Abstract
There is a great interest in developing drug delivery systems that are biocompatible and release the drug in a controlled way. In this study, the release of curcumin from electrospun meshes made from polycaprolactone, polylactic acid, or their blends was studied and compared. The polymers and curcumin were dissolved in a mixture of chloroform and dimethylformamide. The beadless electrospun meshes were obtained under the following conditions: the applied voltage of 22 kV, the tip to collector distance of 13 cm, and the polymer feed rate of 0.2 ml/h. The viscosity of the polymer solution increased by increasing the percentage of polylactic acid in the mixture. As a result, the average fiber diameter of the meshes was increased. The fabricated polycaprolactone mesh showed the minimum average fiber diameter (∼120 nm) and the maximum porosity (∼71%). The hybrid nanofibrous meshes were also fabricated using two counter nozzles on both sides of the collector. The tensile strengths of the polylactic acid and polycaprolactone electrospun meshes loaded with curcumin were 19.86 and 12.64 MPa, respectively. The tensile strain of the meshes increased from 43 to 116% by increasing the polycaprolactone weight percentage in the mixture. The results showed that the physical–mechanical properties of the fabricated electrospun meshes could be tuned by polymer blending. The release of curcumin from these electrospun meshes was monitored in a buffer solution using an UV-Vis spectrophotometer. The critical parameters on the release of curcumin were found to be the type of polymers, the average diameter of nanofibers, porosity, and hydrophilicity of the meshes.
Introduction
The medicines that have a controlled drug release profile can maintain a constant drug concentration in the body for a specific period of time and with minimum side effects. The release of the drug can also be controlled by various environmental stimuli (e.g. pH, temperature, enzyme, etc.) [1]. Encapsulation of drugs in an electrospun nanofibrous mesh is one of the attractive methods to slow the release [2]. The profiles of the drug release from a nanofibrous mesh can be affected by changing its morphology, porosity, and polymer composition [3]. Nanofibers with smaller fiber diameters are good candidates to be used as drug delivery carriers due to their higher surface area to volume ratio [4].
The polymers used in drug delivery systems should be biocompatible, non-toxic, and non-inflammatory. They can be classified into two large categories of biodegradable and stable polymers. Biodegradable polymers are favorable to be used as drug delivery carriers and also in tissue engineering and medical devices (e.g. surgical meshes). This will also guarantee their degradation/hydrolysis and excretion from the body without any further complications [5,6]. Some of the polyesters such as polycaprolactone (PCL) and polylactic acid (PLA) are biodegradable and have hydrolyzable repeating groups in their chemical structures. They are also hydrophobic, semi-crystalline, aliphatic, and non-toxic. Thus, they are good candidates to be used in various biomedical devices and encapsulation of drugs [7]. Moreover, PCL and PLA have good mechanical properties and simple processability (e.g. melt extrusion). They can be stored in solid form without noticeable degradation for two to three years [8]. It should be highlighted that PLA is a sustainable polymer from renewable sources [9,10].
PCL and PLA electrospun (ES) meshes have been used for encapsulation of various drugs. As an example, the release of tetracycline hydrochloride from electrospun PLA, PCL, and PLA + PCL meshes has been investigated. The maximum and minimum release rate was found for the 1:1 mixture and PLA meshes, respectively [11]. The release of thymol from PLA 1:1 PCL blend nanofibers followed a two-stage (bimodal) drug release profile. The burst release time of thymol from the surface of nanofibers was about 12 h followed by a much slower release rate. The PLA and PCL nanofibrous meshes had the lowest and the highest levels of drug release, respectively [12]. The release of 5-fluorouracil from a blend of PCL and PLA increased as a function of the weight percentage of PCL in the composition of the electrospun mesh [13].
There is a growing interest in using natural herbal medicines because their long-term toxicity and harmful side effects are generally much lower than those of synthetic chemical drugs [14]. Curcumin (CM) is a polyphenolic compound extracted from turmeric with a molecular formula of C12H20O6. The main ingredient of CM is curcuminoid (77 wt%) which is also responsible for the yellow color of turmeric. CM has antioxidation, anticancer, anti-inflammatory, wound healing, antiviral, anti-AIDS, and liver protection properties [15].
In this research, the electrospinning technique using one-nozzle or two counter nozzles on both sides of the collector (hybrid) was used for making nanofibrous meshes of PCL and PLA. The morphological, mechanical, and drug release properties of the resulting meshes were studied by various techniques. This will shed more light on the topic of electrospun nanofibrous meshes made from polymer blends with tuned properties for targeted drug delivery.
Materials and methods
Materials
PCL (Mn = 80,000 g/mol), PLA (Mn = 103,000 g/mol), CM (Figure 1), chloroform, dimethylformamide (DMF), and phosphate buffer saline (PBS) tablets were all purchased from Sigma-Aldrich.
Chemical structure of CM with Mn = 368.68 g/mol and λmax = 421 nm.
Methods
Electrospinning
PCL (8% w/v) and PLA (5% w/v) were dissolved in a mixture of chloroform 80:20 DMF. CM 5% w/w was dissolved in the polymer solution and stirred for two hours. The resulting solution was transferred to a 5-ml syringe with a right-angle needle (I.D. 1.9 mm). A laboratory scale Nano-Azma electrospinning machine (Iran) was used for the preparation of nanofibrous meshes. A scheme showing the electrospinning setup has been shown in Figure 2. Table 1 shows the prepared electrospun meshes with their sample codes. The electrospinning conditions for the preparation of beadless nanofibers were found to be V = 22 kV, TCD = 13 cm, feed rate = 0.2 ml/h, and at room temperature (∼23℃). The fabricated nanofibrous meshes were collected on a cylindrical collector (D = 10 cm) covered with a layer of aluminum foil over a period of 40 h.
Electrospinning setup with two counter nozzles on both sides of the collector for the preparation of hybrid nanofibrous meshes (i.e. HY1 and HY2). The same setup with only a single nozzle was used for the preparation of other electrospun meshes. Prepared nanofibrous meshes. CM: curcumin; ES: electrospun; PCL: polycaprolactone; PLA: polylactic acid.

Testing and analysis
Electrical conductivity and viscosity
The electrical conductivity of the polymeric solutions was measured using an AD 8000 electrical conductivity meter, Romania. The viscosity of the polymeric solutions was measured at 25℃ by a DV-E viscometer, England. The measurements were repeated for a minimum of three times.
Scanning electron microscopic studies
The morphology of the nanofibers was investigated using PHENOM PRO X (Netherlands) with 20,000 × and 15,000 × magnification. The fiber diameter of minimum 100 fibers was also measured in the SEM micrographs using image J software.
Measurement of porosity
The apparent density and porosity of the nanofibrous meshes were calculated using equations (1) to (3). The density of PCL, PLA, and CM was 1.14, 1.26, and 0.93 g/cm3, respectively [16–20].
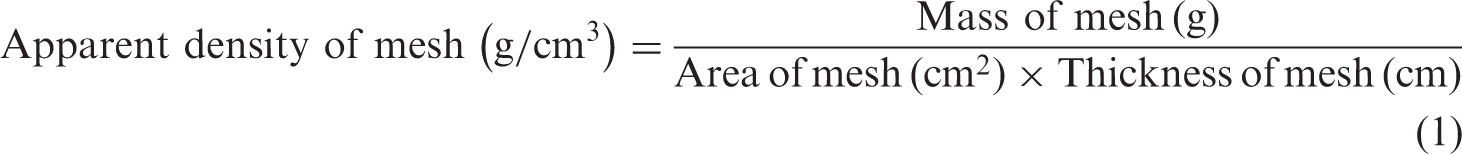


Static contact angle measurement
The static water contact angle (WCA) on the nanofibrous meshes was determined using an optical tensiometer (CA-ES10, Iran). By using a micro-syringe, a water droplet of 4 μl was pumped and deposited on the surface of a horizontally aligned mesh. A picture was taken after 5 s, and the contact angle was measured by the software. The measurements were repeated 5–10 times for each sample to calculate the average.
Determination of the drug release rate
To prepare the buffer solution, one PBS tablet was dissolved in 200 ml of distilled water to obtain the pH of 7.4. A square piece of ES mesh (2 × 2 cm2) was placed in 5 ml of the PBS solution at 37℃ to determine the release rate of CM. At different time intervals, aliquots were extracted for analysis and replaced with the same amount of the buffer solution. The aliquots were characterized by ultraviolet–vis spectroscopy (UV-mini 1240 Shimadzu spectrophotometer). The absorbance of CM in the buffer solution was measured at the characteristic peak of λmax = 421–423 nm. The released concentration of CM was calculated using the corresponding calibration curve.
Tensile properties evaluation
A tensile tester machine (Elima-Iran) was used for measuring the mechanical properties of the meshes. The meshes were cut into rectangular strips of 1 × 3 cm2 and fixed between the mechanical gripping units of the tester. The thickness of each sample was measured by a micrometer screw gauge. The test was carried out at an ambient temperature in three replications at the fixed testing speed of 1 cm/min [12].
Results and discussion
Feature of electrolytic solutions
To obtain beadless nanofibers with minimum fiber diameters, several essential parameters should be optimized simultaneously (e.g. the applied voltage, the polymer concentration, the viscosity, the tip to collector distance, etc.). The type of polymer, solvent, and the polymer concentration affect the viscosity and electrical conductivity of the resulting polymer solution. The effect of PCL:PLA blend ratio on the viscosity of the polymer solutions has been shown in Figure 3. By increasing the amount of PLA in the blend, the viscosity of the polymer solution increases. Considering that the molecular weight of PLA is higher than that of PCL, with an increase of PLA in the blend, the amount of the chain inclusions increases, resulting in an increase in the viscosity of the polymer solution. Similar behavior has been observed and reported for the mixture of these polymers before [21,22].
The viscosity of PCL: PLA polymer blend in chloroform 1: 4 DMF.
The electrical conductivity of the polymer solutions were all very low (<2 µs/cm) which can be explained by the nonionic nature of the polymers and CM.
Average fiber diameter, thickness, and porosity
Table 2 shows the average fiber diameter, thickness, and porosity of the ES meshes. The average fiber diameter of the ES meshes was obtained by analyzing the SEM micrographs (Figure 4). As can be seen from the figure, the ES meshes are all beadless with a normal fiber distribution. The average fiber diameter increased by the percentage of PLA in the blend composition while the pure PLA mesh did not follow this trend probably due to its lower initial polymer concentration. This trend is quite similar to that of polymer viscosity, and it seems that there is a direct relationship between the average fiber diameter and the viscosity of the polymer solution. Under the same electrospinning conditions, the polymer solution with higher viscosity exhibits greater resistance against the stretching in the electric field [22–25]. The average fiber diameter of the hybrid meshes was between 170 and 180 nm which is very close to the mean value of those of PCL (∼120 nm) and PLA (∼233 nm). This is not very surprising as the hybrid meshes are electrospun simultaneously from the individual PCL and PLA polymeric solutions.
SEM images of the ES nanofibrous meshes loaded with 5 wt% CM: (a) PCL, (b) PCL 3:1 PLA, (c) PCL 1:1 PLA, (d) PCL 1:3 PLA, (e) PLA, (f) HY1, and (g) HY2. The thickness and porosity of the electrospun nanofibrous meshes under the following ES conditions: V = 22 kV, TCD 14 cm, feed rate = 0.2 ml/h, duration = 40 h.

The PCL and PLA meshes had the highest and the lowest porosities, respectively. The porosity was decreased by the percentage of PLA in the blend composition. The thickness of the meshes was also related to their average fiber diameter during the fixed rate of polymer feed and the electrospinning duration. The meshes with larger fiber diameters were relatively thicker as can be seen in Table 2.
Water contact angle
The wetting and surface-free energy of the meshes can play an important role in the release of the drug from the matrix into the aqueous solution [24]. In addition to the nature of the polymer, the surface roughness of the mesh plays a significant role in wetting [25].
Figure 5 shows the average WCA on the ES nanofibrous meshes. All of the meshes can be considered as hydrophobic with the WCA above 100°. The WCA of the ES meshes increased by the percentage of PLA in the blend composition. However, HY1 and HY2 meshes had slightly different WCA which was a bit surprising. Probably, the presence of 5 wt% CM in PCL or PLA is affecting the surface-free energy and the surface roughness.
WCA of the ES meshes.
Release of CM from the ES nanofibrous meshes
The release of CM from the electrospun meshes was monitored for 220 h, and the profiles were presented in Figure 6. All of the ES meshes showed a two-stage (bimodal) drug release behavior [26,27]. The initial stage of CM release was relatively faster than the second stage, and 20–30% of the encapsulated CM released gradually over 10 h. The electrospun PCL showed the fastest and highest CM release (i.e. around 87% after 100 h). On the contrary, the electrospun PLA had the slowest and lowest release rate (i.e. around 42% after 100 h). By increasing the percentage of PCL in the blended ES mesh, the total CM release % increased. There are several parameters affecting the CM release in the buffer solution (e.g. fiber diameter, hydrophobicity, porosity, degradation profile of the polymer, etc.).
CM release profile of (a) PCL:PLA blends and (b) PCL, PLA, HY1, and HY2.
The results showed that the lower fiber diameter (i.e. the larger surface to volume ratio), higher porosity, and better wetting (i.e. lower WCA) are responsible for the higher rate of drug release from the PCL meshes [18]. On the other hand, the addition of PLA to the blend increased the average fiber diameter and the hydrophobicity of the meshes which reduced the drug release noticeably.
The nature of these two polymers also played an essential role in the release of CM from the ES meshes. Compared with PCL, PLA had a higher level of crystallinity, hydrophobicity, and slower degradation rate in the buffer solution at pH 7.4 [13,25]. Therefore, the drug release from the PLA meshes was noticeably slower.
The hybrid ES meshes (HY1 and HY2) showed the same bimodal drug release. The total amount of the drug released from HY1 (74%) was higher than that of HY2 (68%) as the CM had been encapsulated in PCL and PLA components of the hybrids HY1 and HY2, respectively (Table 2).
It is worth mentioning that the stability of CM in different solvents and solution media has been previously investigated. The degradation of CM in most of the solvents except water is less than 1% per day. The preparation of polymer solution and electrospinning took typically two days. One can expect 2–3% CM degradation during this process [28].
The ES meshes made from PCL or PLA have already been used for drug delivery in vivo [29–31]. The only concern is the remaining DMF or other solvents in the final mesh which can be washed by water and ethanol or keeping the mesh in the oven at 40–60℃ for several hours. However, the presence of less than 0.1 wt% DMF does not have any cytotoxicity effect [32].
Mechanical properties
The mechanical properties of the electrospun meshes were investigated, and the results are shown in Table 3 and Figure 7, respectively. The electrospun nanofibrous meshes should be robust enough in order to be used for any biomedical applications.
The stress–strain curves of the electrospun nanofibrous meshes. Tensile stress and tensile strain of the drug-loaded nanofibrous meshes.
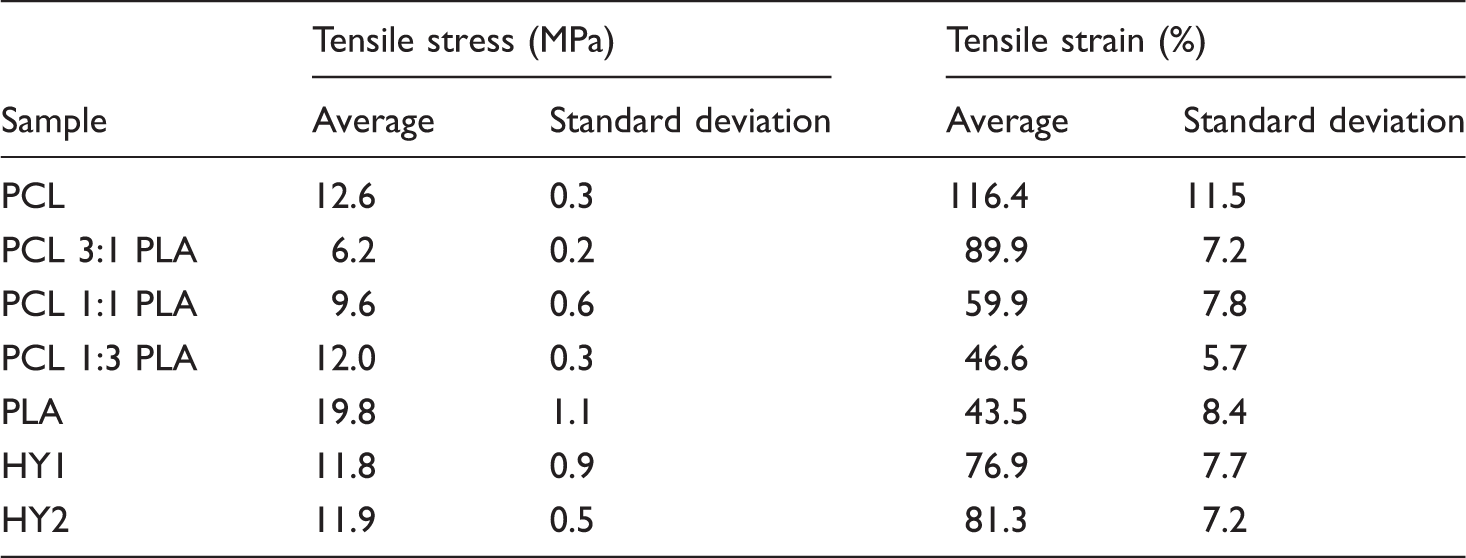
By increasing the percentage of PLA in the blended composition, the tensile strength of the meshes increased noticeably while their elongation at break decreased. The average fiber diameter in the meshes also reduced by increasing the percentage of PCL in the composition. This could be another reason for the lower tensile strength of the meshes containing PCL.
Table 3 indicated that the hybrid meshes (HY1 and HY2) had almost the same tensile strength which was not surprising as they had the same polymeric composition. PCL and PLA were fed separately from two different syringes for electrospinning on the same collector simultaneously. Therefore, the resulting nanofibers had the same properties as those of PCL and PLA. The slight difference in the fiber morphology and the interactions between the nanofibers due to the presence of CM was the reason for their slightly different mechanical properties. The behavior of stress–strain curves of the pure, blend, and hybrid nanofibrous samples was more or less similar (Figure 7).
Conclusion
The electrospun meshes made from PCL, PLA, and their blends were used for the encapsulation of CM. The CM release from the meshes in the buffer solution was investigated. The electrospun meshes were characterized by different techniques. The addition of PCL to PLA (i.e. polymer blending) was found to be an auspicious way for controlling and tuning the average fiber diameter, porosity, wetting (WCA), tensile strength, and the drug delivery. The CM release from the electrospun meshes was in the following order: PCL > PCL3:1PLA > PCL1:1PLA > PCL1:3PLA > PLA. This showed that the electrospun meshes with lower average fiber diameter (i.e. higher surface area to volume ratio), higher porosity, better wetting (i.e. lower WCA), and faster degradation rate in the buffer had higher drug release %. All of the ES meshes showed bimodal drug release profiles with 20–30% CM release in over 6 h. The tensile strain of the electrospun meshes was also in the same aforementioned order due to the presence of PLC with less crystallinity and lower fiber diameter. Thus, the polymer blends of PCL and PLA can be used successfully for the fabrication of nanofibrous meshes with drug release controllability.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
