Abstract
As a natural plant material with unique porous structure, loofah is used in many fields, but there are still defects in fire safety performance in that its flammability. The melamine-modified flame retardant loofah composites (M-FR-PL) were prepared by impregnation baking method. The experimental synthesis conditions were optimized by L9 (34) orthogonal design. The flame retardancy, heat release rate (HRR), thermal stability and smoke density of the composites were analyzed. M-FR-PL6 prepared under the optimum experimental conditions had good properties: the adsorption rate of flame retardant (AR, %) was 6.8%, the limiting oxygen index (LOI, %) reached 27.5%, the vertical combustion level reached V-0, and the amount of residual char was 17.2% higher than that of loofah. The flame retardant mechanism of M-FR-PL6 was thoroughly discussed by various characterization methods. The peak of the HRR curve of M-FR-PL6 declined compared with loofah, indicating the loofah after melamine flame-retardant treatment has good flame retardancy and smoke suppression properties.
Introduction
Loofah is a kind of natural and renewable plant fiber with properties such as unique porous structure, toughness, wear resistance and good elasticity. Loofah is used in the fields of packaging, sound and heat insulation, building and home decoration, adsorption materials and so on.1–5 However, loofah is flammable and can quickly reach the heat release peak. After ignition, it can release a lot of heat and flue gas,6–8 which limits the application of loofah in many fields. Therefore, it is of great significance to explore how to reduce its fire risk. With the popularization and use of environment-friendly and renewable resources, environmental friendly and halogen-free flame retardants have gradually become the focus of attention.9–11 Among them, nitrogen-based flame retardants with high nitrogen content can not only release inert gas and combustible gas in dilution air during pyrolysis, but also be effectively applied to the research of flame retardant materials.12,13
Melamine is a typical nitrogen-based halogen-free flame retardant containing high N element. The thermal decomposition of melamine can produce inert gases such as NH3 and dilute the combustible gases in the system to achieve the effect of flame retardation.14,15 The natural fibers are usually pretreated before surface modification. Alkali treatment is often used to remove not only the colloid and wax on the surface, but also the lignin and hemicellulose inside natural fibers.16,17 Alkalization can change the chemical content of the fiber by breaking the hydrogen bond in the structure of the wood fiber, so as to remove hemicellulose, pectin, wax and lignin during the separation of the fiber bundle in the micro fiber.18,19 The density of the pretreated fiber increases and the structure is closer, which is conducive to improve the mechanical properties of the material. High cellulose content and low hemicellulose after pretreatment can also improve the thermal stability of the fiber.20–22
To improve the flame retardant performance of loofah, the flame retardant loofah composites were prepared by impregnation baking method using melamine as the flame retardant. Scheme 1 presents the preparation process. The optimal experimental preparation conditions were determined by orthogonal design method. The flame retardant, thermal stability and smoke suppression properties of the composites were studied by various characterization methods, and the possible mechanisms of flame retardant and smoke suppression were discussed. Experimental flowchart.
Materials and methods
Materials
Loofah was grown in Nanning, Guangxi. Sodium hydroxide, 30% hydrogen peroxide, phosphoric acid and melamine were purchased from Sinopharm Chemical Reagent Co., Ltd. (China). Sodium hydroxide and 30% hydrogen peroxide were used for loofah pretreatment.
Pretreatment of loofah
Firstly, the dust and impurities of loofah were washed with distilled water, and then dried at 80.0°C for 12.0 h to constant weight. Then, the dried loofah was crushed with a pulverizer, and the loofah was stored in a dry environment. A certain amount of loofah was put into 10%NaOH solution (loofah bath ratio (LBR) was 1:50), and then treated at 98.0° C for 4.0 h. After the reaction, took out the loofah and rinsed it with distilled water until it became neutral. Then 2.0 g/L H2O2 solution was added into the mixture ((LBR was 1:50), refluxed at 80.0° C and mixed for 4.0 h. Finally, the resulting product cooled to room temperature was collected by filtration and multiple washes with distilled water, and then it was dried to constant weight at 80°C and called as pretreated loofah (PL).
Preparation of melamine flame retardant loofah material (M-FR-PL)
Experimental factors and levels in orthogonal design.

The orthogonal design layout and results of adsorption rate (AR) and limiting oxygen index (LOI).

Characterization
Fourier transform infrared (FTIR) spectra were performed on a Spectrum One Autoima spectrometer (Nicolet 50, Thermo Scientific Company) in the range of 4000–400 cm−1.
X-Ray photoelectron spectroscopy (XPS) was carried out using a K-Alpha XPS analyzer (Thermo Scientific, USA), with Al Kα excitation radiation (hυ = 1486.6 eV) in ultrahigh vacuum conditions.
The limiting oxygen index (LOI) test was performed by an oxygen index instrument ZY6155A (Dongguan Zhongnuo Quality Inspection Equipment Co. Ltd, China). According to ASTM D-2863 standard, the LOI value of the samples were tested, and the samples were prepared with a die at 36.0 MPa, with a size of 80.0 mm × 6.0 mm × 3.0 mm. Then, adjusted the proportion of nitrogen and oxygen, ignited the top of the sample. The samples’ LOI were measured for 3 times and the average value was calculated.
The vertical burning (UL-94) test was performed using a vertical burning tester (ZY6017, Dongguan Zhongnuo Quality Inspection Equipment Co. Ltd, China). The samples were prepared with a die at 36.0 MPa, with sample dimensions of 80.0 mm × 13.0 mm × 3.0 mm. Then according to ASTM D-3801 standard the UL-94 values of the samples were measured 3 times and the average value was calculated.
Thermogravimetric analysis(TG) of the composites was carried out by thermogravimeter (TG 209 F1, Netzsch Scientific Instruments Trading (Shanghai) Co. Ltd, China). Under the nitrogen conditions, the gas flow rate of thermogravimeter was 40.0 mL/min, the sample dosage was between 3.0 mg and 5.0 mg, the heating rate was 10°C/min and the heating temperature was from 40°C to 800°C.
The combustion behavior of the samples was studied by a microscale combustion calorimeter (MCC, Suzhou Yangyi Volch Testing Technology Co. Ltd, China). About 5.0 mg sample was heated from 100 to 700°C at a heating rate of 0.5°C/s in a mixed stream of oxygen (20 cm3/min) and nitrogen (80 cm3/min).
Residual char analysis tests were performed on a SUPRA-55 (Carl Zeiss AG, Germany) Scanning Electron Microscope (SEM). The scanning accelerating voltage was 5 kV to investigate the morphologies of the residual char after complete combustion.
The smoke density was measured by using smoke density chamber (ZY6166B, Dongguan Zhongnuo Quality Inspection Equipment Co. Ltd, China) according to GB-T 8624-2007. The smoke generated (flameless combustion mode) during the combustion process of the samples was measured by the change of light intensity.
Results and discussion
Analysis of orthogonal experiment
To investigate the effects of the melamine adsorbed onto loofah surface on LOI variation, the following four factors were considered for synthesis optimization of M-FR-PL: the mass concentration of melamine (ρ m , g/L), loofah bath ratio (LBR), reaction temperature (T r , °C) and reaction time (t r , h). The experimental layout of L9(34) and results of AR and LOI for M-FR-PL samples are shown in Table 2. The LOI values of raw loofah and PL were 16.0% and 17.0% respectively. Range analysis method was used to analyzed the experimental data in Table 2. The subscript K represents a level of the factor. The range value of the four factors was determined by subtracting the minimum value from the maximum value of K. The larger the range, the more important the coefficient was. 23 The AR values of M-FR-PL1 and M-FR-PL3 in Table 2 were negative, which may be due to the dissolution of cellulose in water under acidic conditions and the experimental loss. The range analysis results were similar with AR and LOI. The results showed that the influence degree of various factors on the AR and LOI was as following: ρ m > t r > LBR > T r . According to the K value analysis and combined with the actual situation: the minimum temperature for the complete dissolution of melamine and phosphoric acid in the aqueous solution was 80.0°C; since changes in LBR have little effect on the results, it was of little practical significance to further reduce LBR. Therefore, the experimental conditions for obtaining the best AR and LOI were as follows: 6.0 g/L of ρ m , 1:100 of LBR, 80.0°C of T r , 2.0 h of t r . That was A2B3C1D2, which was the experimental condition of M-FR-PL6. Under the optimum experimental conditions, the quality of each component of the material prepared in this experiment was: 10 g of loofah, 6 g of melamine, 1:100 of LBR, 80.0°C of T r , 2.0 h of t r . After melamine flame retardant modification, compared with PL, the LOI of M-FR-PL6 was increased by 10.5% and the flame retardant performance of M-FR-PL was significantly improved. Due to the thermal decomposition of melamine to produce inert gas and dilute the oxygen around the sample, it can be quickly extinguished when igniting sample M-FR-PL6 in air, which plays the role of gas-phase flame retardant. 24 The other properties of M-FR-PL6 will be shown in the following sections.
FT-IR and XPS analysis
The FT-IR spectra of melamine, PL and M-FR-PL are presented in Figure 1. The peak at 3469 and 3419 cm−1 were ascribed to antisymmetric stretching vibration peaks of NH2 bonds; the absorption peaks at 3410 and 1653 cm−1 were attributed to the stretching vibration peaks of N-H and bending vibration peaks of N-H respectively. In addition, the peaks at 1555 and 1467 cm−1 occurred because of the skeleton vibration peaks of triazine ring.
25
The M-FR-PL modified by melamine retained the characteristic peak of PL and appeared the characteristic peak related to melamine. From these characteristic peaks, it can be inferred that the chemical structure of the compound is basically consistent with that of PL. FT-IR spectra of melamine, PL and M-FR-PL6.
As shown in Figure 2 and Figure 3, the XPS of PL and M-FR-PL and narrow-band spectra of C1s, O1s and N1s also proved that melamine successfully modified loofah. From Figure 2, both PL and M-FR-PL contained two obvious O1s and C1s characteristic peaks. However, after melamine flame retardant modified loofah, M-FR-PL6 has a new peak attributed to N1s at 399.08 eV. The appearance of nitrogen-containing groups shows that melamine had been successfully attached to the surface of loofah complex. It can be seen from Figure 3, the N1s spectrum of M-FR-PL6 shows two peaks at 397.7 and 398.7 eV (Figure 3(e)), which are ascribed to N and C-NH2 in the melamine ring, respectively. The results show that melamine is successfully loaded on loofah surface. XPS full spectrum of PL and M-FR-PL6. XPS narrowband spectra of PL and M-FR-PL6: (a)C1s and (b)O1s narrowband spectra of PL respectively; (c)C1s, (d)O1s and (e)N1s narrowband spectra of M-FR-PL6 respectively.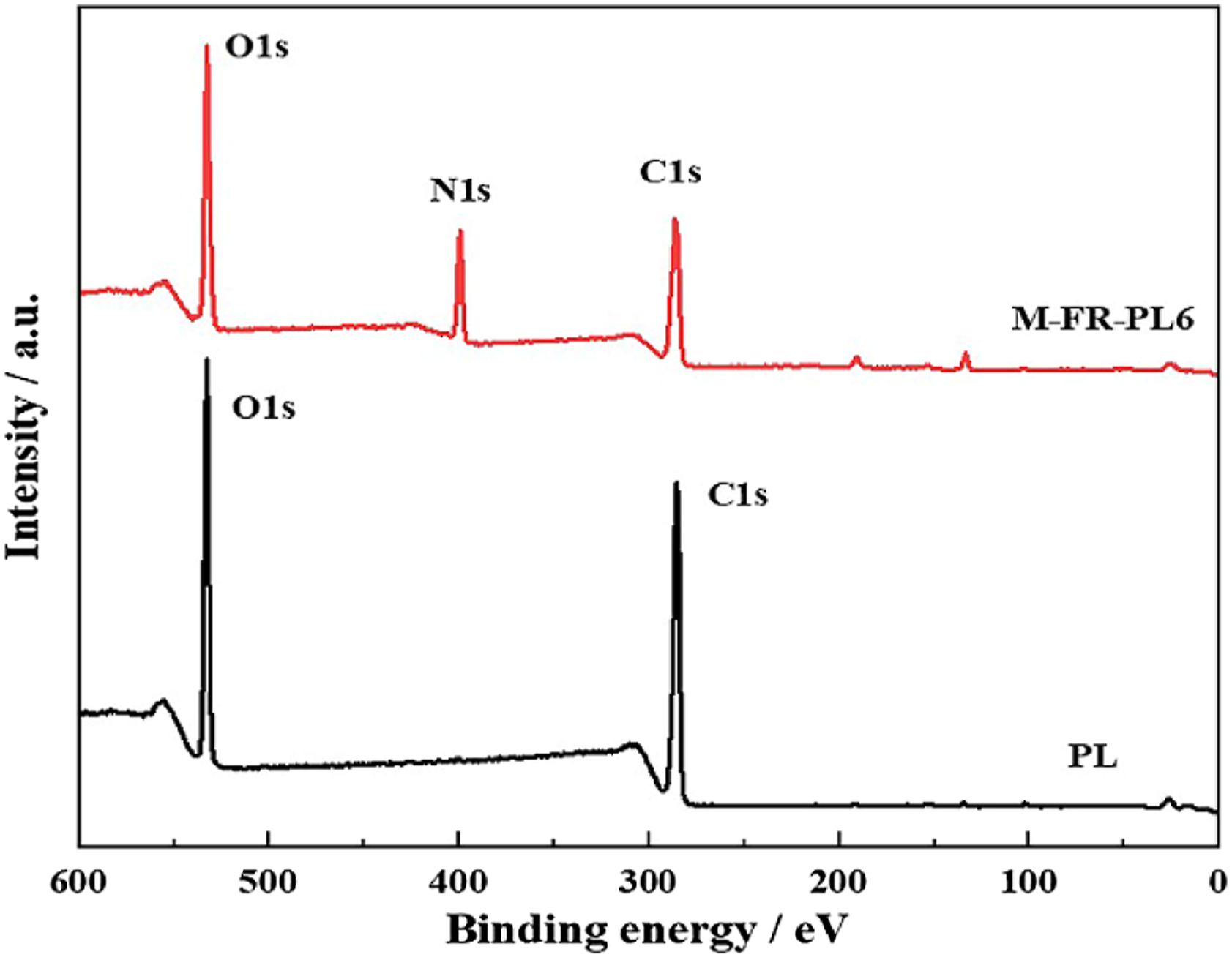

The vertical burning (UL-94) analysis
Vertical burning (UL-94) results including adsorption rate (AR).


Sample M-FR-PL6 before and after combustion: (a)before and (b)after.
Thermogravimetric analysis
The TG and DTG profiles of loofah, PL and M-FR-PL6 are displayed in Figure 5. The initial thermal decomposition temperatures (T5%), the 50 wt% weight loss temperatures (T
50%
), and the temperature (T
max
) at the maximum decomposition rate (R
max
) are summarized in Table 4. The value of T
max
is determined from the point where the slope of the thermogravimetric curve changes sharply. The T
5wt%
, T
50wt%
, T
max
and R
max
results of PL were all lower than those of loofah. The results indicated that the removal of gum and wax from loofah reduced the thermal stability of loofah and the maximum decomposition rate. The alkali treated loofah will be benefit for melamine adsorption after the removal of lignin, hemicellulose and waxy substances from the fiber.
26
The T
5wt%
of M-FR-PL6 was less than that of loofah, and T
50wt%
was greater than that of loofah. This showed that the thermal decomposition rate of M-FR-PL6 was reduced compared with loofah. The TGA results indicated that the initial decomposition temperature of M-FR-PL6 was lower than loofah and PL. It might be contributed to the thermal stability of P-O-C was lower than that of C-C.
27
Thermogravimetric analysis curves of samples: (a)TGA and (b)DTG. Thermogravimetric analysis results.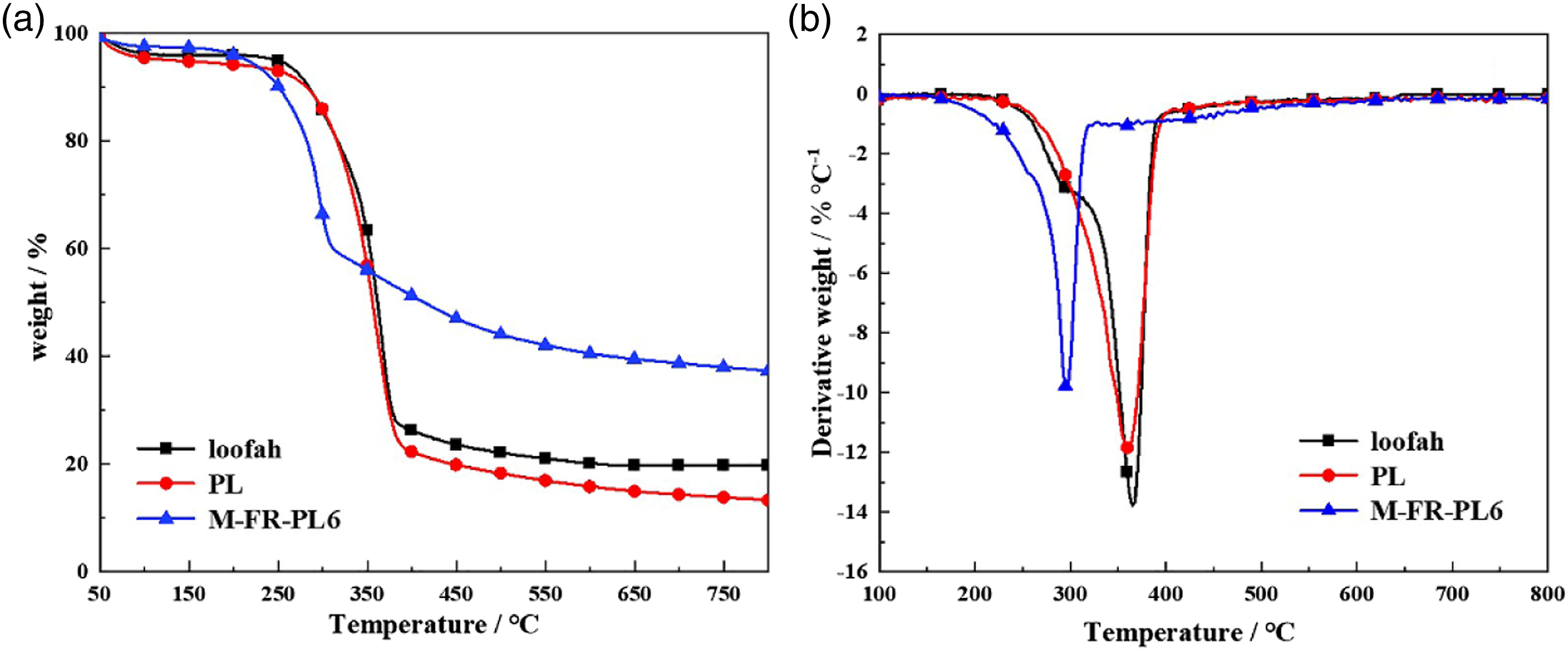

There may be preferential decomposition of phosphorus containing components in flame retardants, which can promote the formation of char layer. 28 The final residual char ratio was M-FR-PL6 > loofah > PL. The final residual char ratio of M-FR-PL6 was 17.2% higher than loofah and 23.2% higher than PL. Melamine could promote the formation of residual char during the thermal decomposition process, and generate nitrogen gas, which takes away part of the heat while blocking the oxygen supply. 29 The final residual char ratio of PL was lower than that of loofah, which was due to the removal of the gum and lignin in the loofah.
Microcalorimetric analysis
Microscale combustion calorimeter (MCC) data of the samples.


Heat release rate curves of samples.
SEM analysis
The digital photo of loofah and its fiber structure is shown in Figure 7, the density distribution of fibers is uneven and the diameter is different. The presence of lumen in the loofah indicates that the porosity of the sample increases when the number of cellulose increases.
31
The SEM micrographs of loofah, PL and M-FR-PL6 are shown in Figure 8. The surfaces of loofah were relatively smooth with shallow grooves Figure 8, The structure is attributed to the colloid, wax and other substances existed in the outer layer. The SEM micrographs indicate that alkali treatment made the surface of the loofah smoother.
32
From Figure 8 (c) the melamine particles attached to the fiber surface were clearly and confirmed the successful preparation of flame-retardant loofah composites. Digital photos of loofah and its fiber structure. SEM of (a)loofah, (b)PL and (c)M-FR-PL6.

The SEM images of residual char after samples combustion are displayed in Figure 9. The residual char surface of loofah after combustion was wavy, with many holes and relatively loose distribution. Although the residual char pores of PL were reduced, the problems of uneven and loose surface still exist. The removal of gum, wax, hemicellulose and lignin from loofah can improve the surface morphology of loofah fibers.
33
The residual char surface of M-FR-PL6 was more smooth and dense. There were many closely packed continuous char layers. The triazine ring existed in melamine was used as a starting char point, which could quickly form the residual char of the polymerized aromatic ring and the polymerized nitrogen heterocyclic ring. The tightly stacked char layer could well isolate oxygen and heat and improve the flame retardancy of M-FR-PL6. Thus, on the basis of the aforementioned results of FTIR, XPS, MCC, TG and SEM analyses, a possible mechanism of the flame retardancy of M-FR-PL is presented in Figure 10. The addition of melamine flame retardant improved the flame-retardant performance of the flame-retardant loofah composites. SEM diagrams of residual char of samples: (a) loofah, (b) PL, and (c) M-FR-PL6. Mechanism of the flame retardancy of M-FR-PL6.

Smoke density analysis
Smoke density analysis results.


Transmittance curves of samples.
Melamine is a kind of gas phase flame retardant, and the function of the gas phase flame retardant is mainly to remove active free radicals (such as H•, OH• and O•) that contribute to flame propagation. 36 This effect can also inhibit the oxidation reaction of the smoke precursor formed during combustion and be beneficial to the decrease of smoke production of M-FR-PL6. Compared with PL, the transmittance at 4 min of M-FR-PL6 decreased by 7.2%, while Dsmax increased by 0.70. Moreover, M-FR-PL6 possessed a slower smoke release rate compared with PL, which also reflect a good smoke suppression performance. 37
Conclusions
The melamine-modified flame retardant loofah composites were prepared by impregnation baking method and the orthogonal design method was used for optimization of the experimental preparation conditions. The following four factors: the mass concentration of melamine (ρ m , g/L), loofah bath ratio (LBR), reaction temperature (T r , °C) and reaction time (t r , h) were discussed. The adsorption amount of melamine has great effect on the flame retardancy of the composites. The results showed that the loofah prepared under the following optimum experimental conditions (M-FR-PL6) had optimal flame retardant performance: ρ m was 6.0 g/L, LBR was 1:100, T r was 80.0 °Cand t r was 2.0 h. Compared with loofah, the flame retardant performance of M-FR-PL6 was significantly improved: LOI was increased by 11.5%, UL-94 reached V0, and the value of Ds max decreased by 128.4. Meanwhile, MCC results indicated that the heat release of loofah was significantly reduced after melamine modification. M-FR-PL6 could form a dense char layer to isolate oxygen and inhibit flame spread during the combustion process. The melamine-modified loofah composites possessed good flame retardancy and smoke suppression performance, which will be beneficial for its application in fire-proof building materials, household products, thermal insulation materials and other fields in the future. In this study, since the limitation of flame-retardant adsorption onto loofah, it is difficult to further improve the flame retardant performance by increasing the flame-retardant concentration.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was financially supported by the National Natural Science Foundation of China (No.21878026), Beijing Key Laboratory of Quality Evaluation Technology for Hygiene and Safety of Plastics (QETHSP2021008), 2020 Industrial Technology Foundation Public Service Platform Project (2020-0107-3-1), Postgraduate Research & Practice Innovation Program of Jiangsu Province (KYCX21_2881, KYCX20_2585), Jiangsu Province University Blue Project, and Project of Higher Education Reform in Jiangsu Province (2019JSJG022).

