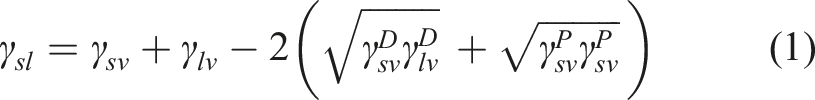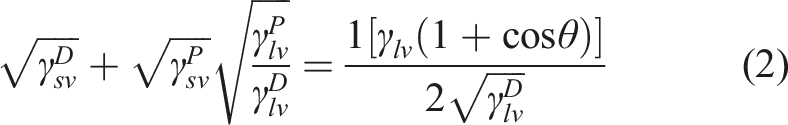Abstract
Segmented polyurethane (PU) block copolymers are widely used in implantable cardiovascular medical devices due to their good biocompatibility and excellent mechanical properties. More specifically, PU Biospan MS/0.4 was used in ventricular assist devices over the past decades. However, this product is being discontinued and it has become necessary to find an alternative PU biomaterial for application in cardiovascular devices. One important criterion for assessing cardiac biomaterials is blood compatibility. In this study, we characterized the surface properties of four medical-grade PU biomaterials: Biospan MS/0.4, BioSpan S, BioSpan 2F, and CarboSil 20 80A, including surface chemistry, topography, microphase separation structure and wettability, and then measured the blood plasma coagulation responses using bovine and human blood plasma. Results showed that BioSpan 2F contains high amounts of fluorine and has the lowest surface free energy while the other materials have surfaces with silicone present. An in vitro coagulation assay shows that these materials demonstrated improved blood coagulation responses compared to the polystyrene control and there were no significant differences in coagulation time among all PU biomaterials. The chromogenic assay showed all PU materials led to low FXII contact activation, and there were no significant differences in FXII contact activation, consistent with plasma coagulation responses.
Introduction
Cardiovascular diseases (CVDs) are the leading cause of mortality globally as well as a major contributor to disability. The number of CVD cases nearly doubled from 271 million in 1990 to ∼ 523 million in 2019 while the number of deaths has steadily increased from 12.1 million in 1990 to 18.6 million in 2019. Furthermore, cardiovascular diseases remain the leading cause of disease burden around the world.1,2
Cardiac implantable devices such as catheters, pacemakers, heart valves, stents, cardiac assist devices, and vascular grafts are modern devices that have been widely used in cardiac therapy to provide patients with numerous benefits including increased survival rate and enhanced quality of life. 3 There are two important factors that must be considered during selection of materials for cardiac implantable devices: (i) physical and mechanical features such as strength and deformation, fatigue, creep, friction and wear resistance, among many others, and (ii) biocompatibility properties with regard to material and tissue/blood interactions. 4 One of the leading causes of cardiac biomaterial failure is the initiation of coagulation and thrombosis by blood contacting devices, arising from the activation of the intrinsic pathway of the blood coagulation cascade when blood is exposed to the surface of biomaterials. The initiation of intrinsic pathway is thought to be through factor XII (FXII, or Hageman Factor) contact autoactivation with a procoagulant surface and generation of the proteolytic enzyme FXIIa, followed by a series of linked zymogen-enzyme conversions, eventually resulting in thrombin production, fibrin formation, and finally blood plasma clotting.5,6 It is important to develop and engineer hemocompatible biomaterials to prevent adverse effects when used for applications in cardiac implantable devices.
While many classes of materials have been used in cardiovascular medical devices, polyurethane (PU) elastomers combine excellent mechanical properties with good blood compatibility which favor their utilization and development as biomaterials in implantable cardiac devices. 4 PUs are a family of heterogenous copolymers containing the urethane linkage within the polymer chain. This functional group is capable of incorporating other functional groups into the polymer network which contributes to the range of properties from rigid hard thermosets to softer elastomers. 7 Well-known properties of PUs include durability, elasticity, elastomer-like character, fatigue resistance, and compliance. Another pivotal property is an acceptable response in the body after implantation. 8 Importantly, PU materials remain the only materials capable of withstanding the many million flexural cycles that are required for application in cardiac support devices for long term use, 9 therefore they are widely employed in the design and manufacture of cardiovascular devices. 10
Among the various medical-grade polyurethanes, poly(urethane urea)s (PUUs) were widely used in a variety of blood-contacting applications in biomedical devices because of good biocompatibility and flexural fatigue characteristics. For example, the Arrow International LionHeart and Abiomed’s AbioCor cardiac assist devices utilized PUU elastomers in the blood pump. 11 Our group at Penn State has investigated ventricular assist systems including implantable left ventricular assist devices 12 and total artificial hearts 13 for adults and a pediatric ventricular assist device for infants 14 for many decades. The PUU material Biospan MS/0.4 (former Polymer Technology Group, now DSM Biomedical Inc. Exton, PA) was used in parts of these devices such as blood sacs and catheters due its high durability and low degradation during long term performance. 15 However, the manufacturer has recently discontinued producing Biospan MS/0.4 and it has become necessary to identify and characterize other medical-grade polyurethanes for application in cardiovascular medical devices.
DSM biomedical commercially provides other medical-grade segmented polyurethanes (SPU) such as other Biospan® materials and Carbosil® as potential replacements for Biospan MS/0.4. Biospan® SPU materials provide excellent performance in many different types of medical devices and have shown the ability to withstand millions of flex cycles, while Carbosil® thermoplastic silicone polycarbonate polyurethanes are biocompatible and well suited to be used in many types of medical devices. In this study, we address important aspects of polyurethane hemocompatibility as we characterized the surface properties of polyurethane samples prepared from four SPU biomaterials: Biospan® MS/0.4, BioSpan® S, BioSpan® 2F, and CarboSil® 20 80A. To evaluate the potential applications of these biomaterials in cardiovascular devices, we measured the plasma coagulation responses of bovine and human blood in vitro and further studied human factor XII contact activation in contact with these polymer surfaces.
Materials and methods
Materials and reagents
Phosphate buffered saline (PBS, 0.01 M, pH 7.4, Sigma) was prepared using purified water (18 MΩ) from a Millipore Simplicity 185 system. Human factor FXII (FXII, Hageman Factor) and human Factor XIIa (FXIIa) were obtained commercially from Enzyme Research lab (South Bend, IN). Pefachrome® FXIIa/TH 5253 for chromogenic assay of FXIIa was purchased from Pentapharm (Switzerland). All proteins were thawed upon receipt and stored as aliquots at −80°C. Prior to use, proteins were thawed at room temperature and diluted with PBS. Formamide and diiodomethane were purchased from Sigma. Bovine blood was collected from healthy animals into CPDA bags containing anticoagulant citrate phosphate dextrose adenine solution under protocols approved by the Penn State College of Medicine (PRAMS201647205). Human blood was collected in Vacutainer® plastic EDTA tubes (Becton Dickinson, Franklin Lakes, NJ) from healthy volunteers under approved protocols in accordance with institutional policies.
Fabrication of polyurethane tubes for coagulation testing
Four medical-grade polyurethane biomaterials, Biospan MS/0.4, BioSpan S, BioSpan 2F, and CarboSil 20 80A, were kindly provided by DSM Biomedical Inc (Exton, PA). Polymers were either dissolved or diluted in dimethylacetamide (DMAC) to obtain a final concentration ∼ 18% (w/v) for fabrication of samples. Nine polyurethane tubes of each polymer were fabricated on 316 stainless steel (316SS) rods by dipping in polymer solution 5–7 times with a 2 h curing step at 40°C in air between coats. Final dimensions of the tubes were diameter of 7.5 mm, length of 11.4 mm and thickness of ∼0.3 mm. After the desired thickness was reached, the tubes were subject to a final curing step at 60°C overnight. PU tubes were removed from rods after soaking in deionized water. The tubes were then placed in a sealed jar with deionized water at 60°C/overnight to extract any residual DMAC.
Surface topography and chemistry characterization of polyurethane tubes
Polymer samples (diameter 10 mm) were cut from tubes and surface topography was assessed with a multimode atomic force microscope (AFM) equipped with a Nanoscope IIIa control system (Veeco, Santa Barbara, CA). AFM was operated in tapping mode in air using a sharp Si probe (NSG30-SS, TipsNano) to collect height and phase image data simultaneously. Each sample was measured at no less than 3 different random locations. AFM images were treated and analyzed by off-line AFM software (version 5.12r3, Veeco, Santa Barbra, CA).
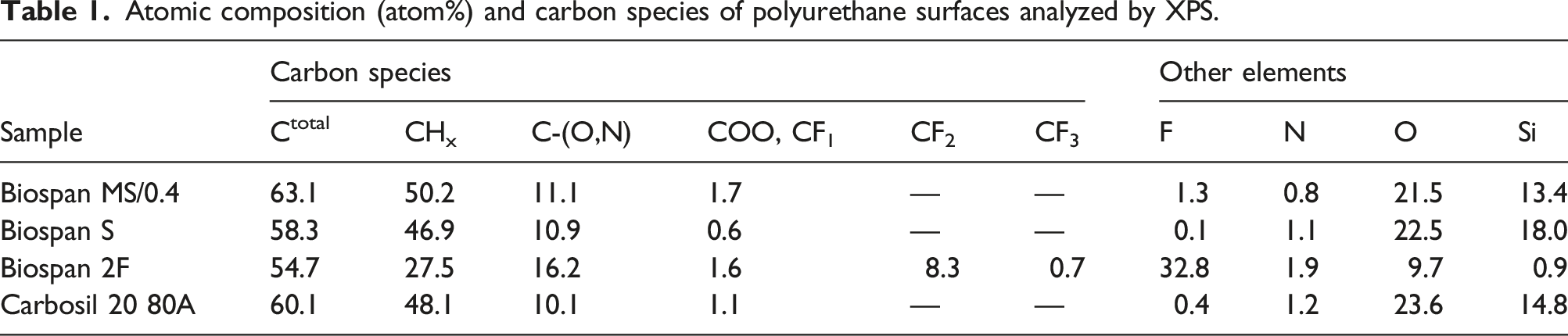
The surface chemical composition of polymer samples was analyzed by X-ray photoelectron spectroscopy (XPS) using a Physical Electronics VersaProbe III instrument equipped with a monochromatic Al kα x-ray source (hν = 1486.6 eV) and a concentric hemispherical analyzer. Charge neutralization was performed using both low energy electrons (<5 eV) and argon ions. The binding energy axis was calibrated using sputter cleaned Cu (Cu 2p3/2 = 932.62 eV, Cu 3p3/2 = 75.1 eV) and Au foils (Au 4f7/2 = 83.96 eV). Peaks were charge referenced to the CHx band in the carbon 1 s spectra at 284.8 eV. Measurements were made at a takeoff angle of 45° with respect to the sample surface plane. This resulted in a typical sampling depth of 3–6 nm (95% of the signal originated from this depth or shallower). Quantification was done using instrumental relative sensitivity factors (RSFs) that account for the x-ray cross section and inelastic mean free path of the electrons. The analysis size was ∼200 µm in diameter.
Surface wettability and surface energy of polyurethanes
The sessile drop measurement of the advancing water contact angle (θ) was used to assess the surface wettability of polyurethane samples using a ramé-hart contact angle goniometer (Succasunna, NJ). The contact angles were obtained by a minimum of six independent measurements at different locations of each sample using an ∼ 4 µL drop of purified Millipore water at each location. Data are presented as mean ± standard deviation.
To further characterize the surface wettability, the surface energy of polymers were also calculated through measurement of contact angles of formamide and diiodomethane on each polymer surface and calculation of the surface tension of each material using the OWRK (Owens, Wendt, Rabel and Kaelble) method.
16
In this method, the total surface energy of the solid surface is defined as the sum of polar and dispersive parts. Three liquids (water, formamide and diiodomethane) with their known dispersive and polar parts of their surface tensions are used to calculate the solid surface free energy, as demonstrated in the equation below:
Blood plasma coagulation assay
All freshly collected blood samples were immediately centrifuged at 460 g for 15 min and the supernatant was then centrifuged twice at 1500 g for 20 min to obtain platelet poor plasma (PPP). Plasma samples were aliquoted into 15 mL conical tubes and stored at −80°C prior to use. An in vitro plasma coagulation assay was performed in polyurethane tubes and also in polystyrene tubes following the procedures described in our previous publication. 17 Briefly, 500 µL plasma and 400 µL of PBS were added in polyurethane tubes (n = 3 of each material), then the sample was recalcified with 100 µL of 0.1 M CaCl2 and a timer was started. The polyurethane tubes were sealed with closure clamps (40/40 mm, Qosina, Ronkonkoma, NY) and rotated at 8 r/min on a hematology mixer, and the corresponding coagulation time (CT) was defined as time to visible clot formation. To ensure the surface contact area of plasma coagulation was the same as the control polystyrene tubes (2 mL, Greiner Bio-One, North Carolina, USA) for all polymer test samples, the working area of the tube was calculated and a closing line was pre-marked on the tube.
Similarly, polymer coagulation experiments were also carried out in polystyrene tubes containing PU material films. Briefly, round polyurethane film discs (n = 4) with diameter of 8 mm were cut from the tubes and added into a polystyrene tube (2 mL, Greiner Bio-One, North Carolina, USA) followed by the addition of 500 µL plasma, 400 µL of PBS and 100 µL CaCl2, and then tubes were capped with parafilm. The tubes were rotated at 8 r/min on a hematology mixer until the formation of clot, and coagulation time was recorded.
FXII contact activation assay and chromogenic assay
To evaluate the human FXII contact activation on each polyurethane surface, polymer films (n = 3 of each material) with diameter of 8 mm were incubated with FXII at physiological concentrations (30 μg/mL) in PBS (total volume 330 µL) in a polypropylene micro-centrifuge tube (1.5 mL, VWR) at room temperature and on a hematology mixer for 60 min. The contact activated solutions were then collected by centrifuge at 2000 r/min for 30 s and a chromogenic assay using Pefachrome was performed to measure the yield of activated FXII (FXIIa) following the procedures suggested by manufacturer. Briefly, 25 µl of FXII activated solution was added to a 96-well microplate containing 175 µL of 50 mM tris-immidazole (pH 7.9, 150 mM NaCl) followed by the addition of 25 µL of Pefachrome FXIIa (4 mM). The reaction was carried out at 37°C for 10 min and then stopped by adding 25 µL of 10% glacial acetic acid. Absorbance at 405 nm was recorded within 5 min to determine the initial color development using a UV/Vis spectrometer (Soft Max Pro UV/Vis spectrometer). A calibration curve was constructed by plotting absorbance for serially diluted [αFXIIa] solutions prepared in PBS with same protocol.
Statistical analyses
Statistical analyses were performed by One-Way ANOVA using VassarStats. The differences were considered statistically different for p < .05. Significant differences are denoted by one symbol (p < .05), two symbols (p < .01), or three symbols (p < .001).
Results and discussion
Surface chemistry and topography of polyurethane surfaces
Atomic composition (atom%) and carbon species of polyurethane surfaces analyzed by XPS.
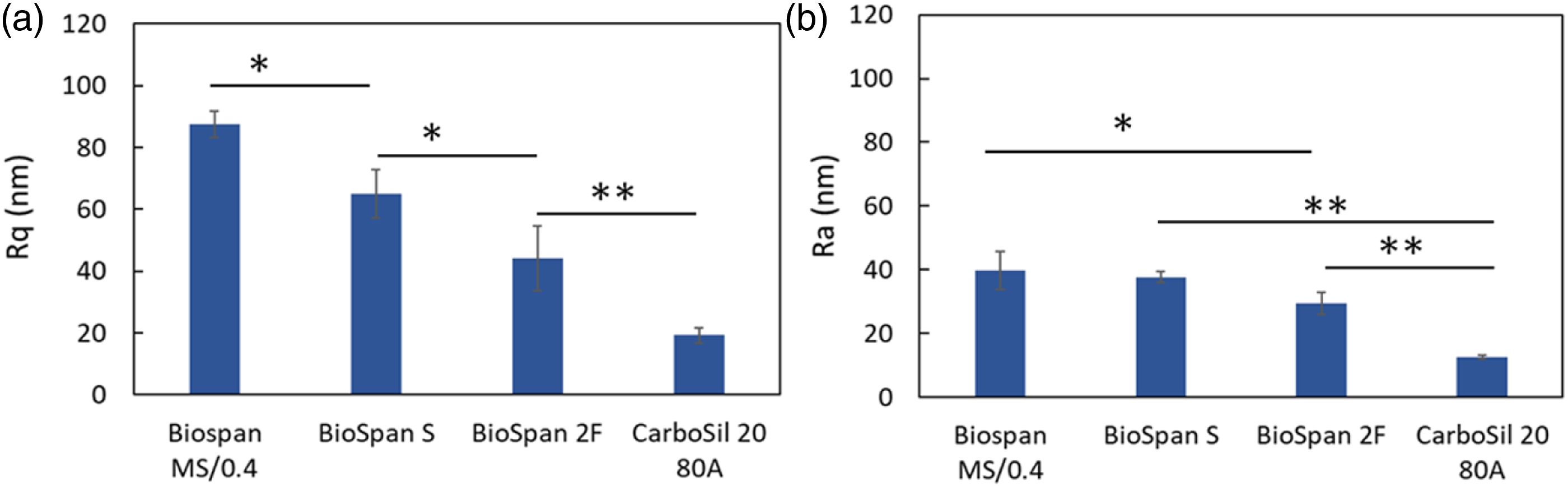
The surface topography of polyurethane tubes was characterized by AFM over a scan area of 50 µm × 50 µm and representative images are shown in Figure 1. Scratch lines were observed on all surfaces which arise from the 316SS rod used to prepare test samples. There were no specific features observed on polymer surfaces except for micro-holes observed on Biospan S surface (Figure 1). The diameters of holes on Biospan S surfaces varied in the range of 1.76–4.88 μm and the depth ranges from 360–700 nm, and which are likely due to the non-uniform evaporation of solvent during fabrication of the polymer tubes (Figure 1(b)). To further characterize the surface topography, the surface roughness of each material was calculated from the AFM height images, represented by Ra and Rq (Figure 2). Rq is the root mean square average of height deviations taken from the mean data plane. Ra is the arithmetic average of the absolute values of the surface height deviations measured from the mean plane. Results showed that Biospan MS/0.4 is rougher than other materials while Carbosil 20 80A is the smoothest among four materials. Representative AFM topography images of four PU tube surfaces (a) Biospan MS/0.4, (b) BioSpan S, (c) BioSpan 2F, and (d) CarboSil 20 80A. scale bar =10 µm, height color scale = 500 nm. Surface roughness (a) Rq and (b) Ra of each PU polymer surface (*: p < .05; **: p < .01).
Segmented polyurethanes are block copolymers containing hard and soft domains. The thermodynamic immiscibility of the polar hard segments and the relatively non-polar soft segments results in an important phase structure of polymers, termed microphase separation structure. The phase separation structure influences the mechanical properties of polyurethanes
18
and also affects the hemocompatibility of polymers in blood contacting applications.
19
Analysis of phase separation structure of the polyurethane biomaterials is important for understanding the blood coagulation responses to polyurethanes, and the microphase separation structure of polyurethane polymers can be visualized by AFM phase imaging mode.20,21 Figure 3 illustrates representative phase images of the four polyurethane biomaterials over a scan area of 1 µm × 1 µm in air. The contrast in AFM phase image arises from the phase lag between the drive oscillation and the actual probe oscillation during scanning, and high phase angle shifts (light color in phase images) generally represent the hard domains. All polymers show the phase separation structure in which the hard domains were dispersed in soft segment matrix, however, the phase separation degree and the domain features are significantly different. Small hard domains were found in Biospan MS/0.4 compared to other polymers, while CarboSil 20 80A demonstrated weak phase separation, with the hard and soft segments appearing to merge together as the images did not show distinct separation of the bright and dark regions. A significant high phase angle shift was observed on Biospan 2F (Figure 3(c)) indicative of a high degree of phase separation in polymers, probably due to the high content of fluorocarbon chemistry in polymer. AFM phase images show the microphase separation structure of four polyurethane polymers, (a) Biospan MS/0.4, (b) BioSpan S, (c) BioSpan 2F, and (d) CarboSil 20 80A. (Scale bar = 200 nm, phase color scale = 25
The wettability and surface free energy of polymers
The contact angles of different solvents and the calculated surface free energy of four PU polymers using OWRK model (n > 6 per polymer. **: p < .01 comparing to other material).
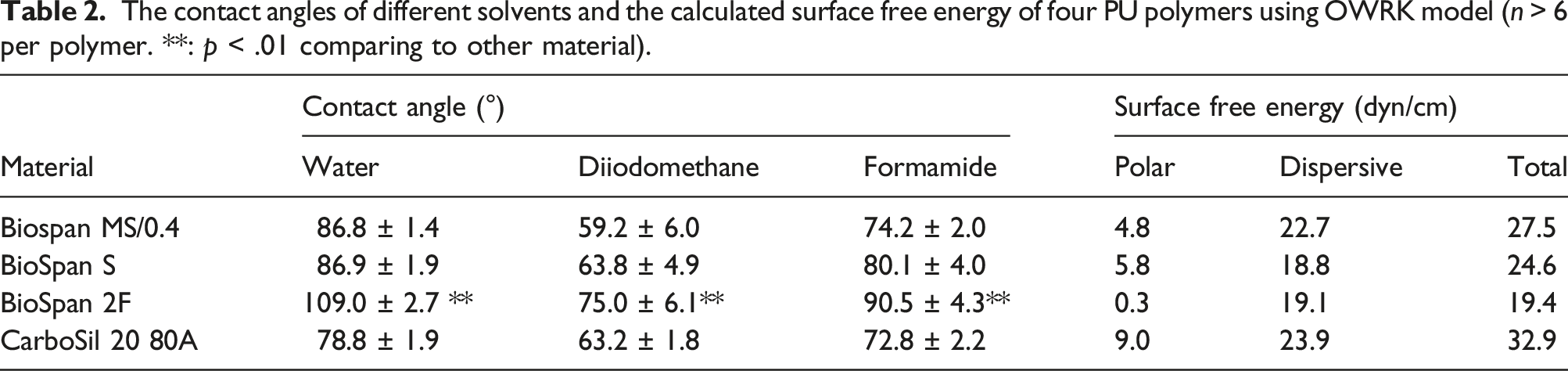
While water contact angle represents the ability of water to spread over a surface, the actual value of surface free energy of a material surface must be measured. Owens, Wendt, Rabel and Kaelble developed a two-component model (OWRK model) and defined the surface energy of a solid as being comprised of two components - a dispersive component and a polar component. 22 To better characterize the wettability of these polyurethane surfaces, we estimated the total surface free energy of surfaces through measuring the contact angles of three liquids (water, diiodomethane, and formamide) with known dispersive and polar parts of surface tensions, and results are shown in Table 2. Similar as water contact angles, Biospan 2F demonstrated significantly higher contact angles of diiodomethane and formamide than other materials. The calculated polar parts of the surface free energy are significantly different across the materials while the dispersive parts of surface energy vary over a small range. The polar component of surface energy of Biospan 2F shows significantly lower values than the others, indicating that high content of fluorine in Biospan 2F significantly reduced polar interactions between the molecular interaction between liquid and materials, and the surface energy of Biospan 2F is largely attributed to dispersive interactions. As a result, the total of surface energy of Biospn 2F is only 19.4 dyn/cm, lower than other PUs.
Blood plasma coagulation and FXII contact activation on polyurethane biomaterial surfaces
Blood plasma coagulation responses to polyurethane biomaterials were performed with bovine and human plasma, and directly assessed in polyurethane tubes which simulate the reactions in implantable devices. In this experiment, the surface contact area of the experimental tubes was controlled to be same as the interior surface area of polystyrene control tubes. Results showed that bovine plasma clotted after contact with polyurethane tubes in the range of 46–49 min, significantly slower than when in contact with control (32.6 ± 0.7 min, p < .01, Figure 4(a)), however, there is no significant difference observed in plasma coagulation time among the different PUs being tested. Human plasma shows a similar coagulation responses as bovine plasma. Coagulation times in contact with polyurethane tubes were significantly longer than that for polystyrene control (p < .01, Figure 4(b)), suggesting all of polyurethane biomaterials have improved blood coagulation responses compared to polystyrene. However, no differences in coagulation time were seen among the polyurethane materials. This is somewhat surprising given that we expected BioSpan 2F would have an improved coagulation responses compared to other three materials since Biospan 2F contains high amount of fluorine and is more hydrophobic, and therefore it is expected to adsorb more proteins such as albumin compared to other less hydrophobic surfaces.
23
Coagulation time of (a) bovine and (b) human plasma in contact with polyurethane tubes (same surface areas as polystyrene tube, n = 3 each, **: p < .01).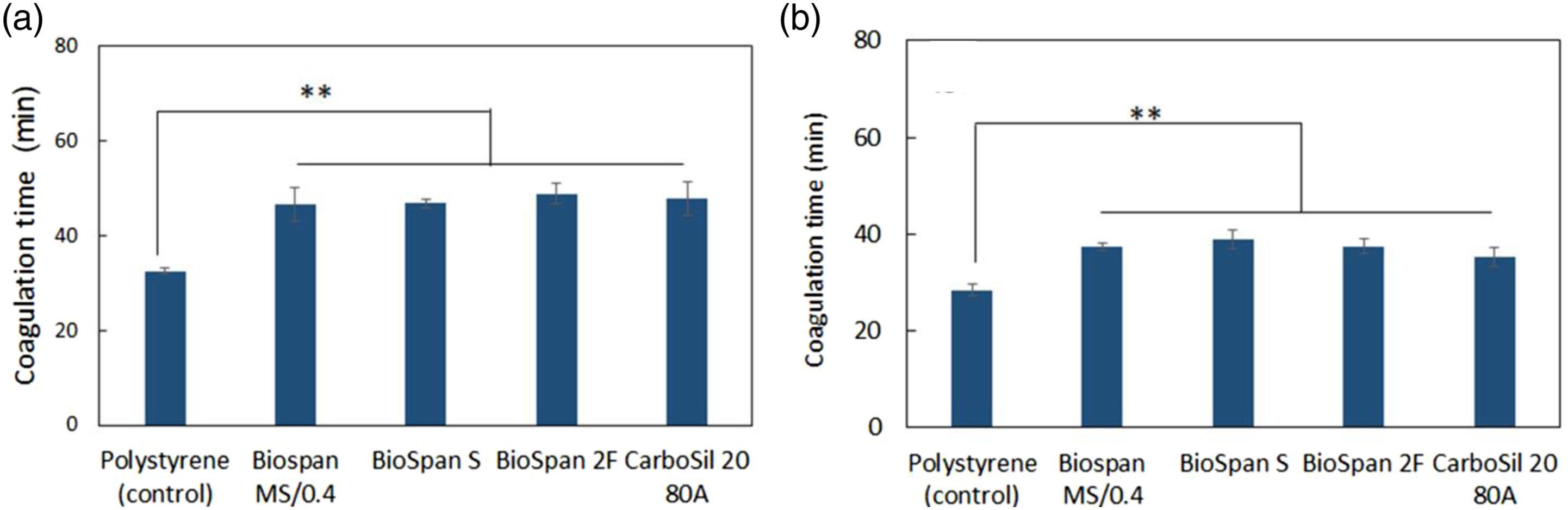
The plasma coagulation time assay was verified by assessing the reaction of polyurethane films and plasma. We prepared polyurethane film discs (diameter of 8 mm) from polyurethane tubes and incubated with plasma in a polystyrene tube. Polystyrene tubes without polymer films served as control. As expected, polyurethane film discs demonstrated a faster coagulation time than control since plasma contacted both the polyurethane material and the polystyrene itself, yielding a shorter coagulation time compared to polystyrene tubes alone (Figure 5). Similar to prior experiments, bovine and human plasma show no significant difference observed among the different polyurethane materials, consistent with the results of coagulation in polyurethane tubes. Coagulation time of (a) bovine and (b) human plasma incubated with polyurethane discs (8 mm) in polystyrene test tubes (n = 4, **: p < .01).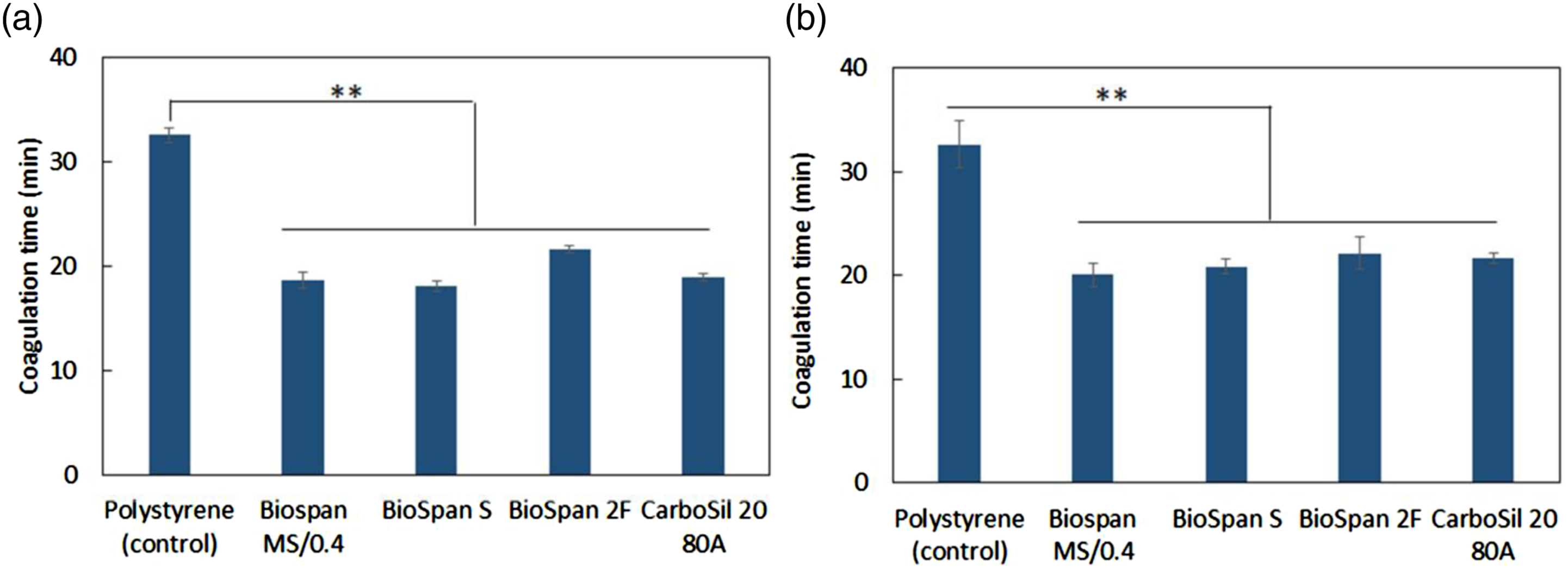
Plasma coagulation in contact with biomaterial surface is initiated by intrinsic contact activation pathway, in which FXII interacts with a procoagulant surface and converts into the active enzyme form FXIIa through autoactivation due to conformational structure changes.
24
The analysis of FXIIa yield after FXII contact with material surfaces is important for understanding the plasma coagulation responses in the above experiments. Using purified human FXII as a model, we measured the human FXIIa yield after FXII contact activation with different materials with a chromogenic assay. Results showed that increased levels of FXIIa were measured in the solutions in contact with polyurethane film discs than that in control because, once again, FXII was only activated by the polypropylene micro-centrifuge tubes in the controls. However, there was no significant difference observed in the amount of FXIIa produced by Biospan MS/0.4, S, 2F, and CarboSil 20 80A (Figure 6(a)), suggesting that all types of polyurethanes have a similar propensity to activate FXII, which would be expected to result in similar coagulation, consistent with the results observed in coagulation time studies (Figures 4 and 5). It should be noted that total FXIIa yield in solutions containing PU discs consists of FXIIa produced from the micro-centrifuge tube and the PU discs. The net yield of FXIIa from PU discs was approximately 0.05 ng/mm2 (∼5 ng in total) when the activation by control is subtracted (Figure 6(b)). This represents only ∼ 0.05% of FXII being activated when compared to the original FXII amount (∼10 µg) in solution. Results suggest that these four medical-grade polyurethane biomaterials have similar blood plasma contact activation and coagulation responses, and the total FXII contact activation in plasma was low. (a) Total yield of activated human FXII (FXIIa) and (b) net yield of FXIIa per surface area from the contact activation of the zymogen FXII with four polyurethane polymers. Control = polypropylene micro-centrifuge without PU polymer (n = 9, NS = no significant).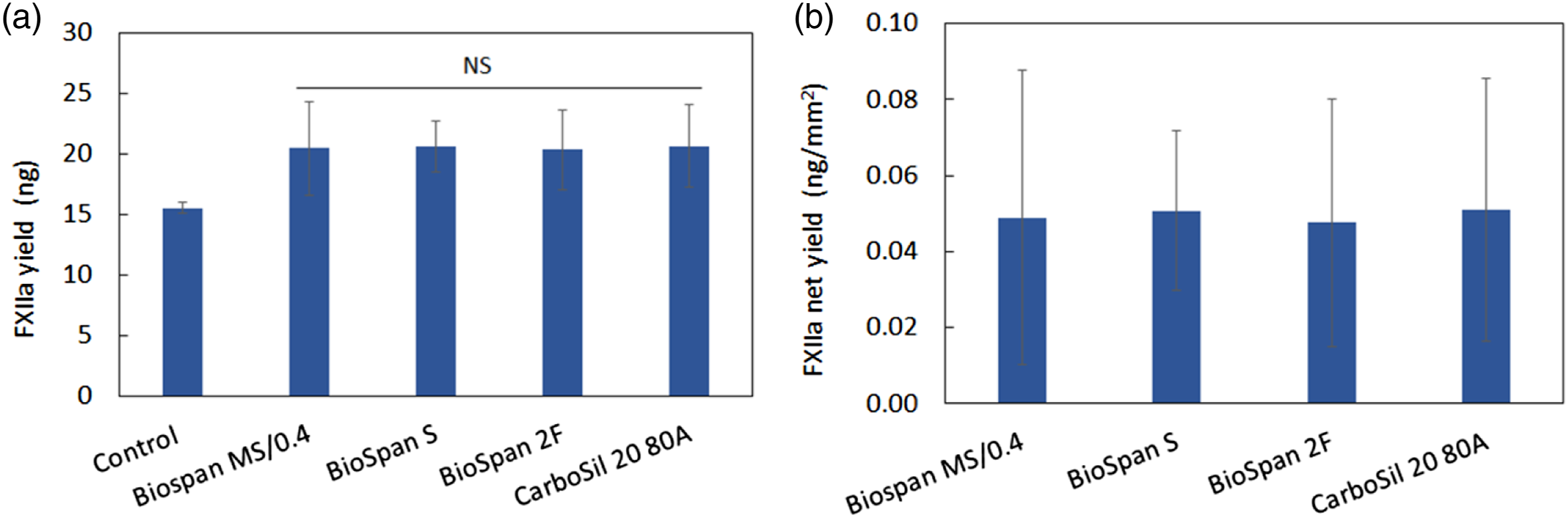
Segmented polyurethane block copolymers are widely used in a variety of blood-contacting medical devices. Understanding the blood coagulation response to material surfaces is essential for the successful application of materials in medical devices. In this study, we only addressed the plasma coagulation responses to four medical-grade polyurethane biomaterials and this was done in an in vitro environment as a preparatory step towards using these materials in vivo. However, it’s important to note that in vivo blood coagulation in the presence of biomaterials is highly complex and the exact role of FXII autoactivation remains unclear. In a model different from the traditional blood coagulation cascade, Hoffman proposed a cell-based model of hemostasis and described that the coagulation takes place on different cell surfaces in three overlapping steps: initiation, amplification, and propagation. 25 This cell mediated coagulation model presents a more complex understanding of how hemostasis works in vivo. Gorbet and Sefton 26 proposed in a 2004 paper that contact phase activation of proteins might not be important in the activation of coagulation by biomaterials. Rather, they proposed that monocytes are activated to produce tissue factor in the presence of biomaterials, and that this tissue factor leads to activation of the extrinsic pathway of coagulation. They further expanded on this thought and proposed that both the intrinsic and extrinsic pathways might both be involved, with the relative contributions of each dependent on the local flow environment and the time frame of coagulation. In 2019, these same authors contributed to a review article that noted that “overall, evidence suggests that FXII activation by biomaterial contact activates the intrinsic coagulation cascade, leading to formation of a clot”, with an important qualifier that other pathways may augment or replace the contact activation step during biomaterial contact with blood. 27 More recently, there has been effort to develop FXIIa inhibitors including antibodies directed against FXIIa as a means to control clotting in biomaterials systems. 28 These molecules reduce fibrin deposition in extracorporeal membrane oxygenators (ECMO systems) but appear to have no effect on bleeding in animal models. The complex process of FXII activation, including for both coagulation and the inflammatory pathways through leukocyte activation, demonstrate that there is still much that is not understood of this mechanism. 29
In addition to the above noted limits of in vitro testing, there are other important questions of blood responses that are not addressed in this study. For example, platelet adhesion and activation, and plasma protein adsorption are other important topics in blood contact. Furthermore, it is also important to investigate more about the benefits of fluorinated and silicone-based polyurethanes and compare the two to identify which type of modification demonstrates optimal stability as well as the best capability of preventing blood clotting and thrombosis. In addition to blood coagulation and thrombosis, significant issues arise from the activation of other factors in response to materials, while microbial infections due to the biofilm formation on the surface of biomaterials are also problematic. Therefore, future studies will be performed to further understand the biological responses to PU polymers such as protein adsorption, platelet adhesion and activation as well as pathogenic bacterial adhesion responses. Finally, when in vitro testing and initial screening of these materials is complete, in vivo animal testing will be required to verify that results translate into this more challenging environment. Overall assessment of polymers will help contribute to the selection and application in blood contacting medical devices.
Conclusions
In this study, we characterized medical-grade polyurethane biomaterials: Biospan S, Biospan 2F, and CarboSil 20 80A for evaluation as potential candidates for replacement of discontinued Biospan MS/0.4 for circulatory support devices, vascular grafts and heart valves. Biospan MS/0.4, Biospan S and Carbosil 20 80A are all silicone modified polyurethanes while Biospan 2F is a fluorinated polyurethane with the lowest surface free energy. In vitro plasma coagulation assays with bovine and human blood demonstrate that all four of polyurethane biomaterials have improved blood coagulation responses compared to the polystyrene, however, there were no significant differences in plasma coagulation and the yield of FXIIa after contact activation with each polyurethane polymer. Biospan S, 2F and Carbosil 20 80A demonstrated low FXII contact activation and low coagulation responses similar to Biospan MS/0.4.
Footnotes
Acknowledgements
The authors would like to thank the assistance of MCL from Penn State University Park for the surface study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We also acknowledge the support from Penn State College of Medicine Summer Undergraduate Research Internship Program, NIH grant R01 HL153231-02 and NIH grant R01HL151736-01.