Abstract
In this work, silver (Ag) self-metallization on a polyimide (PI) film was prepared through autocatalytic plating. PI films were prepared through the solution casting method, followed by etching with potassium hydroxide (KOH) solution, sensitization with tin chloride (SnCl2), and the use of palladium chloride (PdCl2) to activate the surface of PI. Energy-dispersive X-ray analysis (EDX) showed the highest peak in the (Ag) region and confirmed the presence of AgNPs. The diffraction peaks at 2θ = 38.2°, 44.5°, 64.6°, and 78.2° represented the 111, 200, 220, and 311 planes of Ag, respectively. The FT–IR analysis for Ag-metalized PI showed that the =C-O-C= stretching absorption bands at 1735 cm−1 had no changes in position, only a significant difference in peak size at the deposition time increase. The formation of new bands (N–H stretching absorption band and N–C stretching band) assigned at 2325 and 955 cm−1 indicated strong coordination between N atoms and silver nanoparticles. The C–C stretching and = C–H plane vibration band at 1488 and 1117 cm−1 are shifted to 1413 and 1112 cm−1, indicating the silver nanoparticles' interaction with the polymer backbone. The thermal stability of PI- and Ag-metalized PI films at various deposition times (5, 10, and 15 min) was examined using thermogravimetric analysis (TGA). For PI, T0, T5, T10, and Tmax were observed to be 388°C, 402°C, 414°C, and 515°C, respectively. When the deposition time increased, the thermal stability increased. As a function of the deposition, the thickness and surface morphology of the copper layer on the PI films were characterized using scanning electron microscopy (SEM).
Introduction
Polymer metallization has attracted attention in recent years due to its wide use in microelectronics, electronics technology, and the automotive industries.1–3 Polyimide (PI) has excellent thermal and chemical stability, and it has good adhesion and electrical properties long-term operation ability.2,4 On the other hand, silver (Ag) is the most conductive noble metal. It has unique electronic and optical features, and these features make it ideal for use in coating polymer in electronic material. 5
Metallization is the best method for Ag plating on polymer films when aiming to improve properties that are equivalent to those of Ag powder. 6 However, the adhesion of passive metals, such as Ag, Au, and Cu, to polymers is notoriously poor.6,7
Metallization, or the autocatalytic metal plating of polymers, can be achieved using the following methods: solid and molten metal-based deposition, gaseous phase-based metal deposition, and solution-based metal deposition. 7 Because nonconductive materials—primarily synthetic polymers—cannot be metalized through simple electrolytic deposition, the surface of these materials must be activated with a metal-reducing catalyst through a process called autocatalytic plating.8,9
Nickel, gold, Ag, platinum, palladium, and copper are frequently deposited through this process to produce industrial products, such as printed circuit boards, hard memory disks, automotive parts, and decorative elements.10,11 Additionally, many researchers have prepared metallized polymer films by heating positive metal ions and polymer solutions to stimulate the reduction of metal ions. 12 However, the polymers were already deformed or cracked before the reduction of metal ions at a high temperature, and adhesion problems still existed.13,14 In view of the shortcomings of the above methods, developing a more efficient method to prepare metallizing polymer films is of great significance.15,16
In recent years, researchers have been focused on polymer surface modification and metalized it by using different metals. Ji et al. prepared the metal-plated polyimide and treated (etching) it by base solution and then activated by silver. The modified polyimide film was immersed into the electroless copper plating solution, which has different molar ratios of nickel in the solution. The thickness and surface morphology of the copper layer on the polyimide films were characterized with scanning electron microscopy (SEM). 17 Wu et al. metalized polyimide (PI) by using nickel (Ni) and formed it using a solution-based process including imide-ring opening reactions, the implanting of Ni ions, the reduction of catalytic Ni nanoparticles, and the electroless deposition of a Ni film. They report that the KOH concentration significantly affects the implanting, reduction, and deposition behavior of Ni. A uniform Ni layer can be grown on a PI film with full coverage through electroless deposition with a KOH concentration of 0.5 M and higher. 18
In this research, Ag-metalized PI films were prepared using the autocatalytic deposition method. The quality of Ag coatings improved after etching by KOH solution, SnCl2 sensitization, and PdCl2 activation of the PI surface. The metalized films were characterized as a function of different deposition times.
Experimental
Materials
The following PI materials were purchased from Sigma–Aldrich: 4,4'-(hexafluoroisopropylidene)diphthalic anhydride (6FDA), 4,4’-oxydianiline (ODA), and dimethylacetamide (DMAc). The following chemical plating components were all used as received and purchased from Sinopharm Chemical Reagent Co (Shanghai/China): tin chloride (SnCl2), palladium chloride (PdCl2), potassium hydroxide (KOH), acetone (CH3COCH3), sodium hydroxide (NaOH), silver nitrate (AgNO3), formaldehyde (HCHO) and disodium ethylenediamine tetraacetate (EDTA-2Na). Deionized water was used to rinse and prepare all the solutions.
Preparation of PI
PI was prepared by placing 5 g of 6FDA in a beaker containing 20 mL of DMAc solution and stirring it for 40 min. Then, 5 g of ODA was added to the solution in a partition and stirred for 4 h at room temperature to prepare a polyamic acid (PAA) solution. The PAA solution was heat-treated at 250°C for 1 h to produce PI. The preparation of PI is illustrated in Figure 1. Schematic illustration of 6FDA–ODA PI synthesis.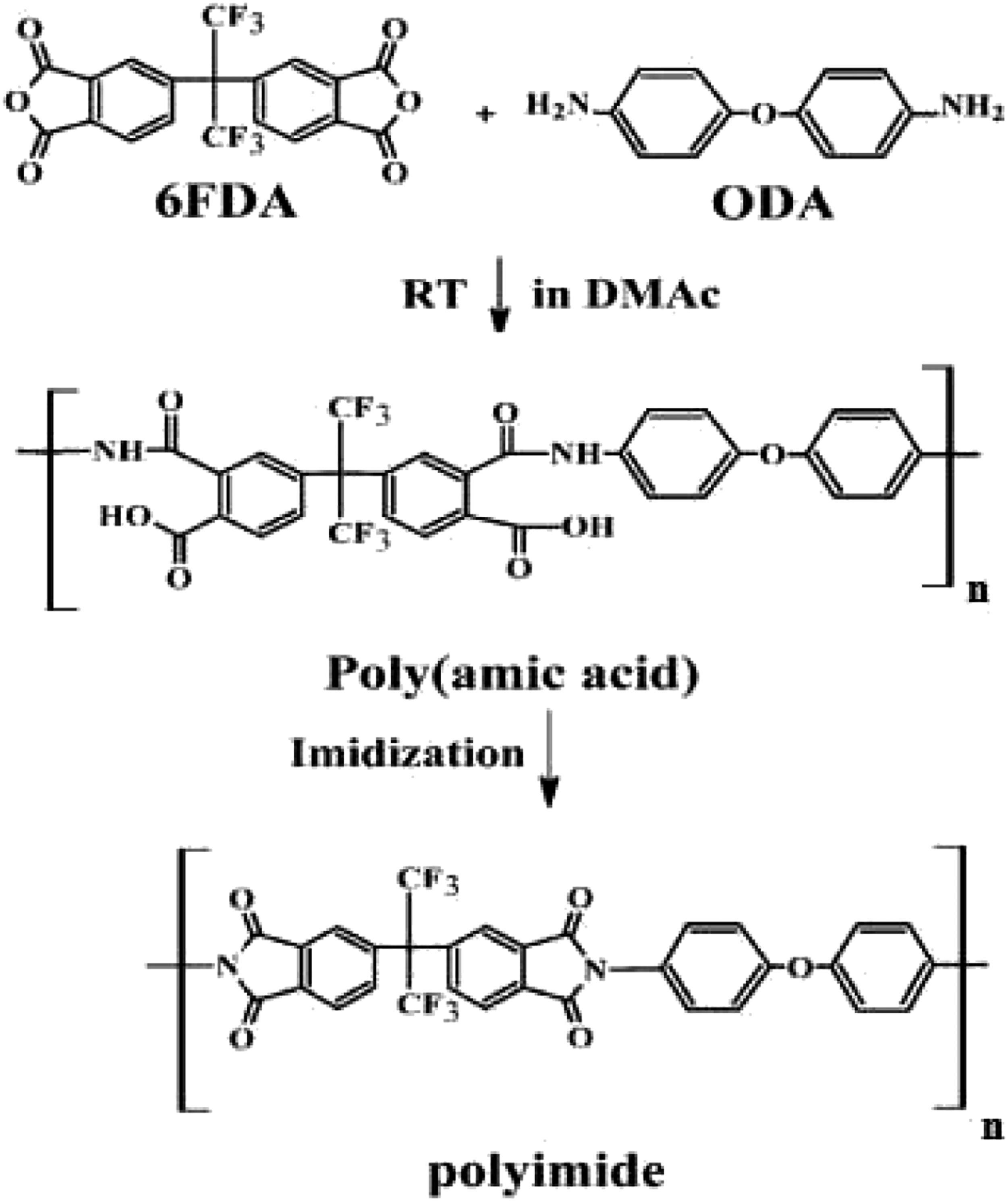
PI surface modification
The PI films were cleaned with distilled water and CH3COCH3. They were then etched and immersed in an aqueous 1 M KOH solution, rinsed with DI water, and dried at 35°C for 30 min. The etched PI was immersed and stirred in a sensitization solution of 45 mL of SnCl2, and then a PdCl2 solution was used to activate the surface of the PI. Finally, the PI films were dried at 25°C for 25 min.
Ag metallization plating
The Ag plating bath was composed of 15 g/L AgNO3, 2.6 g/L EDTA, 1 g/L NaOH and 10 mL/L HCHO. The mixture was stirred until it became transparent with yellow color at a pH value of 12, and then the PI samples were rinsed in the solution and dried under vacuum at 50°C. The Ag metallization process on the PI films is schematically illustrated in Figure 2. Schematic illustration of Ag metallization on PI films.
Characterizations
The surface properties were investigated with scanning electron microscopy (SEM), energy-dispersive X-ray spectroscopy (EDX), and X-ray diffraction (XRD).
The XRD pattern was recorded on an XRD-D80 supplied by a SHIMADZU focus diffractometer that used CuKα radiation with a wavelength of λ = 1.54 Å.
The composition of the deposited Ag was obtained from energy-dispersive spectroscopy, and the morphology and thickness of the Ag deposited on the PI films were measured using SEM. Observation and analysis were performed with a Hitachi SU-1500 SEM.
The FTIR spectra were recorded with an FT-IR infrared spectrophotometer (Shimadzu, made in Japan) in the wavenumber range of 500–4000 cm−1. The thermal stability was measured over a temperature range of 50–800°C using a thermogravimetric analyzer model (TGA7-USA) under N2 gas and at a heating rate of 10°C/min.
Results and discussion
Figure 3 illustrates the EDX analysis, which showed a strong signal in the Ag region and confirmed the formation of Ag nanoparticles. The Ag nanoparticles typically showed an optical absorption peak at approximately 3 keV, and Yuen et al. presented a similar absorption peak.
19
Other peaks were observed in the C, O, and Cl biomolecules involved in capping the surfaces of these nanoparticles. EDX spectrum of Ag nanoparticles.
The XRD patterns of the Ag deposited at different deposition times are presented in Figure 4. These XRD patterns exhibited only the peaks of metallic silver (JCPDS 04–0783) without any other signals, indicating that the synthesized samples are highly purified and face-centered cubic (fcc)-phased Ag NPs, which is in agreement with Zhang et al.
20
XRD patterns of silver deposits at different deposition times: (A) 5 min, (B) 10 min, and (C) 15 min.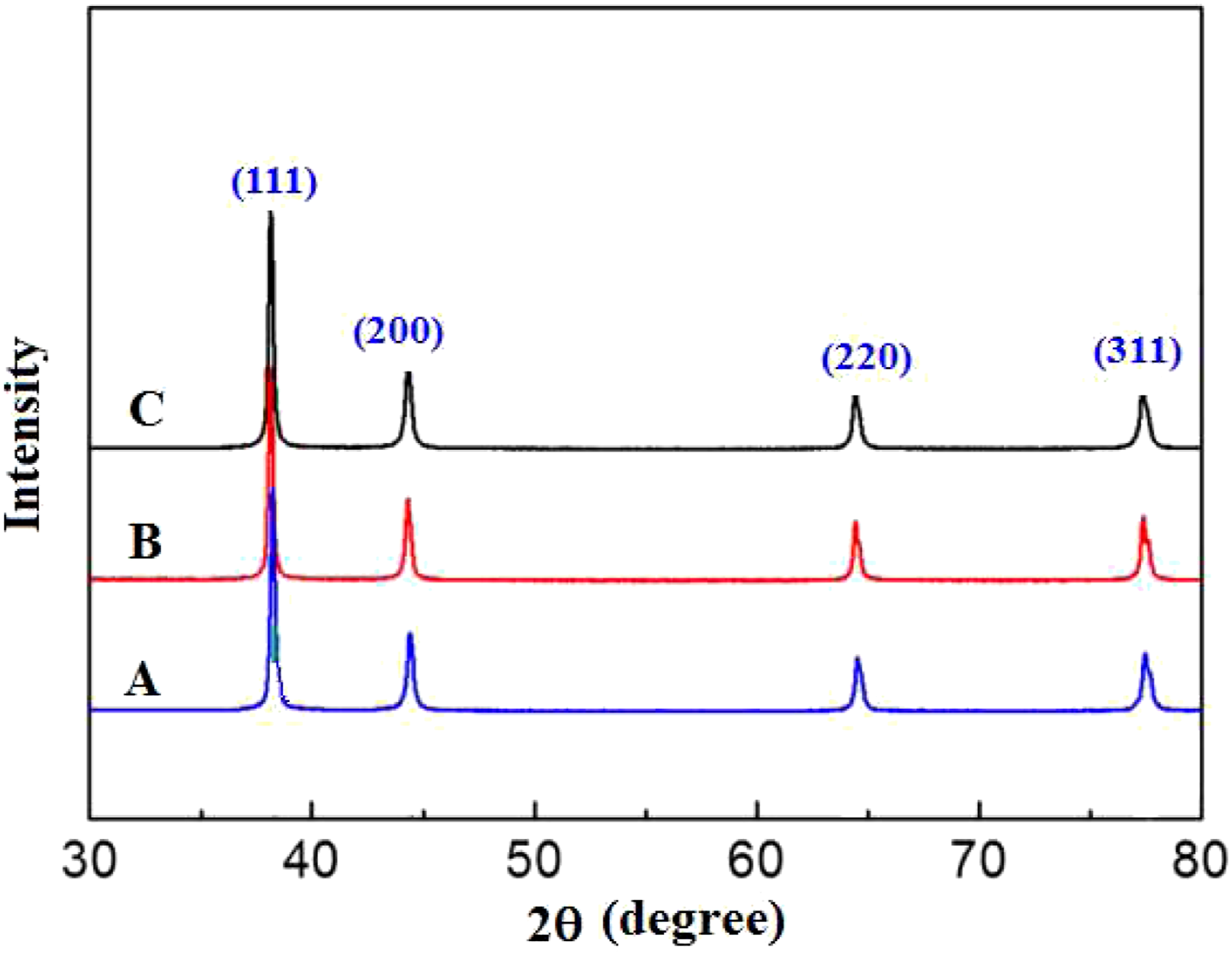
The diffraction peaks at 2θ= 38.2°, 44.5°, 64.6°, and 78.2° represent the 111, 200, 220 and 311 planes of silver, respectively. The silver oxide phase was not detected in the deposits. Figure 4 also shows that the corresponding peaks became sharp and the peak width became slightly narrower, indicating that the peaks for the crystalline structure of the silver deposits at different times became more vivid.20,21
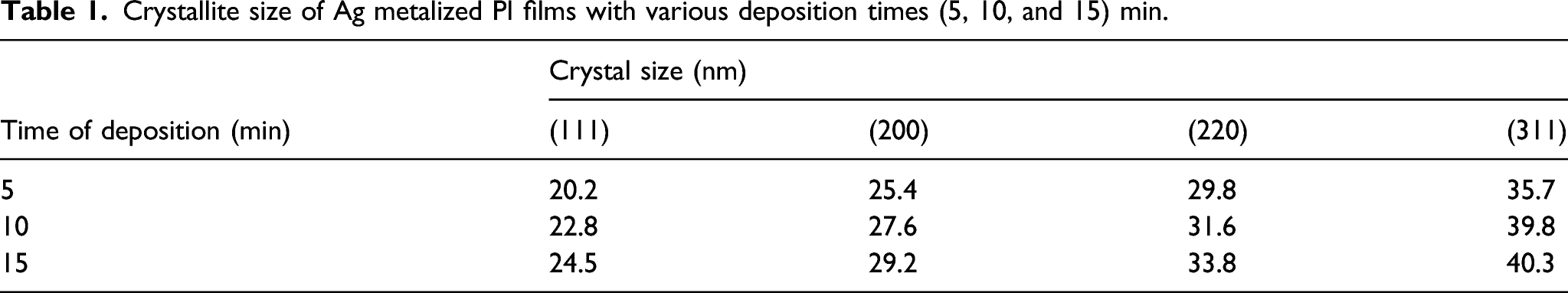
The position of the peaks obtained from the diffraction pattern also provides information about the translational size and shape of the crystal. The crystallite size of the silver nanoparticles was calculated using Scherrer’s equation
Crystallite size of Ag metalized PI films with various deposition times (5, 10, and 15) min.
FTIR analysis for polyimide and metalized polyimide at different deposition times are shown in Figure 5. For the pure PI spectrum, the main characteristic bands of PI at 1952 and 1602 cm−1 unsymmetrical and symmetrical for C=O stretching of imide groups, whereas the band at 1488 cm−1 is due to stretching of the C–C bond of the imide ring representing the successful chemical imidization process of PAA conversion to polyimide.22,23 Fourier transform infrared (FT-IR) spectra of A) PI and Ag metalized PI films with various deposition times (5, 10, and 15 min for B, C, and D, respectively).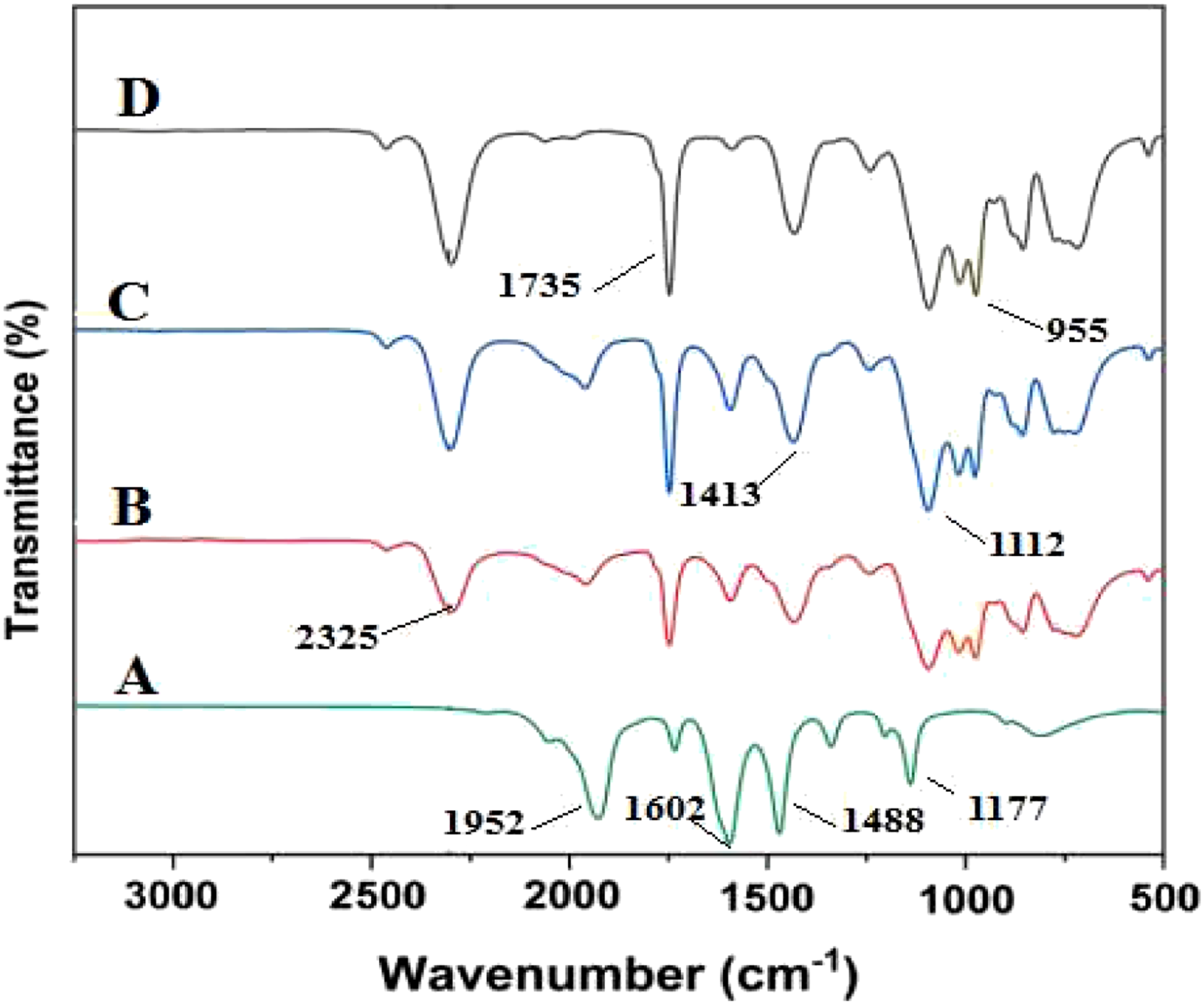
The FT-IR analysis for Ag metalized PI at different deposition times (5, 10, 15) minute is shown in Figure 5 (B, C, and D). We can see that the position of =C-O-C= stretching absorption bands at 1735 cm−1 had no changes in position, only a significant difference in peak size at the deposition time increase. The formation of new bands (N–H stretching absorption band and N–C stretching band) assigned at 2325 and 955 cm−1 indicated strong coordination between N atoms and silver nanoparticles. 18 The C–C stretching and = C–H plane vibration band at 1488 and 1117 cm−1 are shifted to 1413, and 1112 cm−1 indicates the interaction of the silver nanoparticle with the polymer backbone.24,25
Thermal analysis of PI and Ag metalized PI films with various deposition times (5, 10, and 15) min.
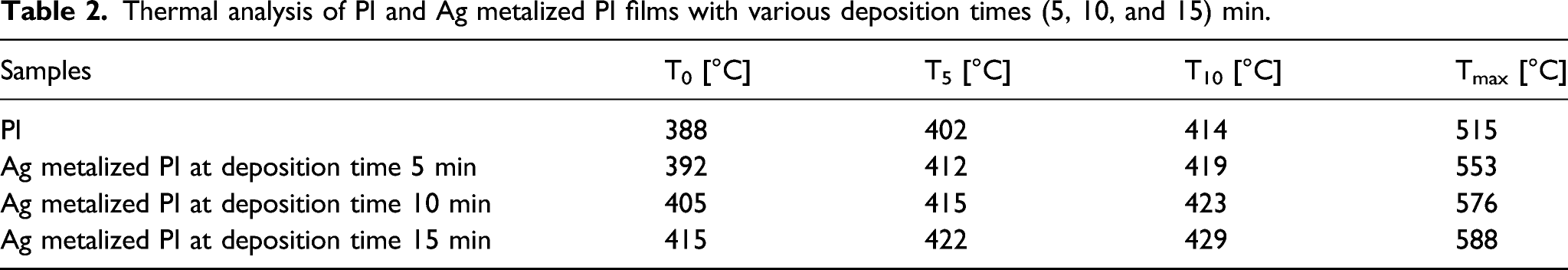
As shown in Figure 6, PI completely decomposes at temperatures above 550°C, and this behavior agrees with ref. [26,27]. PI, T0, T5, T10, and Tmax were observed at 388, 402, 414, and 515°C. As the deposition time increased, the thermal stability increased, and Ag deposited on polyimide at 5 min resulted in T0=392, T5=412, T10=419, and Tmax=553°C. Ag-metalized PI at a deposition time of 10 min increased the thermal properties compared to PI and Ag-metalized PI at a deposition time of 5 min. TGA plots of PI and Ag metalized PI films with various deposition times (5, 10, and 15) min.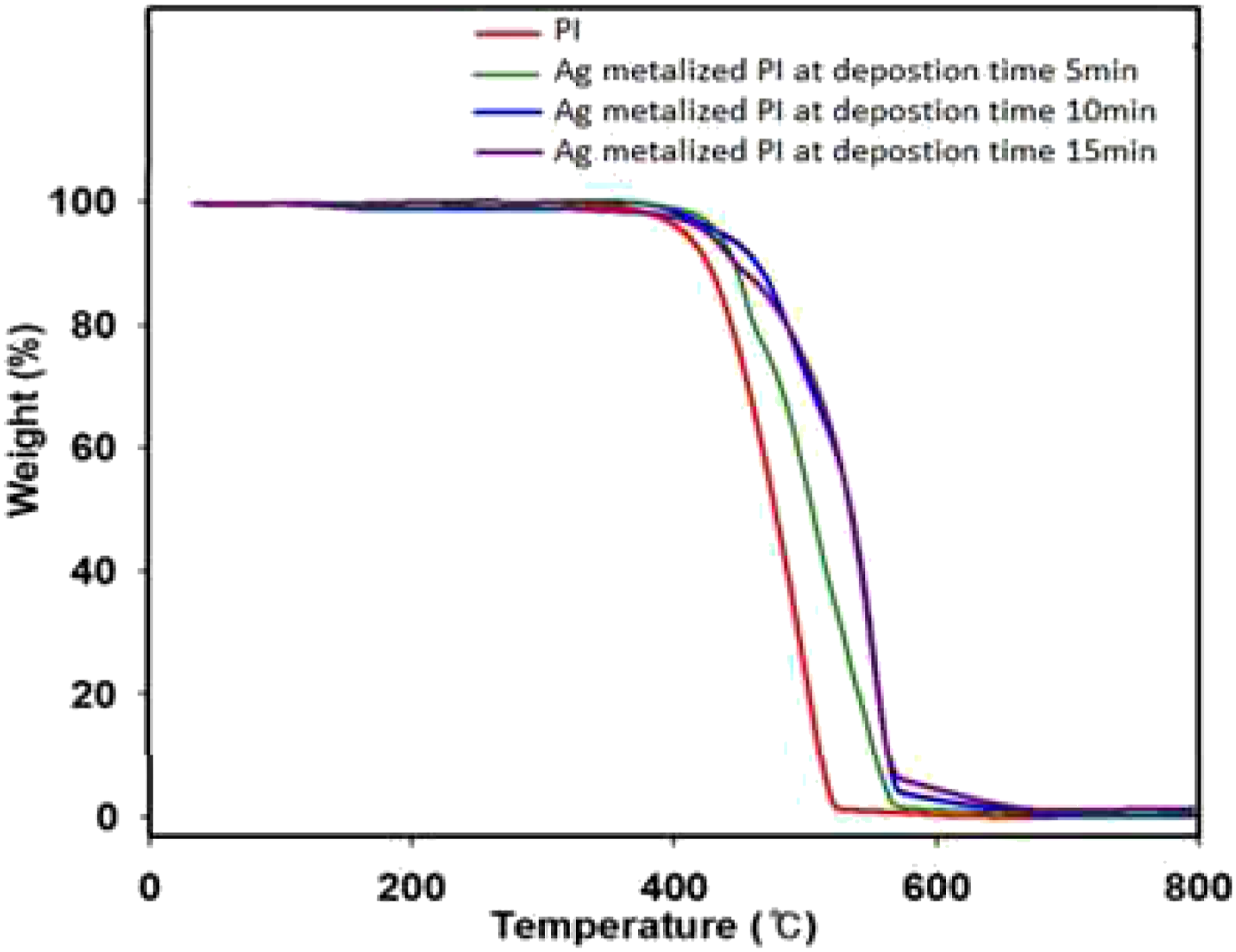
Consequently, Ag-metalized PI at a deposition time of 15 min further increased the thermal stability, which would benefit electronic devices.28,29
Figure 7 shows cross-sectional SEM micrographs of the deposits as a function of deposition time. The surface morphology studies imply that the deposited films are uniform, smooth, homogeneous, and nearly densely packed and free from voids, cracks, or pinholes.19,24 However, the surface of the PI substrate was comparatively rough due to chemical etching. The rough surface could have been caused by the strong interfacial interaction between Ag and the PI substrate.18,30 Shows cross-section SEM micrographs of Ag metalized PI films with various deposition times (5, 10, and 15) minute for (A, B, and C).
The surface morphology of the films shows that the Ag NPs are closely packed and form continuous films along with the PI substrates, and the thickness was highly dependent on the deposition time. We can see that the thickness increased from (5–9.5) μm as the deposition time increased from 5 to 15 min.
The surface morphology and the average grain size of the films are shown in Figure 8 from the SEM images. SEM images of Ag metalized PI films with various deposition times (5, 10, and 15 min) for (A, B, and C).
The obtained specific grains are the fastest, favoured growing with no preferential growth direction.20,25 We can see crystal growth at increasing deposition time via a slight increase in grain size.31–33 The development and formation of granular structures can be observed, and the grains become tight (closely connected) and regularly shaped, but Ag deposited at 15 min, the grain size increases rapidly.19,34
Conclusion
In this study, Ag metalized an autolytic plating technique fabricated PI film. The quality of Ag coatings improved after etching by KOH solution, SnCl2 sensitization, and PdCl2 activation of the PI surface. FT-IR analysis showed that the modification opened the ring structure of the imide of the film and formed a new ring between Ag and polyimide. The results show that the thermal stability increases as the deposition time increases.
Surface characterization of silver metalized PI showed that the coating surface morphology became more compact, regular, and intense, and optimized as the plating time reached 15 min.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
