Abstract
Over the last few decades, micropollutants have been found in the natural surface water, and their presence poses a series of risks to living organisms in the aquatic biotope. Nanofiber membranes have been shown as a new strategy for separating micron-size pollutants due to their tight pore size and high water permeability. Here, a high-efficient multi-layered nanofiber membrane with improved mechanical strength was prepared using the lamination technique. First, polyamide 6 (PA6), polyacrylonitrile (PAN), and polyvinylidene fluoride (PVDF) nanofibers webs were selected. Then nanofibers were laminated on support under various temperatures. Prepared membranes were characterized using SEM, contact angle, pore size, and air permeability tests. The significance of lamination condition on membrane structure was investigated experimentally and statically. The membranes were examined for the treatment of synthetic micropollutants. According to the results, PAN and PA6 nanofiber membranes exhibited high water permeability (over 3000 L/(m2hbar)) under low pressure, whereas PVDF membranes showed zero permeability. Besides, the prepared nanofiber membranes could separate micropollutants from water with up to 99.91% separation efficiency. Prepared nanofiber membranes hold great promise for practical applications in environmental remediation.
Introduction
Water is essential for all living organisms all over the world. However, due to rapid growth in population and industrialization, climate changes, and contamination of available freshwater resources, the accessible freshwater sources have reduced drastically. Currently, one billion people do not have clean water, and 2.3 billion live in water-stressed areas, which is expected to be 3.5 billion by 2025. 1 As a result of contaminated water, people suffer from many diseases, such as cholera, diarrhoea, typhoid, dysentery, and polio. It is estimated that every year 485,000 diarrhoea cause deaths because of contaminated drinking water. 2
Moreover, more than 2 million people die yearly from contaminated water pathogens. A considerable amount of fresh water is used for industrial, agricultural, and domestic purposes, resulting in contaminated water with various synthetic and geogenic compounds. Therefore, removing micropollutants and chemical compounds from contaminated water is a big challenge for safe reuse.
Membrane separation is an efficient technology for the remediation of contaminated water and reducing the impact of micropollutants. Compared to other remediation technologies, membrane technology offers high performance, selective separation, low cost, ease of maintenance, operation, low chemical consumption, and less space. Even though the many benefits of this technology, membranes are suffering from fouling, which increases the cost of operation and reduces membrane lifespan. Fouling forms when the foulants are collected on the membrane surface or inside the membrane pores. It is a great challenge to overcome membrane fouling. Several strategies have been tried to overcome membrane fouling, such as membrane physical and chemical cleaning, modified membranes, and optimization of operating conditions and feed solution.3–6 Mohammadi et al. 7 used various chemical cleaning agents to recover fouled polysulfone ultrafiltration membrane. The membrane was fouled by the precipitation of milk components. The optimum recovery rate was achieved by combining sodium dodecyl sulfate and sodium hydroxide. In another study, a different strategy was used to reduce membrane fouling. Surface-modified polyvinylidene fluoride/polyacrylonitrile (PVDF/PAN) nanofiber membranes were used to minimize membrane fouling for the oil-water emulsion. 4 The separation results showed that, after low vacuum microwave plasma treatment with Argon (Ar) and chemical modification with an alkaline solution, the membranes had excellent hydrophilicity due to the formation of active carboxylic groups. Membrane permeability was tested 10 times without any cleaning process between each cycle. Results indicated modified membrane showed an excellent anti-fouling property. The research and development behind the membrane technology show that there is still a huge interest in newly developed membranes for better performance and fouling resistance.
Recently, electrospun nanofiber membranes have gained huge attention due to their high specific surface area, tight pore size and pore size distribution, and interconnected and highly porous structures. Nanofibers’ structural architecture and physicochemical properties allow use in water domain applications. The nanofibrous structure can be utilized to enhance the membrane’s adsorption capacity and separation efficiency. Electrospun cellulose acetate (CA) nanofiber membrane prepared as affinity membrane for separating many biomolecules. 8 The CA nanofiber was first treated with heat to improve mechanical properties, and then alkaline treatment was done to obtain regenerated cellulose (RC) nanofiber mesh. The surface of RC is functionalized with Cibacron Blue F3GA (CB) by a covalent bond. Nanofiber membrane showed abilities to capture bovine serum albumin (BSA) or bilirubin with capacities of 13 and 4 mg/g, respectively. After regeneration with elution buffer, the membrane showed reusability. Veleirinho et al. 9 fabricated poly(ethylene terephthalate) (PET) nanofiber membranes for the apple juice clarification process. Compared to the commercially used ultrafiltration technique, the electrospun PET nanofiber web allowed a faster juice flux with lower time consumption. The study showed the filtration potential of PET nanofiber membrane, which opened new approaches for the juice processing industry. PVDF is one of the most used polymers in membrane technology due to its outstanding chemical and thermal stability. However, PVDF is hydrophobic characteristics that reduce the membrane flux. Li et al. 10 improved PVDF nanofiber membrane flux and fouling resistance by blending PVDF with hydrophilic polyvinyl alcohol polymer during the electrospinning process, which created ridge-and-valley structures on the surface. Blended nanofibers improved super-hydrophilicity as well as the wettability of the membrane. PAN is another interesting polymer in membrane technology due to its good chemical stability. The surface of PAN nanofiber membranes was modified by dual-vinyl and tri-vinyl monomers polymerization. 11 The modified PAN nanofiber membranes effectively removed bacteria (by size exclusion) and viruses (by adsorption) from contaminated water. The modification process reduced membrane pore size, which increased particle rejection. Modified PAN showed 2–3 times higher flux than the commercial membrane. Polyamide (PA) nanofiber membranes offer the advantage of being a hydrophilic characteristic.12,13 PA6 nanofiber membrane was used to remove microorganisms taken from wastewater from a general hospital (107–108 CFU/100 mL). 14 The removal of microorganism efficiency for the bactericides incorporated nanofibers and the Ag functionalized fibers were compared. Unfunctionalized PA6 nanofiber membranes showed a low reduction of microorganisms, while opposite results were achieved for the functionalized membranes. In another study, PA6 and chitosan (CH) polymers were blended during electrospinning to increase adsorption behavior. These membranes remove anionic dyes Solophenyl Red 3BL and Polar Yellow GN. 15 After optimization of electrospinning time, CH concentration, and solution parameters, the removal of Solophenyl Red 3BL and Polar Yellow GN was found at 96% and 95%, respectively.
Although the nanofiber membrane looks like a great candidate for wastewater remediation, research and development are needed to carry membranes to an industrial scale. One of the biggest problems of nanofiber is its poor mechanical and abrasion resistance under load. Scientists focus on several techniques to overcome the mechanical weakness of nanofiber membranes. Those strategies includes, fiber orientation and alignment, 16 fiber blending,17,18 reinforcement,19,20 additives,21,22 epoxy composite,23,24 carbonization,25,26 inter-fiber bonding by using chemical crosslinking or heat treatment,27,28 ultrasonic welding, 29 dip-coating,30,31 and thermal lamination and heat-press.32–34 Among all, thermal lamination and ultrasonic welding processes are more promising for the industrial scale of membrane production. Other strategies are time-consuming, require expensive chemicals, are not eco-friendly, are costly, and some are not effective for long-term application.
Mechanical properties of poly(trimethylene terephthalate) (PTT) nanofiber membranes were enhanced by applying heat and crosslinking points by blending polypropylene (PP) nanofibers. 35 Membranes showed great flux with high rejection of TiO2 nanoparticles (100 nm) above 99.6%. The maximum pure water flux of PTT, heat-treated PTT, and PP/PTT composite nanofiber membranes was found at 6579 L/(m2h), 6126 L/(m2h), and 4126 L/(m2h) at 0.6 MPa pressure, respectively. Heat treatment caused compactness and softening of PP melts to block the membranes’ pores, reducing flux. Moreover, heat treatment encouraged fusion at inter-fiber contact points and increased the mechanical strength of the membrane. Similarly, post-heat treated PVA nanofiber web with temperature below and above Tg to improve the mechanical properties. 36 However, the higher the heat treatment temperature caused more melting and recrystallization in the polymer, and the material became more brittle. Recently,34,37 it was reported that the heat treatment improved the mechanical properties of PVDF and PAN nanofiber membranes while increased applied temperature reduced the air permeability of the membranes. The reason was that softening of the melt adhesive web between support and nanofiber filled the pores of the fibrous structure and reduced air permeability. Heat treatment improves nanofiber membranes’ mechanical strength, membrane compactness, and chemical stability, reduces residual charges on the fiber surface, and reduces delamination of layers. However, the conditions of the process need to be optimized.
In the current study, three polymeric nanofiber membranes (PVDF, PAN, and PA6) were systematically laminated under various temperatures. The relationship between lamination condition, liquid filtration, and particle separation performance of the membranes was investigated. Importantly, this work provides extensive information to the literature by highlighting the importance of the lamination process for various polymeric nanofibers to separate micropollutants from water. Many suggested methods for forming nanofiber membrane composite in the literature are costly, time and energy-consuming, require chemicals, and are not eco-friendly. Herein, an easy-to-use, less energy and time-consuming, and easy-to-scale-up lamination process have been offered to laminate nanofiber webs. Additionally, nanofiber layers are produced by using an industrial-scale production method. The findings in the current study demonstrate that both the lamination method and the nanofibers used in this work provide a convenient and cost-effective strategy for wastewater remediation in industrial-size applications.
Materials and methods

Schematic diagram of lamination process. The sample abbreviations are shown in Table 1 Abbreviations of nanofiber membranes.
Samples were kept under various temperatures and applied force for 3 min (first 2 min 30 kN, last 1 min 50 kN).
Characterization
The surface morphology of the prepared samples has been observed using a scanning electron microscope (SEM; TESCAN VEGA, Brno, Czech Republic). No damage has been observed after the lamination process. Fiber diameter was measured using the Image-J software program.
The water contact angle of membranes was measured using The Krüss Drop Shape Analyser DS4 (Krüss GmbH, Hamburg, Germany) against distilled water (surface tension 72.0 mN m−1). In order to measure contact angle, ASTM D 5725-99 (Standard Test Methods for Surface Wettability and Absorbency of Sheeted Materials Using an Automated Contact Angle Tester) was used. Each measurement was done from the right and left sides of the droplet, and their average was taken. At least 5 measurements were done from different places on the samples, and an average of 5 measurements was taken.
ATLAS 128 Air Permeability Tester (SDL ATLAS, Rock Hill, SC, USA) was used to test air permeability which was set at 200 Pa and 20 cm2. At least 5 measurements were done, and an average was taken. In addition, ASTM D737 Standard (Air Permeability of Textile Fabrics) was used to determine the air permeability of samples.
Membrane pore size was determined by the bubble point method using a pore size analyzer (Porometer 3G, Anton Paar GmbH). At least 3 measurements were done, and the average pore size was evaluated. Pore size measurement was done according to ASTM F316-03 (Standard Test Methods for Pore Size Characteristics of Membrane Filters by Bubble Point and Mean Flow Pore Size).
Tensile properties of the membranes were measured according to information from literature 38 by a universal testing machine (Labor-Tech s.r.o., CR). In summary, the test was done with the extension rate of 10 mm/min at room temperature for the sampling size of 100 mm long and 25 mm wide, and the distance between the two clamps was 50 mm. In addition, the prepared membranes were tested according to the ISO 9073-18:2007 (Textiles—Test methods for nonwovens) to determine their mechanical properties.
Statistical analysis
Statistical analyses were performed in Python programing language using the StatsModels package.
Two-way ANOVA was used to assess the effects of raw material type and temperature and their interaction. The factorial experimental design was conducted with two factors, “Type” and “Temperature” with 3 levels of Type (PA6, PVDF, PAN) and 3 levels of Temperature (110°C, 125°C, 135°C). There are 3 levels for Type and 3 levels for Temperature, and this is a 3 × 3 factorial design yielding 9 unique combinations for measurement of the response variable. Each sample unit size is chosen to be equal (
The CV value of the fiber diameters at different lamination temperatures is given in Supplementary Information S1. Boxplots of the independent variables provide a graphical summary of the distribution of each sample. The boxplot graphs are given in Supplementary Information S2.
In ANOVA Tables, the Source of variation means the source of the variation in the data, both main factors and interaction level of factors. Sum of Squares (SS) is the sum of squares due to the source where the sum of squared differences between the grand mean and modelled value. The sum of squares shows a measure of variation or deviation from the mean. The sum of square is calculated as a summation of the squares of the differences from the mean. Degree of Freedom (Df) is the degrees of freedom in the source. F Value Significance is the
Filtration test
A lab-scale dead-end 50 mL Amicon stirring cell was used for the test (Merck, Millipore). The 13.4 cm2 filtration area with 0.02 bar pressure was used. In the first filtration test, 40 mL distilled water was used for all membranes, and membrane flux and permeability were calculated according to equations (1) and (2).

In the second filtration test, polydispersed polystyrene (PS) microparticles (particle size about 0.503 μm) were mixed with distilled water and used as feed solution. 5 μL of PS particles were mixed into 1,000 mL of distilled water and shaken for 10 min. Membrane permeability was calculated according to equation (2).
Membrane rejection was measured using a turbidity meter (Livibond m/c in NTU unit), and rejection was calculated according to equation (3).
Results and discussion
The surface morphology of nanofiber membranes was investigated using Scanning Electron Microscope (SEM). SEM images of the nanofiber membranes are given, as shown in Figure 2. SEM images of PVDF, PAN, and PA6 nanofibers before and after various lamination temperatures.

| Before lamination | 110°C | 125°C | 135°C | |
|---|---|---|---|---|
|
|
155 ± 44 | 173 ± 43 | 180 ± 52 | 185 ± 39 |
|
|
137 ± 43 | 177 ± 30 | 186 ± 40 | 179 ± 35 |
|
|
135 ± 40 | 157 ± 34 | 144 ± 29 | 178 ± 41 |
ANOVA table (Fiber diameter: Type + Temperature + Type: Temperature).

Pore size, air permeability, and water contact angle of the samples.
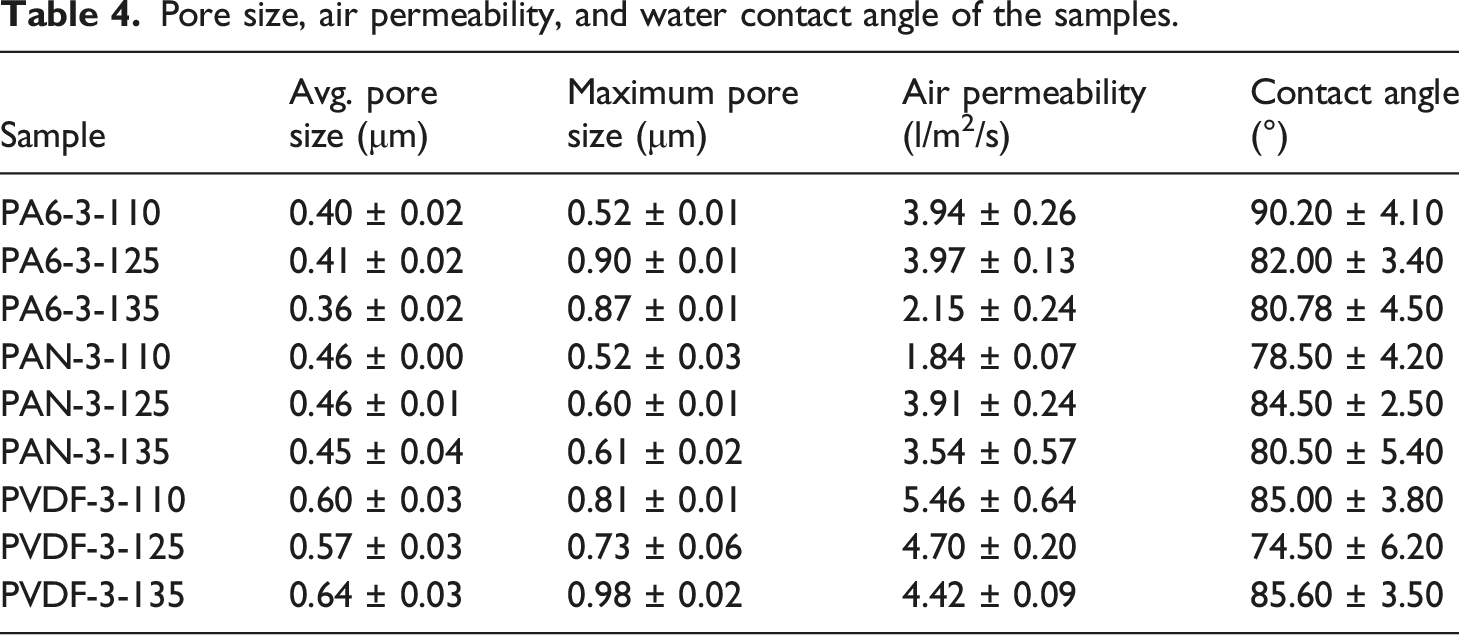
During the lamination process, the molten adhesive web partly covered the nanofiber surface and attached it to the support layer. As a result, the surface wettability of nanofibers changes. Herein, three polymeric membrane shows similar water contact angles after the lamination process. The huge effect of the composite structure is visible in the material wettability. All three different polymeric membranes showed similar water contact angles. The lower water contact angle means higher wettability, enhancing the membrane water flux. However, the contact angle is not the only phenomenon driving the membrane’s water flux. Membrane type (pore size, configuration) and several physical and environmental operating conditions (type of waste, feed temperature, feed viscosity, system operating conditions) influence the membrane flux.46–49 In this work, the physical and environmental conditions were kept the same. The only difference between samples was the membrane type and lamination temperature. Since the membrane water contact angle is more or less similar, and all membranes were used as flat-sheet, the differences between membranes were their pore size, porosity, and water affinity of the overall composite structure. The differences in membrane water permeability are expected to be related to pore structure, interconnection of pores, and affinity to water absorption.
The lamination process can bring the fibers closer together. As a result, smaller pores and a smoother surface with a much more regular structure can be form. 41 Nanofibers under heat and force can exhibit thicker and flatter fibers with some melted and fused nodes, resulting in smaller pore size. 50 Unlike the given literature, we observed that the average nanofiber pore size slightly increased under lamination. A similar case was observed by Hughes et al. 51 It was found that raising the membrane temperature caused the expansion of fibrous PTFE material and, subsequently, pore size increased. Heat treatment can be said to cause nanofibers’ expansion, which can affect the pore size diameter.
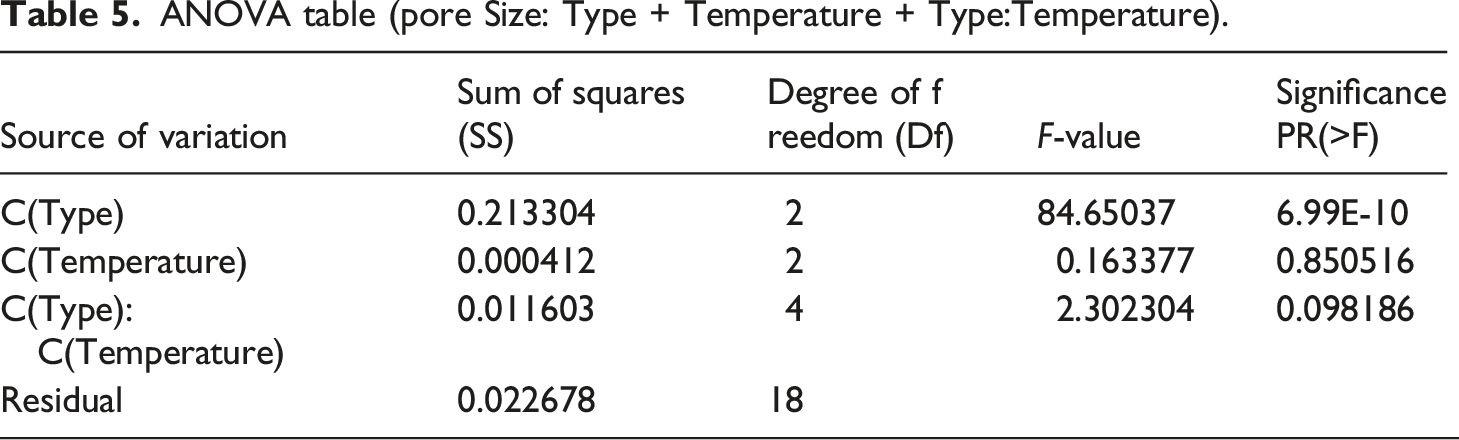
ANOVA table (pore Size: Type + Temperature + Type:Temperature).
ANOVA table (water contact angle: Type + Temperature + Type: Temperature).

ANOVA results indicated that temperature does not significantly affect pore size, while the type of polymer has significance. For pore size, the interaction effect and Temperature main effect are statistically non-significant (
The pore size of PA6, PAN, and PVDF membranes change between 0.36–0.41, 0.45–0.46, and 0.57–0.64 μm, respectively. The changes in pore size are due to fiber diameter and lamination condition of membranes. PA6 and PAN nanofiber membranes have the narrowest pore size compared to PVDF. PVDF nanofiber membranes have the largest pore size, which is even twice higher than microparticles separated (size 0.5 μm). Based on the pore size of PVDF, it can be expected that these membranes can have high water permeability with significantly less selectivity. However, water filtration test results indicated that PVDF has the lowest water permeability due to the intrinsic hydrophobic property of the membrane and the charge effect. The high fluorine content with polar C-F bonds increases PVDF membrane intrinsic hydrophobicity.
Moreover, it is necessary to emphasize that nanofiber webs do not have straight-through pores. Instead, they have tortuous pathways. The particles can be caught within the membrane structure.
Na et al. 52 applied continuous hot-press at elevated temperatures up to 155°C to PVDF electrospun membranes. SEM images showed no significant changes in fibrous membrane morphology when the temperature changed from 25 to 130°C. However, larger pores were formed due to the melting and bonding of fibers under higher temperatures. The mechanical properties were improved, whereas the porosity and the liquid absorption decreased under the heat press. Wang et al. 53 used the hot-pressing method to adjust and control the PAN membrane porosity. Using the hot-pressing method, the membrane porosity decreased by 40% from the original membrane, while the particle rejection increased from 0% to ∼100% for 0.2 μm particles. The fibers kept their shape/morphology under low pressure (5 MPa) and kept their shape/morphology without distinguishable deformities, while hot-pressed PAN membranes under the highest pressure (20 MPa) became flattened.
Figure 3 shows how the hydrophilic adhesive covered the surface of hydrophobic PVDF nanofiber. The low water contact angle of PVDF could be due to the adhesive web effect. The lighter part in Figure 3 shows the nanofibers, whereas the darker parts are the adhesive layer over the surface of nanofibers and form a film layer. Surface morphology of laminated PVDF nanofibers (dark spots indicate the adhesive web over the PVDF nanofiber).
Lamination temperature is an important parameter. The adhesive layer must heat enough to be molten and adhere to surfaces. Overheating of the layer may cause damage to the supporting layer and nanofiber layer. The adhesive molten can easily move toward composite layers at the proper temperature and combine them. Depending on the applied force on the layers, the molten structure can distribute a higher area between the composite layers and over the surface of nanofiber webs which generally has a thickness of a few micrometers.34,37 Liao et al. 54 applied a heat-press post-treatment to PVDF nanofiber to improve membrane integrity and mechanical strength. The membrane kept its morphology as the fresh membrane after 160°C heat pressing. When the heat-pressing was arranged to 170°C, the thickness of the membrane was reduced with a lower contact angle than the fresh membrane. Moreover, the heat-press post-treatment decreased membrane pore size, porosity, and water contact due to the compaction. In our case, the final thickness of laminated membranes is important. The finished membrane thickness influences membrane flux. The laminated membrane thickness was measured for selected membranes. Results indicated that the membrane type and lamination temperature did not influence the thickness. The membranes showed an average thickness of around 28 mm.
Herein, the contact angle between each polymer laminated at various temperatures significantly affects the contact angle according to the ANOVA test (Table 6).
When the ANOVA test results are examined, both Type and Temperature statistically affect the water contact angle. At the same time, the Type and Temperature interaction effect are also statistically significant on the water contact angle (Table 6).
The pressure and temperature employed during the heat pressing can change the degree of crystallinity and surface roughness of polymeric material. As a result, the water contact angle changes.55–57 In this work, not only the polymeric material of nanofiber web but also adhesive plays a huge role in contact angle. As shown in Figure 3, adhesive molten covered the surface’s huge part, creating changes in contact angle.
ANOVA table (air permeability: Type + Temperature + Type:Temperature).

As the ANOVA test results indicate, Type and Temperature have a statistically significant effect on the air permeability as well as the interaction effect (
The membrane tensile testing showed that all the membranes have more or less the same tensile properties as the base supporting the nonwoven layer. It was found that in the machine direction 133 N/5 cm, and in the counter direction 107 N/5 cm. The role of the supporting layer is huge on overall membrane structure and integrity. The selection of a support layer is crucial to preparing nanofiber membranes.
Different areal weights of the same nanofiber membrane can change membrane water permeability. For example, it was demonstrated that 1.12 g/m2 PA6 nanofibers result in 2-fold higher water permeability than 2.31 g/m2 PA6 under the same conditions. 38 However, the particle rejection was lower. The areal weight is essential in the nanofiber layer’s porosity and bubble point. The thicker layer caused a less porous structure and high tortuosity. Herein, the areal weight of the nanofiber web was kept constant to compare each under various lamination conditions.
The water filtration and the microparticle separation are crucial parts of this study. Therefore, water permeability and microparticle rejection rate have been calculated to understand the separation performance.
Water was contaminated with PS micropollutants, water flux was measured, and permeability was calculated according to equation (2). Permeability results of all membranes have been given in Figure 4. Contaminated water permeability of PA6, PAN, and PVDF membranes laminated at various conditions.
The water contact angle of all polymeric membranes is more or less similar. As explained above, the adhesive web played a significant role in the water contact angle since it covered the surface of nanofibers. In this case, judging the contact angle effect on water permeability is not easy. Even though samples have similar contact angles, PVDF membranes showed zero water permeability for PVDF-3-125 and PVDF-3-135. Among all the nanofiber webs, only PAN nanofiber shows super hydrophilic characteristics before the lamination process. After the lamination process, both PA6 and PAN nanofiber membranes have high water permeability.
PS microparticles are prepared using potassium persulfate as the initiator has sulfate groups on their surface.59,60 Consequently, these particles are charged negatively. Since PVDF has positively charged C–H bonds, the partial positive CH2 dipoles of the PVDF chains can interact with negatively charged PS microparticles.61,62
PVDF membranes fouled from the very beginning. Only PVDF membrane laminated with low temperature showed water permeability at the beginning. However, the membrane fouled in time. The interaction between PVDF and PS microparticles increases membrane fouling. Moreover, PVDF membranes have larger pore sizes than tested microparticles. As a result, particles penetrated and filled the pores and blocked the water permeability. PVDF-3-125 and PVDF-3-135 did not show any water permeability. For this reason, the PVDF has been excluded from the 2-way ANOVA test.
On the other side, both PA6 and PAN membranes showed high water permeability. The rejection mechanism is mainly governed by size exclusion. Particle size is around 0.5 μm, while membranes have an average pore size less than the size of microparticles. Therefore, the particles accumulate on the membrane surface in time. As a result, the permeability decreased in time due to membrane fouling. In general, the permeability of flux decline continues until the fouling reaches its steady-state thickness. Herein, both PA6 and PAN membrane permeability declined steadily.
Water permeability: PA6 + PAN + PVDF.

Water permeability: PA6 + PAN.

As the ANOVA test results indicate, temperature itself is not significant on water permeability while type and Type + Temperature interaction have a statistically significant effect on the water permeability (
Only PA6 and PAN raw material types are used in the 2-way ANOVA analysis with 3 levels of temperature (110, 125, 135). Every unit sample size was chosen an equal number of samples (
As shown in Table 9, at least one Type group has a statistically significant main effect on water permeability, and the interaction term also reveals statistical significance. At the same time, the temperature is not statistically significant.
It can be concluded that both PAN and PA6 membranes have high water permeability. However, it was generally observed that PAN membranes have a lower permeability decline than PA6. The reason could be due to negatively charged PAN membranes. PAN is a negatively charged synthetic membrane63,64 that can repel the PS microparticles. For this reason, the membrane fouling is reduced for PAN nanofiber membranes.
In summary, the lamination temperature did not significantly affect the water permeability. On the other hand, the type of polymeric material was more predominant for water permeability. The membranes' water contact angles were similar after the lamination process. The reason was that the adhesives covered the surface of the membrane. We found that the pristine nanofiber web’s water contact angle was effective on water filtration performance. The lowest water contact angle led to higher water permeability. Membrane pore size played a crucial role in water permeability. Even though the highest pore size membranes were PVDF based, they showed the worst water permeability due to their affinity to microparticles, resulting in membrane fouling.
The water permeability test is not the only criterion to determine if a membrane is suitable for the remediation of water or not. A particle rejection test has been run to observe membrane separation performance. The rejection rate was calculated according to equation (3). Particle rejection rate of membranes is shown in Figure 5. The particle rejection rate of membranes.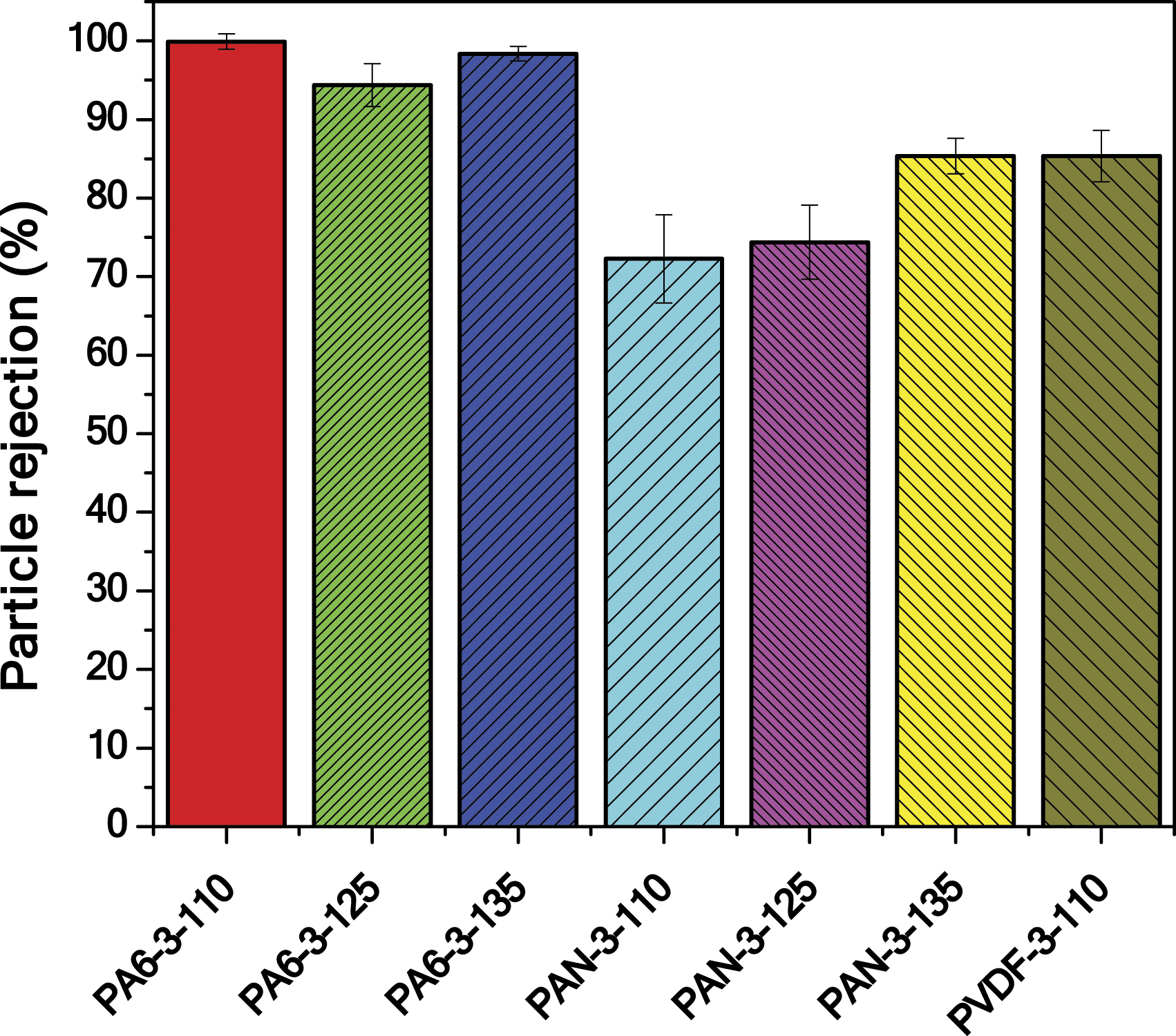
The particle rejection of PA6 nanofiber membranes is higher than PAN and PVDF membranes. The main mechanism behind the particle rejection is size exclusion related to the pore size of the membranes. However, the tortuosity structure of the nanofiber web also plays a huge role in the rejection of microparticles. PA6 has the narrowest pore size among all membranes, while PVDF has the largest pore size. Similarly, it was found in the literature that the PA6 nanofiber membrane has higher waste rejection compared to PAN nanofiber membranes. 38 The maximum pore size of the membranes is given in Table 4. Maximum pore size provides the information if there is any damage, big holes, or large pores on the surface of the membrane. Even though PA6 has some large pores on the membrane surface, the number of large pores is probably less and has a highly tortuous pathway within the membrane structure. As a result, the rejection of PA6 membranes drops to till 94%. By contrast, PAN membranes showed the lowest particle rejection. It might be due to the higher number of large pores on the membrane surface. Unfortunately, it was impossible to measure the number of large pores using a porometer.
Conclusion
A conversive study on lamination conditions of the nanofiber membrane was demonstrated in this study. Three different polymeric membranes were selected and laminated under various temperatures. The effect of temperature on membrane pore size, contact angle, air permeability, and water permeability has been investigated. The results were statically analyzed.
In summary
— The fiber diameter of the nanofiber web increased after the lamination process. Under various lamination temperature, the fiber diameter changes slightly. The fibres seem flattened after heat treatment under applied force and become more stable. The results were controlled statically, and ANOVA results showed both polymeric material type and lamination temperature influence the final fiber diameter of the membranes. — In the beginning, only PAN nanofiber webs showed hydrophilic characteristics before lamination. After the lamination process, all samples showed similar water contact angles due to adhesive material. It proves that each layer of the structure is quite important for the final characteristics of the membrane. Besides the nanofiber material, the adhesive type and supporting layer must be selected carefully for forming a membrane. — Changing the lamination temperature did not significantly affect pore size between each temperature value. On the other hand, the type of polymeric material plays a significant role in membrane pore size. Using PAN or PA6 nanofibers, it is possible to get microfilters with pore sizes less than 0.5 μm. — Both polymeric material type and lamination temperature play a significant role in air permeability. During lamination, applied force reduces the compactness of fibrous material, where the heat treatment influences the fiber diameter and the molten adhesive. This work aims to use membranes in water filtration, not in air filtration. However, an air permeability test is needed to observe if molten adhesive totally blocks all the membranes' pores. — It was investigated that the mechanical properties of nanofiber membranes mainly depend on the support layer. It is crucial to select suitable support to improve membrane tensile properties. — Water filtration test results indicated that PAN and PA6 had high water permeability, whereas PVDF had almost zero. Only the PVDF-3-115 membrane showed permeability at the beginning, and then permeability stopped due to membrane fouling. The effect of temperature on PAN and PA6 membranes was not significant. Based on the water filtration test and particle rejection, the optimum lamination temperature value for PAN and PA6 nanofibers could be suggested as 135°C and 110°C, respectively. Each polymeric membrane showed different filtration behavior under various temperatures. — Average pore size showed a dominant effect on micro-particle rejection. Due to the narrowest pore size, PA6 nanofiber membranes' selectivity was highest at 99.91%.
Our findings showed the potential of using nanofiber membranes as pre-filters prior to ultrafiltration or nanofiltration to separate contamination from microparticles. The membranes prepared and laminated in this study were in the size of 80 x 80 cm2. These membranes can be counted as the next generation of filtration media for industrial application.
Supplemental Material
Supplemental Material - Multi-layered nanofiber membranes: Preparation, characterization, and application in wastewater treatment
Supplemental Material for Multi-layered nanofiber membranes: Preparation, characterization, and application in wastewater treatment by Nazrul Islam, Bekir Yıldırım, Jiri Maryska and Fatma Yalcinkaya in Journal of Industrial Textiles
Footnotes
Acknowledgements
The authors also acknowledge the assistance provided by the Research Infrastructure NanoEnviCz (Project No. LM2018124), supported by the Ministry of Education, Youth and Sports of the Czech Republic and Mobility Fond in Technical University of Liberec.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Industry and Trade––TRIO project FV40421. Recycling of technological waters in the beverage industry.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
