Abstract
A novel melamine-phosphate tri-functional acrylate (MPTO) was successfully synthesized via cyclization reaction of hexamethylolmelamine (HMM) with phosphorous oxychloride (POCl3) followed by addition reaction of hydroxyethylmethacrylate (HEMA). The molecular structure of MPTO was identified by FTIR and 1H-NMR, 13C-NMR, and GC-MS spectra. The synthesized MPTO oligomer was impregnated with polyurethane acrylate to make various formulations and UV cured on wood and galvanized steel panels. The results of UV cured films from differential scanning calorimeter (DSC) and thermo-gravimetric analysis (TGA) exhibited increase in thermal stability and highest char yield of 18.4% at 800°C. The diffused diffraction peak appearing around 2ϴ = 16–22° corresponds to an amorphous structure which leads to irregular arrangement with formation of strong cross-linking polymer network. Furthermore, all coating films show prominent flame retardancy with UL-94 V-0 rating and maximum limiting index (LOI) values of 34.8%. The polyurethane acrylate (PU) coatings cured with MPTO that exhibited excellent mechanical properties were estimated using various tests such as adhesion, pencil hardness, solvent resistance, flexibility, and corrosion test. The coating performance revealed that MPTO can improve the mechanical, thermal, and flame retardant properties because of its unique structure identity of melamine-phosphate moiety and long aliphatic segment of an acrylate ester.

Introduction
The various treatments using resin or chemicals have been widely studied to enhance the properties of wood or metal surfaces. A variety of resin and polyurethane foam can be used to increase dimensional stability, thermal, mechanical, and chemical resistance. The systems used for coating include epoxy resin, phenolic resin, polyurethanes, melamine resin, melamine formaldehyde resin, and unsaturated polyesters.1–6 Epoxy and polyurethane acrylate have been widely used because of their outstanding performance toward adhesive, dielectric properties, microelectronic encapsulated materials, and other coating properties.7,8 The specific curing agents or nanoparticle are impregnated to obtained desired properties in the coating systems. The specific curing agents are responsible for increased cross-link network which has a major influence on the chemical and physical performance of the coating materials.9–11 As far as wood materials are being widely used in furniture, decorative, architectural use, and other devices. The fire degrades as well as certainly decomposes the wood material. Thus, the impregnation of flame retardants with polyurethane coating to the surface is an effective way to increase global flame retardancy. The commercial use of halogenated flame retardants has been restricted due to the emission of toxic gases during the curing process which affects the environment. As a result, the flame retardant compounds having high flame retardant properties but barely contaminate the environment are predominantly needed.12,13 The phosphorous-nitrogen-containing flame retardants are predominantly replacing halogenated flame retardant because these types of compounds perform condensed and vapor phase mechanism. In general, phosphorous-based flame retardant (-PO., -PO2., HPO2., etc.,) radicals act in gas-phase has a mechanism via inhibition of chain reaction. However, the phosphonates and phosphates act by promoting char residue exhibit as condensed phase mechanism. The phosphorous-nitrogen chemistry has been studied in the last decades owing to its capability to reduce flammability.14–17 It is well known that in fire, the destruction of products occurs with the evolution of smoke and many toxic gases. Fire retardant polyurethane is preferred due to minimum or not producing smoke while they are burning. Hence, polyurethane coating is used in a variety of products like rubbery, plastics, adhesive, insulation, and packaging industries. 18
In the earlier reports it was found that, melamine polyphosphate and melamine cyanurate act as effective flame retardants in various polymeric coatings. The coating materials include ethyl-vinyl acetate copolymer, polypropylene, epoxy compounds, polyamides, and melamine polyphosphate in polyurea’s.19–22 There is narrow research on the application of melamine derivatives as flame retardant materials in polyurethane foam. The effects of additive ammonium polyphosphate mixed with the melamine cyanurate on physical-mechanical properties and flame retardant performance of polyisocyanurate–polyurethane foams blown with n-pentane.23,24 The hyperbranched polymers have a faster curing rate and lower viscosity as compared with linear co-polymer and have great applications in UV-curable coating systems.25–27
Here, novel tri-functional melamine-phosphate oligomer was design and synthesized via melamine diol as a monomer followed by the reaction of (hydroxyethyl)-methacrylate (HEMA). Synthesis of the tri-functional polymer (MPTO) via polymerization of AA’A’’ monomer with B3 monomer was achieved with higher conversion. It has very high commercial value for monomers and gives superficial preparation and easy-to-tailor structures of desired products. 28 Then it was impregnated into polyurethane acrylate to prepare various coating formulations and executed in a UV-curing system. The flame retardant properties were studied by vertical burning test and limiting oxygen index (LOI). Thermal degradation was analyzed by thermogravimetric analysis (TGA) and differential scanning calorimeter (DSC).
Experimental
Materials
Melamine was purchased from Gujarat chemicals, Gujarat, India. Formaldehyde solution (37%) and phosphorous oxychloride were purchased from Sigma Aldrich, Mumbai, Maharashtra, India. Hydroxyethyl methacrylate, triethylamine, and methylene dichloride were purchased from Merck India Pvt. Ltd. All the chemicals were used without further purification in the synthesis of MTPO, and distilled water was received from Pratap College Amalner, Maharashtra, India, for all the experiments. Commercial polyurethane acrylate and galvanized steel panels used as a substrate for the coatings were provided by Champion Advance materials Pvt. Ltd. Jhagadia, Gujarat.
Synthesis of hexamethylolmelamine
In four-necked round bottom flask equipped with a magnetic stirrer, charge melamine (10.0 g, 0.079 mmol) and formaldehyde solution (37%) (142.72 g, 1.42 mmol). Stir reaction mass for 10 min and adjusted pH to 8.5 to 9.5 using 10% sodium hydroxide (1.2 mL) solution. Stir and maintain the reaction temperature at 60–65°C. Clear reaction mass gets solidify represents the HMM formation monitor by using thin-layer chromatography. Cool the reaction mass to 25–30°C. The product was filtered and washed with distilled water several times, and then dried in oven at 70°C for 8h to obtained white product (yield: 93.54%).29,30 The reaction scheme for the synthesis of HMM is shown in Scheme 1. Synthetic route of hexamethylolmelamine.
Synthesis of HMC
In 50 mL three-necked flask equipped with stirring rod, thermometer sensor, and reflux condenser, charge HMM (5.0 g, 0.016 mol) and add phosphorous oxychloride (45.06 g, 0.293 mmol). Stir and heat the reaction mass to 75–80°C for 4h. Distilling out excess phosphorous oxychloride under vacuum at 50–55°C gives pale yellow color oil as product of HMC (yield: 98.87%). The literature found that HMC is chloro-compound which is highly reactive and moisture sensitive. Hence it is used in the next stage immediately.
31
The reaction scheme for the synthesis of HMC is shown in Scheme 2. Synthetic route of HMC.
Synthesis of MTPO
In 100 mL three-necked flask equipped with stirring rod, thermometer sensor, and nitrogen inlet, charge hydroxyethyl methacrylate (5.09 g, 0.039 mmol), charge methylene dichloride (50 mL), and triethylamine (7.89 g, 0.087 mmol). Stir and cool the reaction mass to 5–10°C, and add HMC (5.0 g, 0.006 mmol) drop wise over 10–15 min. Monitor the progress of the reaction by using thin-layer chromatography (Mobile phase—methylene dichloride: ethanol 8.5:1.5) and charge distilled water (100 mL) on completion of the reaction. Separate the lower organic layer and wash with distilled water. Finally, the clear pale yellow oil as a product (MTPO) was obtained on evaporation of the solvent (yield 91.35%). The reaction scheme for the synthesis of tri-functional melamine-phosphate oligomer (MPTO) is shown in Scheme 3. Synthetic route of melamine-phosphate tri-functional acrylate
Synthesis of cured polyurethane acrylate (PU-MPTO)
Formulations of polyurethane acrylate with MPTO.

Characterization
The chemical structure of HMM and MPTO was identified by Fourier transform infrared (FTIR) on Shimadzo 8400s. The spectra were scan from 4000 to 400 cm-1 wavelength range. The structural conformation of HMM and MPTO was done by gas-chromatography mass spectral analysis (GC-MS) on Shimadzu (QP 2010. The proton nuclear magnetic resonance (1H-NMR, 13C-NMR) spectra were conducted on Advance AV500WB Bruker at 400 MHz spectrophotometers in solvent CDCl3.
Thermogravimetric analysis (TGA) of UV-cured films was performed on TGA Q500 V20.13 build 39, Universal V4.5A TA instrument under the nitrogen atmosphere. Thermal analysis monitored in the temperature range 0–800°C with 10 °C/min heating rate. The glass transition temperature (Tg) of UV-cured films was performed via differential scanning calorimetry (DSC) (TA Q100 analyzer, USA). The film sample was weighed accurately in an aluminum pan and heated in a 0–100°C temperature range with a heating rate of 5 °C/min. The flame retardancy of coated films was analyzed by using the underwriter’s laboratories (UL-94) vertical burning test and limiting oxygen index (LOI) measurements as per ASTM D3801 and ASTM D 2863–97, respectively. The qualitative methods for flammability and thermal behavior of the materials were achieved by LOI analysis. The coated films were ignited on the LPG Bunsen burner vertically and ignited for 10 s. The coating’s performance was evaluated by various mechanical properties as per ASTM standards. The adhesion of coating panels was examined according to the cross-cut test ASTM D-3359. On the surface of coated panels, 1 cm2 lattice marking was made until the surface was exposed over the adhesion tape. The adhesion tape was then dragged over the surface and failure in adhesion was observed on the lattice marking. The pencil hardness test was performed according to ASTM D-3363. Scratch was made over the panels using pencil 6B–6H range with an angle of 45°. The performance on the coatings was carried by the weighted ball of 1.46 kg ranging from extreme height 60 cm over the surface. Rhopoint gloss meter was used to perform gloss of coated panels at 60° angle according to ASTM D523-99. The gel content of UV-cured films was determined as per ASTM D2765-16. The gel content of the cured film was weighed carefully deep in xylene at room temperature for 24 h and then the films were removed and dried at 75°C until constant weight achieved. The contact angle measurement of UV-cured PU-MPTO coatings was analyzed by using Standard Goniometer, Model 21 AC, Rame-Hart instruments company, New jersey, USA, by applying sessile drop method.
Results and Discussion
Structural Characterization
Functional group analysis by FTIR
The FTIR spectra of HMM are shown in Figure 1. The spectra of (1,3,5-triazine-2,4,6-triyltrinitrilo) hexamethanol (HMM) show a characteristic absorption band at 3199.91 cm−1 corresponds to stretching vibrations of alcohol -O-H. The peak at 2962.67 cm−1 is due to the C-H stretching vibration of N-CH2 group. The sharp peak at 1554.63 cm−1 is due to -C=N ring vibration. The peak at 1492.91 cm−1 and 1390.68 cm−1 corresponds to the methylene -C–H bending vibrations. The absorbance peak at 1193.94 cm−1 corresponds to aliphatic -C–N stretching vibration. The peak observed at 975.98 cm−1 and 869.9 cm−1 corresponds to the out-of-plane bending vibrations of the triazine ring of melamine derivative.
32
From this spectrum of HMM, it is concluded that the melamine has completely reacted with formaldehyde and there is no residue of melamine and formaldehyde. Fourier transform infrared spectra of hexamethylolmelamine.
The FTIR spectrum of MPTO is shown in Figure 2. The weak absorption peak at 3427.51 cm−1 corresponds to =C-H stretching of terminal methylene group of MPTO. Furthermore, methylene bending vibration of –C-H appeared at 1454.33 cm−1. The strong absorbance at 1714.72 cm−1 corresponds to stretching vibrations of –C=O ester group. The characteristic peaks obtained at 1296.17 cm−1 and 1319.31 cm−1 are of -P=O, and the peaks at 1161.15 cm-1 attributed to the absorption of -C–O–C- groups.
33
Fourier transform infrared spectra of melamine-phosphate tri-functional acrylate.
There is a decrease in the intensity of the peaks at 3427.51 cm−1 and 1161.15 cm−1 that corresponds to the terminal methylene stretching vibrations. This decrease in intensity may be due to the extent of cross-linking. From these FTIR spectra, it was confirmed that the MTPO has reacted with polyurethane acrylate and formed strong cross-linking during the UV-curing process. The FTIR spectra of HMM, MPTO, and UV-cured film are shown in Figure 3. Fourier transform infrared spectra of hexamethylolmelamine, melamine-phosphate tri-functional acrylate, and UV cured PU-MPTO.
Structure confirmation of MPTO using 1H-NMR and 13C-NMR
The structural conformation of MPTO was given by 1H-NMR spectroscopy, and the spectra obtained are shown in Figure 4. The singlet around 1.96–2.0 ppm (a, 9H) is associated with the terminal methyl groups of olefinic carbon. The characteristic peaks of protons to attached O-CH2-CH2-O linkage observed triplet in the region at 3.72–3.88 ppm (d, 6H), similarly another triplet at 4.28–4.20 ppm (e, 6H). Supplementary Figure S2 shows brief expansion of MPTO. The methylene protons of the cyclic ring show peaks at 5.80–6.15 ppm (c, 12H) doublet due to germinal coupling. The peak at 7.27 ppm attributed to the olefinic protons of MTPO terminals. However, from the data of 1H-NMR spectra, it is concluded that MPTO was successfully synthesized. 1H-NMR of melamine-phosphate tri-functional acrylate in DMSO.
Figure 5 shows 13C-NMR spectra of MPTO. The chemical shift value of terminal carbon of methyl group is at 18.42 ppm (C1). The intense peaks observed at 61.3 ppm (C4) are due to -CH2 bonded to oxygen atoms, while 66.51ppm (C5) is due to a carbon atom of a cyclic ring. The chemical shift values assign for olefinic carbon atoms at 126.18ppm (C2). The tertiary carbon attached to methyl shows the peak at deshielded region around 136.19ppm (C3) and carbon atoms of the heterocyclic aromatic ring show peak at 167.93ppm (C6). Hence, from the spectra of 13C-NMR spectra, it is concluded that MPTO was successfully synthesized. 13C-NMR of melamine-phosphate tri-functional acrylate.
Structural confirmation by Gas Chromatography-Mass spectroscopy (GC-MS)
Figure 6 reveals the GC-MS spectrum of HMM. The molecular ion peak observed at m/z = 306.89 indicates the formation of melamine-hexanediol having a molecular weight of 306.27. Hence it confirms that all primary amines of melamine that reacted with formaldehyde converted to HMM. Figure 7 shows the GC-MS spectra of MPTO. The peak at maximum intensity m/z = 827.53 confirms the formation of tri-functional MPTO having molecular formula weight of 828.54. Thus, from mass spectral data, the structure of oligomer MPTO was confirmed. GC-MS spectrum of hexamethylolmelamine. GC-MS spectra of melamine-phosphate tri-functional acrylate.

Thermal analysis of the cured films
The thermal decomposition of the cured PU-MPTO systems was studied via thermogravimetric analysis (TGA) as shown in Figure 8. The specific degradation temperatures and the final char yield at 800°C are summarized in Table 2. Thermogravimetric analysis of cured films. Thermal degradation of cured PU-MPTO coatings.

As seen, the onset decomposition temperature of MPTO was 276.7°C and presented mainly three-stage decompositions with 15 wt% residual mass at 428.3°C. The temperatures at the maximum mass loss rate (Tmax) of MPTO were 279.6°C, 398°C, and 502°C, respectively. The thermal behavior of the cure PU-MPTO coatings was studied by the TGA. All the cured PU-MPTO coatings showed three steps of degradation pattern. The first, second, and third steps of decompositions are shown in Table 2. All cured PU-MPTO show the first, second, and third step decomposition at temperature ranged from 250°C to 304°C, 371°C to 426°C, and 453 to 539°C, respectively. The degradation in the first initial step was obtained due to the decomposition of polyurethane. The second step was shown due to the decomposition of an ester of MPTO. The third step was obtained due to the degradation of aliphatic and dehydrogenation of melamine moieties present in all coatings.34–36 All cured PU-MPTO coatings show a high degree of cross-linking and greater thermal stability than polyurethane acrylate.
Differential Scanning colorimeter (DSC)
DSC thermogram of cured polyurethane-MPTO oligomer for different formulations is shown in Figure 9. The glass transition temperature (Tg) performance of cured PU-MPTO coatings with growing MPTO was investigated by using DSC. The Tg of cured PU-MPTO coating films shows an increasing trend with increasing MPTO content. From Figure 9, it is clear that the glass transition thermogram of PU-MPTO shows single glass transition regions, which proves that MPTO is molecularly miscible with polyurethane acrylate. The increase in Tg values is due to higher conversion of unsaturated, brittleness, and presence of rigid melamine-based structure, which leads to an increase in cross-link polymer network of cured PU-MPTO coatings. Therefore, more heat energy is required to break the molecular segments of PU-MPTO materials to move from hard glassy to soft rubbery region.37–39 DSC spectra of UV cured-films.
Flame retardancy and combustion behavior of cured PU coating
UL-94 and LOI results of cured PU-MPTO.

Study of the crystallinity of the coatings by XRD
The crystal structure of the cured PU-MPTO composites was studied by X-ray diffraction (XRD) measurement. As shown in Figure 10, the XRD profile of the PU-MPTO confirms the presence of amorphous nature with a diffractogram. The diffused diffraction peak appearing around 2ϴ = 16–22° corresponds to an amorphous structure. The hybrid materials cured PU-MPTO forming a strong interpenetrating polymer network for 10% PU-MPTO, 20% PU-MPTO, and 30% PU-MPTO, respectively. The literature studies found that systematic arrangement of any polymeric compounds enhances the crystallinity, and irregular arrangements show amorphous nature of coating films. The amorphous nature in cured PU-MPTO was due to a hyperbranched and cross-linking polymer network.42,43 This indicates that PU-MPTO has been chemically incorporated into the hybrid materials and formed a cross-linked network between PU and MPTO consistent with the results of FTIR analysis. The X-ray diffractogram of UV cured films.
Gel content
The gel content in the cured films was determined by the weighting method as per ASTM D2765-16. We know that cross-linked gel can only be swelled, while those uncross-linked moieties like diluents and photo-initiators can be dissolved in acetone. The UV-cured films were immersed into a 30 mL glass vial containing xylene for 48 h at room temperature. Then, xylene was decanted and the left swelling gel of films was dried in an oven at 70°C till constant weight achieved. Finally, the gel content in each cured film can be calculated by the equation
36
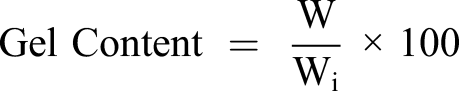
Literature studies found that the properties of UV-cured coating films are directly related to gel content.44,45
The gel contents of UV-cured PU-MPTO films are illustrated in Figure 11. The result reveals that PU without MPTO films had lower gel content than the films impregnated with tri-functional melamine-phosphate, indicating the introduction of tri-functional melamine-phosphate facilitated cross-linking and thus boosted cross-linking density. Gel content of UV cured films.
Application performances for UV-coated galvanized steel
Coating properties studied for hybrid PU-MPTO are crosshatch adhesion, pencil hardness, solvent resistance, and flexibility test. The crosshatch adhesion, pencil hardness, methyl ethyl ketone (MEK) double rubs, and flexibility were measured as per ASTM D3359, ASTM D3363, ASTM D4752, and ASTM D4145, respectively.
Coating properties of cured PU-MPTO.

a HB—H Poor Soft film. Prone to rapid wear and ease of scuffing, 2H–3H Fair Moderate performer, 4H–6H Good Durable film with projected good wear.
b Salt spray results are expressed using a comparative scale: 1 is the best corrosion protection, 3 is the worst one.
The corrosion protective properties were characterized by the salt spray test and results were illustrated in Table 3. The mechanism of corrosion coatings can be explained as barrier creation between substrate materials, environments, inhibition of the corrosion processes, and coating acting as sacrificial materials. The coated PU-MPTO acting as barrier layers that will not allow the permeation of corrosive agents (salt solution) to the metal surface. It has been found that novel tri-functional melamine-phosphate can reduce the amount of corrosion and is strongly impacted by the MPTO.
Gloss of UV-cured coatings
The gloss of cured PU-MPTO coatings is depicted in Figure 12. The gloss is related to the surface smoothness of the materials. The effect of tri-functional melamine-phosphate oligomer on the surface smoothness behavior of the coating materials was determined by gloss measurement at 60° angle. Figure 12 shows that the gloss of the coating increases with MPTO content. This could be due to the higher extent of photo-polymerization and degree of cross-linking of the UV-cured PU-MPTO increasing with MPTO oligomer concentration, which improves the gloss of coatings. After examining the films at 60° angle, it is observed that all the coating panels were glossy. Coating films of 30% PU-MPTO show the maximum gloss. Gloss of cured coated steel panels.
Contact angle analysis
The PU-MPTO films were examined through water contact angle measurements by using standard goniometer. The relationship between roughness and wettability of the surface can be characterized by contact angle measurement. The results show that, increase in the concentration of MPTO in PU acrylate linearly increases contact angle values as illustrated in Figure 13 Contact angle analysis of UV-cured coatings.
Conclusion
The current research demonstrated the synthesis of novel melamine-phosphate-based tri-functional acrylate oligomer (MPTO) and incorporated in PU acrylate for UV-curing technology. The UV cured MPTO-PU containing 30% of MPTO could reach a high LOI value of 34.8%, UL-94 V0, and performed as excellent flame retardant materials. Identical results were reflected in the case of DSC, and TGA with all different formulations shows improved thermal stability at a high temperature which may be due to dense cross-linking and rigid melamine-phosphate moiety. Exponential relation has been found between the contact angle and gloss of all coating formulation which has in favor of increasing durability, smooth-look, and stability of the coated materials. XRD spectrum of cured MPTO-PU had shown amorphous nature due to uniform cross-linked network during UV-curing process. The insertion of MPTO into PU acrylate concludes that mechanical properties like, adhesion, pencil hardness, chemical resistance, flexibility, and anti-corrosion behavior were superior as compared to pure PU. This may be attributed to a high degree of branching, higher extent of polymerization, and enhanced cross-linking density in MPTO-PU. Therefore, tri-functional melamine-phosphate oligomer due to its structural features will be one of the promising materials for coating industries.
Supplemental Material
sj-pdf-1-ppc-10.1177_09673911221079116 – Supplemental Material for “UV-curing of novel tri-functional melamine-phosphate oligomer: An effect of coating with polyurethane acrylate toward mechanical, thermal, and flame retardant properties”
Supplemental Material, sj-pdf-1-ppc-10.1177_09673911221079116 for “UV-curing of novel tri-functional melamine-phosphate oligomer: An effect of coating with polyurethane acrylate toward mechanical, thermal, and flame retardant properties” by Pundalik Mali, Narendra Sonawane, Nilesh Pawar and Vikas Patil in Polymers and Polymer Composites
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
