Abstract
Melamine formaldehyde (MF) resin is a thermosetting polymer with superior flame retardancy and high thermal stability and is widely used in fire-retardant coatings, flame-retardant sheets, heat-resistant filter materials, and so on. However, its further application is restricted by its extreme brittleness and poor impact strength. In this article, polyvinyl alcohol (PVA), a water-soluble polymer with high toughness and elasticity, which can be well blended with MF prepolymer in hot water, was used to improve the toughness of MF resin. The nano-silicon dioxide (SiO2) particles modified by KH-550 were used to improve the compatibility between MF prepolymer and PVA. Moreover, the modified nano-SiO2 can simultaneously enhance the toughness and impact the strength of MF resin. The interaction of components, fractography, thermal stability, flame retardancy, and mechanical properties of the modified MF resin was studied systematically. Fourier transform infrared results indicate that KH-550 was successfully grafted onto the surface of nano-SiO2. The grafted nano-SiO2 shows better dispersion in the matrix than the unmodified one, furthermore improving the compatibility and interface adhesion between MF and PVA. When PVA content is 20%, the obtained material has good thermal stability (T max > 350°C), flame-retardant properties (UL-94 V-0 rating and limited oxygen index = 42%), and toughness (elongation at break > 20% and unnotched impact strength > 13 kJ m−2).
Introduction
Melamine formaldehyde (MF) resin has been widely used in industry and our daily lives, mainly as flame-retardant materials because of its high thermal stability, low thermal conductivity, self-extinguishing performance, low smoke emission, nonformation of molten drops, and noncontraction in flames. 1 -3 With personnel and property losses caused by fire increasing all over the world, the requirements for flame-retardant materials continue to improve year-by-year. Benefiting from excellent flame retardancy, MF resin has aroused much interest in the industry and research fields. 4 However, due to the 3-D nonlinear structures of triazine rings joined by methylene and dimethylene ether, poor toughness is a critical drawback to MF resin, which severely limits its further applications, such as fiber, foam, and so on. 5 To overcome this contradiction, many studies have been carried out, indicating that blending with a flexible polymer like polyvinyl alcohol (PVA) and polyethylene glycol (PEG) was a practical way. 1,6 Moreover, considering the multihydroxyls of PVA and the water solubility of PVA and MF prepolymer, PVA is usually used as a toughening agent for MF resin. 7 -9 However, the low compatibility between MF resin and PVA at room temperature will always result in phase separation, thereby reducing the mechanical properties of the prepared material. 10 Therefore, the MF/PVA composite fiber must be produced as soon as the MF prepolymer/PVA blend is prepared; 11 to increase its toughness (the elongation at break > 30%), relatively large volumes (50 wt%) of PVA must be used as raw materials. 9 Yu et al. investigated the effect of the multiphase structure of the MF/PVA blend system on the properties of MF/PVA composite fiber. 8 Xu et al. reported that the MF resins with different hydroxymethylation degree have different compatibilities with PVA, and MF/PVA composite fibers were prepared by wetting spinning. 9,10 Kakkar et al. found that MF/PVA composite film formed by casting the blend of MF resin and a small proportion of PVA has a high level of antimicrobial activity. 12,13
PVA, a typical water-soluble polymer, shows high toughness and is convenient for mixing with hydrosoluble MF prepolymer. 14,15 Nevertheless, MF prepolymer water solution has poor miscibility with PVA at room temperature. In addition, numerous studies have shown that the toughness of polymer composites can be considerably improved by introducing nano-silicon dioxide (SiO2). 16 -21 In this work, MF resin composites with improved toughness and high flame resistance were successfully prepared through compounding MF prepolymer with a relatively small amount of PVA and 3-aminopropyltriethoxysilane (KH-550) grafted nano-SiO2. Modified nano-SiO2 can greatly enhance the compatibility of the MF prepolymer and PVA, which promote the storage stability of the MF prepolymer/PVA mixture and the interfacial bonding between MF resin and PVA in the final material. The results show that the modified MF resins with high thermal stability, good flame retardancy, and enhanced toughness were obtained. And the precured MF/nano-SiO2/PVA mixture is suitable for using as the spinning solution process for the production of MF composite fiber.
Experiment
Material
Melamine was supplied by Chengdu Yulong Chemical Co., Ltd (China). Paraformaldehyde, trimethylamine, glacial acetic acid, KH-550, and deionized water were obtained from Kelong Chemical Industries Reagent Co., Ltd (China). Nanosilica (TG96) was purchased from Jingjiang Tonggao Chemical Co., Ltd (China). PVA (1799) was provided by Sinopec Sichuan Vinylon Works Chemical Plant (China).
Preparation
KH-550 volume fraction 15.75%, deionized water volume fraction 15.75%, and anhydrous ethanol volume fraction 68.5% were mixed in a 100-mL glass beaker with a magnetic stirrer. KH-550 was hydrolyzed at room temperature for 1 h under stirring and then a colorless transparent solution was obtained. Meanwhile, nano-SiO2 was treated in a muffle furnace at 200°C for 2 h. Calcined nano-SiO2 and toluene dried over 3-Å molecular sieves were added to a 500-mL three-necked round-bottomed flask, which was equipped with a constant pressure dropping funnel, a mercury thermometer, and a mechanical agitator. At 80°C, the hydrolyzed KH-550 was slowly dropped into the mixture of nano-SiO2 and toluene under agitation and refluxed for 4 h. With the hydrolyzed KH-550 added, the formed liquid gradually turned into the pale yellow transparent solution. The modified SiO2 nanoparticles were filtered and washed with absolute alcohol for five times, after that, they were dried in a vacuum oven at 120°C for 8 h.
Modified SiO2 nanopowders (2% of the added solid materials), PVA (5%, 10%, 15%, and 20% of the added solid materials), 101 g melamine, 48 g paraformaldehyde, and proper amount of deionized water were mixed in a 500-mL three-necked flask fitted with a reflux condensing tube, a thermograph, and an electric blender. The mixture was adjusted to pH 9–10 with triethylamine and agitated at 80°C under ultrasonic waves for 4 h, and a “foggy” solution including MF prepolymer, modified nanosilica, PVA, and deionized water was formed, as can be seen from Figure 1(b). After this operation, a small amount of acetic acid was dropped into the flask, blended for a few minutes. Next, the resulting composites slurry were poured into polytetrafluoroethylene molds. Finally, the molds were put into a blower oven and heated at 60°C for 5 h. Under this condition, the cross-linking reaction of MF prepolymer occurred and the composites were obtained after solvent drying.

Photographs of the (a) suspension and (b) misty solution.
A series of MF/modified nano-SiO2/PVA composites, MF resin, and MF/modified nano-SiO2 composites have been fabricated. MF resin and MF/nano-SiO2 were used as contrastive samples. Their production progress was almost the same as for making MF/nano-SiO2/PVA composites. The differences among them are just the raw material ingredients whether contained nanosilica and/or PVA. The proportions of nano-SiO2 powder added were 2% and that of PVA were 5%, 10%, 15%, and 20% of the amount of added solid, and the corresponding products were named MFNSPVA5, MFNSPVA10, MFNSPVA15, and MFNSPVA20, respectively. The control samples named MF and MFNS did not contain nano-SiO2 and/or PVA. The sample names including abbreviations were listed in Table 1.
Abbreviated names of the samples and their weight percentage of the total solid content.

SiO2: silicon dioxide; PVA: polyvinyl alcohol.
Measurements
A Nicolet 6700 Fourier transform infrared (FTIR) Spectrometer (Thermo Fisher Scientific, Waltham, MA, USA) was utilized to identify the chemical structure of the pure MF resin, MF/nano-SiO2 composite, and MF/nano-SiO2/PVA composites, which were ground with potassium bromide in advance.
The microfractography of the samples was observed by scanning electron microscopy (SEM, JEOL JSM-5900LV, Tokyo, Japan) with a thin gold layer at an accelerating voltage of 10 kV.
Wide-angle X-ray scattering was carried out using two-circle diffractometer X-ray diffraction (XRD) 3003 θ/θ (GE Inspection Technologies/Seifert-FPM, Freiberg, Germany), with copper K α radiation (K = 0.154 nm) at 40 kV and 30 mA in 2θ range of 10–60°.
The thermal stabilities were characterized with a TA Q-500 (TA Instruments, New Castle, DE, USA) thermogravimetric analysis (TGA) thermal analyzer under nitrogen (N2) as a purge gas with a flow rate of 40 mL min−1. About 5 mg samples were placed in an alumina pan and measured from ambient temperature to 700°C with a heating rate of 10°C min−1.
Limited oxygen index (LOI) measurements were performed on the Oxygen Index Flammability Gauge JF-3 (Jiangning Analysis Instrument Co., Ltd, Nanjing, China) according to ASTMD2863-97. The dimensions of the samples were 130 × 6.5 × 3.2 mm3.
The UL-94 vertical burning level was measured on HK-HVRA (HK Detection Equipment Co., Ltd, Zhuhai, China) instrument according to ASTMD3801. The samples had a size of 130 × 13 × 3.2 mm3.
Tensile strength of the samples was tested at room temperature using a universal testing machine RGM-4010 (Reger Instrument Co., Ltd, Shenzhen, China) according to ASTMD638.
The unnotched Izod impact strength of the samples was conducted with Izod impact machine XBJ-7.5/11 (Testing Machine Co., Ltd, Chang Chun, China) according to ASTMD256.
The dynamic mechanical properties of the samples were examined by a dynamic mechanical thermal analysis apparatus (TA dynamic mechanical analysis (DMA) Q800, TA Instruments, NewCastle, DE, USA) according to ASTMD 4065-2012. The specimens who had dimensions of 3 × 13 × 30 mm3 were measured by three-point bending mode in the temperature range from −20°C to160°C at a heating rate of 5°C min−1 under the frequency of 1 Hz.
Results and discussion
Interactions among MF, PVA, and nano-SiO2
The preparation processes of modified nano-SiO2 and the formation of MF resin are shown in Figure 2. The chemical structures of the used compounds and the composites were identified by FTIR (Figure 3 and Table 2). From the FTIR spectra of nano-SiO2 and KH-550 modified nano-SiO2 in Figure 3(a) and (b), the absorption peaks at 3423, 1090, 800, and 467 cm−1 in the IR spectra are ascribed to the vibrations of –OH and Si–O–Si groups in SiO2. The absorbance at 1390 cm−1 corresponds to the asymmetric stretching vibration peak of C–H. Two new absorption peaks at 2854 and 2922 cm−1 in Figure 3(b) are attributed to the vibration of –CH2. The band at 1544 cm−1 is due to KH-550 N–H in-plane bending vibration. 22 -24 These results suggest that KH-550 was successfully grafted onto the surface of nano-SiO2. Figure 3(c) shows an IR absorption spectrum of MF resin. The bands observed at 1494 cm−1 and 1339 cm−1 are related to C–N stretching vibration of triazine ring. The peak at 1160 cm−1 may be assigned to C–O or C–O–C stretching modes. 25,26 The above analysis indicates that MF resin was successfully prepared. FTIR spectrum of PVA is shown in Figure 3(d). IR spectrum of the composite MFNSPVA20 is shown in Figure 3(e). Because the peaks at 2917 cm−1(–CH2, PVA), 2942 cm−1(–CH, PVA), and 2972 cm−1 (–CH, MF prepolymer) overlap each other, one peak appears at 2967 cm−1 in the IR spectra of MFNSPVA20. The occurrence of peaks at 1023 cm−1 and 1258 cm−1 may be due to the superposition and splitting of the peaks at 999 cm−1 (–CH2OH, MF resin) and 1333 cm−1 (C–O, PVA). The peak around 1402 cm−1 is only observed in the IR spectra of MFNSPVA20, which maybe on account of the blue shift of C–O group in PVA (1333cm−1) forming hydrogen bonds to hydroxymethyl in MF resin.

Preparation of KH-550 modified nano-SiO2 and MF resin.

FTIR spectra of (a) nano-SiO2, (b) KH-550 modified nano-SiO2, (c) MF resin, (d) PVA, and (e) MFNSPVA20.
IR spectral data of nano-SiO2, KH-550 modified nano-SiO2, MF resin, and PVA.

MF: melamine formaldehyde; PVA: polyvinyl alcohol
The morphologies of MF, MF/nano-SiO2, MF/modified nano-SiO2, and MF/PVA/modified nano-SiO2 were investigated by SEM. As can be seen from Figure 4(a), the neat silica nanoparticles with a mean diameter of 1.34 µm (marked by yellow circles) were agglomerated in the MF matrix, while KH-550 modified nano-SiO2 particles with a mean diameter of 0.17 µm were dispersed homogeneously in Figure 4(c) to (g). It is obvious that the average size of the unmodified nano-SiO2 is bigger than that of modified nano-SiO2. The situation has arisen because nanosilica treated with KH-550 can reduce its surface energy and improve its dispersibility in the matrix. This shows that it is necessary to modify the surface of silica nanoparticles using KH-550. Both the size distributions of unmodified nano-SiO2 and modified nano-SiO2 are close to the Gauss distribution. With the increase of PVA content (from Figure 4(c) to (g)), it becomes more and more difficult to find nano-SiO2 in the fractography of the samples, which may be on account of its good dispersion in the MF resin and the PVA matrix. Note that there is a trend that the fractography smoothness of the samples reduces as the content of PVA increases. This may have occurred because the viscosity of the system increases with increasing the content of PVA, and then, the surface tension of the system increases. 27
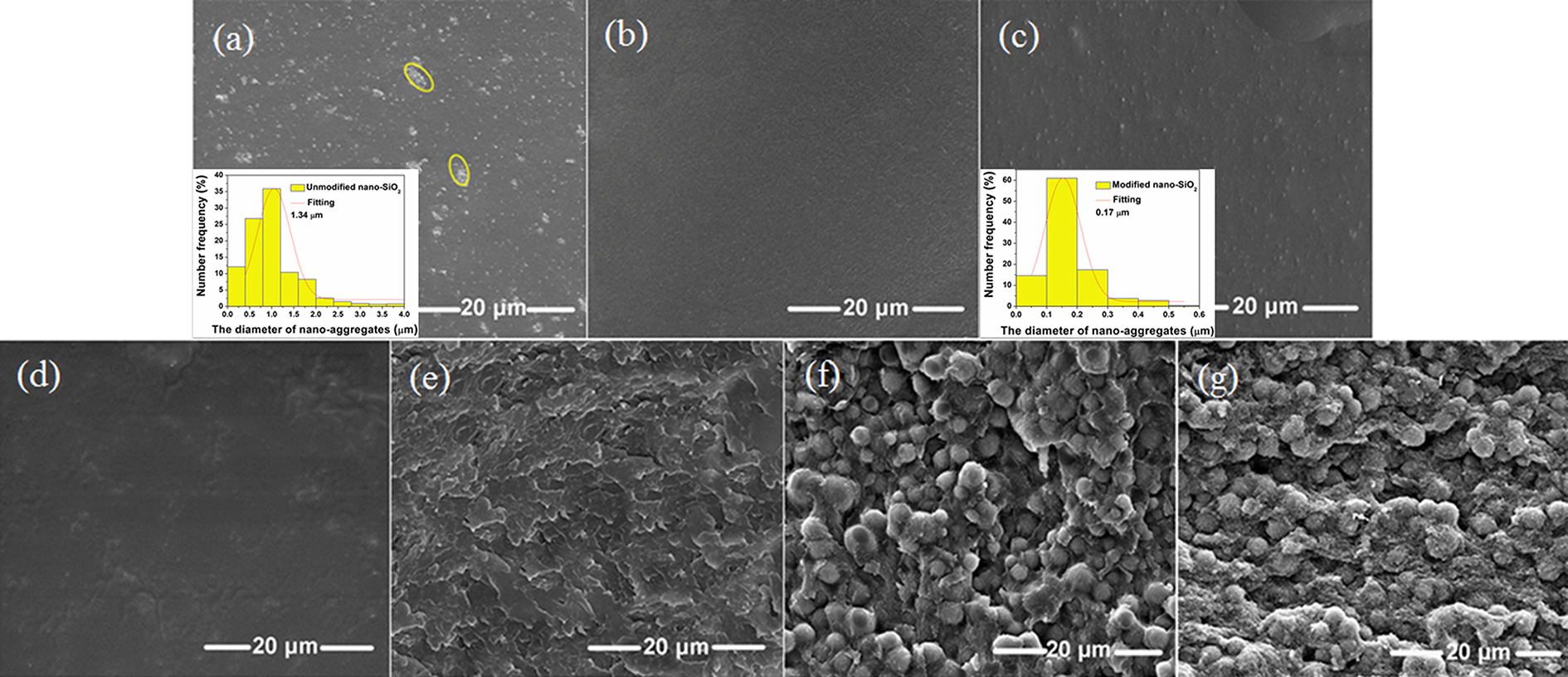
(a) SEM fractography of the composite containing unmodified nano-SiO2 and MF resin and the aggregate size distribution of unmodified nano-SiO2 in MF resin matrix; (b) SEM fractography of MF resin; (c) SEM fractography of MFNS and the aggregate size distribution of modified nano-SiO2 in the MF resin matrix; (d) MFNSPVA5; (e) MFNSPVA10; (f) MFNSPVA15; and (g) MFNSPVA20.
Figure 5 shows the XRD patterns of the samples. It cannot be seen by any obvious diffraction peaks of PVA, which would appear around 11.13° (100), 19.17° (101), 22.19° (200), 28° (201), 32.15° (002), and 40.19° (111), 28 owing to the 3-D skeleton networks of MF resin destroying the crystal structure of PVA. In other words, PVA in the prepared compounds was in an amorphous state. This result is also verified by the DMA results in Figure 9(b), because the Tg (at about 60–65°C) of PVA in MFNSPVAs can be easily observed.

XRD spectra of (a) MF resin, (b) MFNS, (c) MFNSPVA5, (d) MFNSPVA10, (e) MFNSPVA15, and (f) MFNSPVA20.
Thermal properties
Thermal degradation behaviors of MF, MFNS, and MFNSPVAs were studied by TGA under N2 atmosphere. Figure 6 shows their thermogravimetry (TGA) and differential thermal gravity (DTG) curves. The decomposition data of MF, MFNS, and MFNSPVAs were summarized in Table 3. From the TG and DTG curves, the pyrolysis of MFNSPVAs can be divided into three major stages. The first stage ranging from 105°C to 250°C is attributed to water and formaldehyde volatilization and thermal degradation of PVA; the second stage around 250°C to 450°C is due to the removal of hydroxyl groups in PVA chains and the destruction of triazine network structures; and the third stage in the temperature range from about 450°C to 600°C is related to the fracture of triazine rings. 29 For MF and MFNS, their pyrolysis also proceeds in three stages: the first from 300°C to 460°C, the second from 460°C to 560°C, and the third from 560°C to 700°C, and corresponds to the pyrolysis of triazine-based frameworks, the thermal cracking of triazine rings, and the carbonization of PVA and MF resin. Maybe because the interface bonding between functionalized nano-SiO2 and matrix was well, there are no apparent steps of decomposition in TG curves. The values of onset decomposition temperature (T 5%), temperatures at maximum rate of weight loss (T max), the rate of T max and the char residues gradually decrease with the increase of PVA content. The T 5% of MF is 180.1°C, lower than MFNS, because of the release of small molecules like formaldehyde and water. The values of T max are all above 350°C, which meant high thermal stability. The char yield of MFNS at 680°C was 30.9%, the highest among the materials, and the maximum decomposition rate of it is the lowest, about 0.41% °C−1, suggesting that nano-SiO2 and MF resin both have good heat resistance.

(a) TG and (b) DTG curves of MF, MFNS, and MFNSPVAs.
TGA and DTG results of MF, MFNS, and MFNSPVAs.

MF: melamine formaldehyde; PVA: polyvinyl alcohol; MFNSPVA: melamine formaldehyde resin/KH-550 modified nano-SiO2/PVA composite; MFNS: melamine formaldehyde resin/KH-550 modified nano-SiO2 composite; TGA: thermogravimetric analysis.
Flame retardancy
UL-94 vertical burning test and LOI measurement, as two common indicators, are frequently adopted to investigate the flame retardancy characteristics of materials. The vertical burning test procedure of MFNSPVA10 is shown in Figure 7. And the real combustion processes of other samples are similar: only red hot phenomena occur when the specimens are placed in a flame, without melt-dripping, once leave fire, the afterglow is extinguished immediately (no smoldering) with a small amount of smoke. All materials met UL-94 V-0 flammability rating at a thickness of 1.6 mm, and their LOI values are above 42% (Table 4), which is due to their main ingredient—MF resin having 3-D networks of the methylene- or/and oxymethylene-linked triazine ring. As is widely known, tertiary nitrogen triazine ring structures are very easy to form stable carbon layers during the burning, which can prevent flames from spreading; 30 moreover, the released small molecules such as water, carbon dioxide, and ammonia take away some of the heat, which promotes the carbonization of the matrix at the same time, hence, MF resin shows self-extinguishment. Therefore, LOI values decrease with the decreasing of the content of MF resin in the samples, and among them, MFNS has the highest LOI value of 49%. In comparison with MF and MFNSPVAs, MFNS shows better flame retardancy and thermal resistance due to the high thermal resistance of nanosilica.

Pictures of MFNSPVA10 during vertical burning test.
Flame retardance of MF, MFNS, and MFNSPVAs.

MF: melamine formaldehyde; PVA: polyvinyl alcohol; MFNSPVA: melamine formaldehyde resin/KH-550 modified nano-SiO2/PVA composite; MFNS: melamine formaldehyde resin/KH-550 modified nano-SiO2 composite; LOI: limited oxygen index; +: the amount of smoke.
Static and dynamic mechanical properties
Tensile strength, elongation at break, and impact strength of the specimens were presented in Table 5. Pure MF resin is very brittle and shows poor mechanical properties. Comparing with the mechanical data of pure MF resin, MFNS has higher strength and toughness, which is most likely caused by some compatibility and certain bonding force between the modified nano-SiO2 and the matrix, as shown in Figures 1 and 4. When a composite material is impacted by a foreign object, the inorganic rigid nanoparticles (modified nano-SiO2) absorb some of the energy of collision, which produces plastic deformations and microcracks or crazes among the matrices surrounding the particles. Moreover, if the microcracks or crazes propagate, the nanoparticles will hinder this occurrence and passivate crazes, so as to achieve a certain toughening effect. Furthermore, physical interaction and/or chemical bonding between modified nanosilica particles and matrix resin enhance interfacial adhesive effect, thereby the nanoparticles can bear a part of the load and then the stretching strength is increased. 31,32 With the increase of PVA content, the tensile strength of MFNSPVAs decreases gradually from 7.4 ± 0.5 MPa to 5.7 ± 0.3 MPa, while the elongation at break and unnotched impact of the samples increase from 5.7 ± 0.9% to 25.6 ± 4.7% and from 4.6 ± 0.3 kJ m−2 to 14.8 ± 1.1 kJ m−2, respectively. It is clear that the modified nano-SiO2 particles have positive impact on both tenacity and intensity of the materials, and the addition of PVA contributes significantly to improve their toughness and reduce brittleness and strength.
Mechanical properties of MF, MFNS, and MFNSPVAs.
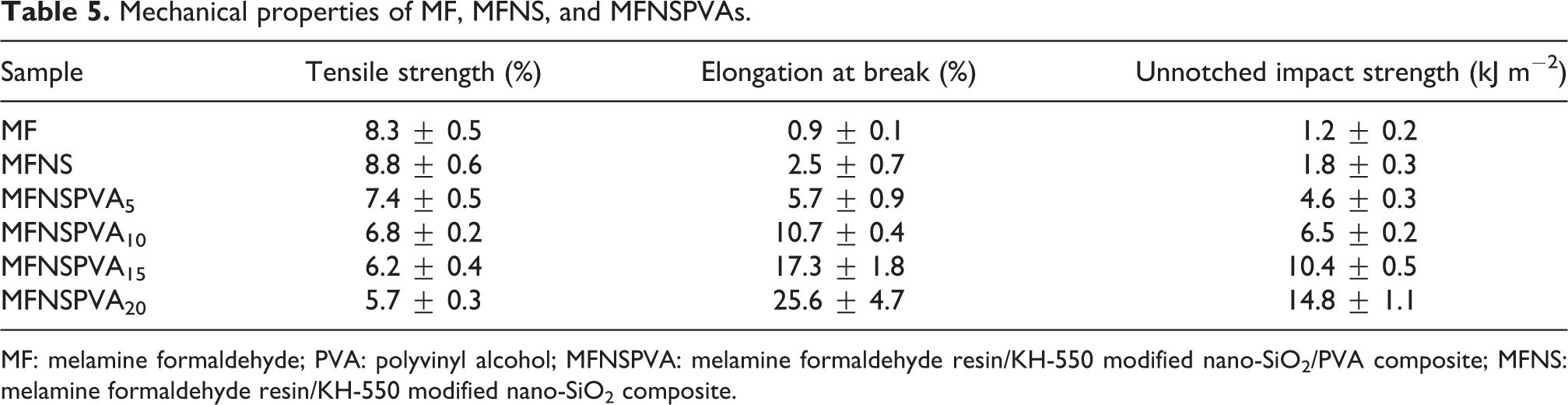
MF: melamine formaldehyde; PVA: polyvinyl alcohol; MFNSPVA: melamine formaldehyde resin/KH-550 modified nano-SiO2/PVA composite; MFNS: melamine formaldehyde resin/KH-550 modified nano-SiO2 composite.
The stress–strain curves (see Figure 8) indicate the decrease of tensile strength and increase of flexibility as PVA concentration increases. The reason is most likely that the –OH groups in the molecular chains of PVA react with the –OH and –NH2 groups of MF resin and nano-SiO2 particles, and hydrogen bonds can form between these groups. In general, MF, MFNS, and MFNSPVAs demonstrate brittle behavior. Because there are no apparent yield points and obvious plastic deformation stages in the curves, meanwhile, the specimens are pulled off when the strain is low. Although MFNSPVA20 still belongs to a brittleness material, contrasting to MF, its ductility has been improved to some extent from 0.9 ± 0.1% to 25.6 ± 4.7%.

Stress–strain curves of MF, MFNS, and MFNSPVAs.
To investigate the effect of nanofiller and PVA content on viscoelasticity of composite materials, the dynamic mechanical behaviors of MF, MFNS, and MFNSPVAs were measured using DMA. Figure 9(a) to (c) show storage modulus (E′), loss modulus (E″), and tan δ as a function of temperature for each sample in the temperature range of 20–155°C, respectively. It is seen from Figure 9(a) that the overall trend of storage modulus of the materials decreases as the temperature increases, and there are characteristic drops in storage modulus around the glass transition temperature (T g) of MF propolymer. Owing to incorporation of 2% nano-SiO2, the storage modulus values of MFNS are higher than that of MF, but that of MFNSPVAs decreases with the increasing concentration of PVA. In Figure 9(b), there are two loss modulus peaks (the one at about 60°C–65°C is the T g of PVA and the other at about 124°C–130°C is the T g of MF pro-polymer) on curves of MFNSPVAs and one peak on curves of MF and MFNS, which proves that PVA exists in an amorphous state in the sample and MF prepolymer has thermoplasticity. And the magnitude of loss modulus is in the order of MFNSPVA20 > MFNSPVA15 > MFNSPVA10 > MFNSPVA5 > MFNS > MF. As described in Figure 9(c), the magnitude of the tan δ peak gradually increases with the increasing of PVA—a viscoelastic polymer, whereas nanofillers have little effect on it. In addition, the interfacial bonding between MF, nano-SiO2, and PVA is well without obvious transition layer (Figure 4). Thus, when the composite materials are subjected under external force, PVA absorbs the vibration energy and converts mechanical energy into heat energy, and the toughness of the materials has been improved on the macro level.

DMA results for MF, MFNS, and MFNSPVAs.
Conclusion
In summary, this study provided a method of producing MF resin composites with improved toughness and high flame retardancy. Siloxane coupling agent KH-550 was successfully grafted onto the surface of nano-SiO2 and then incorporated into MF and PVA via ultrasonic dispersion in aqueous solution to produce MFNSPVA composite materials. The 5% weight loss temperatures and maximum thermal decomposition temperatures of MFNSPVAs decrease comparing with those of the MF resin. MFNSPVAs have good flame-retardant properties as the LOI values for all samples are above 42% and UL-94 reach V-0 rating (1.6 mm). Although the modified nano-SiO2 cannot greatly improve the flame retardancy and mechanical properties of pure MF resin, it is useful to promote the compatibility of MF prepolymer and PVA. Comparing with the pure MF resin and MFNS, MFNSPVAs display better comprehensive performance. Specially, MFNSPVA20 achieves UL-94 V-0 rating, LOI value of 42%, elongation at break of 25.6 ± 4.7%, and impact strength of 14.8 ± 1.1 kJ m−2, which can be used as the raw materials for preparation of MF composite fiber.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the National Key R&D Program of China (2017YFB0309001) and State Key Laboratory of Polymer Materials Engineering [Grant No. sklpme2018-3-01].
