Abstract
The present research paper explores the potential of electrospun nanofibers in the promising field of skin tissue engineering. Specifically, we propose an advanced preparation and characterization of an electrospun Polyurethane/Calcium Chloride (PU/CaCl2) nanocomposite scaffold, devised to boost the scaffold’s physicochemical and biological properties for skin tissue regeneration. By incorporating CaCl2 into the PU matrix using an electrospinning process, we were able to fabricate a novel nanocomposite scaffold. The morphological examination through Field Emission Scanning Electron Microscope (FESEM) revealed that the fiber diameter of the PU/CaCl2 (563 ± 147 nm) scaffold was notably smaller compared to the control (784 ± 149 nm). The presence of CaCl2 in the PU matrix was corroborated by Fourier-Transform Infrared Spectroscopy (FTIR) and Thermogravimetric Analysis (TGA). Furthermore, the PU/CaCl2 scaffold exhibited superior tensile strength (10.81 MPa) over pristine PU (Tensile −6.16 MPa, Contact angle - 109° ± 1° and Roughness - 854 ± 32 nm) and revealed enhanced wettability (72° ± 2°) and reduced surface roughness (274 ± 104 nm), as verified by Contact angle and Atomic Force Microscopy. The developed scaffold demonstrated improved anticoagulant properties, indicating its potential for successful integration within a biological environment. The improved properties of the PU/CaCl2 nanocomposite scaffold present a significant advancement in electrospun polymer nanofibers, offering a potential breakthrough in skin tissue engineering. However, additional studies are required to thoroughly evaluate the scaffold’s effectiveness in promoting cell adhesion, proliferation, and differentiation. We aim to catalyze significant advancements in the field by revealing the creation of a potent skin scaffold leveraging electrospun nanofibers. Encouraging deeper exploration into this innovative electrospun composite scaffold for skin tissue engineering, the PU/CaCl2 scaffold stands as a promising foundation for pioneering more innovative, efficient, and sustainable solutions in biomedical applications.
Keywords
Introduction
The emergence of tissue engineering as a viable solution to the challenges in regenerative medicine has led to the need for developing versatile and effective scaffolds. These scaffolds aid in regenerating various tissues, including skin, which is a primary protective barrier in the human body and often the subject of various ailments and injuries. 1 In the pursuit of ideal scaffolding materials, electrospun polymer nanofibers have gained significant attention due to their structural similarities to the natural extracellular matrix, high porosity, and an extraordinary surface-to- volume ratio, which collectively enhance cell attachment and proliferation. 2
Electrospun polymer nanofibers are instrumental in the multidisciplinary realm of tissue engineering. A fusion of engineering principles and life science, tissue engineering aims to restore or enhance the function of tissues and organs using highly specialized structures known as scaffolds. 3 These biodegradable scaffolds, typically crafted from biomaterials, are designed to support cellular growth and tissue repair while maintaining the necessary transport of nutrients and waste products.4,5
However, despite their considerable potential, there are existing issues which obstruct the practical application of electrospun polymer nanofibers in biomedical fields. One such problem is related to the hydrophobic nature of certain commonly used biocompatible polymers, like Polyurethane (PU). 6 Despite its mechanical properties and compatibility with biological tissues, the hydrophobic nature of PU hampers cell attachment and proliferation, thereby limiting its application in tissue engineering. 7 Various metallic nanoparticles, including zinc, gold, silver, and copper have been utilized in wound dressing applications to support the wound closure. 8 In this study, we have introduced CaCl2 into the polyurethane matrix, marking the first instance of its use in developing scaffolds for wound healing applications. The physicochemical alterations resulting from the introduction of CaCl2 into the polyurethane matrix, along with its impact on blood compatibility and fibroblast growth, have not been explored in previous research to the best of our knowledge. This study introduces an innovative approach by combining PU with CaCl2 for the design and fabrication of electrospun scaffolds.
Pivotal to this design is the development of porous structures, balancing mechanical robustness and biological compatibility to stimulate cell proliferation and propel tissue regeneration. 9 Electrospinning serves as a highly versatile technique for creating such nano-/microfibrous porous structures, making it an invaluable asset in the creation of wound-healing scaffolds.10,11 This technique offers the potential for fabricating scaffolds from a wide range of biopolymers and nanocomposites, thus extending its reach to a multitude of medical applications.12,13
This study primarily explores the potential of PUs in the electrospinning process. PUs, with their inherent toughness, abrasion resistance, and durability in watery solutions, are a favorable choice for medical implants and wound dressings. 14 The hydrophilic surface functionalization of electrospun nanofibers was shown to enhance the biological properties resulting in the wound closure.15,16 Such integration leads to enhanced wound healing properties, as affirmed by prior research that confirms the beneficial effects of electrospun PU mats in wound healing.17,18 This research presents an innovative strategy of combining PU and CaCl2 for the design and fabrication of electrospun scaffolds. We aim to develop scaffolds with improved thermal and mechanical properties and porous structures through the integration of CaCl2 into the PU composite. By doing so, we aim to pave the way for advancements in tissue engineering specifically related to skin regeneration applications.
Materials and methodology
Materials
The study employed medical grade Tecoflex polyurethane (molecular weight (Mw) 1000 g/mol) obtained from Lubrizol, USA, dimethylformamide (DMF) purchased from Merck, USA, and anticoagulant reagents obtained from Thermo Fisher Scientific, Selangor, Malaysia.
Preparation of electrospinning solutions
To prepare the solutions for electrospinning, Tecoflex PU pellets were dissolved in DMF solvent at a concentration of 9 wt% by stirring the mixture overnight to ensure complete dissolution of the polymer. Meanwhile, CaCl2 salt was separately dissolved in DMF solvent at a concentration of 4 wt% under constant stirring for 1 h. After that, the PU/CaCl2 solution was prepared by mixing the two solutions at a ratio of 7:2 (w/v%) and further stirred for an hour to ensure the uniform dispersion of CaCl2 salt within the PU matrix. The resultant solution was then loaded into a syringe and used for electrospinning to produce the skin scaffold.
Fabrication of scaffolds
The preparation of the PU/CaCl2 solution intended for electrospinning (Progene Link SdnBhd) involved several carefully managed steps. Initially, Tecoflex PU pellets were immersed in a Dimethylformamide (DMF) solvent, aiming for a final concentration of 9 wt%. To facilitate complete dissolution of the polymer, this mixture was stirred continuously overnight. Simultaneously, CaCl2 salt was dissolved in a separate container of DMF solvent at a concentration of 4 wt%, requiring an hour of continuous stirring to ensure complete dissolution. Subsequently, the two solutions were blended in an 8:2 v/v ratio, resulting in a PU/CaCl2 solution. To ensure the uniform dispersion of salt within the PU matrix, the stirring was continued for an additional 1 hour. The resulting solution was then loaded into a syringe with an attached needle in preparation for electrospinning. The electrospinning process took place under specific conditions - a flow rate of 0.2 mL/h, an applied voltage of 10.5 kV, and a collector distance of 20 cm, culminating in the formation of a nanofibrous scaffold.
Surface characterization studies
Field emission scanning electron microscope
The preparation of samples for Field Emission Scanning Electron Microscope (FESEM) analysis (Hitachi SU8020, Tokyo, Japan) involved an initial step of coating the membranes with a thin gold layer employing a sputter coater. Following this, the gold-coated samples were subjected to scrutiny via FESEM. The microscope’s versatility was utilized by capturing images from numerous locations and at diverse magnifications, which afforded a comprehensive understanding of the sample’s surface morphology. The fiber diameter was measured via Image J by choosing 30 locations randomly in the captured image.
Fourier transform infrared spectroscopy
The composition of the fabricated nanofibers was assessed using Fourier-Transform Infrared Spectroscopy (FTIR) (Nicolet iS 5, Thermo Fischer Scientific, Waltham, MA, USA.). Sample preparation involved positioning a minute quantity of the nanofiber onto the FTIR unit’s diamond crystal. Thereafter, the spectra were collected within a wavelength span of 400 to 6000 cm−1 and a resolution of 4 cm−1. This data acquisition process facilitated the identification of functional groups present within the sample.
Uniaxial mechanical testing
The assessment of the scaffolds' tensile strength was conducted via uniaxial mechanical testing, utilizing a Gotech Mechanical Tester (AI-300).Segments of the scaffold (4 cm × 1.5 cm) were secured in the tensile compartment of the tester, following which tension was exerted at a crosshead speed of 10 mm/min until the point of failure was reached. The data collected in the form of stress-strain curves during these tests facilitated the computation of the scaffold’s average strength.
Thermal analysis
The scaffolds' thermal stability was evaluated using a Thermogravimetric Analyzer (TGA) (PerkinElmer, Waltham, MA, USA). For this analysis, individual specimens, each weighing 5 mg, were positioned in the TGA unit. Subsequently, these samples were subjected to a heating process under a nitrogen atmosphere, ranging from 30 to 1000°C, at a progressive rate of 10°C per minute. The record of the specimen’s weight reduction in relation to temperature allowed for the determination of the scaffold’s thermal stability.
Atomic force microscopy
Surface characterization of the nanofibers was carried out with an Atomic Force Microscope (AFM) (NanoWizard®, JPK Instruments, Berlin, Germany). The assessments were executed in tapping mode, with the scanning dimensions set to 20 μm × 20 μm. During this process, the AFM tip was maneuvered to make contact with the nanofibers' surface. The subsequent recording of the interaction between the tip and the sample provided insight into the surface roughness and morphology of the scaffold.
Contact angle measurement
The samples' contact angle measurements were executed via the liquid drop method, utilizing Video-based Contact Angle (VCA) equipment (AST products, Inc., Billerica, MA, USA). A drop of deionized water, precisely 0.5 µL, was delicately positioned on the sample membrane’s surface. Subsequently, a still image of the water droplet was swiftly captured using a video camera. Using computer-integrated software, the average contact angle was calculated from this static image. This process facilitated the determination of the sample’s wettability and surface energy.
In-vitro degradation behaviour
The electrospun membranes measuring 1.5 cm * 1.5 cm were placed in PBS at pH 7.4 to examine the degradation of the membranes in vitro. Every sample was stored at 37°C after being separately placed in PBS-filled centrifuge tubes. The membranes were collected at day 1, 3 and 5 completely cleaned with distilled water, and oven-dried to determine the amount of weight reduction. The following equation was utilized to calculate the weight loss percentage of developed electrospun membranes:
Blood compatibility analysis
Activated partial thromboplastin time and prothrombin time
Experiments were conducted to measure the blood clotting times, to understand the anticoagulant behavior of the developed nanofibers. The Activated Partial Thromboplastin Time (APTT) test was conducted by first incubating a small piece of the sample with 50 µl of Platelet-Poor Plasma (PPP), then introducing 50 µL of rabbit brain cephaloplastin reagent and allowing it to incubate for 3 min at 37°C. Initiation of clotting was done by adding 50 µL of CaCl2 to the mixture and the time taken for blood clot formation was recorded as the APTT time. Likewise, for the Prothrombin Time (PT) assay, a small piece of the sample was incubated with 50 µl of PPP, followed by the introduction of 50 µL of Sodium Chloride-Thromboplastin reagent (Factor III). The time required for the blood clot to form was recorded as the PT time. These experiments shed light on the potential utility of the developed nanofibers in wound dressings or other medical applications where blood clotting could be a significant consideration.
Hemolysis percentage
Hemolysis testing serves as a critical step in assessing the biocompatibility of medical materials, including the developed nanofibers, as it gauges the possibility of red blood cell disruption and lysis. Hemolysis can result in the dispersal of intracellular components into the bloodstream, leading to potential health complications such as renal failure, thrombosis, and cardiovascular events. In this study, freshly separated blood was diluted with saline, and a small piece of the sample was left to incubate with the diluted blood at 37°C for a duration of 60 min. The negative control comprised a blend of blood and normal saline, while the positive control consisted of a mix of blood and distilled water. Afterward, the samples were centrifuged at 3000 r/min for 15 min, and the absorbance of the supernatant was measured at 542 nm using a UV spectrophotometer. The hemolysis percentage was determined using a previously reported equation, thereby providing crucial data concerning the biocompatibility of the nanofibers for diverse biomedical applications.
Cell viability analysis
Human dermal fibroblast (HDF) cells were used to test the cell viability of the electrospun membranes. These cells were grown in DMEM media with 10% fetal bovine serum and 5% antibiotic penicillin. Every 3 days, the culture media was changed and incubated at 37°C with 5% carbon dioxide (CO2). The created electrospun scaffolds were divided into small pieces and put in 96-well plates before cell seeding. They were also cleaned with PBS and sterilized with 75% alcohol. The samples were then seeded with HDF cells at a density of 10*103 cells/cm2 and housed in a CO2 incubator for 72 h. The medium was supplemented with 20% MTS after a 72-h culture and placed in the 5% CO2 for 4 h. To ascertain their viability rates, absorbance at 490 nm was measured using a spectrophotometric plate reader after 4 h.
Statistical analysis
Statistical analysis of results performed were analyzed using GraphPad Prism and presented as mean ± standard deviation. A one-way ANOVA test was employed for statistical analysis with p value less than 0.05 was considered statistically significant./
Results and discussion
FESEM investigation
The results of the FESEM analysis presented in Figure 1 showed that the prepared PU and its nanocomposites had a finely formed fiber structure with a randomly oriented matrix. The mean diameter of pristine PU was found to be 784 ± 149 nm. In contrast, the prepared PU/CaCl2 displayed a significantly reduced fiber diameter of 563 ± 147 nm compared to the control.It has been reported by Shi et al. a decrease in the polymer concentration could result in the small diameter of fibers.
19
The addition of CaCl2 resulted in the reduced polymer concentration which might have favored the decrease in fiber diameter.Further, the fiber reduction of PU may alsoattribute to the increase in conductivity of the solution upon adding CaCl2 to the pure polymer matrix. These findings are consistent with previous reports by Jaganathan et al.
20
and Lv et al.
21
who demonstrated a reduction in fiber diameter with the addition of Zinc Nitrate andursolic acid into the polymer matrix, respectively. Moreover, cell viability in the ursonic acid loaded chitosan-polyvinyl alcohol scaffolds was higher compared to the pure chitosan-polyvinyl scaffolds, suggesting the potential of the developed nanocomposite with smaller diameter for skin tissue regeneration.
19
The presence of Ca in the PU matrix was confirmed through an Energy Dispersive X-ray Analysis (EDX) study, with pure PU showing no trace of Ca, while the PU/CaCl2 displayed a Ca content of 1.8%. The observed reduction in fiber diameter and the presence of Ca in the PU matrix suggest that the developed nanocomposite scaffold may have improved mechanical and biological properties for various biomedical applications, including skin tissue engineering. FESEM images of (a) PU and (b) PU/CaCl2.
Fourier transform infrared spectroscopy
The functional groups of the prepared PU and its nanocomposites were analyzed using Fourier-transform infrared (FTIR) spectroscopy. Figure 2 shows the spectra of the samples. The peak at 3318 cm−1 represents the NH stretching and the peaks at 1597 cm−1 and 1531 cm−1were due to the vibrations of the NH group. The peaks at 2920 cm-1and 2852 cm-1denote the CH2 stretching and its vibrations were observed at the peak 1414 cm−1. The C = O stretching of carboxylic groups was shown by a twin peak at 1730 cm−1 and 1701 cm−1, and its vibrations corresponding to alcohol were found at 1221, 1105, and 770 cm-1.
22
The spectra of PU/CaCl2 showed no new peak but changes in the peak intensity (wide deviation in NH peak) were observed. The CH peak in PU at 2920 cm-1 was shifted to 2943 cm-1and 2937 cm-1in PU/CaCl2, respectively. Further, a new peak was also formed in a region of 2916 cm−1. The formation of a hydrogen bond, changes in peak shift, and new peak formation indicate the presence of CaCl2 in the PU matrix.23,24 The FTIR results confirm the successful incorporation of CaCl2 into the PU matrix, which was also observed in the EDX analysis. FTIR images of electrospun membranes.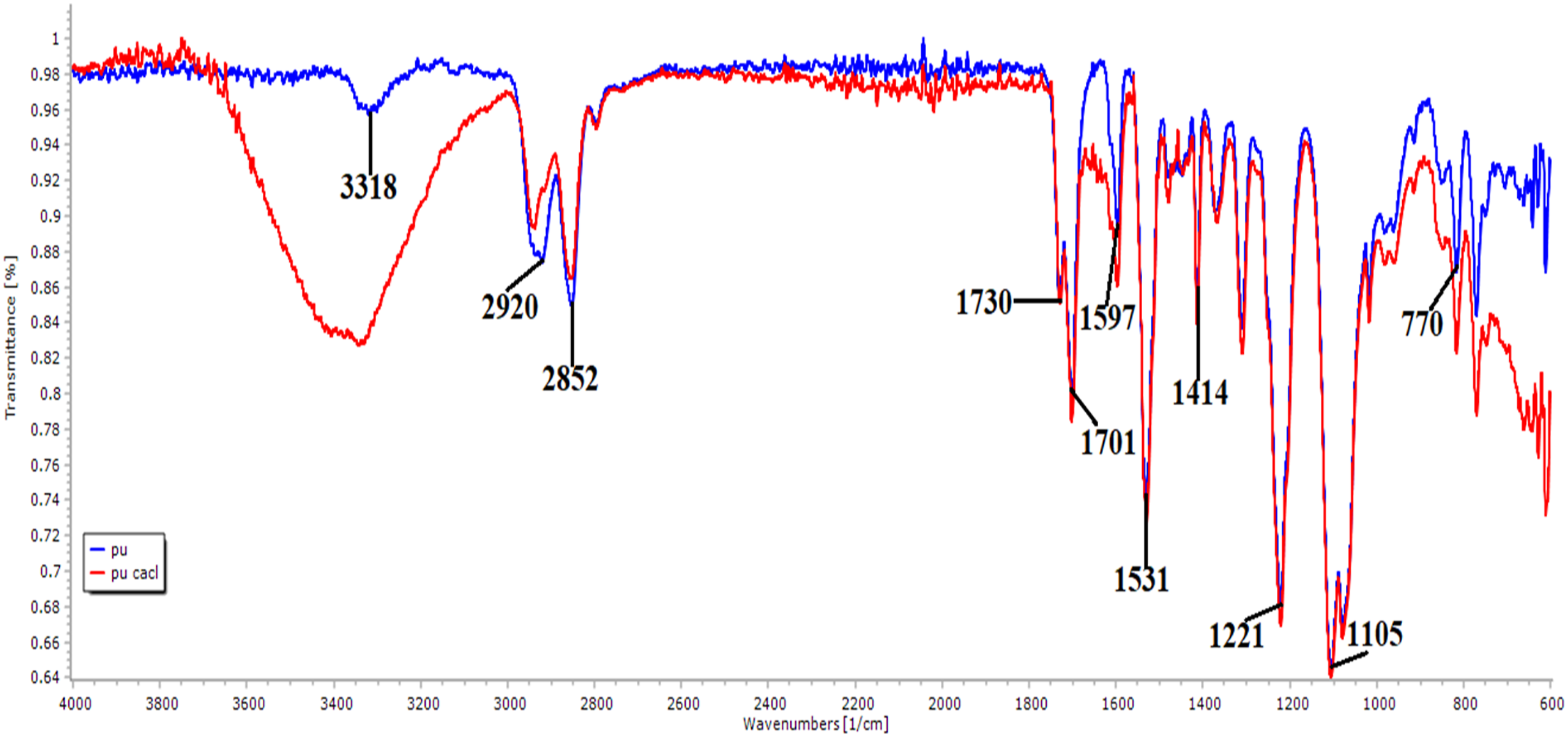
Uniaxial mechanical testing
The tensile strength of the prepared PU and its nanocomposites were analyzed and discussed. The tensile curves for both the PU and PU/CaCl2 nanocomposites are shown in Figure 3. The results indicated that the addition of CaCl2 into the PU matrix resulted in an increase in the average tensile strength. The pure PU showed a tensile strength of 6.16 MPa, while the PU/CaCl2 nanocomposites exhibited a higher tensile strength of 10.81 MPa. This increase in strength may be attributed to the strong interaction between the PU matrix and CaCl2. Jaganathan et al. also reported an increase in tensile strength for their skin scaffold comprising PU added with Zinc Nitrate. It was found that the composite scaffold with smaller fiber diameter displayed higher tensile strength which resembles our findings.
20
Further, they concluded that the enhanced strength of the scaffolds is conducive to skin tissue growth. Therefore, the increased tensile strength of our developed nanocomposites may be suitable for skin tissue engineering applications. Tensile curves of electrospun membranes.
Thermal analysis
The thermal stability of the developed nanofibrous scaffolds was evaluated using thermogravimetric analysis (TGA). Some researchers conducted thermal analysis of materials in order to understand how produced composites degrade. The thermal stability, interaction and integrity of the produced materials were predicted using thermal analysis.
25
The thermal stability of the developed scaffold would be conducive for tissue regeneration process.
26
Figure 4 shows the TGA curves for pure PU and its nanocomposites. The onset degradation temperature for the pure PU was 284°C, whereas for the PU/CaCl2 nanocomposites, it was found to be 223°C. The results revealed that the initial degradation temperature of the pure PU was reduced with the addition of CaCl2 The reason for the reduction was due to the evaporation of water or moisture present in the developed composites.
27
The derivative weight loss curves for the pure PU and its nanocomposites are shown in Figure 5. The pure PU exhibited three weight loss peaks at temperatures of 218°C to 360°C, 360°C to 518°C, and 518°C to 745°C, while the PU/CaCl2 displayed six weight loss peaks at temperatures of 35°C to 106°C, 106°C to 244°C, 244°C to 348°C, 348°C to 554°C, 554°C to 665°C, and 665°C to 862°C. The nanocomposites showed an increase in weight loss peaks compared to the pure PU, and but weight loss peak intensity was found to be lowered in the nanocomposites with the addition of CaCl2 indicating higher stability. These results suggest that the presence of CaCl2 in the PU matrix affects the thermal stability of the nanocomposites. Thermogravimetric analysis (TGA) curve of electrospun membranes. Derivative thermogravimetric 

Atomic force microscopy
Atomic force microscopy (AFM) was employed to investigate the surface morphology of PU and its nanocomposites. As shown in Figure 6, the surface roughness of the pristine PU was found to be 854 ± 32 nm, while the prepared PU/CaCl2 composite scaffold exhibited significantly reduced surface roughness of 274 ± 104 nm. The incorporation of CaCl2 into the PU matrix was found to be responsible for the smoother surface of the nanocomposites. This might be due to the molecular structure of CaCl2, which has a planar shape that could reduce the surface roughness of the composite scaffold. These findings are in line with the study conducted by Jaganathan et al. who reported a decrease in surface roughness upon the addition of Zinc Nitrate in PU nanocomposites .
20
Additionally, Chou et al. prepared microporous poly (hydroxybutyric acid) membranes and reported a decrease in surface roughness, which led to higher attachment of fibroblast cells.
28
Therefore, the PU/CaCl2 composite scaffold with a smoother surface could be a promising candidate for skin tissue engineering. AFM images of (a) PU and (b) PU/CaCl2.
Contact angle measurement
The contact angles of PU and PU/CaCl2 composites were measured using VCA equipment. The results indicated that the mean contact angle of the PU was 109° ± 1°, while the prepared PU/CaCl2 nanocomposites showed a contact angle of 72 ± 2.The corresponding images were indicated in Figure 7. The nanocomposites exhibited hydrophilic behavior compared to the control. This finding is consistent with the study by Jaganathan et al. who developed a skin scaffold comprising PU added with Zinc Nitrate, which showed hydrophilic behavior.
20
It has been reported that hydrophilic behavior is favorable for fibroblast cell adhesion and proliferation.
23
Therefore, the developed nanocomposites with hydrophilic behavior might be suitable for skin tissue engineering. Contact angle images of (a) PU and (b) PU/CaCl2.
In-vitro degradation analysis
Weight loss measurement of PU and PU/CaCl2 at different time intervals.


Graphical representation of weight loss percentage of electrospun membranes.
Blood compatibility analysis
The APTT and PT assays were performed to investigate the clotting time and anticoagulant properties of pure PU and PU/CaCl2 as indicating in Figure 9. The results showed that the developed PU/CaCl2 composites exhibited prolonged blood clotting time compared to pure PU membrane, indicating their potential as anticoagulant materials. The values of APTT were found to be significantly higher for PU/CaCl2 (p < .05) compared to PU, while the values of PT were also higher for PU/CaCl2 (p < .05) compared to PU.These findings suggest that the incorporation of CaCl2 into the PU matrix might have altered its coagulation properties. Blood compatibility measurements of developed electrospun membranes.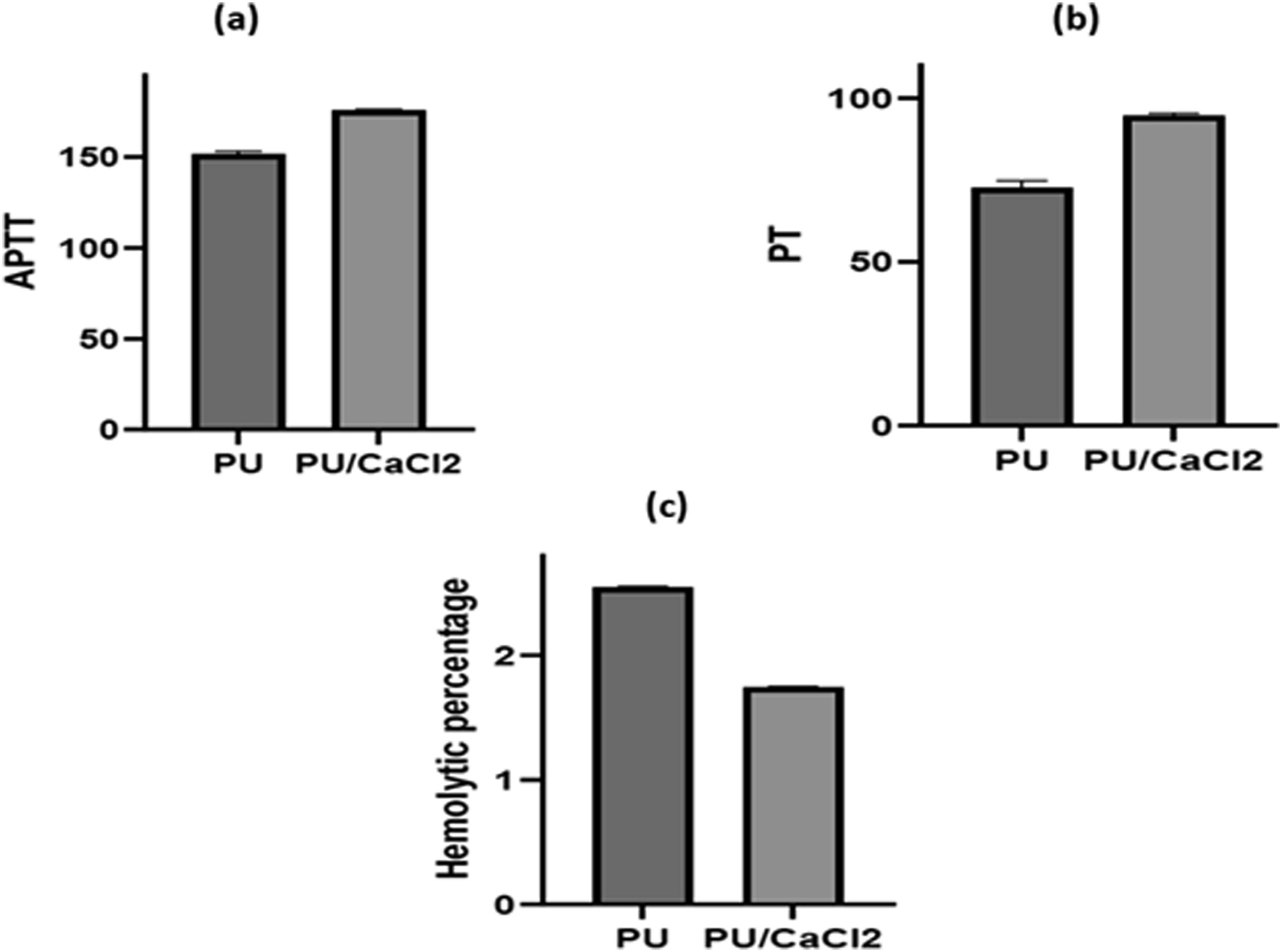
The hemolytic assay was carried out to evaluate the safety of the developed nanofibers against red blood cells. The results revealed that the hemolytic value of PU was higher than that of the PU/CaCl2 composites. The fabricated PU/CaCl2 nanocomposite scaffold material displayed a hemolytic percentage of 1.74%, while for pure PU the index was observed to be 2.55% as shown in Figure 8 (p < .05).According to the ASTMF756-00 (2000) standard, a scaffold is considered non-hemolytic if its index is less than 2%. 30 These findings suggest that the PU/CaCl2 composites have better blood compatibility compared to pure PU.Studies have been reported that the scaffolds with smaller fiber diameter and smoother surfaces favoring the enhanced blood compatibility. 30 Our developed composites showed both smaller fiber diameter and smoother surfaces which might have resulted in the enhanced anticoagulant nature.
Overall, the APTT and PT assays along with the hemolytic assay results indicate that the developed PU/CaCl2 composites exhibit improved anticoagulant and blood compatibility properties compared to pure PU. These findings have important implications for the use of these materials in skin tissue engineering applications, where it is crucial to ensure compatibility with the host’s blood.
In-vitro cell viability analysis
Figure 10 illustrates the fibroblast cell viability of electrospun PU and PU/CaCl2 composites. After a 3-day cell culture period, fibroblast cells were observed to be attached to both electrospun PU and PU/CaCl2. The cell viability rate for PU membrane was 126 ± 5%, while the electrospun PU combined with CaCl2 displayed 138 ± 3%. Clearly, the cell viability of PU/CaCl2 was significantly higher than that of pure PU. Literature suggests that scaffolds with smaller fibres and hydrophilic properties are favourable for fibroblast cell adherence and growth.
31
The smaller fibre diameter and increased hydrophilicity due to the CaCl2 addition resulted in higher fibroblast cell adhesion and proliferation. Cell viability of developed electrospun membranes.
Conclusion
In conclusion, this study elucidates the enhanced characteristics of electrospun PU nanofibers achieved by creating a novel PU/CaCl2 nanocomposite. The incorporation of CaCl2 into the PU matrix resulted in improvements in fiber properties, including reduced diameter and increased tensile strength, as confirmed by scanning electron microscopy and tensile strength analysis. Additionally, these enhancements led to improved blood compatibility, demonstrated by favorable results in APTT and PT assays and positive hemolysis tests indicating a non-hemolytic nature. HDF cell viability, assessed via MTT assay, underscored the composite scaffold’s biocompatibility. This innovative composite shows significant promise for biomedical applications due to its enhanced morphology and potential to enhance cell attachment, proliferation, and differentiation.
Footnotes
Acknowledgments
The authors would like to extend their heartfelt gratitude and acknowledgment to Miss Darsana Gopinathan and Dr. Ngo Thi Minh Thu for their invaluable assistance in the review of this manuscript. Their efforts and contributions were integral to the refinement of our work, and we are deeply thankful for their support and expertise. Their insights have significantly enhanced the quality of this research, and we would like to express our special thanks for their dedication and commitment to this project.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

