Abstract
The functional fabric with high electromagnetic shielding (EMI) performance, durability as well as good handle has been drawing increasing attention. Herein, AgNPs@MXene of high conductivity was prepared by in-situ reduction of AgNO3 onto MXene. Then, boron-enhanced AgNPs@MXene-PDA coating fabric (BAMP-F) of high properties was fabricated through polydopamine (PDA) coating, AgNPs@MXene assembly, and finally sodium tetraborate (Na2B4O7) densification. Results showed that the surface resistivity of AgNPs@MXene arrived at 7.22 × 10−4 Ω·m and decreased by one order in comparison with pure MXene. After 0.6 mol/L PDA coating, AgNPs@MXene deposition for 6 times, and 0.02 mol/L Na2B4O7 densification, the surface resistivity of BAMP-F reached 7.08 × 10−3 Ω·m and its EMI values of one layer attained 34.51 dB. Compared with AgNPs@MXene-PDA coating fabric (AMP-F), the electrical conductivity of BAMP-F improved by 47.6 % and its electromagnetic shielding performance increased by 42.8 %. BAMP-F possessed good handle and maintained excellent EMI performance after bending for 600 times, washing for 60 min, and tearing.
Introduction
Electromagnetic radiation is considered as the fouth environmental pollution following air, water, and sound pollution, which causes serious harm to human being’s health. Electromagnetic shielding fabrics have been attracting much attention due to the flexibility and the protection of people from electromagnetic waves. 1 The primary focus on electromagnetic shielding fabric lies in how to impart excellent conductivity and durability. The construction of electromagnetic shielding fabric with high performances has become a hot topic.
Electromagnetic shielding fabrics primarily rely on the reflection loss to electromagnetic waves. Their conductivity has the positive correlation with the electromagnetic shielding effectiveness. 2 So far, most of conductive fabrics have been fabricated by the load of conductive materials at one-, two- or three-dimensional states. Based on percolation, tunneling and field-emission effects, the conductive path was formed onto conductive fabric.3–5 Conductive materials are mainly divided into four categories: metal material, 6 carbon-based material, 7 conductive polymer, 8 and composites. 9 Though the metal conductive materials (Au, Ag, Cu) possess high conductivity, their adhesion fastness onto fabrics are poor. While carbon-based materials have the problems of dispersion stability and conductive polymers exhibit the lower electrical conductivity. Consequently, the composite of different conductive materials could achieve the best performances.
MXene (Mn+1XnTx) was a two-dimensinal transition metal carbonitride, where n was 1–3; molar denotes transition metal; X denotes C or N; T means fuctional groups −OH, −O, −F. 10 MXene shows the theoretical conductivity of 105 S/m. Due to the narrow band-gap semiconductor characteristics, it possesses excellent electromagnetic shielding and photothermal convension functions.11,12 In addition, the existence of fuctional groups −OH, −O, −F imparts certain hydrophilcity and bonding with other materials via hygrogen bond. Compared with graphene, MXene has not only covalent bond, but also metal bond and ionic bond, which provides the advantages for composite with other materials. The insertion of metal ions can realize the high conductivity of MXene. 13 Except Li+, other ions (Ag+, Mg2+, Na+, NH4+) can insert between MXene sheets. 14 The strong reducibility of low valent titanium induces the in-situ growth of nano-silver (AgNPs) onto MXene.15,16 It not only improves the aggregation of AgNPs, but also promotes the electron transimission by means of Ag+ insertion. The electromagnetic shielding efficiency presents positive correlations with the materials’ conductivity. At present, the conductivity of MXene has been influenced by the interspace among MXene sheets. 17 In order to further improve the conductive properties of materials, some researchers induced the densification method through bridging bonds.18,19 Sodium tetraborate (Na2B4O7) can be used for densification of MXene film by the bonding between borate ions and −OH of MXene. 20 The MXene lamella touched closer to form a more compact conductive network.
Electromagnetic shielding fabrics on the human body are vulnerable to chemical and mechanical attacks in practice, such as sweat, water, bending, and severe cycle washing.21,22 These greatly hinder their application in daily use. Electromagnetic shielding fabrics should have flexibility and durability under strong external forces such as bending, tearing, washing, rubbing, and so on. There are always lack of firm adhesion between conductive materials and fabric. Under the action of external forces, conductive materials are easily detached from the fabric surface, which leads to the decrease of conductivity. Moreover, owing to the rough and porous texture of fabrics, it is difficult for conductive material to contact closely and form complete conductive network. Several researches have been carried out to improve the conductivity and fastness of conductive fabrics, including the modification of fabric surface, 23 complex of conductive particle and conductive polymer, 24 and in-situ deposition of conductive materials onto fabric. 25 Among these methods, modification of fabrics may be the most effective route. 26 Polydopamine (PDA) possesses ultra-strong adhesiveness due to the rich hydroxyl and amino groups of its structure. 27 Pyrocatechol structure can coordinate with metal ions. Accordingly, it is the best candidate to modify the surface of fabric and further bond the conductive materials.
Herein, AgNPs@MXene was prepared by in-situ reduction of Ag+ onto MXene. The conductivity of AgNPs@MXene were optimized. AgNPs@MXene was characterized by XRD, SEM-EDS, TEM, and XPS. Then, boron-enhanced AgNPs@MXene-PDA coating fabric (BAMP-F) was fabricated through PDA coating, AgNPs@MXene assembly, and finally Na2B4O7 densification. Effects of dopimane concentration, impregnation times of AgNPs@MXene, and Na2B4O7 concentration on the conductivity of conductive fabric were investigated. The electromagnetic shielding performance, durability and handle of the conductive fabric were evaluated for the potential application.
Materials and methods
Materials
Titanium carbide (Ti3AlC2, 200 mesh) was purchased from Bowarth Nanotechnology Co., Ltd Lithium fluoride (LiF), dopamine hydrochloride (DA), tris(hydroxymethyl)aminomethane, and sodium tetraborate (Na2B4O7) were supplied by Shanghai Macklin Biochemical Technology Co., Ltd Hydrochloric acid (HCl, 37%) was provided by Huadong Medicine Co., Ltd Silver nitrate (AgNO3) was provided by Changzhou Guoyu Environmental Protection Technology Co., Ltd. All chemical reagents were of analytical grade and used without further purification.
Cotton fabric (yarn count: 40 × 40, warp density: 133 threads/10 cm, weft density: 72 threads/10 cm) which went through scouring, mercerization, and bleaching, was obtained from Shanghai Hualun Printing and Dyeing Co., Ltd.
Preparation of AgNPs@MXene
25 mL HCl was poured into a PTFE beaker. Then 1 g LiF was slowly added into HCl at 45°C under stirring. After 5 min, 1 g Ti3AlC2 powder was put in the above solution. After reaction for 36 h, the solution was centrifuged at 3500 r/min for 5 min, washed repeatedly until pH surpassed 6, treated by ultrasound for 40 min, and freeze-dried to obtain MXene powder. Subsequently, MXene podwer was uniformly dispersed in the distilled water. AgNO3 solution was dripped into MXene dispersion. After stirring for 30 min, AgNPs@MXene dispersion was obtained. The preparation route of AgNPs@MXene is shown in Figure 1a. Schematic diagram of preparation for (a) multilayer MXene, (b) few-layer MXene, (c) AMP-F, and (d) BAMP-F.
PDA coating on cotton fabric
Tris(hydroxymethyl)aminomethane and HCl adjusted pH value of the solution to 8.5. Then certain amount of dobutamine hydrochloride were added into the solution. Cotton fabrics were soaked into DA solution and stirred at room temperature for 6 h. Finally, PDA coating cotton fabric was rinsed several times to remove the residual PDA, and dried at 60 °C. PDA coating on cotton fabric was shown in Figure 1b.
Fabrication of conductive cotton fabric
First, the PDA coating cotton fabric were immersed into 19 g/L AgNPs@MXene dispersion liquid for 30 min, and dried at 60°C. The above step was repeated for several times to obtain AgNPs@MXene-PDA coating fabric (AMP-F). Second, AMP-F was impregnated into Na2B4O7 solution at different concentrations for 6 h to make the borate ions fully enter into the fabric. Boron-enhanced AgNPs@MXene-PDA coating fabric (BAMP-F) was gained after drying at 60 °C. The fabrication of AMP-F and BAMP-F was shown in Figures 1(c) and (d). The cotton fabric which was directly treated by AgNPs@MXene dispersion liquid was marked as AM-F.
Characterizations
The morphology of MXene, AgNPs@MXene, and cotton fabric were observed by Field Emission Scanning Electron Microscope (FE-SEM) (Altra 55, Carl Zeiss, German) and analyzed by Transmission Electron Microscope (TEM) (JM-2100, JESCO, Japan). X-Ray Diffraction Spectometry (XRD) (Arl Xtra, Thermo ARL, Switzerland) was used to characterize the Cu Kα radiation (λ = 1.5406 A) at scanning speed of 5 °/min under 40 kV and 200 mA. The energy spectra was determined by X-ray Photoelectron Spectrometer (XPS) (K-Alpha, Thermo Fisher Scientific, USA) equipped with an Al-Ka X-ray source. The functional groups of modified fabric were characterized by Fourier Transform Infrared Spectrometer (Nicolet iS-20, Thermo Fisher, USA). Surface resistivity of the sample was measured using Four-probe Resistance (RTS-9, China). EMI shielding effectiveness (EMI SE) of the circular sample with a diameter of 10 cm was measured via Fabric Electromagnetic Radiation Tester (FY800, China). The washing resistance of the conductive fabric was tested for 10, 20, 30, 40, 50, 60 min by ultrasonic cleaning machine at 240 W (JP-040s, China). The surface resistivity and EMI SE of the conductive fabric was determined after bending for 50, 100, 200, 300, 400, 500, 600 times according to the literature. 22 According to AATCC Test Method 202, the hand parameters of the sample were measured through a fabric sensory instrument (PhabrOmeter, Nucybertek, USA).
Results and discussion
AgNPs@MXene with high conductivity
Micro-structure analysis of AgNPs@MXene
MXene was prepared by selective etching and assisted ultrasonic peeling. Then, silver ion was in-situ reduced on MXene in order to further improve the conductivity of MXene. The micro-structure of AgNPs@MXene composite are shown in Figure 2. Compared with Ti3AlC2, (104) diffraction peak of MXene almost disappeared and (002) diffraction peak moved towards left (Figure 2(a)). It indicated the removal of Al layers into Ti3AlC2 and the increased interlayer spacing between sheets. From XRD spectra of AgNPs@MXene, four new peaks appeared, corresponding to (111), (200), (220) and (311) crystal plane of AgNPs with the face-centred cubic structure. SEM-EDS exhibited the abundant AgNPs on MXene sheets (Figures 2(b) and (c)). The pure MXene sheets possessed the limited conductivity due to the inadequate contact with each other. AgNPs/Ag+ could enter into the gap among MXene sheets to enhance the electronic transmission. Consequently, AgNPs@MXene displayed better conductivity than MXene alone. TEM images also demonstrated the successful composite of AgNPs@MXene (Figures 2(d) and (e)). Else, AgNPs had good monodispersion onto MXene. Their size was concentrated on around 50 nm and lattice fringe spacing was 0.24 nm. (a) XRD of Ti3AlC2, MXene, and AgNPs@MXene; (b) EDS of AgNPs@MXene; (c) SEM images of Ti3AlC2 and AgNPs@MXene; (d) and (e) TEM images of AgNPs@MXene and AgNPs.
Conductivity of AgNPs@MXene
The conductivity of AgNPs@MXene at different mass ratios of AgNPs and MXene are displayed in Figure 3a. When the mass ratio of AgNPs and MXene was 3: 10, the sheet resistivity of AgNPs@MXene arrived at the minimum value of 7.22 × 10−4 Ω·m. Compared with the surface resistivity of pure MXene 5.02 × 10−3 Ω·m, the sheet resistivity of AgNPs@MXene decreased by an order of magnitude, which manifested the greater improvement of conductivity. It may be attributed to the filling of AgNPs/Ag+ into the interspaces among MXene sheets for the complete conductive path. With the mass ratio of AgNPs and MXene increasing, the sheet resistivity of AgNPs@MXene had the sharp increases. It was attributed to the obvious agglomeration of AgNPs, leading to larger size and uneven distribution of AgNPs. It would affect the improvement of conductivity of AgNPs@MXene. (a) Surface resistivity of AgNPs@MXene at different mass ratios of AgNPs and MXene; (b) XPS survey spectra for AgNPs@MXene, MXene and Ti3AlC2; (c) Ti 2p spectra for AgNPs@MXene and MXene, (d) Ag 3days spectra for AgNPs@MXene.
To elucidate the possible self-reduction mechanism, the surface composition and chemical states of AgNPs@MXene, MXene, and Ti3AlC2 were displayed in Figure 3(b)–(d). Compared with Ti3AlC2, F1s and Ag 3D signals were clearly observed in AgNPs@MXene (Figure 3(b)). Seen from Figures 3(c) and (a) distinct peak of MXene at 464.1 eV was classified as Ti(IV) 2p3/2. From the wide peak of 453–462 eV in MXene, Ti(II) (∼457.4 eV) and Ti(III) (∼461.6 eV) appeared. 13 The self-reduction process onto AgNPs@MXene made the transformation of Ti (II) and Ti(III) into Ti(IV). It was observed that the peaks of Ti(IV) at ∼460 eV and ∼465 eV. 28 At the same time, the representative peaks of Ag 3d5/2 and Ag 3d3/2 (Figure 3(d)) were located at ∼ 367.6 and ∼ 373.7 eV, respectively, and cleaved at ∼ 6.0 eV, 29 which indicated the successful formation of Ag(0). Almost all the silver particles are in the Ag(0) state, and the peak characteristics of Ag+ and Ag2+ were not obvious, suggesting the Ag(0) state. It further proved that the low Ti(II) and Ti(III) species had strong reduction activity.
Conductive fabric based on AgNPs@MXene
In order to obtain conductive fabric of high conductivity and fastness, the adhesive layer and conductive layer was incorporated onto the surface of cotton fabric. Conductive fabric was first coated by PDA. Then AgNPs@MXene was assembled onto the surface of PDA modified cotton fabric by Van Der Waals force, hydrogen bond and metal chelation. Finally, AgNPs@MXene-PDA coating fabric was treated by Na2B4O7 to further improve its conductivity.
Effects of dopamine concentration on the conductivity of AMP-F
The structure of PDA has rich amino and hydroxyl groups. Therefore, it shows the strong adhesion ability and can make the firm binding between conductive material and fabric. In addition, the functional groups of PDA coordinated with silver ion of AgNPs, which enhanced the binding of AgNPs@MXene. The concentrations of dopamine influenced the conductivity of AgNPs@MXene-PDA coating fabric (AMP-F).
Figure 4a exhibits the surface resistivity of AMP-F at different dopamine concentrations for one impregnation of AgNPs@MXene. It was found that the surface resistivity of AMP-F decreased, reached the lowest value of 8.96 × 10−2 Ω·m at 0.6 mol/L dopamine, and then increased. Compared with the surface resistivity of AgNPs@MXene treated fabric (AM-F) 50.4 × 10−2 Ω·m, the surface resistivity of AMP-F decreased by 82.2 %. It demonstrated that PDA coating contributed to the improvement of conductivity for the conductive fabric based on AgNPs@MXene. It might be attributed to the roughness chang of the fabric’s surface, which was confirmed in SEM images of the fabrics. Seen from Figure 4(c), the surface of cotton fabric after PDA coating became smoother. It was also observed from Figures 4(d) and (e) that AMP-F possessed smoother surface than AM-F. The smoothness of the substrate contributed to the completeness of conductive path formation for conductive materials.
30
Accordingly, AgNPs@MXene would form good conductive path on the surface of PDA coating fabric. (a) Surface resistivity of AMP-F at different dopamine concentrations for one impregnation of AgNPs@MXene; SEM images of (b) cotton fabric, (c) PDA coating fabric, (d) AM-F, and (e) AMP-F.
Effects of AgNPs@MXene impregnation times on the conductivity of AMP-F
The amount of conductive materials onto the fabric influence the conductivity. Surface resistivity of AMP-F and AM-F after multiple impregnation of AgNPs@MXene are presented in Figure 5. It demonstrated that the surface resistivity of AMP-F decreased with impregnation times increasing and reached the lowest value of 1.35 × 10−2 Ω·m after six impregnation times. It existed the firm binding between AgNPs@MXene and PDA coating fabric due to hydrogen bonds of functional groups. More AgNPs@MXene onto PDA coating fabric contributed to the construction of the integral conductive path. Besides, compared with AM-F, the surface resistivity of AMP-F had sharp decreases regardless of impregnation times. The surface resistivity of AMP-F decreased by 65.5 % after six impregnation times in comparison with AM-F. It also verified that PDA coating was beneficial for the formation of complete conductive path for AgNPs@MXene onto fabric. Surface resistivity of AMP-F and AM-F under different AgNPs@MXene impregnation times.
Effect of sodium tetraborate concentration on the conductivity of BAMP-F
In order to further improve the conductivity of AMP-F, Na2B4O7 was employed. Figure 6a gives the surface resistivity of boron-enhanced AgNPs@MXene-PDA coating fabric (BAMP-F) after the treatment of Na2B4O7 at different concentrations. It arrived at the lowest value of 7.08 × 10−3 Ω·m at 0.02 mol/L Na2B4O7. Compared with AMP-F and AM-F, the surface resistivity of BAMP-F decreased by 47.6 % and 81.9 %, respectively. SEM images of the cross section of conductive layer onto BAMP-F were displayed in Figures 6(b) and (c). There were pores among MXene sheets and the stack was not dense (Figure 6(b)). After Na2B4O7 treatment, the conductive layer had fewer voids and a denser structure (Figure 6(c)). Certain amount of borate ion reacted with –OH of MXene surface, which made the MXene lamella closer. The denser conductive network was formed. (a) Surface resistivity of BAMP-F treated by Na2B4O7 at different concentrations; SEM images of the cross section of conductive layer onto BAMP-F: (b) without Na2B4O7 treatment and (c) after 0.02 mol/L Na2B4O7 treatment.
XRD diagram in Figure 7a showed that the peak (002) of MXene existed before Na2B4O7 densification, indicating the existence of gaps between MXene sheets. However, the peak (002) further weakened after Na2B4O7 densification, suggesting the closer stack between MXene sheets. As seen in Figure 7(b)–(d), boron element appeared in boron-enhanced AgNPs@MXene. A new C-O-B peak at 281.8 eV presented in boron-enhanced AgNPs@MXene.
20
It elucidated that the covalent bridging between boron ions and −OH of MXene surface. (a) XRD of boron-enhanced AgNPs@MXene and AgNPs@MXene; XPS spectra of boron-enhanced AgNPs@MXene and AgNPs@MXene: (b) the total spectra, (c) carbon spectra of AgNPs@MXene, and (d) carbon spectra of boron-enhanced AgNPs@MXene.
Electromagnetic shielding performance of BAMP-F
In order to meet the requirements for daily use, electromagnetic shielding performances of BAMP-F and AMP-F at bands ranging from 30 MHz to 3030 MHz were tested in Figures 8(a) and (b). At 200 MHz, EMI SE values of BAMP-F and AMP-F were 34.51 dB and 24.16 dB, respectively. Compared with AMP-F, it increased by 42.8%. Under three layers of fabrics, EMI SE values of BAMP-F and AMP-F increased to 58.70 dB and 57.43 dB, respectively. It implied that 99 % electromagnetic waves could be shielded. BAMP-F would meet the commercial applications on electromagnetic shielding (≥20 dB). Electromagnetic shielding mechanism of BAMP-F was presented in Figure 8(c). Most incident electromagnetic energy was reflected by AgNPs@MXene conductive layer due to impedance mismatch. Reflection loss of BAMP-F to electromagnetic waves played the main role. Residual electromagnetic waves penetrated into conductive layer, experienced multiple refelection and energy adsorption until being fully dissipated.
31
Moreover, the abundant polar groups and local defect of MXene produced dipole to enhance the electromagnetic shielding performance. Furthermore, Table 1 lists the comparison for EMI shielding property of different fabrics. It was found that EMI shielding performance of BAMP-F in this work lay in the medium level in comparison with other fabrics reported in literature. Electromagnetic shielding performance of (a) AMP-F and (b) BAMP-F at different layers; (c) Electromagnetic shielding mechanism of BAMP-F. The comparison for EMI shielding property of different fabrics.

Durability of BAMP-F
MXene possessed a few functional groups –OH, –O, and –F which could form hydrogen bonds with amino and pyrocatecdiol groups of PDA. Moreover, the functional groups of PDA coordinated with silver ion of AgNPs. As a result, it existed the firm binding between AgNPs@MXene and PDA coating fabric. AgNPs@MXene could construct the integral conductive path onto the fabric. Durability of BAMP-F was investigated and compared with cotton fabric directly treated by AgNPs@MXene (AM-F).
Figure 9 shows the stability of surface resistivity and EMI SE of BAMP-F and AM-F after bending, ultrasonic washing, and tearing. After 600 bending, washing for 60min, and tearing, surface resistivity of BAMP-F had a slight increase and EMI SE values of BAMP-F lowered a little bit. However, surface resistivity of AM-F exhibited sharp increase and EMI SE values of AM-F decreased to a large extent. It demonstrated that BAMP-F could remained the performance stability under external action, showing good durability. It was ascribed to the hydrogen-bonding interactions between AgNPs@MXene conductive layer and PDA adhensive layer. Stability of surface resistivity and EMI SE of BAMP-F and AMP-F: (a) bending, (b) ultrasonic washing, (c) tearing, (d) photos before and after tearing.
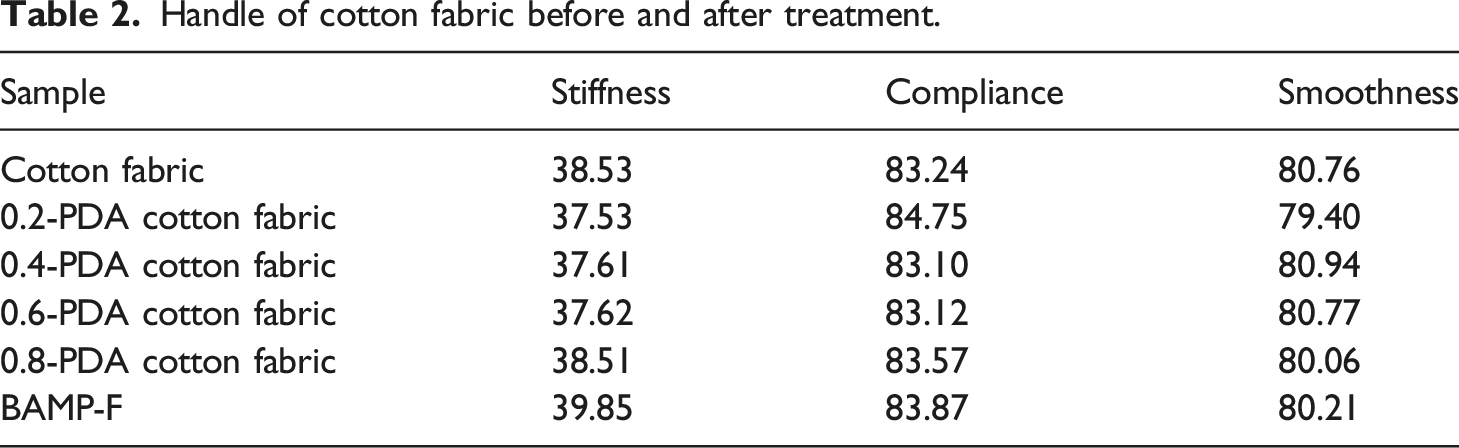
Handle of BAMP-F
Handle of cotton fabric before and after treatment.
Conclusion
AgNPs@MXene composites with excellent electrical conductivity were successfully prepared by in-situ reduction of AgNO3 by MXene, and its surface resistivity reached 7.22 × 10−4 Ω·m.
The optimum surface resistivity of BAMP-F arrived at 7.08 × 10−3 Ω·m after coating by 0.6 mol/L dopamine, impregnating by AgNPs@MXene for 6 times and densifying by 0.02 mol/L Na2B4O7. Compared with AMP-F and AM-F, the surface resistivity of BAMP-F decreased by 47.6 % and 81.9 %, respectively. Its electromagnetic shielding performance arrived at 34.51 dB and increased by 25.8 % in comparison with AMP-F. BAMP-F also exhibited good handle and durability such as bending-resistance, water-resistance, and tear-resistance.
Footnotes
Acknowledgements
This work was financially supported by National Natural Science Foundation of China (NO. 51703202) and the Fundamental Research Funds of Zhejiang Sci-Tech University (No. 2021Q003).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (NO. 51703202) and the Fundamental Research Funds of Zhejiang Sci-Tech University (No. 2021Q003).
