
Characterization
All material characterizations and performance tests follow international or industry-wide common standards in the relevant fields. The morphology and microstructures of the CPCM were characterized by scanning electron microscopy (SEM, thermo scientific Apreo 2S, FEI Corporation, USA). The gold spraying treatment of the sample is carried out in accordance with the conventional SEM sample preparation process. Transmission spectra were collected in the range of 4000–500 cm−1 using Fourier transform infrared spectroscopy (FT-IR, Thermo Fisher Scientific, Nicolet IS50) with a resolution of 10 cm−1 and scanned 40 times. Crystal structure analysis was conducted by X-ray diffraction (XRD, Bruker, D8A A25X) using Cu Kα radiation (λ = 1.5406 Å), with a scanning range of 10°–80°, a step size of 0.02°, and a scanning speed of 5° min−1 in accordance with ISO 20203:2005. The thermal conductivity of the material was measured by using a laser flash thermal conductivity meter (C-THERM TCI type). The test was conducted in accordance with ASTM E1461-13 standard. Each sample was measured five times and the average value was taken. The absorption spectra of the samples in the wavelength range of 200–4000 nm were measured by using the ultraviolet-visible-near-infrared (UV-Vis-NIR) spectrophotometer (Agilent Technology Kari Series), and the integrating sphere accessory was used to reduce the scattering influence. The thermal decomposition behavior was evaluated in accordance with ISO 11358-1:2014 when the temperature was raised from 20°C to 800°C at a heating rate of 10°C min−1 using a thermogravimetric analyzer (STA2500, NCI Scientific Instrument Trading Co., LTD.) in the N2 atmosphere. The infrared thermal imaging spectrometer recorded the prepared sample’s surface temperature changes, heat storage, and heat release (EVERTE, EN60825-1). The test method refers to ASTM G173-03. The contact Angle between the sample and deionized water was measured by the static drop method using the contact Angle measuring instrument (JC2000DS2B). Referring to the ASTM D7334-08 standard, the average values at five different positions were taken. Compression tests were conducted using a universal testing machine (CMT2502, China MTS System Co., LTD.), with the loading rate set at 0.5 mm·min−1. The compressive stress-strain behavior of the material was determined in accordance with the ISO 604:2002 standard. The differential scanning calorimeter (DSC, pyris1DSC, PerkinElmer) was used to scan from 20°C to 90°C in the N2 atmosphere at a heating/cooling rate of 5°C·min−1, and the phase transition temperature and enthalpy were calculated in accordance with ISO 11357-3:2018. The temperature-time curves under the simulated solar light illumination were recorded by a solar-thermal test system (solar simulator and data collector), and the temperature-time curves of the CPCM under-connected to a DC power were obtained using an electro-thermal test system (DC power and data collector). The experimental setup complies with the conventional Joule heating test specifications.
Results and discussion
Structural characterization of PU-rG/Cu/C-PW
Figure 2(a)–(c) shows the surfaces of CuNWs, GO, and CNTs obtained by preparation. The PU has a 3D continuous network structure, and with the increase of magnification, the skeleton surface presents a smooth structure (Figure 2(d)). The surface pretreatment results in wrinkling and a rough texture of the skeleton surface from smooth but hardly affects the porous PU structure (Figure 2(e)). As shown in Figure 2(f) and (g), the physical entanglement of GO, CuNWs, and CNTs formed a homogeneous composite material. These hybrid composites and the PU skeleton were impregnated and dried several times and adhered well to the skeleton of PU, thus establishing a uniform thermal conductivity network (Figure 2(h)). Hydrophobic rGO surface enhances the hydrophobicity and oil wettability of PU-rG/Cu/C. There are many -OH and -COOH groups in the structure of rGO and CNTs, and these functional groups can directly form hydrogen bonds with C=O in PU molecules. In addition, the Cu-O chemical bond was formed by combining CuNWs and -COOH of CNTs. As a bridge between CuNWs and PU, rGO and CNTs firmly connected CuNWs and PU skeleton under the synergistic action of van der Waals forces, physical entanglements, and chemical bonds, providing excellent mechanical properties for composite materials. Furthermore, CuNWs, as a one-dimensional nanomaterial with high thermal conductivity, can prevent thermal conductivity damage under high strain and provide high thermal performance for CPCM. The higher CuNW activity promoted the adsorption and entanglement of CNTs and, as a rigid structure, avoided agglomeration between CNTs. Figure 2(i) shows that PW is wholly impregnated into PU porous material coated with rG/Cu/C. Due to the large number of adsorption active sites such as -OH and -COOH on the surface of carbon material, the leakage problem of traditional PCM can be solved, and PU-rG/Cu/C has good compatibility with PW, which can ensure the stability of the composite material. Figure 2(j)–(l) shows the mapping of the elements in the PU-rG/Cu/C-PW material section. These indicate the presence of C, O, and Cu elements, with C being the most abundant element owing to the high content of rGO. The small O element originated from the PU-PW mixture but is present in deficient concentrations. Meanwhile, the Cu element arises from the CuNWs mixture. Morphological characteristics of PU-rG/Cu/C-PW composites; (a), (b) and (c) are CuNWs, GO and CNTs surfaces; (d) and (e) are smooth surface network structure and pretreated rough surface of PU; (f) and (g) GO/Cu/C hybrid composite; (h) surface structure of PU-rG/Cu/C material; (i) surface structure of PU-rG/Cu/C-PW; (j), (k) and (l) are the mappings of C, Cu and O elements.
The structural evolution of PU-based CPCM was observed by X-ray diffraction (XRD) (Figure 3(a)). The strong diffraction peak of PU appears at 2θ = 22.1°, while the typical diffraction peak of PW occurs at approximately 2θ = 21.5° and 23.7° and is narrow; those of CuNWs appeared at 2θ = 43.4°, 51.5°, and 74.5°, and CNTs had a broad and robust diffraction peak at 2θ = 26.2°.
43
In the CPCM, the characteristic peaks of PU, rGO, CuNWs, and CNTs are all present, indicating that PU and CuNWs were present in the samples of PU-rG/Cu/C and PU-rG/Cu/C-PW film layers, respectively. Adding PU and CuNWs does not affect the crystal structures of individual components. It has not been altered, which indicates that PU and CuNWs have fantastic chemical compatibility with PW. Both PW and PU can crystallize, but PU tends to have a lower degree of crystallinity, resulting in a lower enthalpy. However, during the preparation process, the diffraction peak strength of the composites decreased after CuNWs, CNTs, and rGO films were coated on the PU surface. This was due to the solid capillary interaction force leading to the effective absorption of PW into the inner pore of PU, the formation of hydrogen bonds between PU and PW, and the adsorption of PW on PU. Together, they restrict the movement of the PW molecular chain and hinder crystallization. It should be emphasized that the intense peaks of CuNWs and PW cover the carbon peaks so that the carbon material has no prominent characteristic peaks. (a) XRD pattern of CPCM; (b) FT-IR spectrogram of CPCM.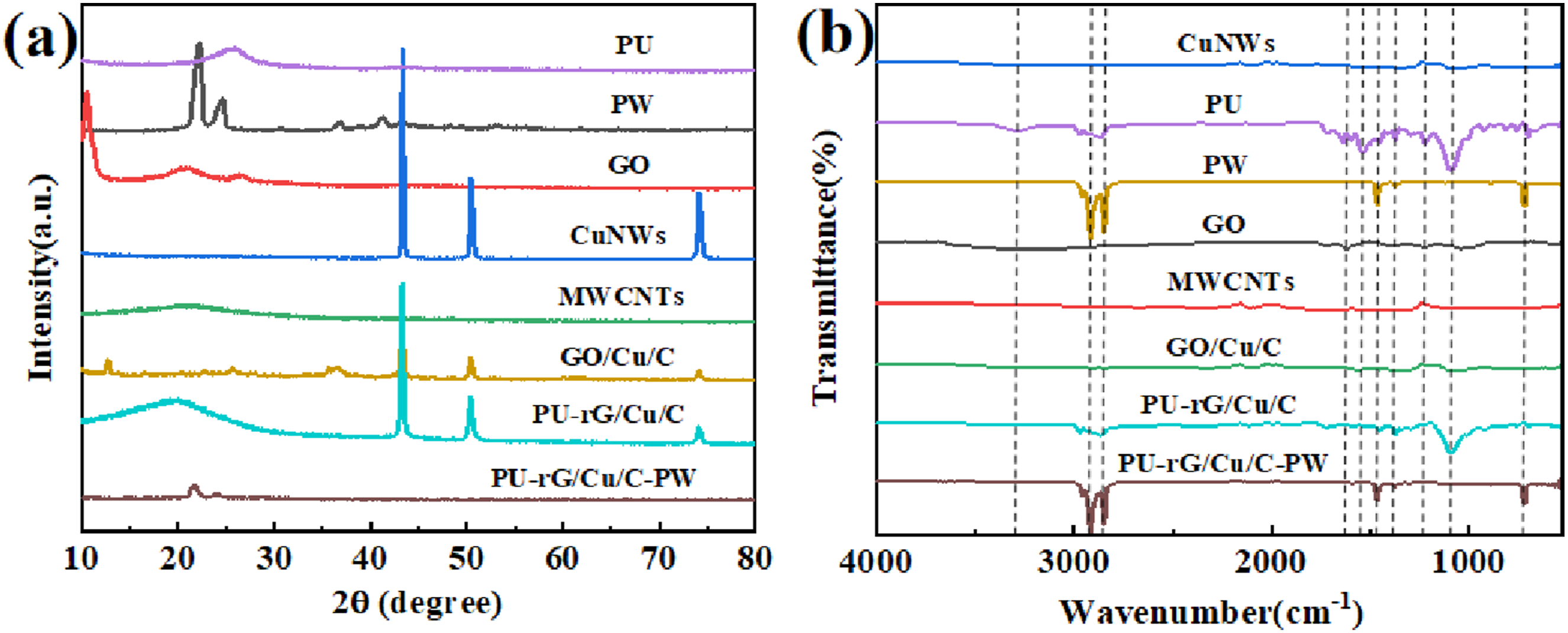
The chemical structure of CPCM was analyzed by FT-IR spectroscopy. Figure 3(b) presents the FT-IR spectra of PU, CuNWs, CNTs, GO, PW, PU-rG/Cu/C, and CPCM. A broad absorption peak at 3440 cm−1 is observed in the GO spectrum, attributed to the stretching vibrations of the -OH and O-H bonds. The PU and PW spectra exhibit peaks at 2918 cm−1 and 2849 cm−1, respectively, attributed to the C-H asymmetric and symmetric stretching vibrations of -CH3 and -CH2 groups. The absorption peaks at 1463 cm−1 and 1378 cm−1 are caused by the C-H bending vibrations of -CH3 and -CH2. The absorption peak 719 cm−1 is C-H in-plane rocking vibration.44–47 Additionally, the PU spectra show characteristic absorption peaks at 1530 cm−1 and 1341 cm−1, arising from the N-H deformation vibration and C-N tensile vibration of the -NHCOO group. Notably, the FT-IR spectra of PU-rG/Cu/C-PW composites display all the characteristic peaks of PU and PW without any shifts or generation of new characteristic peaks. However, the GO characteristic peaks at 1042 cm−1, 1623 cm−1, and 1725 cm−1 disappeared utterly, caused by C-O stretching, O-H bending vibration, and C=O stretching, proving that GO is reduced to rGO. 48 The characteristic peaks of all PW confirm the presence of long alkyl chains and are consistent with the structure of PW. FT-IR analysis indicates that no strong covalent bonds occur between PU and its components, but various weak chemical interactions, such as hydrogen bonds and van der Waals forces, make significant contributions to the physical integration and stability of the interface.49,50
Mechanical property and affinity characterization of PU-rG/Cu/C
Thanks to the physical combination between rGO and the PU skeleton, the high flexibility of PU is retained in the PU-rG/Cu/C composite material. To systematically evaluate its compression performance, we conducted static and cyclic compression tests on all samples. In the static compression test, five parallel specimens were prepared for each group of samples to eliminate individual differences. The compressive strength of the measured PU-rG/Cu/C-PW composite material was 128.44 kPa. The stress-strain curve and mechanical parameters were the average values of the five tests, and the relative standard deviation (RSD) was less than 5%. As shown in Figure 4(a), PU-rG/Cu/C exhibits superelastic behavior similar to that of pure PU: it can rebound rapidly at 90% compressive strain without plastic deformation. To further evaluate the mechanical durability of the material in dynamic deformation scenarios, we conducted 10 cyclic compression tests with strain reaching 90% on PU-rG/Cu/C. As shown in Figure 4(b), the stress-strain curves of 10 cycles highly overlap. After each unloading, the sample completely returns to its original state without any plastic deformation or structural damage. This indicates that the synergistic effect of the rGO/CuNWs/CNTs multi-component network significantly enhances the mechanical stability of the PU matrix and ensures the integrity of the thermal conductivity network of the composite material under repeated deformation. It demonstrates excellent and reliable mechanical resilience. This mechanical property shows significant advantages when compared with the carbon-based composite phase change materials reported in recent years. For instance, He et al. constructed a hybrid aerogel using GO, carboxylated CNTs, and polyvinyl alcohol, which exhibited a compressive strength of 16.14 kPa at 50% compressive strain. The structure maintained excellent stability even after impregnation with polyethylene glycol.
51
Chen et al. reported that the compressive strength of the rGO/CNTs composite aerogel reached 73.6 kPa, which fully confirms the synergistic effect between rGO and CNTs in constructing an enhanced network.
52
In contrast, the PU-rG/Cu/C composites prepared in this study maintain high flexibility while having competitive structural strength compared with the above-mentioned systems, and even perform better in some aspects. This is mainly attributed to the synergistic enhancement effect formed by the multi-component thermal conductive network we designed in 3D space. (a) Compressive stress-strain curves of PU, PU-rG/Cu and Pu-rG/Cu/C at 90% strain; (b) stress-strain curves of Pu-rG/Cu/C at 90% strain for 10 cycles; (c) digital photographs of Pu-rG/Cu/C deformation tests; (d) surface hydrophobicity of PU, (e–g) digital photographs of PU and PU-rG/Cu/C lipophilic hydrophilic tests.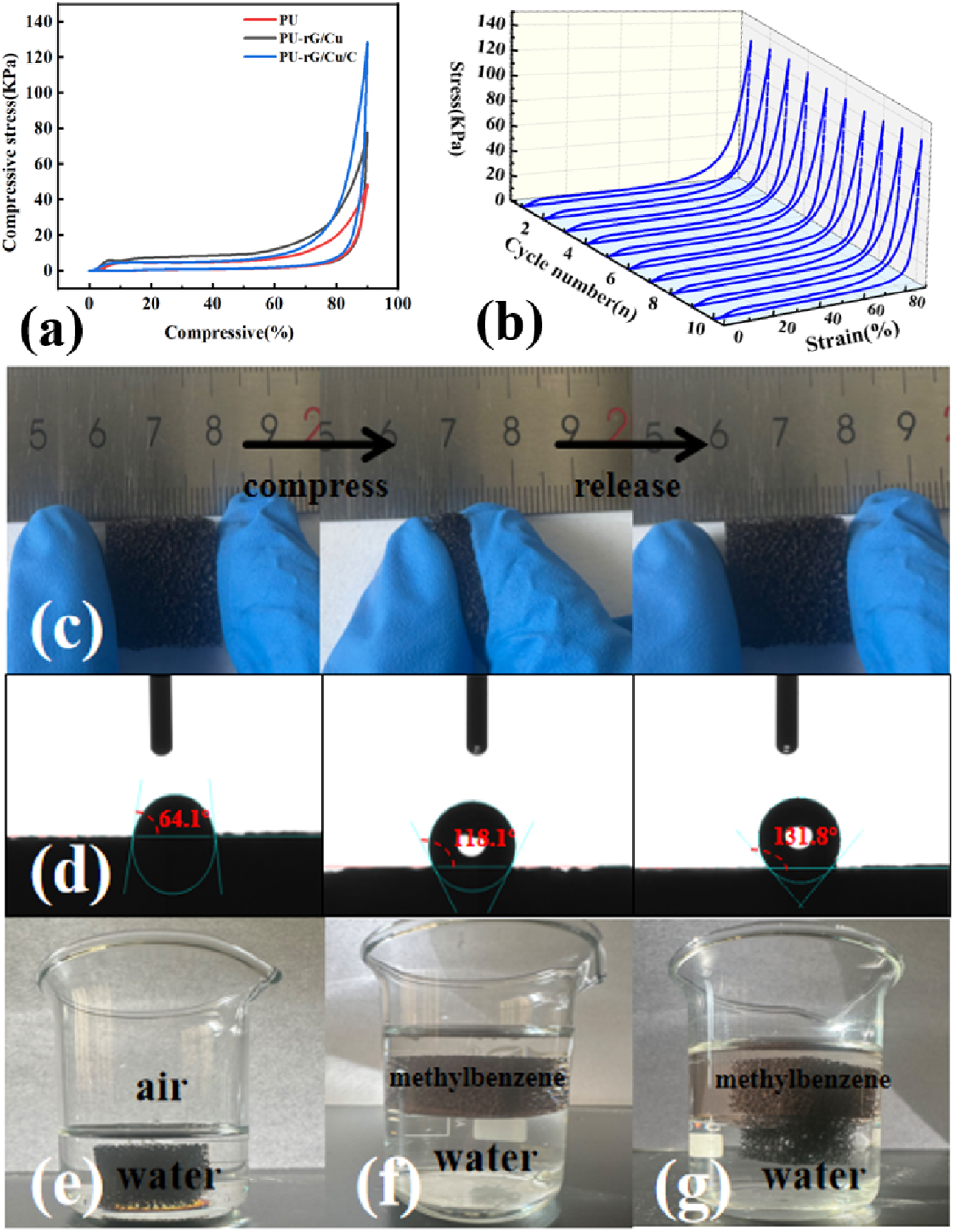
Figure 4(c) shows the digital photographs of the deformation test. When the PU-rG/Cu/C carrier is compressed and released with fingers, its porous material is quickly recovered without shedding any impurities, and the three-phase nanomaterials are tightly attached to the surface of PU. In addition, the good mechanical properties of the PU skeleton can maintain the stability of CPCM, such as preventing the volume contraction of PW during the melting/solidification process and damaging the structure of the entire composite material. The exceptional plasticity of PU can realize the synchronous contraction of the three-phase nanomaterial rG/Cu/C, effectively avoiding the structural collapse of PU-rG/Cu/C-PW. The results show that the PU-rG/Cu/C carrier can maintain the thermal conductivity pathway of CPCM stably under deformation conditions.
It can be seen from Figure 4(d) that PU is a porous hydrophilic material when water droplets drip onto the surface of PU with a contact Angle of 64.1°. However, when water drips onto the PU-rG/Cu/C surface, the water droplets stay on its surface, and the contact Angle is 118.1°, indicating the hydrophobic property. Thus, the hydrophobic property of the PU-rG/Cu/C composites is attributed to the evenly and continuously distributed rGO. When the water drips onto the PU-rG/Cu/C-PW surface, the contact Angle is 131.8°, and the water droplets stay on the surface and remain spherical for at least 15 min, with super-hydrophobic properties. The polarity of PU modified by carbon material is conducive to the adsorption stability of PCM. That is, rGO is conducive to the stable adsorption of PW. This result shows that the polarity of the PU surface can be well adjusted by a simple rGO modification method, and a large number of polar and non-polar CPCM can be generated. In addition, we also put the PU and PU-rG/Cu/C samples in beakers containing water and toluene or only water, respectively. In the beaker (Figure 4(e)–(g)), the PU sample is completely submerged in water, but the PU-rG/Cu/C sample can float stably on the surface of water or be immersed into the region of toluene solution. The lipophile hydrophobic characterization of PU-rG/Cu/C-PW can be utilized by the hydrophobic property of PU-rG/Cu/C, and it is of great significance in extending the potential application of composite CPCM.
Thermal performance and cycle stability
Shape stability is an essential characteristic of CPCM in practical applications, as it prevents solid-liquid leakage and impacts their long-term reliability during thermal cycling storage. Leakage of different samples at various temperatures is confirmed by heating samples on a heating table and capturing results with a digital camera. All samples have well-defined shapes at room temperature (Figure 5(a)). As the temperatures are higher than the phase transition temperature (∼50°C), significant liquid leakage occurs in both pure PW and PU-PW. However, the leakage of modified carbon material is significantly reduced. In particular, PU-rG/Cu/C-PW only has a slight leak of PW at continuous high temperature, which may be due to the solid capillary force of PU structure caused by the hydrophilicity of rGO modified PU and the molecular interaction between rGO and PW further improves its limiting ability. Figure 5(b) records the instantaneous temperature response when the infrared thermal imaging spectrometer simulates solar illumination. The color of PU-rG/Cu/C-PW was brighter than the other three samples at 15 s, and the more colorful the color, the higher the surface temperature. DSC tests are performed to present the thermophysical properties, including enthalpy change and phase transition temperatures of the PU-rG/Cu/C-PW. Typical DSC curves are presented in Figure 5(c), which consists of a melting/solidification cycle. The corresponding thermal parameters are illustrated in Table 1. For PW, a strong peak can be observed at 62°C. Compared with the PW, the PU-rG/Cu/C-PW sample has a similar curve with two-phase transition peaks, in which the weak peak at the low temperature corresponds to the solid-solid phase transition of PW. In contrast, the strong peak with more considerable latent heat corresponds to the solid-liquid transition of the PW.
53
Interestingly, the peak positions of the PU-rG/Cu/C-PW show a specific deviation compared to pristine PW. Compared with the supercooling range of pure PW, the reduction in the supercooling of CPCM should be because PU-rG/Cu/C acts as a heterogeneous nucleating agent and reduces the nuclear energy, thus accelerating the phase change of the PW.
54
Besides, the PU-rG/Cu/C provides efficient continuous heat transfer channels and accelerates the rate of thermal transfer of the composite, which also results in the phase transition of the PU-rG/Cu/C-PW occurring earlier than that of pristine PW. At the same time, the DSC melting and solidification cycle curves of PU-rG/Cu/C-PW almost completely overlap after 100 cycles, and the enthalpy value does not change significantly (Figure 5(d)), indicating that PU-rG/Cu/C-PW has good thermal reliability during long-term energy storage cycles. (a) Digital photographs of shape stability of pure PW and CPCM at different temperatures; (b) IR diagram of heat transfer evolution in solar-thermal process of PW, PU-PW, PU-rGO/Cu-PW, PU-rGO/Cu/C-PW; (c) DSC heating and cooling curves of pure PW, PU-PW and CPCM; (d) 100 phase change cycles in the melting and solidification process of PU-rG/Cu/C-PW; (e) thermogravimetric curves of PU, GO, PW and CPCM; (f) thermal conductivity of CPCM. Summary of DSC heat storage and heat release characteristics of PW, PU-PW, and CPCM. Tpeak: peak temperature; Ton: onset temperature; Tend: end temperature; M: melt process; S: solidification process.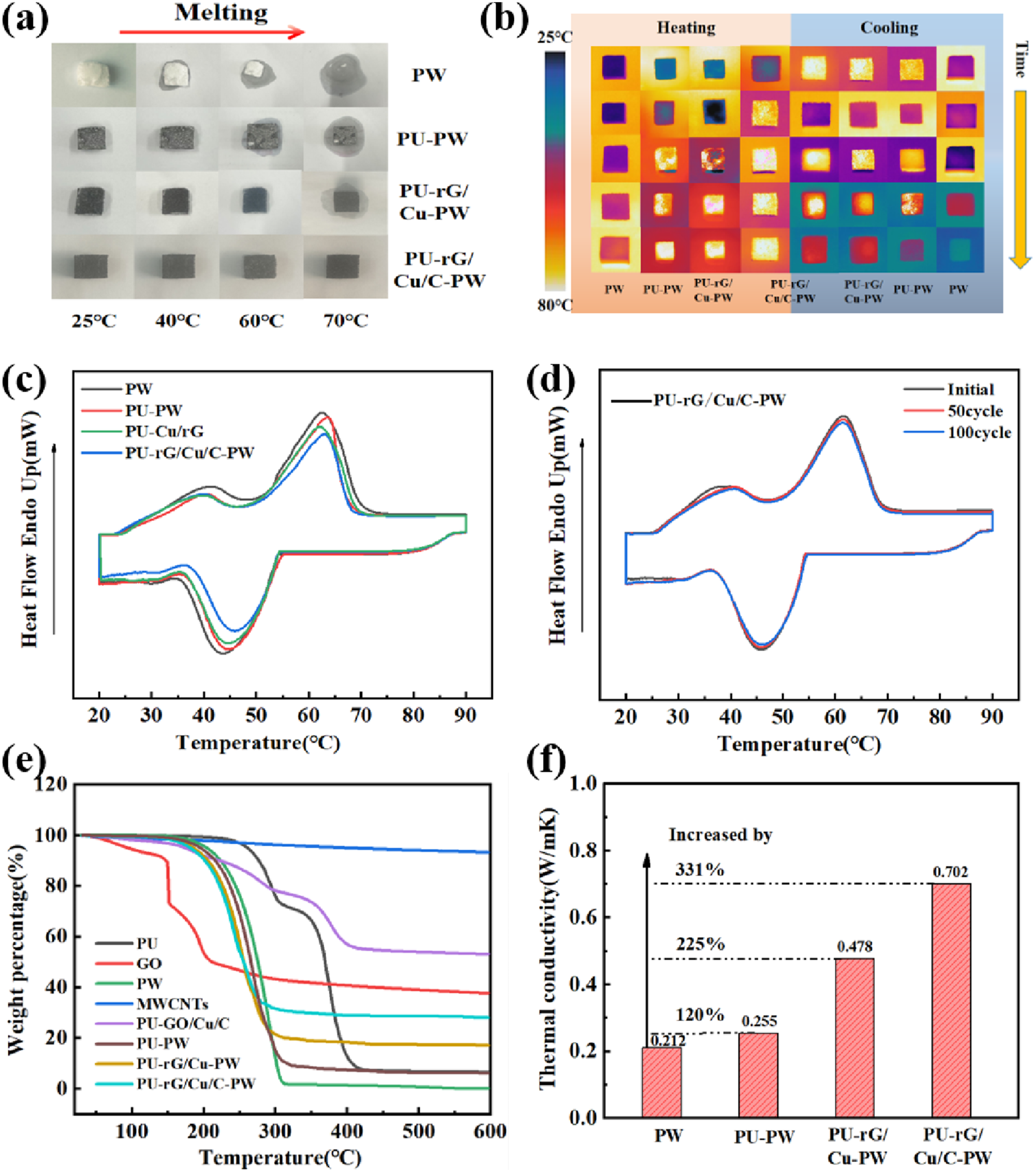
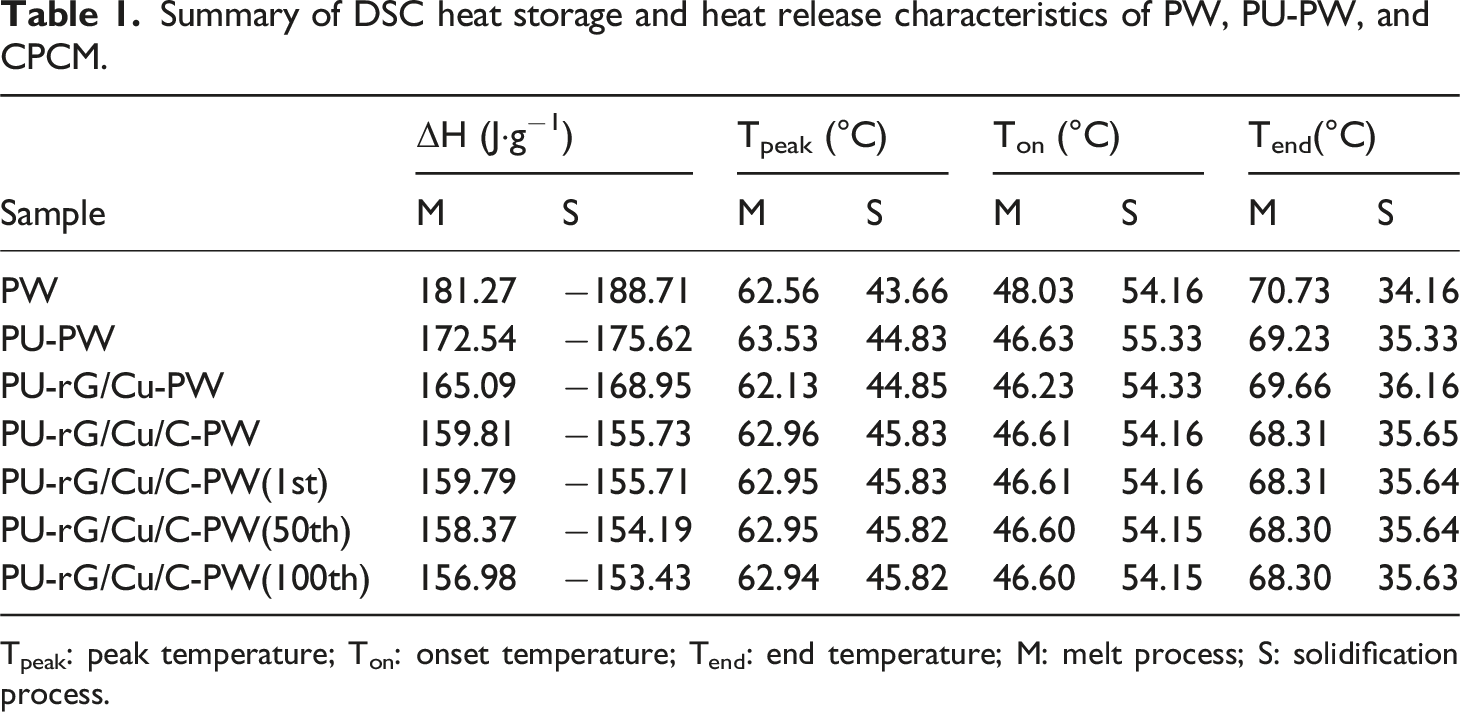
It can be seen from Figure 5(e) that the decomposition temperature of PW is between 180∼312°C, the mass loss is sharply at 180°C, the total oxidation decomposition is near 312°C, and the mass loss rate is between 180°C and 312°C is about 99%. In contrast, PU exhibits two distinguishable plateaus in their curves. The decomposition temperature of PU is in the temperature range of 247∼417°C. Its thermal decomposition can be divided into two steps; the first step occurs at 247∼309°C, with a mass loss of 28%, and the second step occurs at 352∼417°C, with a mass loss of 63%. The mass of GO loss rapidly in the (150∼200°C) range is due to the reduction of GO at 200°C. It is worth noting that the final mass loss of PU-PW is about 94%, and PU-rG/Cu-PW and PU-rG/Cu/C-PW are about 83% and 72%. Because GO, CuNWs, and CNTs did not decompose at high temperatures, the residual mass of CPCM was relatively high. This lower PCM filler content avoids the great sacrifice of fusion enthalpy and imparts a higher energy density to PCM. These results further prove the homogeneity of PU-rG/Cu/C-PW composites, indicating that the CPCM has good thermal cycle reliability and stability.
In addition to composite materials’ high latent heat and stabilized properties, thermal conductivity is crucial to the composites. The thermal conductivity of pure PW is only 0.212 W·m−1·K−1 Figure 5(f), and the melamine PU loaded with PW (PU-PW) has a thermal conductivity value of 0.255 W·m−1 K−1, which means the PU plays less effect on thermal transfer property of composites. This is due to the low thermal conductivity of the polymer skeleton of PU and the low thermal conductivity of the air in the pores not being occupied by the PW. With the addition of rG/Cu composites, the thermal conductivity of PU-rG/Cu-PW composites increases to 0.478 W·m−1 K−1, which is 225% higher than that of pure PW because the thermal conductivity of CuNWs is related to the porous interconnected thermal conduction path formed by PU skeleton. After CNTs were increased, the thermal conductivity of PU-rG/Cu/C-PW was increased dramatically to 0.702 W·m−1 K−1, which was 331% higher than that of pure PW. This is due to the CNTs attached to the GO surface, which increase the thermal conductivity adsorption site and help to improve the thermal conductivity of the PW. For each of the above composite materials, we prepared and measured at least five independent samples. All samples were carefully crafted to ensure consistent geometry and surface flatness, with a relative standard deviation not exceeding ±0.05%, indicating that the measurement results have good repeatability. Overall, PU-rG/Cu/C-PW has exceptional thermal conductivity and heat transfer performance. This demonstrates that a 3D continuous framework structure can improve heat transfer performance. The continuous porous 3D thermal conductivity structure of the PU-rG/Cu/C provides an effective channel for phonon transfer, which is beneficial for thermal transmission, reducing the strong interfacial thermal resistance between the matrix and the filler. 55
Solar-thermal conversion performance
A solar-thermal conversion system was assembled to verify CPCM’s solar-thermal conversion and heat storage performance, as shown in Figure 6(a). The solar-thermal conversion unit comprises a solar simulator (CEL-S500 Xenon lamp), samples, thermocouples, data acquisition instruments, and a computer. CEL-NP2000 verified the Solar illumination intensity. The temperature variation of the CPCM was monitored by a T-type thermocouple and data acquisition instrument, Which records the temperature every 5.0 s. Additionally, considering that the CPCM is affected by the error of simulated sunlight irradiation, this paper adopts the average algorithm of each temperature point to analyze and calculate its storage efficiency and other fundamental evaluation indexes. A schematic diagram of the energy storage process of the PCM is displayed in Figure 6(b). The heat storage capacity of CPCM can be expressed as follows
56
: (a) Image of a solar-thermal test system for CPCM; (b) schematic diagram of solar-thermal energy storage process of CPCM; (c) UV-Vis absorption spectra of CPCM; (d) the solar-thermal conversion temperature curve of the CPCM under the simulated sunlight of 1.5 KW·m−2; (e) temperature-time curves of CPCM under different simulated solar illumination; (f) heat storage and change of heat storage rate of CPCM; (g) solar-thermal conversion efficiency of PW and CPCM; (h) η of PU-rG/Cu/C-PW at the 1st, 50th and 100th cycles of thermal cycling; (i) digital photos and infrared thermal images of PU-PW, PU-rG/CuPW, PU-rG/Cu/C-PW control group under sunshine.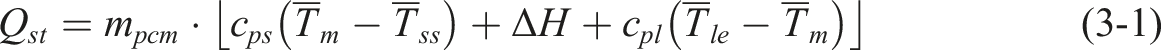



The average heat storage rate of the equipment q

Solar-thermal conversion efficiency (η) is another crucial evaluation index of solar heat utilization in the process of heat storage, and it was calculated as follows
57
:
In the study of PU-rG/Cu/C-PW, it is essential to use materials with high absorption capacity to achieve excellent solar energy absorption and conversion. Figure 6(c) shows the absorption capacity of different materials using a UV-Vis spectrophotometer. The results show that the light absorption capacity of pure PU and PW is fragile. Compared with PU and GO, only using rG/Cu load PU-PW, the light absorption capacity of the material is improved. The surface modification of PU-rG/Cu-PW with the addition of CNTs enhanced the light absorption of the CPCM (samples of the same diameter and thickness) over the entire UV-Vis range. Especially in the visible range, due to the low reflectance and practical light capture ability of the rGO film layer, the composite loaded with 10 rGO already exhibits a primarily saturated black color on its surface; Its behavior is similar to that of a black body, and its light absorption ability is improved.58,59 At the same time, due to the pore network of PU, after the incident light reaches the surface of the CPCM. Most of the incident light will be internally reflected several times in the porous network so that the CPCM can rapidly absorb solar radiation and convert it into thermal energy. The results indicate that PU-rG/Cu/C-PW composites have various application prospects, especially solar-thermal conversion and thermal energy storage.
Figure 6(d) and (e) depicts the temperature-time profiles of various samples exposed to the same simulated solar illumination, along with the temperature-time profile of a single sample under varying simulated solar illumination. The figures indicate that under the light intensity of the solar simulator (1.5 KW·m−2), the temperature rise curve of CPCM based on PW has undergone three processes: solid-solid heating stage, equilibrium phase transformation stage, and solid-liquid heating stage. At this radiation intensity, the solar-thermal energy storage and energy storage rates of PU-rG/Cu/C-PW reach 1185.3 J and 2.37 J·s−1, 1.57 times that of PU-PW (Figure 6(f)). After 500 s of irradiation, the temperature of PU-rG/Cu/C-PW is 77.07°C. At the same time, that of PU-rG/Cu-PW, PU-PW, and PW are 68.39°C, 57.66°C, and 46.47°C, respectively. Compared with pure PW, the thermal response time required for PU-based CPCM at 50°C is significantly reduced. The figures indicate that the Component and the simulated solar illumination’s light intensity significantly influence the composites’ solar-thermal absorption and conversion. Specifically, the high thermal conductivity of the rGO, CNTs, and CuNWs has a significant effect on enhancing solar energy absorption of CPCM, that is, better solar-thermal conversion performance under the same light, while a more substantial light intensity results in better solar-thermal absorption and conversion performance. These findings suggest that CPCM can rapidly convert solar energy into thermal energy, facilitating the storage and release of thermal energy.
In Figure 6(g), we present the maximum temperature attained by samples PW, PU-PW, PU-rG/Cu-PW, and PU-rG/Cu/C-PW under the simulated solar irradiation intensity is 1.5 KW·m−2. Additionally, we investigate the storage efficiency of the CPCM for solar thermal energy, which is found to be as follows: At the simulated solar irradiation intensity of 1.5 KW·m−2, η of PU-PW, PU-rG/Cu-PW, and PU-rG/Cu/C-PW reached 55.8%, 72.5%, and 87.8%, respectively, benefit from rGO and CNTs ultra-high solar-thermal conversion ability. As shown in Figure 6(h), after 100 thermal cycles, η value decreased slightly, which may be related to the effect of PW repeatedly penetrating rG/Cu/C in porous PU on light scattering, but still showed outstanding solar-thermal energy conversion efficiency. Moreover, after thermal cycling, PU-rG/Cu/C-PW maintains a fast thermal response speed during the test, indicating that the composite has favorable thermal cycling stability.
To demonstrate the practical photothermal application potential of composite materials, on a sunny day in Baotou, Inner Mongolia, with an average ambient temperature of 33.5°C and an average solar irradiance of 925 W·m−2, we applied three samples, namely PU-PW, PU-rG/CU-PW and PU-rG/Cu/C-PW, to the back of the hand for solar photothermal tests. The digital photo and infrared thermal imaging images of the samples in the outdoor environment are shown in Figure 6(i). In this case, the areas where no material is attached are used as a blank control. As seen, the PU-rG/Cu/C-PW and PU-rG/Cu-PW reaches 45.6°C and 42.8°C with 120 s of sunlight exposure, while the PU-PW (40.3°C) does not differ much from the blank group (38.5°C). This directly demonstrates the significant heating advantage of the multi-element nanocomposite system under real lighting conditions.
Conversion and storage of electrical energy
Because of the intermittency of solar energy, electric conversion is also essential. The electro-thermal conversion device uses a DC power supply to provide energy. For the electro-thermal conversion, rGO and nanomaterials give the CPCM excellent electrical conductivity, the PU-PW blend provides the CPCM with a specific resistance, and the high electrical conductivity and low resistance of the composite give it an excellent electro-thermal conversion. Figure 7(a) shows the schematic diagram of the electro-thermal conversion equipment. A DC power supply system investigates CPCM’s electro-thermal conversion and storage performance. GO, CuNWs, and CNTs were entangled in forming a uniform composite material. Uniform CNTs caused by van der Waals forces are the main adhesion force of mixed composites adsorbed on the surface of the PU skeleton. At the same time, GO, CuNWs, and CNTs have fantastic electrical conductivity (Figure 7(b)), and conductive rG/Cu/C foam is connected to an external power supply to make the small lamp emitting light. Figure 7(c) and (d) depicts the temperature-time profiles of various samples at the same DC voltage and the temperature-time profile of a single sample at different voltages. The results show that the temperature of PU-rG/Cu/C-PW increases faster with time compared to PU-rG/Cu-PW and PU-PW at 10 V, demonstrating that the addition of rGO and nanomaterials at the same voltage, the quicker the electro-thermal conversion. The higher the voltage of the same sample, the higher the temperature achieved simultaneously. They further indicate that CPCM can convert electrical energy into thermal energy for storage and release. (a) Schematic diagram of experimental device for electro-thermal conversion of CPCM; (b) digital image of a small lamp emitting light from a CPCM under direct current; (c) the change of temperature and time of thermal transformation of CPCM at 10 V voltage; (d) temperature-time curves of CPCM at different voltages; (e) the maximum temperature achieved by the CPCM and the electro-thermal conversion efficiency diagram of CPCM; (f, g) two integrated and portable CPCM modules for personal hyperthermia; (h) digital and infrared thermal imaging images of PU-rG/Cu/C-PW placed on the finger.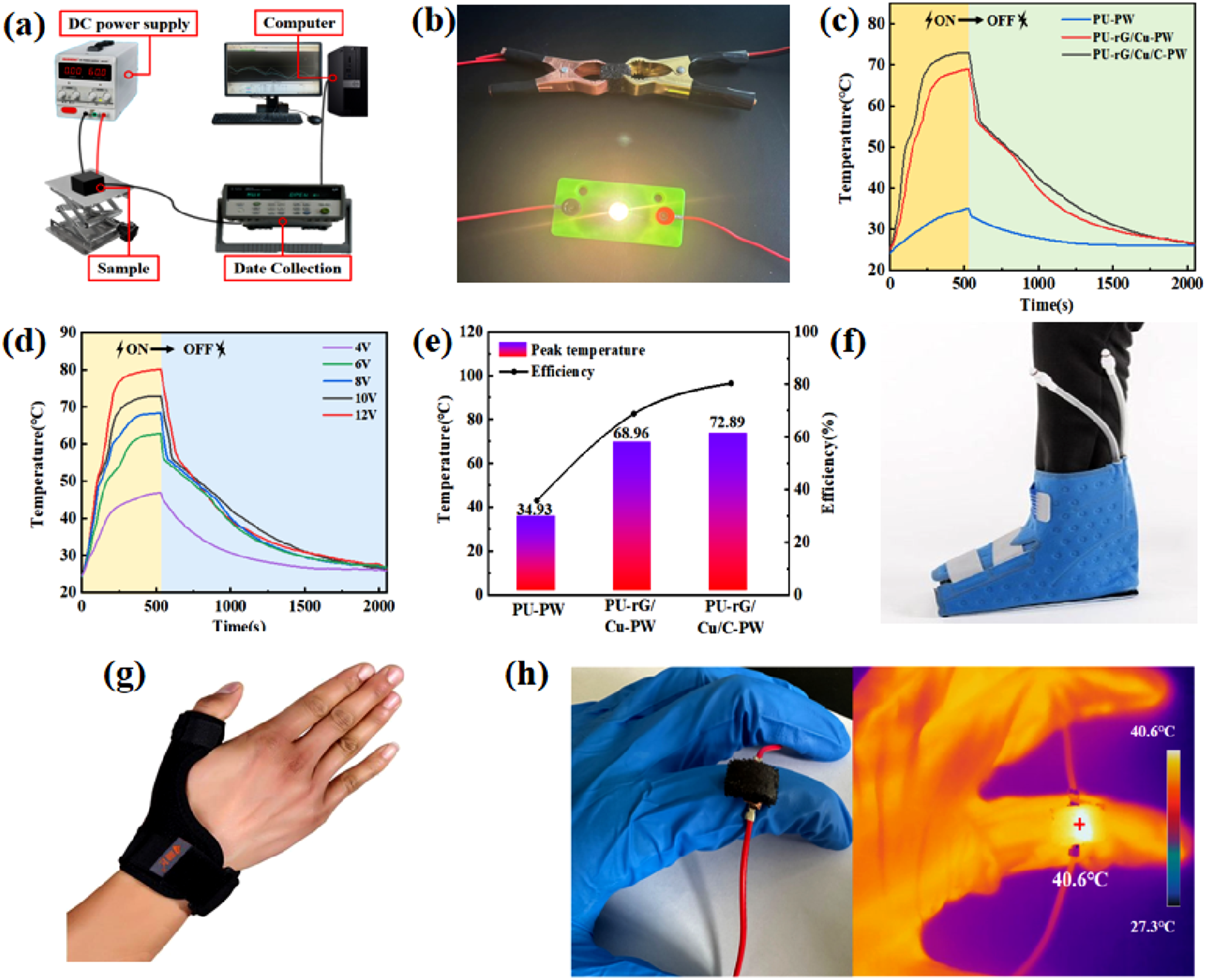
The CPCM in this study has excellent electro-thermal conversion efficiency, which is calculated as follows:
As shown in Figure 7(e), the maximum temperature of PU-rG/Cu/C-PW after 500 seconds of being connected to a DC power can reach 72.89°C at 10 V. The temperature of PU-rG/Cu-PW and PU-PW can reach 68.82°C and 34.78°C, respectively, and the electro-thermal conversion efficiency is 80.4%, 68.82%, and 35.8%, respectively. This is due to the rGO ultra-high thermal conversion performance of over 97%. Therefore, the CPCM in this study has excellent electro-thermal conversion and storage capabilities, thus enabling a multi-response CPCM that provides a pathway for the conversion and storage of thermal energy.
With somewhat excellent temperature control performance, we further applied the PU-rG/Cu/C-PW module to the human foot joint (Figure 7(f)). Furthermore, when combined with commonly used rehabilitation equipment, modules with controllable thickness can be fixed on different human body parts, including knees, fingers, wrists, arms, etc. As shown in Figure 7(f)–(h). Several integrated and portable PU-rG/Cu/C-PW temperature control modules for personal thermotherapy have been designed with excellent fitting and wearability. They can also be bent to accommodate human joint curvature without leakage. Compared with the split PCM and heating device structure, the modular design form here greatly simplifies the structural process, reduces the weight, and shortens the manufacturing cycle. Notably, the thermal charging of PU-rG/Cu/C-PW modules is not limited to electrical energy. It can also be heated by low-grade waste heat or solar power. It also has excellent potential for battery thermal management, solar-thermal conversion, and storage.
Conclusion
This study proposed a triple-network collaborative strategy based on the PU skeleton and successfully constructed a CPCM with high flexibility, leak-proof and multi-response characteristics. Through the cyclic impregnation - gradient reduction process, 1D CuNWs, CNTs and 2D rGO were integrated into a 3D porous structure to form a composite skeleton with the characteristics of “hard core-soft shell”. This design effectively fixes PW through capillary action and interface effects, and constructs continuous and efficient phonon and electron transport channels, thereby synergistically enhancing the material’s flexibility, light/electrical-thermal conversion efficiency, thermal conductivity and cycling stability, breaking through the balance limitations among multiple properties of traditional single or binary systems. It is worth noting that for wearable and medical application scenarios, the biocompatibility of materials, user safety and large-scale cost are the keys to achieving practical application. This study significantly reduced the risk of direct skin contact of active components through the stable encapsulation of nanomaterials in the polymer skeleton, providing a structural basis for biosafety. However, systematic in vitro biocompatibility evaluation and long-term wearing safety verification still need to be carried out as key focuses in the future. In terms of cost, the nanomaterials currently adopted in the strategy can further optimize the loading capacity and synthesis path through sustainable processes. Future research will focus on developing low-content, high-efficiency composite systems, and combine circular design and recyclability assessment to promote the development of this material towards low cost, environmental friendliness, and safety and reliability.
In conclusion, this study provides an expandable design paradigm for high-performance polymer-based CPCM, which have clear application potential in fields such as personal thermal management, smart textiles, and flexible electronics. Subsequent work will focus on the performance optimization of materials in low-grade thermal energy collection, integration verification in actual wearing scenarios, and comprehensive assessment of environmental and economic benefits throughout the entire life cycle, thereby promoting the practical application process of the next generation of multi-functional thermal management materials.
Footnotes
Acknowledgements
The authors would like to acknowledge the support of the Inner Mongolia Major Science & Technology Innovation Demonstration Project Foundation (2023JBGS0012) and the Inner Mongolia Applied Technology Research & Development Foundation (2021GG0043) for this work.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Inner Mongolia Major Science & Technology Innovation Demonstration Project Foundation (2023JBGS0012) and the Inner Mongolia Applied Technology Research & Development Foundation (2021GG0043).
Data Availability Statement
Data will be made available on request.
