Abstract
The present study aimed to develop wearable, electrically conductive hygienic electrodes and their use for Transcutaneous Electrical Nerve Stimulation (TENS) application. The development of electrodes was done in two stages. Firstly, the dispersion of conductive activated carbon particles was done into flexible elastomer followed by its silver electroplating. The Orthogonal Array Testing (OATS) technique was used to optimize the silver-plating recipe. The Fourier transform infrared spectroscopy (FTIR) analysis confirmed the impact of activated carbon by increase in intensity of stretching bands. To improve electrode properties in response to various human body movements, resistivity changes due to stretching and repeated extension were tested on conductive elastomers. Increasing the extension degree allowed a very minute change in electrical resistivity. Therefore, the resistivity can be considered almost constant within the 0-60 % stretch range. While, a notable increase in electrical resistivity was observed after 70 % of stretch. However, the resistivity of elastomers remained stable even after repetitive extension (for over 100 cycles). Conversely, no significant change in resistivity was observed over time when subjected to a constant current. Furthermore, to minimize the effect of skin electrode resistivity during electrotherapy, the range of different pressure was applied over the surface of electrodes. Minimum value of resistivity about 1 Ω.mm was obtained at 6 N/cm2 of applied pressure. Additionally, hygienic properties, such as antiviral, antibacterial and antifungal were examined using different pathogens to assess the impact of the deposited silver particles. In the end, the durability of electrodes against washing and rubbing was confirmed. The potential applications of prepared electrodes are in the field of electrostimulation and electrotherapy etc.
Keywords
Introduction
In recent times, electrotherapy has been increasingly utilized in rehabilitation and physiotherapy to reduce pain, promote healing, and to improve the patient mobility. 1 TENS is a non-invasive (non-surgical), simple analgesic (pain relief) technique that is widely used in healthcare centers by nurses, midwives, and physiotherapists. 2 TENS is used mainly to manage non-malignant and acute chronic pain. Different action mechanisms are involved in TENS treatments, but 'gate control theory’ is commonly applied. 3 The gate control system is accountable for events of inhibitor interneurons that are placed within substantia gelatinosa. According to this theory, large alpha and beta nerves (responsible for pain) are stimulated through electrical stimulation. As a result, the intensity of pain signals traveling through smaller nerves is reduced before being transmitted to the brain. Consequently, this process prevents the perception and sensation of pain, providing relief. Moreover, small C and A-delta nerves activate the opioid system and open up the spinal gate system. Thus, patients feel much better with the secretion of the endorphin hormone. TENS therapy is applied normally using conductive hydrogel electrodes to the treatment zone whereby body nerves are stimulated through electrical current. 4 The treatment is often associated with harmful side effects and is effectively reported for patients suffering from peripheral neuropathic pain, 5 diabetic neuropathy, 6 or patients with different origin pain. Still, it is less effective in patients with central neuropathic pain. 7 Electrodes made from carbon rubber, coated metals, gel based, and conductive fabric based have also been utilized. 8 Additionally, some metal-based hard surface electrodes, and needle electrodes are also used for electrotherapy. However, there are concerns about many issues associated with the electrode surface when placed on the skin or inserted into the body. Despite these problems, such electrodes are still continues to be used in various TENS applications. 9 One of the most commonly used electrodes for TENS applications is a self-adhesive hydrogel-based electrode. 10 Many disadvantages have been linked to self-adhesive electrodes due to their viscous nature, i.e., creating redness, discomfort, sweat, rashes, being difficult to wash, having less service life, being unhygienic and having improper compatibility with the body structure. 8 In a few cases, patients are recommended electrotherapy for hours and they are encouraged to use them in the presence of pain. It indicates the condition for ongoing chronic pain, which means, patients have to utilize TENS for the whole day. A study reported by Ashton et al., 11 for long-term TENS users, where users use the TENS therapy over 49 h per week. If TENS is continuously used in in this manner, it is strongly recommended to monitor the skin condition regularly, beneath the electrodes. Thus, they need to pause the therapy and start to operate after constant intervals. Few patients reported post-stimulation analgesia due to the electrotherapy of more than 49 h. 12 Due to long-term TENS application, patients might experience skin maceration, skin irritation, swelling, and reddening around or beneath the electrodes, which can be attributed to dermatitis at the contact side with electrodes resulting from the electrode gel, adhesive tape, or electrode constituents. 13 Therefore, it is important for patients to wash the body parts after applying gel electrodes. Consequently, there has been a growing interest in developing electrode pad materials with enhanced physical and conductive properties. Recently, a novel category of electrodes fabricated by coating of metal particles over the textile fabrics has been foreseen to play a significant role in developing the flexible textile electrodes for electrotherapy. Previously, textile based flexible electrodes were fabricated by incorporating the silver nanoparticles on cotton-lycra structure. 14 Because of the unique physical and chemical properties, silver nanoparticles have been used for several different applications such as in household and healthcare products, in consumer products, as antibacterial agents, coating of various medical devices, in cosmetics, in pharmaceutical and food industry, in diagnostics, optical sensors, drug delivery and as anticancer agents etc. These nanoparticles have also been used and applied in many textiles such as in wound dressings for the purpose of assessing their compatibility with skin and wound healing properties. The silver-coated electrodes were quite flexible and stretchable so fulfil the demand of comfort. The silver-coated electrodes have markedly reduced the incidence of contact dermatitis. They can be conveniently applied to injured or wounded skin, such as cuts, scratches, scrapes and punctures because developed electrodes possess antipathogenic properties due to silver coating. Normally silver metal is inert but when it comes in contact with skin aqueous it ionizes yielding Ag, which are believed to be quite active antimicrobial agent. 15 The death of microbial cell may proceed through different ways. 16 Various studies have been conducted to incorporate the silver particles into polymeric structures to obtain flexible electrically conductive polymers. In a recent study, stretchable silver embedded silicon elastomers were developed for low-voltage nerve stimulation. The electrodes performed well in terms of electrical performance under multiple stretches, and showed minimum electrical resistivity about 0.3 Ohm sq−1. However, the author not study the antipathogenic properties of silver incorporated polymer also, they did not focus on achieving the electrical properties and used the silver-embedded polymers as electrodes for electrotherapy applications. 17
In this study, we tried to develop polymeric and hygienic electrodes for electrotherapy applications. The silicon polymer was embedded with activated carbon particles. Subsequently, electroplating of silver was performed over the carbon-filled elastomers to increase their electrical conductivity and enhance their hygienic properties (antimicrobial, antifungal, and antiviral properties). According to our knowledge, this is the first time we made a conductive elastomeric-based electrode with excellent hygienic properties coupled with compatible electrical resistivity recommended in electrotherapy. Moreover, the prepared conductive electrodes are flexible and stretchable; fulfilling the demand of comfort. The developed electrodes easily conform their shapes in accordance with the movement of the human body during electrotherapy treatment. Additionally, the functional properties which are most required for an electrotherapy electrode were studied deeply, such as durability to friction and washing, surface roughness and conforming, performance of electrodes under moisture conditions and with different sizes. Electrodes have many other benefits such as good washing fastness and prevention of cracks during stretching, as well as good drape, softness and hand feel. Elastomers were prepared by porous carbon particles, which stabilize the sweat and exudates coming from the surface of the skin, so they will maintain hygienic contact if applied for a long time over the skin. Electrodes have significantly reduced the incidence of contact dermatitis and can be easily used over wounded or injured skin (such as cuts, scrapes, scratches, and punctures), as the developed electrodes are antipathogenic and have hygienic properties by using silver coating. The durability of electrodes was tested against electrical, mechanical and washing.
Experimental
Materials
Properties of used silicon rubbers.

Methods
Fabrication of electrically conductive elastomers
It is universal fact that carbon (non-polar) cannot be dissolved into water (polar). Hence it was necessary to dissolve it onto polar solvent such as methanol. However, methanol contains several hydroxyls (-OH) groups. At first, 2 g of CaCO3 stones were added into 100 mL of methanol for 24 h to absorb the -OH content. Subsequently, CaCO3 was removed from the reaction media and 8 g of carbon particles were added into it. The obtained solution was then ultrasonicated for 50 min. After that, 200 mL of liquid silicon elastomer was added slowly. The reaction mixture was further ultrasonicated for 1 h to produce a conductive elastomer. After sonication, the methanol was evaporated by placing the beaker in the oven at 50°C followed by the dropwise addition of 15 drops of curing agent to the solution of silicon elastomer with continuous stirring. The product was then kept at room temperature for 15 h for proper curing. Then, the elastomers were added with carbon particles (8 g) based on the optimized concentration documented in our previous work.19,20 The elastomer samples were made by hand lay-up technique by spreading the conductive elastomer paste over the glass panel and subsequently cured.
Electroplating of silver over carbon black imbedded elastomeric structures
The surface must be electrically conductive prior to the process of electroplating. Electroplating can only be done on material that is already conductive. Hence, the carbon embedded electrically conductive elastomers prepared in the previous sections were used for further silver electroplating. At first, elastomers made of carbon black were immersed in a 70% nitric acid solution for 10 min. Subsequently, the elastomers were added to a solution containing 10 g/L stannous chloride (SnC12) and 4 g/L 40% HCl to sensitize them for 20 min. The sensitized elastomer was transferred to a solution containing 15 g/L AgNO3 and 30 g/L glyoxylic acid to activate it. The activated elastomer was subsequently electroplated with silver coating. Hence the adequate amount of silver was compactly deposited on the substrate during plating. Different concentrations of silver salt (10 g, 20 g, and 30 g) were dissolved in the solution of 1 L of distilled water. The power supply was set at 5 V/0.5 A for varying intervals of time and varying temperatures ranges. The experiment was conducted with a constant current. The electrically conducive carbon elastomer was connected to the cathode, while a commercially available silver electrode served as the anode. The entire system, which involved continuously circulation of the solution, is seen in Figure 1. Schematic illustration of silver electroplating over carbon embedded silicon elastomer.
Parameters and levels used for optimization.

Characterizations
Surface morphology test
The analysis of surface morphology of conductive elastomers (embedded with carbon particles) was conducted by using a scanning electron microscope (SEM). For this, SEM (Tescan VEGA III TS5130) was utilized which was operating at an accelerated voltage of approximately10 kV.
Structural studies
In order to study the appeared peaks at different intensities of functional groups, Fourier transform infrared spectroscopy (FTIR) was analysis was performed. The analysis of infrared ray spectroscopy of silicon elastomer before the embedment of carbon particles and after the embedment of carbon was also determined. It was recorded through spectrometer (Nicolet Nexus 470) which was equipped with an attenuated Total Reflection Pike-Miracle accessory. A diffractometer with a Cu ka1 irradiation (k 5 1.5406 A) and a power supply of 40 kV/30 mA was used to conduct the XRD experiment. The X-ray diffraction patterns were measured in the 10-80 range with a step size of 0.02 step.
Electrical resistivity evaluation
The volume resistivity of elastomers was measured with concentric electrodes using the ASTM D257-07 standard approach. A 100 V DC power supply with a 2.3 kPa pressure was employed at T = 21°C. All samples were maintained in the room for 24 h before testing, with the humidity level kept at 54%. The net current through the sample was calculated by applying an electrical potential across opposite sides of the surface. Equation (1) was used to evaluate the volume resistivity, ρV [mm].
Here, S indicates the surface area electrode [mm2] (πD22/4) and RV [Ω] denotes the volume resistance value. D2 denotes the inner diameter of outer ring electrodes [mm] Figure 2. Electrical resistivity measuring instrumental setup.
Change in resistivity at different pressure
The dry electrodes faced many problems in collecting the biopotential signals from the body. In particular various impedances are involved during skin-electrode contact and electrotherapy, such as noise interference, balance interference between electrode and human skin, also including static impedance (SI), alternating current impedance (ACI), and polarization voltage (PV) etc. Therefore, to record the high quality biopotential signals an adequate amount of proper pressure is necessary on the applied electrodes. In present study, we investigated the influence of 200 g, 400 g and 600 g load (3 cm × 3 cm of sample), which implies to calculated pressure of 2 N/cm2, 4 N/cm2 and 6 N/cm2.
Moisture regains
Moisture regain testing was performed on all elastomer samples according to ASTM D5229. The sample was put into standard environmental chamber to attain maximum moisture. Then after specific time it was taken out and weighted. Subsequently the sample was dried in a drying chamber at 120°C for 30 min. Hence the elastomer mass was calculated at regular intervals of time. The initial weight was determined after the sample was dried, and it was then put into the environmental system (65 ± 5% relative humidity at 20 ± 2°C). The change in mass was calculated using a weighing balance. The specimen’s weight was measured at specific time intervals until equilibrium was reached. The graph shows the changes in the weight gain over time.
Antimicrobial properties
Using conventional test procedures, the antibacterial effect of all samples was evaluated qualitatively and quantitatively.
Zone of inhibition test (qualitative measurements)
Preparation of bacterial strain
Masaryk University Brno, in the Czech Republic, gave the Gram-positive S. aureus (CCM 3953) and Gram-negative E. coli (CCM 3954) bacterial cultures used in this work. The most common method for growing fresh bacterial cultures was to cultivate a single colony in broth culture for a whole night at 37°C. Before performing the antibacterial test, the sample turbidity was adjusted to an optical density of 0.1 at 600 (OD600). Just before the antimicrobial tests, each agar plate was freshly formed. After being put with a sterile cotton ball into the culture medium, the cells became uniformly dispersed across the agar media. The samples were transferred instantly onto prepared plates for extensive analysis.
Determining zone of inhibition
The technique is described in great depth by (Padil, Nguyen, 2015). 21 We tested the antibacterial effect of silver-plated elastomers, which were placed onto agar plates containing contaminated bacteria in 3 × 3 mm squares. Elastomer samples filled in agar plates were heated at 370°C for 24 h. The entire diameter (mm) of the coated sample and the peripheral zone, where no bacterial growth was seen, were used to estimate the area of inhibition (ZOI). The procedure was carried out three times, and the average was determined.
Reduction factor (quantitative test)
The quantitative antibacterial examination of silver-plated elastomers was conducted using the transfer method according to ISO 20743:2013.
22
As stated in the methodology, agar plates were formed and inoculated with 1 mL of inoculum. The control and 3 silver plated specimens were placed on prepared agar medium, and the samples were squished with a weight of 200 g. The control (samples) and silver-plated both samples were 38 cm in size. One sample from each sample was taken off the agar plates and put on a different Petri dish with the transferred side pointing up. The samples would be next incubated for 24 h at 37 degrees Celsius. To obtain a bacterial colony estimate at 0 h, the second specimen of the control and colored samples was simply transferred to 2 reagent bottles that each contained 30 mL of saline solution (0.85% Sodium Chloride). This saline solution is generated in 8 serial dilutions after being shaken in bottles for 15 min, and all of the serial dilutions were plated on agar growth media in accordance with ISO 20743. The same method was used for samples that were incubated for 24 h to determine the number of bacterial colonies present. Equation (2). Formula was used to calculate the colored cotton sample’s antibacterial activity (A). To check the outcomes, each sample was run three times.
F= (log Ct - logC0) and C0& Ct is the bacterial count of control cotton fabric at 0 and 24 h and G= (log Tt – log T0) and T0 & Tt is the bacterial count of control fabric at 0 and 24 h.
Antifungal activity assessments
The antifungal characteristics of both the raw sample (uncoated) and coated samples was measured according to the standard test procedure, AATCC 100-2004. A specific species of fungus, named as A. niger was selected to perform the test. The antifungal performance was calculated in reduction percent by using the following equation (3).
Antiviral activity
Different conditions for testing.

Method and preparation of growth medium
TCID50 methodology was adopted to assess the dilution of virus suspension, which in turn generates a CPE (cytopathic effect) in almost 50 % of cell unit. For this purpose, 96-well microplates were taken with some other sterilized equipment. The nutrient media was prepared by following the recipe given in ISO 18184 (Grade 3 water, Kanamycin sulfate, Eagle’s essentials, sodium bicarbonate, inactivated fetal bovine serum).
Firstly, the cryopreserved host cells were defrosted. After that, a new flask for nutrient agar was made 20 mL of growth medium). The host cells were added into the new flask. This new flask was placed in CO2 incubator for 24 h and at temperature of 37°C in order to grow the cells. Consequently, the grown cells were further cultured by following the same procedure as described by ISO 18184.
About 20 ml of agar media was added to a beaker along with 1 mlof subculture cells. Secondly, 0.1 ml of solution was taken in each well i-e total of 96 wells of microplates. After that, these plates were placed in incubator for 5 days, at about 37°C. After the specified time period, cell multiplication was observed under microscope. Then, the surface of cells was washed with maintenance media which is mentioned in Table 1. This solution was named as Flask A.
Preparation of viruses
Coronavirus was also defrosted by applying the same process. This defrosted coronavirus was added to a new test tube where it is diluted with maintenance media. Therefore, the concentration of virus was also adjusted to TCID50/mL. The 1 mL of coronavirus was inoculated and was spread uniformly on the cell surface in flask A. Then, this flask was again placed in incubator at 34°C for approximately 1 h in order to completely absorb the virus into cell. After an hour, 20 mL of maintenance medium and 30 µL of trypsin was added to it and the flask was again shifted to incubator at 37°C for almost 1-3 days. After 3 days, the culture of grown viruses was added to a centrifugal tube at 9°C for 15 min. The suspension obtained was separated from the tube (virus suspension), and was left in test tubes at −80°C. It was observed and confirmed that the concentration of virus was greater than TCID50/mL.
Principle
A sample of size 20 mm × 20 mm was cut (but the total mass should be 0.40 g and can be settled with extra/less material if required). There should be total of 6 treated and 9 control pieces. Out of these, 3 pieches of each substrate were taken to observe the effect of fabric on cells without virus (cytotoxicity) and remaining 3 pieces were taken to recover initial titer of virus. The remaining pieces were then inoculated in 200 μL of virus at ∼107. TCID50 concentration (giving a final concentration of 105) and was left for contact time. By this contact time, the piece of electrode was recovered in 20 mL of culture media estimated on the cell line. Further, TCID50 was also recorded soon after following the incubation time. The antiviral activity was also assessed by comparing the antiviral test material and the recovered sample from control. Moreover, the quantitative standard test methodology was utilized to calculate the antiviral effectiveness of prepared silver nanocomposite coated electrodes.
Durability of electrodes
Durability under multiple stretching cycles
Conductive elastomers typically show a change in resistivity when stretched and recovered for a variety of cycles. During any service application, conducting elastomer could be subjected to mechanical reactions such as recovery, folding, stretching, jerking, twists, and release. We subjected them to about 75 cycles to test the durability of our developed samples. The electrical resistivity was determined for each level of deformation (stretching and releasing) throughout these cycles. The samples were released after being extended to 60 % of their initial length (one cycle). The experiment was carried out three times on each sample to confirm that the outcomes were consistent.
Durability against friction
Martindale (device M235) rubbing method was adopted to evaluate the resistance against friction of prepared electrodes. The sample were cut into circular form, each of them having diameter about 140 mm, and placed in a sample holder. Similarly, three more samples were cut from same sample and fixed to the machine table. The test was conducted under a load of 600 g and machine was adjusted at 300 rubbing cycles. Subsequently, the electrical properties were evaluated.
Durability against multiple washing cycles
The standard test method ISO 105-C01 was used to examined the washing durability of prepared electrodes sample. At first, 5 g of standard detergent was dissolved in 1 L of deionized water. Then sample was dipped into the solution with a liquor ratio about 50:1, and applied sever stirring. Subsequently, samples were rinsed at 40°C with a stirring speed of 750 rpm for 30 min. Each sample was washed separately without acidification. The surfaces of all samples were then physically assessed, and their electrical resistivity was measured.
Results and discussion
Electrical resistivity results of elastomers embedded with particles
All elastomer samples (simple silicon, silicon filled with carbon, and silver-plated samples) were tested for their volume resistivity, along with their evaluation of resistivity at different stretching percentages. The volume resistivity of each sample was calculated using the average outcomes of three experiments. A percolated network forms as a result of the continual connectivity that is built inside the embedded carbon particles. The resistivity is reduced with the increase in the concentration of carbon particles. The minimum resistivity was observed at 8 % of carbon particles (1183 Ω.mm).
Measure the optimum conditions for the silver electroplating plating
Experimental arrangement for optimizing silver plating against volume resistivity.
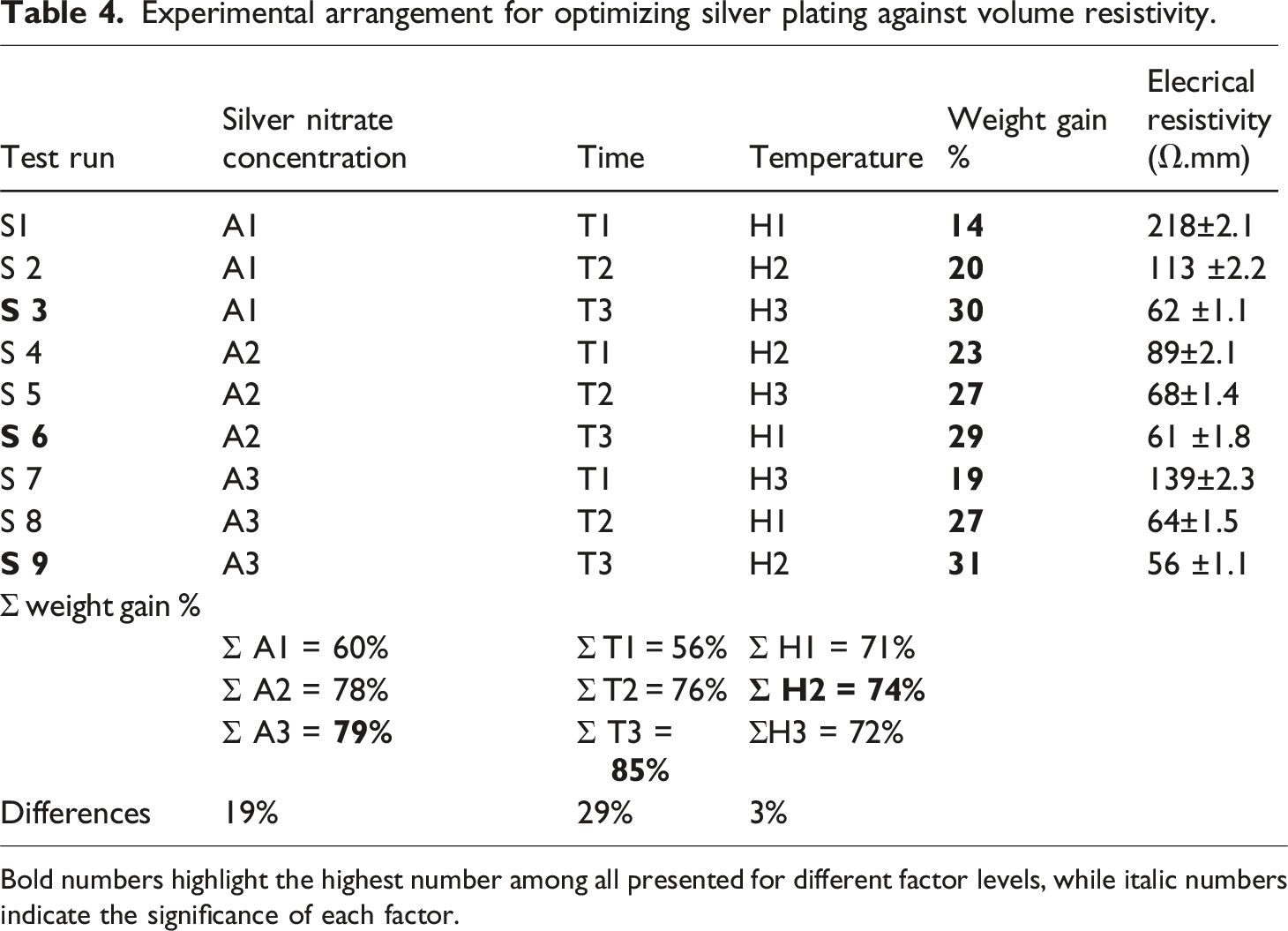
Bold numbers highlight the highest number among all presented for different factor levels, while italic numbers indicate the significance of each factor.
The effect of time on silver plating
Time is the most significant parameter during the silver plating. The maximum value of time produces the finest electroplating due to the more weight gain after 30 min, or value T3. The reason is that either hydroxyl ions (OH−) or protons (H+) are produced during the deposition process, which affects the pH of the solution. The redox process generates H+ or OH− ions, additional time will function as a buffer to manage pH; in fact, the longer time will balance pH and keep the coating uniform. As hydrogen liberation in anodic reactions lowers pH, a longer electroplating period will allow for the production of positively charged ions (H+). This shows that after 30 min of electroplating, the maximum coating was attained and resistance was at its lowest level. It indicates that the silver has finished depositing after roughly 30 min. Therefore, 30 min were chosen for the fabrication of the optimum sample.
Effect of concentration of silver nitrate
The effect of AgNO3 concentration is less important when compared to the electroplating time. However, it is more important than the plating temperature. The concentration of silver nitrate effect is about 79 %. This shows that at 30 g/L of AgNO3, the resistivity reached its lowest value and the maximum coating was attained. It indicates that the silver deposition is finished at around 30 g/L. Therefore, 30 min were specified for the fabrication of optimum sample.
Effect of temperature
The temperature has the least noticeable impact on the silver-plating process out of all the variables. Furthermore, 30°C, or H2, was thought to be the appropriate temperature. A higher weight deposition of Ag for roughly 74 % was seen at 30°C. This shows that the highest amount of silver was deposited within 30°C of temperature. The plating is achieved at this point. In fact, due to saturation at 45°C, the silver that has been deposited on the elastomer would dissolve, diffuse back, and then be discharged to solution from the elastomer, resulting in weight loss.
Furthermore, Figure 3 is showing the effect of each parameter on electrochemical deposition of metal over the elastomer surface. Effect of each plating parameter.
Electrical resistivity
.Parameters decided for optimum recipe.

Figure 4 shows the resistivity value graphs for each sample. The graph was initially examined for the Ag plating on elastomers with incorporated carbon particles. The trend lines of the resistivity values for each sample demonstrate that in samples 1, 2, and 3, the concentration of AgNO3 was kept constant at A1 while the other two parameters varied (time and temperature). The lowest resistance was attained for sample 3. Similarly, with fix A2, sample number 6 has the minimum resistivity, and for fix A3, sample 9 has the minimum values. The resistivity values for samples S3, S6, and S9 were very low which indicates that these samples have excellent electrical conductivity. The sample S 10 was developed using an optimized recipe because the main purpose for the application of the OATS technique was the optimization of the recipe and achievement of the lowest resistivity. The resistivity values for S 10 are mentioned in the graph. The values for resistivity are quite low they showed the best fit of our OATS technique. Moreover, the maximum weight gain of silver corresponding to sample 10 was about 34 %. Electrical resistivity trend for copper plating over silver coated fabric.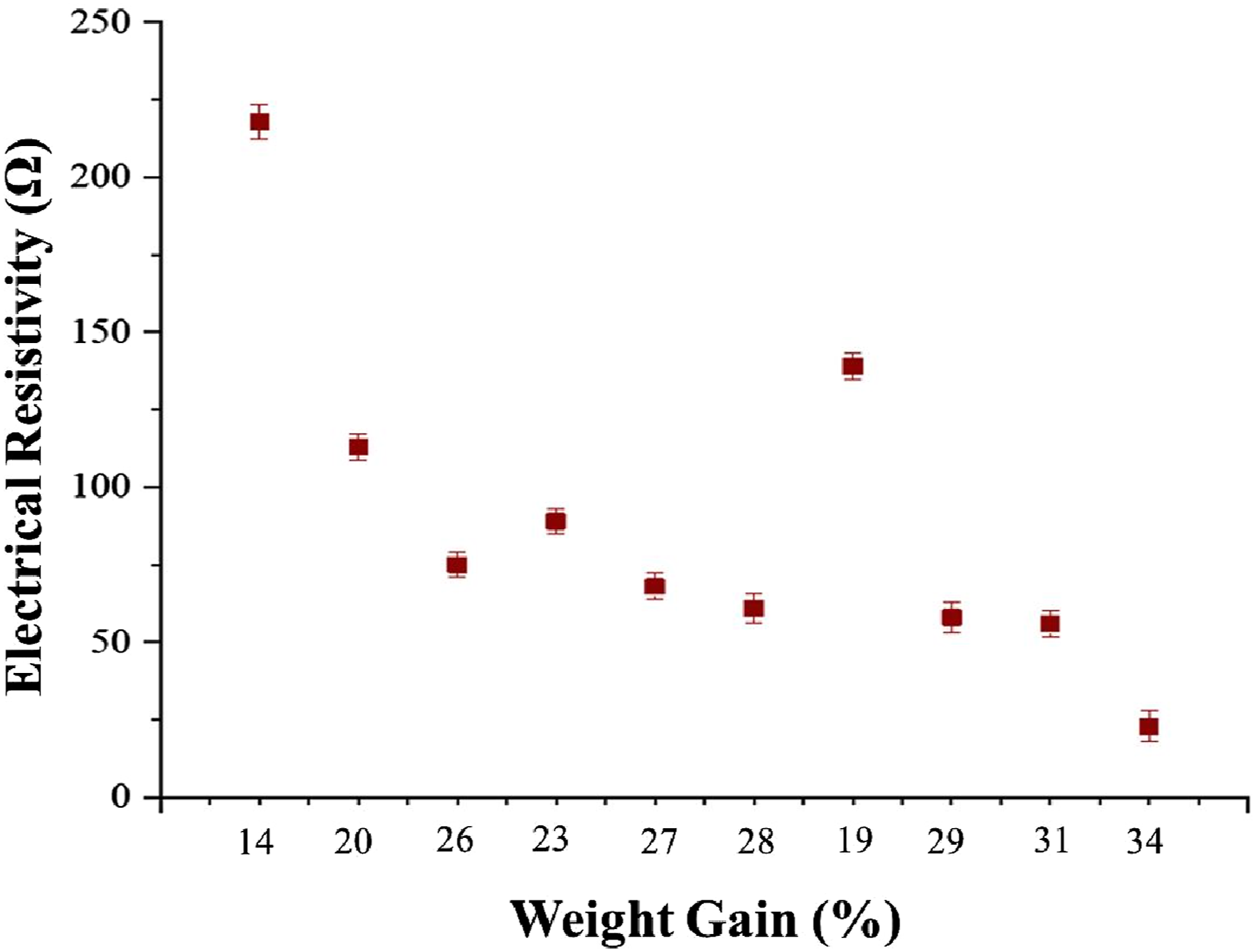
Surface morphology
As, the electrotherapy involves applying electrical currents to the body for therapeutic purposes, and the effectiveness of this technique depends significantly on the properties of the conductive electrodes. The prepared conductive electrode should have homogeneously dispersed electrically conductive fillers (nano or microparticles). Figure 5 displays the morphological characteristics of carbon particles, carbon-embedded elastomers, and silver-plated elastomers (a, b, and c). High-resolution SEM images are shown in Figure 5(a). The microstructures of the carbon particles after 60 min of dry milling showed a combination of nanoparticles and nano-segments. The size of the particles was in the micro range with a high aspect ratio after half hour of dry milling. The increase in temperature of ball-milling was avoided to attain further homogeneous refinement. The SEM results of silicon elastomer embedded with carbon are shown in Figure 5(b). The particles are properly dispersed and connected so that the incorporated active carbon particles can conduct an electrical current to make electrical thresholds. Moreover, an additional layer of silver was coated on the surface of the elastomer after embedding the carbon particles Figure 5(c). The sample was developed through the deposition of carbon particles and electroplating followed by the electroplating of a thick layer of silver. Surface morphology of (a) carbon particles, (b) carbon embedded elastomers and (c) silver plated elastomers.
The additional coating of silver serves dual functions. Firstly, its homogeneity and dense coverage enable the proper and even flow of electrical current. Secondly, the silver layer is effective against pathogens. The image also shows that over the particle-covered area, electroplating is conducted in an orbicular manner. It possesses crocodile skin-like features. For an effective electrotherapy function the electrode surface should have homogenous coating of conductive material. Hence, the homogenous coating of silver was observed over the carbon embedded polymeric electrodes, with results similar to those reported previously upon coating of Neural Electrodes with platinum nanoparticles reduces and stabilizes resistivity In Vitro and In Vivo in a Rat Model. 25
Surface roughness
Moreover, the surface roughness was also measured, as smooth surface on electrotherapy electrodes is essential for enhancing electrical conductivity, improving patient comfort, optimizing therapeutic effectiveness, ensuring durability, and maintaining safety. In the prepared conductive electrode, the surface of elastomeric silver coated electrode was observed to be mostly smooth, only showing very less beads of silver bumps on the coating at some points. The surface roughness was measured with confocal microscopy. From Figure 6, it is clear that the majority of the surface area has a smooth coating, with only a few micro beads observed at certain points under detailed analysis. This may due to the accumulative coating points. However, most of the surface was even and smooth. The average parameters of beads and plane area are also demonstrated in given image.26,27 Confocal microscope image of electrode surface.
FTIR analysis
Characteristic FTIR absorption bands for PDMS before and after carbon fillers.


FTIR spectra of (a) silicon elastomer with activated carbon filler and (b) plain silicon elastomer without activated carbon particles.
XRD analysis
The X-ray diffraction (XRD) analysis was performed for the S 10. XRD analysis was used to identify the crystallographic structure of the plated silver and carbon. The XRD spectra were recorded in the range of 20 to 80° with a step size of 0.02 and results are shown in Figure 8. The precise indexing of each diffraction peak reveals the phase purity of the as-synthesized Ag-NPs. The signals occurred at 2θ values of 38.1, 64.5, and 77.5° explained the structure of silver. Additionally, three new signals at 2θ values of 25.6, 43.8, and 52.7° could be ascribed to the incorporated carbon particles. These signals were referred to the diffraction lines of the (001), (100), and (004) planes of carbon, respectively.
28
XRD analysis of silver-plated carbon embedded elastomers.
Electrical resistivity at different extension percentage
Effect of stretch percentage on electrical resistivity
The electrically conductive elastomeric substrates embedded with 8 g of carbon particles and subsequently silver plated for 10, 20, and 30 min have shown the lowest value of electrical volume resistivity. The volume resistivity, at normal condition (without applying any stretch) were measured about 23 Ω.mm. Then the prepared elastomeric substrates were applied different levels of stretch. The electrical volume resistivity (Ω.mm) against different stretch percentage is shown in Figure 9. At 0 % of stretch (at normal state), the resistivity for the selected sample was recorded as 23 Ω.mm, which steadily increased and reaching 32 Ω.mm at 60 % stretch. In the stretch range of 0 to 60 % there is insignificant change in volume resistivity was observed and resistivity nearly remained constant. However, a drastic change in the resistivity was observed after 60 % of stretch, at 70 % and 80 % a significant increase in resistivity values was recorded. Hence till 100 % of steadily increased of stretch and maximum resistivity was approximately 45 K Ω.mm. This is attributed to the fact that, at higher stretch % there is continous disconnection of conductive network of particles and also of silver plating. Additionally, the silver coating turned into cracks at some points, which results in further lowering of electrical conductivity. Electrical volume resistivity of silver electroplated elastomers, as a function of different stretching percentage.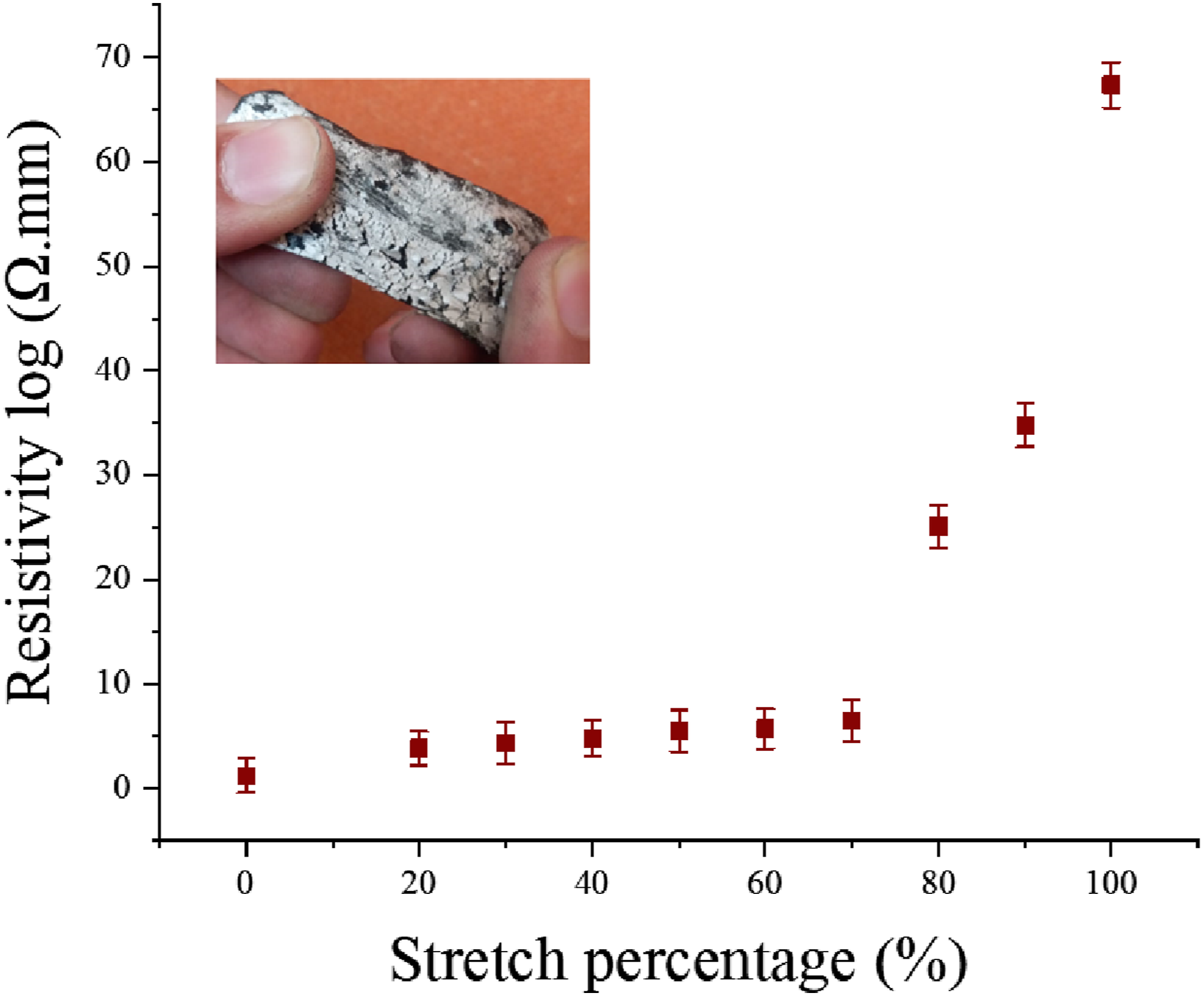
Durability of electrical resistivity across multiple stretching cycles
The durability of the silver-plated sample S 10 was also checked under multiple stretch and release cycles. The resistivity of sample was 23 Ω.mm (at relaxed position i-e 0 % stretch), then the sample was stretched up to 50 % and volume resistivity was noted (32 Ω.mm). The stretch was released and sample reverted back to original position (again % stretch). Three readings were noted of resistivity and mean was calculated during one complete cycle (position 1 at 0 %, position 2 at 50 % and position 3 again at 0 % stretch). Figure 10(c) is showing a more comprehensive analysis of complete one cycle. A total of 150 cycles were subjected and resistivity was measured at each stage. It was worth noting that the sample S10 showed good recovery in resistivity and there is insignificant change in values 150 cycles, as illustrated in Figure 10(a). The developed elastomers are highly stable in electrical conductivity and justify the re-establishment of conductive network (during stretch and recovery). Consequently, it can be claimed that the fabricated electrically conductive rubber substrates have excellent stability in terms of recovery in resistivity values. This behavior attributes the reconnecting and restoration of conductive network of coating, once the fibers return to their original (normal) form. Conductive elastomers remain stable in terms of the resistivity values (all cycles have shown constancy).
17
The stretching and releasing phases are also described by extending the first five cycles (see Figure 10(b)). Sustain in electrical resistivity with multiple stretch and release cycles.
Moreover, the insignificant change in the resistivity was observed between the resistivity values (23 Ω.mm) at 0 % stretch during 1st cycle and resistivity values (24.3 Ω.mm) at 0 % stretch after 150 cycles. Same in between resistivity values (32 Ω.mm) at 50 % stretch during 1st cycle and resistivity values (38.8 Ω.mm) at 50 % stretch after 150 cycles as shown in Figure 11. The graph implies that the volume resistivity tested Sample increased very less even upto 150 cycles of stretch and release. The small change in resistivity values of electrodes is due to the multiple stretching actions, which in turn deteriorates conductive properties and result of breaking single conductivity paths during the action of mechanical forces. Difference between the resistivity values at 0 % and 50% stretch during 1st and 150 cycles.
Impact of different dimensions of electrodes on volume resistivity
The commercial electrodes for TENS applications are available in various sizes, though they are most commonly found in standard sizes ranging from 2×2 cm2 to 3×3 cm2. The different size of electrodes may affect the electrical properties of electrodes during their service. To examine the impact of different sizes over the resistivity and performance, electrodes of various dimensions were also developed. The sample S10 was composed up of optimized recipe and its original size was around 3×3 cm2. Hence, we developed two more samples with sizes 2×2 cm2 and 2.5×2.5 cm2. The sample S10 was termed as (3×3 cm2) S10 A, (2.5×2.5 cm2) S10 B and (2×2 cm2) S 10 C. Their response against electrical resistivity was measured both in normal (unstretched) and stretched conditions as shown in Figure 12. Electrical resistivity both in normal (unstretched) and stretched conditions.
The effect of applied pressure on electrode resistivity during electrotherapy
The high potential epidermal biopotential signals are recorded by wet and dry electrodes, during application on all types of skin, and even during moving body parts. Since, there is not any adhesive material to attach these electrodes with skin, because these adhesives cause uneven and enhanced skin-electrode resistivity. Therefore, to record the high quality biopotential signals an adequate amount of proper pressure is necessary on the applied electrodes. In present study, we investigated the influence of 200 g, 400 g and 600 g load (10 cm × 10 cm of sample), which implies to calculated pressure of 2 N/cm2, 4 N/cm2 and 6 N/cm2. The trend line values of electrical resistivities with respective applied pressure are shown in Figure 13. It was noticed that the pressure has a significant effect on the electrical resistivity of the prepared electrodes. A substantial drop in resistivity was observed by increasing the pressure. The initial resistivities if samples S3, S 6, S 9 and S 10 were 75 Ω.mm, 61 Ω.mm, 56 Ω.mm, 23 Ω.mm, and at 6 N/cm2 the values decreased and noticed about 23 Ω.mm, 17 Ω.mm, 14 Ω.mm and 1 Ω.mm. The same procedure had applied by a previous research work, to investigate the effect of pressure in dry textile electrodes to obtained the ECG signals. Where pressure of 30 mmHg (3.99 KPa) was selected for compression applications of electrodes.29,30 Another, research work chose the pressure of 392 N/m2 for effective electrostimulation of TENS electrodes. The also observed the increase in pressure cause lower in resistivity. In particular, the resistivity is dropped due to the increase in contact area of conductive network and skin. As, the polymers and textiles are soft materials and on squeezing the interface of contact improves, which leads to obtained better signals.
31
Trend line values of electrical resistivity with respective applied pressure.
Moisture regain percentage
The graph of moisture regain versus time for the elastomer sample S10 is shown in Figure 14. The regain in moisture was rapid during the first 10-h period, moisture regain was noted significant for this period. The sample is fully dried in initial stage and tends to attain maximum moisture in beginning. After 10 h (when saturation immense) the rate of moisture regains became slow. At this stage the trend line of graph is showing the almost minor increase in moisture uptake. As the experiment was further proceeded, after 70 h to 130 h. There were almost constant values in moisture uptake (sample are fully saturated). Here sample is almost in equilibrium about moisture uptake. However, the test was further continued to 130 h (nearly 5.5 days) and it was ensured that specimens regained the highest level of moisture. Therefore, it can be concluded that carbon particles incorporated and silver-plated elastomeric electrodes will retain less moisture and remain functional even in wet or humid environments. Regain in moisture in terms of percentage over time.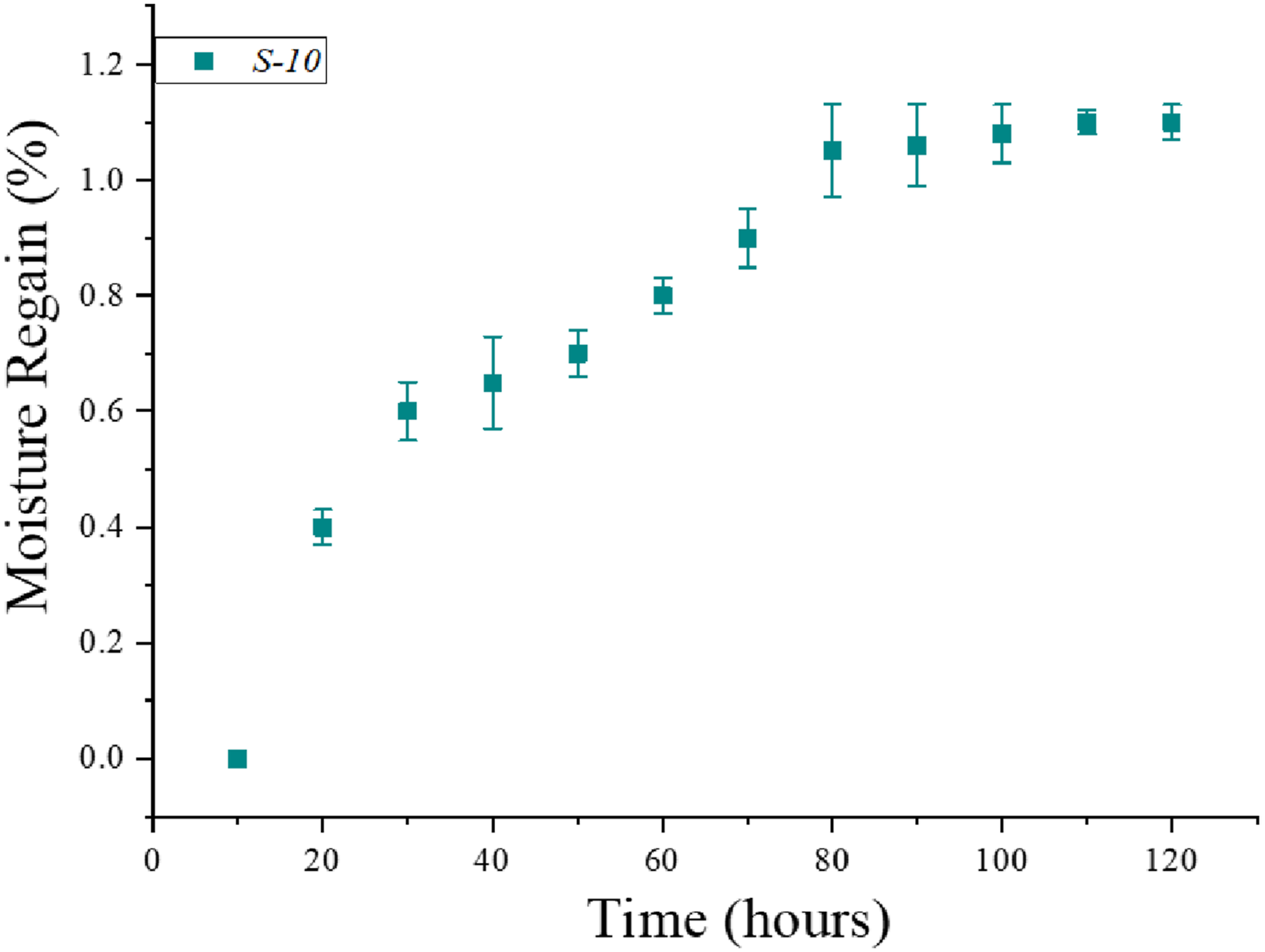
Resistivity changes with time
Change in resistivity at constant current was also measured as shown in Figure 15. For medical purpose normally, direct current is used. The direct current of medical use is normally electrified to nerves or muscle in the range of 4.5 to 45 voltage and the current from 0.1 to 10 mA. Therefore, the further experiment was conducted at 20 mA to analyze the variation in current duration. The conductivity significantly remains unchanged with time over 800 s at the constant intensity of current. Therefore, during the electrical nerve stimulation therapy the impact of prolonged current apply (without change in electrical resistivity) can be negligible. Change in resistivity with time at constant current.
Antipathogenic properties of developed electrodes
Antibacterial properties
The Gram-negative bacteria Escherichia coli (E. coli) and Gram-positive bacterial strains Staphylococcus aureus (S. aureus) were used for testing the antibacterial activity of silver-plated elastomers. Figure 16 display inhibition zone around the substrates S8, S9 and S10 after 1 day of incubation in dark at temperature 37°C. It is clearly proving that the zone of inhibitions was against both type of bacteria S. aureus and E. coli due to silver coating. Additionally, S. aureus shows maximum sensitivity with respect to E. coli. The zone of inhibitions enhanced from 6 to 11 mm in case of S. aureus and 5 to 9 mm for E. coli. The antibacterial property of coated elastomers can be ascribed due to the amalgamation of chemical and physical interactions of bacteria with silver. The silver particles infused through the is the cell by a process of endocytotic mechanisms. Hence, the cellular uptake of ions is enhanced due to the increased amount of released ionic species in cells by the metal dissolution process
32
which leads to gain in higher intracellular concentration and caused immense oxidative stress. Antimicrobial effectiveness of silver coated elastomers.
The antimicrobial effect of metal coating having various unique properties, including microparticles and biomolecules such polynucleic acids and proteins. 32 The antibacterial impact of silver coating may result from chemical interactions, physical interactions or by a combination of both. So, in future the prepared electrodes are highly hygienic and are most effective for hospital environment.
Antifungal activity of treated samples
The antifungal activity was calculated through the standard test method of qualitative measurement. For this activity, the fungi Aspergillus niger (A. niger) was used. The activity was examined for uncoated elastomer and for silver plated (S 10) elastomer sample. Figure 17 display the percentage reduction of fungi A. niger with raw sample and samples loaded with silver. It was observed that the simple raw sample without any antifungal agent can provide a suitable environment for microorganism’s growth. The silver-plated sample exhibit maximum antifungal activity because the metal particles coated elastomer having suitable antibacterial properties. Hence, the percentage reduction of fungus A. niger by silver nanoparticles is higher as compared to extracted phytochemicals.
32
Antifungal activity of sample against A. niger.
Antiviral effectiveness
The Figure 18(a) describes the infectivity titer change of corona virus (0 h and 60 min) at 25°C for uncoated elastomer and silver coated elastomer S 10. It was examined that there is a major decrease in infectivity titer for fabrics coated sample after 60 min instead of 0 h whereas no reduction in virus activity titer was calculated in vase of uncoated elastomer sample. While, Figure 18(b) exhibit virus percent reduction for uncoated and coated S 10 sample. The elastomer treated with silver reduces 85 % in virus titer separately uncoated elastomer remained ineffective against virus. The antiviral action exhibited through elastomer treated with silver could be due to the binding of metallic surface with glycoproteins at the viral surface working as an inhibitory action for viruses.
33
The studies from past explained that synthesized metallic particles (Ag-NPs) showed antiviral effectiveness against coronavirus.
34
The investigation showed that antiviral activity of silver nanoparticles against various other viruses was because of the direct attachment of silver nanoparticles to viral envelope glycoproteins, which ultimately inhibits the viral penetration into host. However, the mechanism of action was not explained clearly in depth.
35
The particle size effect on the antiviral activity was slightly observed, which indicates the spatial restriction of binding between Ag-NPs and virions.
36
Reduction in concentration of infectious virus particles (viral infectivity titer) (a) and percentage reduction (b) Determined from viral infectivity for control (untreated) and treated fabrics (coated) after 0 and 60 min of contact time.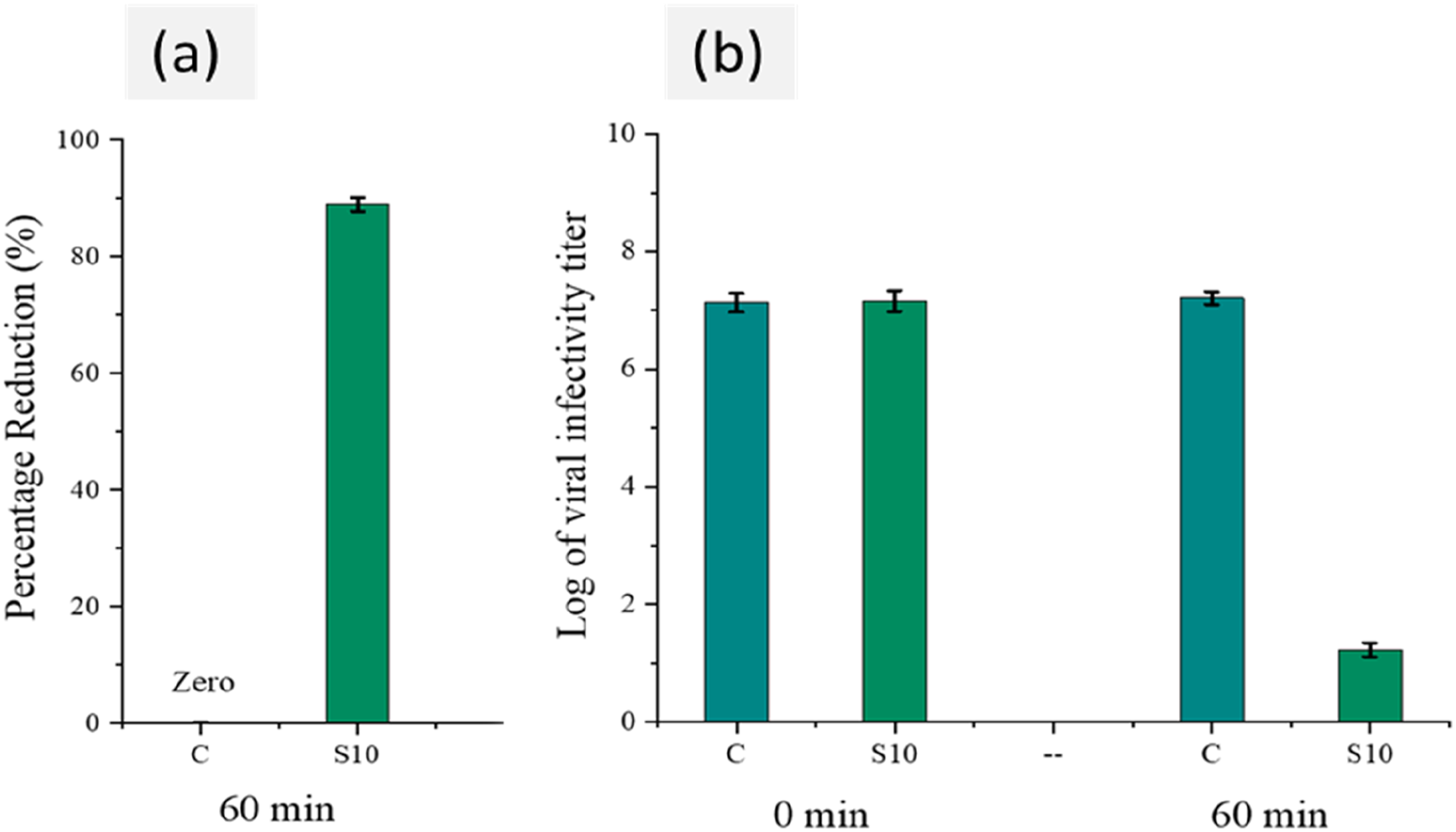
Durability against washing and rubbing
TENS electrodes that can be washed multiple times offer a reliable and affordable way to improve the effectiveness and safety of TENS therapy. Their durability ensures they last longer, perform consistently, stay hygienic, are convenient for users, and are better for the environment.37,38 Figure 19 implies that the volume resistivity of conductive electrodes was insignificantly affected even after 20 washing and 20 rubbing cycles. The retention of the silver coating and consistent resistivity values ensure the durability and performance of the electrodes. The electrodes showed good washing durability as compared to rubbing durability. The reduced rubbing durability of silver-coated electrodes arises from factors such as weak adhesion and poor stability against sever mechanical action. However, the overall electrical resistivity values remain quite suitable even after severe washing and rubbing. (a) Washing and (b) rubbing durability of conductive electrodes.
Conclusion
Electrically conductive, highly stretchable electrodes for TENS machines with acceptable comfort properties were developed. The electrodes were made by incorporating electrically conductive (activated carbon) fillers to the silicon rubber and followed by silver metal electroplating. The OATS technique, revealing that the time factor (duration of electroplating) had a significant impact on the results. The carbon particles embedded elastomers provided the minimum volume resistivity of 1183 Ω.mm at 8 % of carbon particles. Whereas, there was a significant decrease in the volume resistivity was observed by applying further silver electroplating over carbon-embedded elastomers. The minimum resistivity of about 23 Ω.mm was recorded for sample S 10 (even during connected to a TENS machine). Moreover, the silver-plated elastomer sample showed very less moisture regain about 1.1 %, (even the sample was retained in water for many days). In further section, hygienic properties (antibacterial, antiviral and antifungal) were measured against silver plated elastomers. The sample S10 showed a significant effect against each pathogen in all antipathogenic testing. In the end, the durability of electrodes against washing and rubbing was confirmed. The performed work has numerous advantages because of silver electroplating over the carbon-embedded electrodes. The potential applications of prepared electrodes are in the field of electrostimulation and electrotherapy etc.
Footnotes
Author Contributions
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported by the project ‘Advanced Structures for thermal insulation in extreme conditions (Reg. No. 21–32510M) granted by the Czech Science Foundation (GACR). The work was also supported by the Ministry of Education, Youth and Sports. INTER-ACTION project number LUAUS23054 “Textile-derived micro-plastics in aquatic ecosystems: identification, characterizations, and effect assessment”.
Data availability statement
The authors confirm that the data supporting the findings of this study are available within the article. The data will be available on request.
