Abstract
Textiles play a vital role in assuring the thermal stability of the human body. The absorption of perspiration or moisture from a humid environment might result in wet clothing. Protective and sports apparel are often wet when the wearer is involved in high level of activity; thereby affecting the comfort. In order to understand the comfort behaviour of textiles in wet state, most of the researchers used the distilled water or NaCl aqueous solution. However, human sweat is a very complex aqueous mixture of chemicals comprising of salts, lipids, urea, lactic acid, carbohydrates, and minerals. In the present study, the effect of simulated sweat solution on the thermal behaviour of multi-layered fabric assembly is studied and compared with that of distilled water. The multi-layered fabric assembly consists of breathable PU-coated nylon as an outer layer; polyester knitted fabric as an inner layer; and micro-polyester wadding/hollow-polyester wadding/spacer fabric as the middle layer. In addition, the inner layer (next to the skin) is also tested for thermal comfort for both dry and wet states. It is observed that thermal properties of fabrics are drastically affected under wet conditions. The inner layer of fabric wetted with distilled water shows higher thermal conductivity as compared to that wetted with simulated sweat. Among various multi-layered assemblies, the thermal resistance and thermal diffusion of multi-layered assembly having hollow polyester wadding as a middle layer is high as compared to that having spacer fabric in the middle, under both dry and wet conditions.
Introduction
The term “comfort” refers to a pleasant condition of physiological, psychological, and physical harmony between a person and his surroundings. 1 The thermo-physiological and sensory aspects of clothing comfort interact to provide a subjective experience of good performance. The thermo-physiological refers to how clothing protects and dissipates metabolic heat and moisture,2,3 while the sensorial refers to how clothing interacts with the wearer’s senses.4,5 The strongest tactile and pressure comfort felt by individuals during exercise is thermal-wet comfort. 6
In a clothing arrangement, the human body works as an internal heat source, creating a temperature differential between the body and the surrounding environment. Unlike other engineering heat transfer issues, however, the sign of this gradient often changes when the ambient temperature changes. Perspiration is produced by the human body, which is then evaporated, resulting in an efficient body cooling mechanism through phase change. 7 Clothing functions as a barrier to the outside world as well as a heat transporter from the body to the environment. The degree of thermal comfort for the human body is determined by the heat and moisture balance between the body and the surroundings. The capacity of the body to adjust to heat and cold is critical for survival. The body cannot adjust if environmental circumstances become too severe, and death may ensue. Maintaining a somewhat constant body temperature, even under a range of external situations, is crucial and may be regulated by correct clothing. For sportswear and apparel worn in cold climates, moisture accumulation (i.e., absorption or condensation) within clothes is a severe concern.8–12 The decline of clothing thermal insulation due to condensates within clothes and the heat absorption by the moisture desorption re-evaporation of the condensates can produce “chilling” discomfort or even hypothermia as the user stops exercising and his metabolic heat output decreases. As a result, it’s critical to optimize clothing assembly structure to maximize moisture transport through clothes while minimizing moisture absorption and condensation within clothes.
Perspiration heat loss reduces thermal insulation and cooling efficiency when moisture is absorbed by the cloth from the skin before it evaporates. Sportspeople or military personnel can sweat at a rate of up to 2 L per hour.13,14 In the cold season, the highest sweat rate is 1000–1200 g/h, and in the hot season, it is 1500–2000 g/h. 15 The steady air has a thermal conductivity of 0.026 W/m.K at 20°C, but distilled water has a thermal conductivity of 0.6 W/m.K, which is 25 times higher. Therefore, excessive sweat generation due to physical activity causes progressive reduction in thermal insulation due to condensation of sweat, which eventually wets the cloth. Chen et al., reported that during sweating water accumulation (5–20%) within garment ensemble which reduces its thermal insulation, and the degree of decline ranges from 2 to 8%. 16
Layering fabrics that are expected to be worn as clothing has a significant impact on attributes such as air and water vapor permeability, thermal resistance, and nominal thermal conductivity.
17
Multilayer clothing, such as jackets and parkas, is often used in cold climates. These multi-layered assemblies are typically three-layered, with each layer serving a specific purpose and working together to achieve thermal physiological equilibrium: 1. The major function of the inner layer is to drain moisture away from the skin and transport it to the next layer. In addition, it traps body heat to minimize heat loss. Knitted structures are used. Quickly dry materials are preferred for this layer. Polypropylene, Polartec®, and polyester microfiber are preferred. 2. The core material of the multi-layered assembly is primarily used to provide thermal insulation to the whole fabric assembly. Feathers, wadding, and other materials are used to shield the human body from excessive heat loss. 3. The outer layer protects against the influence of wind and rain. It should be breathable with good water vapour permeability. Various materials which can be used including Gore-Tex®, Cloudburst®, Windstopper®, Trinity®, eVent® and Precip® windstopper (PTFE laminate), PU-coated nylon fabric and others.
Several types of research have been carried out to determine the effect of fiber, yarn, and fabric constructional parameters on fabric thermal characteristics.18–26 According to the studies, fabric thickness, weight, cover factor, and porosity have a strong relationship with thermal resistance. The heat and moisture transfer qualities of layered clothes are extremely important because they play a significant role in determining thermal comfort. When it comes to outdoor garment performance in cold and wet environments, they are the most important consideration. It becomes more difficult if sweat produced by the body condenses within the clothing system, especially in cold weather garments. 27 As the stored moisture freezes when it reaches a certain temperature, it might become trapped within the garment system, reducing heat resistance and increasing user discomfort. Therefore, appropriate sweat regulation from multilayer clothing components and their microclimate to the external environment is a major challenge. The material composition and structure of each layer in a multilayer garment system have a significant impact on moisture transmission through the fabric. Differential thermoregulatory responses, thermal strain, and thermal comfort may be affected by changes in layered structural components during activity and rest times. 28
Boguslawska-Baczek & Hes (2014) reported the effect of saline water on Nomex fabric’s thermal conductivity and resistance. It was discovered that when the moisture content of Nomex increases, its thermal insulation reduces. As the percentage of the aqueous solution grows, the amount of sodium chloride in the solution increases, causing the wetted fabric’s thermal conductivity to increase and its thermal resistance to decrease. Saltwater is used to study the effect of the salinity of real sweat or seawater on fabric thermal behaviour. Actual sweat, on the other hand, is a colourless, clear liquid produced by eccrine sweat glands in the dermal layer of the skin and composed of salts, lipids, urea, lactic acid, carbohydrates, and minerals such as potassium, calcium, magnesium, and iron. 29 Lactate (or Lactic acid) and ammonia, two primary solutes are released in sweat after prolonged exercise or intense activity. Lactate is primarily produced by anaerobic glycolysis inside the sweat glands. Lactic acid exists in the form of two enantiomers: D-lactic acid (Dextro lactic acid) and L-lactic acid (Laevo lactic acid). While L-lactic acid is a common compound of human metabolism, D-lactic acid is produced by some strains of microorganisms or by some less relevant metabolic pathways. 30 Whereas no such chemical compounds are present in distilled water or in NaCl aqueous solution. By using simulated sweat solution and water, a study is done to examine the moisture transmission and drying behaviour of multi-layered/individual fabric. 31 It is found that there is a significant difference in moisture properties of fabric with sweat solution and pure water. However, the research is limited to the moisture properties of textile materials, and no emphasis is laid on the thermal behaviour of fabric.
The impact of sweat solution on clothing thermal behaviour is yet unknown. As discussed earlier the composition of sweat is very different from that of distilled water. So, for more realistic approach toward the thermal comfort of multi-layered fabric in wet state sweat solution is used. Till date, no comprehensive research has been done on the differences in thermal behavior of a fabrics or multilayered assemblies with sweat and distilled water. In this paper, an exhaustive study on determining the wet state thermal comfort properties of multi-layered fabric assemblies using both sweat solution and distilled water has been done.
Material and methods
Material
Specifications of fabrics used.

Note. For breathable PU coated nylon porosity (%) is calculated using capillary flow porometer.
f -The number of filaments in the yarn cross-section;
( )-the values in parenthesis indicate the standard deviation.
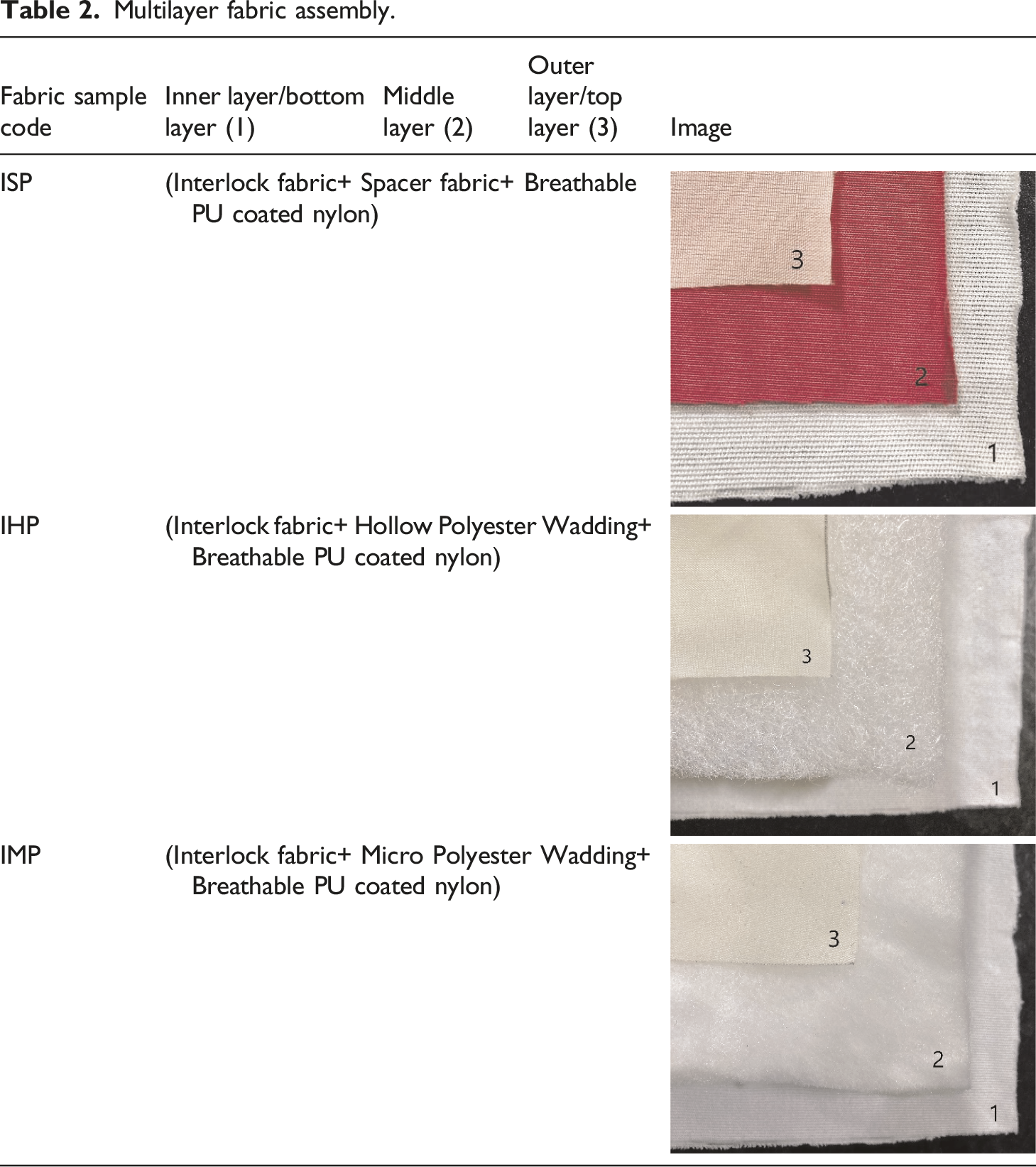
Multilayer fabric assembly.
Simulated sweat preparation
ISO standard 3160/2 is used for preparing simulated sweat solutions. It comprised 20 g/L NaCl, 17.5 g/L NH4Cl, 5 g/L acetic acids, and 15 g/L DL-lactic acids with the pH adjusted by NaOH. For making a 100 mL simulated sweat take a beaker and add 2 g NaCl, 1.75 g of NH4Cl, 0.5 mL of acetic acid, and 2.5 mL of DL- Lactic acid. After that add distilled water to a level of 100 mL in a beaker and continuously stir the solution. The pH of 2.0 is obtained which is checked with the help of pH paper. The average normal pH of human skin is 4.5 32 and it is adjusted by adding NaOH pellets to the solution.
Methods
Fabric parameters such as thickness, mass per unit area, bulk density, and porosity were measured for all the fabric specimens used in this research work. An average of 15 readings were taken for each test. For fabric bulkiness, mass per unit area and thickness is the important physical characteristic taken into consideration. Fabric thickness measurement is done as per DIN EN ISO 5084 standard (0.5 KPa for the woven and knitted fabrics; 1 KPa for the high-density nonwoven fabrics). Fabric mass per unit area is measured as per ASTM D3776 standard. Bulk density of fabric (g/cm3) is calculated by equation (1)
Here, M-mass per unit area (g/cm2); T-thickness of the fabric (cm)
The porosity of the fabric is determined by the volume of air trapped in the material, which may be calculated using the following equation (2).
The thermal properties were evaluated using an Alambeta tester (Figure 1) discovered by Hes & Dolezal, which provides a quick assessment of both steady-state and transient-state thermal characteristics.
33
This device approximates the heat flow (q) [W/m2] from the human skin to the fabric during brief initial contact in the absence of body movement and external wind flow to some degree. Due to the differing temperatures of the bottom measuring plate (22°C) and measuring head (32°C), this device’s concept relies on mathematical processing of the time course of heat flow passing through the tested fabric. The measurement takes just a few minutes. Thus, accurate measurements on wet textiles are feasible, since the sample moisture remains almost constant during the test. Alambeta instrument for thermal testing.
Thermal conductivity (λ), thermal resistance (R), thermal absorptivity (b), heat flow (qm), thickness (h), and thermal diffusivity (a) are the parameters evaluated in the Alambeta instrument under a 200 Pa contact pressure. The term “thermal resistance” [m2KW−1] is used to describe the heat-insulating characteristics of the material. The thermal resistance of textiles is influenced by many factors, including fiber conductivity, fabric porosity, and fabric structure. As demonstrated by equation (3), it is also a function of fabric thickness and has an inverse relationship with thermal conductivity
Thermal diffusion is a property of fabric structures that refers to the movement of heat through the air. It is directly proportional to the thermal conductivity of the material. It is defined by the following equation (4)
Another property measured in the Alambeta instrument is thermal absorptivity [W. s½/m2.K]. The range of thermal absorptivity is 20–600.
34
It is the objective measurement of a warm-cool feeling of the fabric. Fabrics with a lower thermal absorptivity value have a warm feeling and vice versa. Equation (5) is used to measure the thermal absorptivity of any fabric
The inner layer (interlock polyester) of the fabric assembly is tested for thermal properties in two different conditions: normal state and saturated state. The saturation level is achieved using the ISO-20,158 standard, which measures the fabric’s maximum water holding capacity. The dry weight (m1) of cloth (10 cm × 10 cm) is measured and it is immersed for 180 s in a container with a minimum of 100 mL of distilled water. After that, gently lift the specimen from the water by one corner and hang it upright for 60 ± 2 s to allow the water to drain. Weigh the damp specimen (m2) to the nearest 0.01 g (i.e., two decimal places). For estimating water absorption capacity equation (6) is used
The value of the water absorption capacity of the inner layer (interlock fabric) is, 314% (4.8 g) calculated from equation (6) is further used for wetting the multi-layered fabric assembly. During experiments in wet state, 4.8 g of water is spread over the surface of inner layer (which makes the top surface of the assembly), while testing on Alambeta, with the help of a syringe. For wetting the samples both distilled water and simulated sweat solution are used. Furthermore, the impact of wet fabric in steady-state on the thermal comfort characteristics of the multi-layered fabric is studied over time.
Results and discussion
Protection from the cold in protective clothing is a result of the design of the multi-layered system; where each individual component has an important role to play. These multi-layered assemblies are typically three-layered, with each layer serving a specific purpose and working together to achieve thermal physiological equilibrium. The major function of the inner layer is to drain moisture away from the skin and transport it to the next layer. In addition, it traps body heat, which is why a knitted polyester structure is chosen. Three distinct fabrics (Spacer fabric, Micro polyester wadding, Hollow polyester wadding) are utilized as the core material to make a sandwich structure of multi-layered assembly, with breathable PU coated nylon as the outer layer to limit the influence of wind and rain. Individual fabric layers were assessed for their thermal properties in order to establish the ensemble’s overall performance. The next section delves further into the thermal comfort of individual components and their ensembles in dry and wet conditions using water and simulated sweat solution.
Thermal comfort properties of individual fabric layers
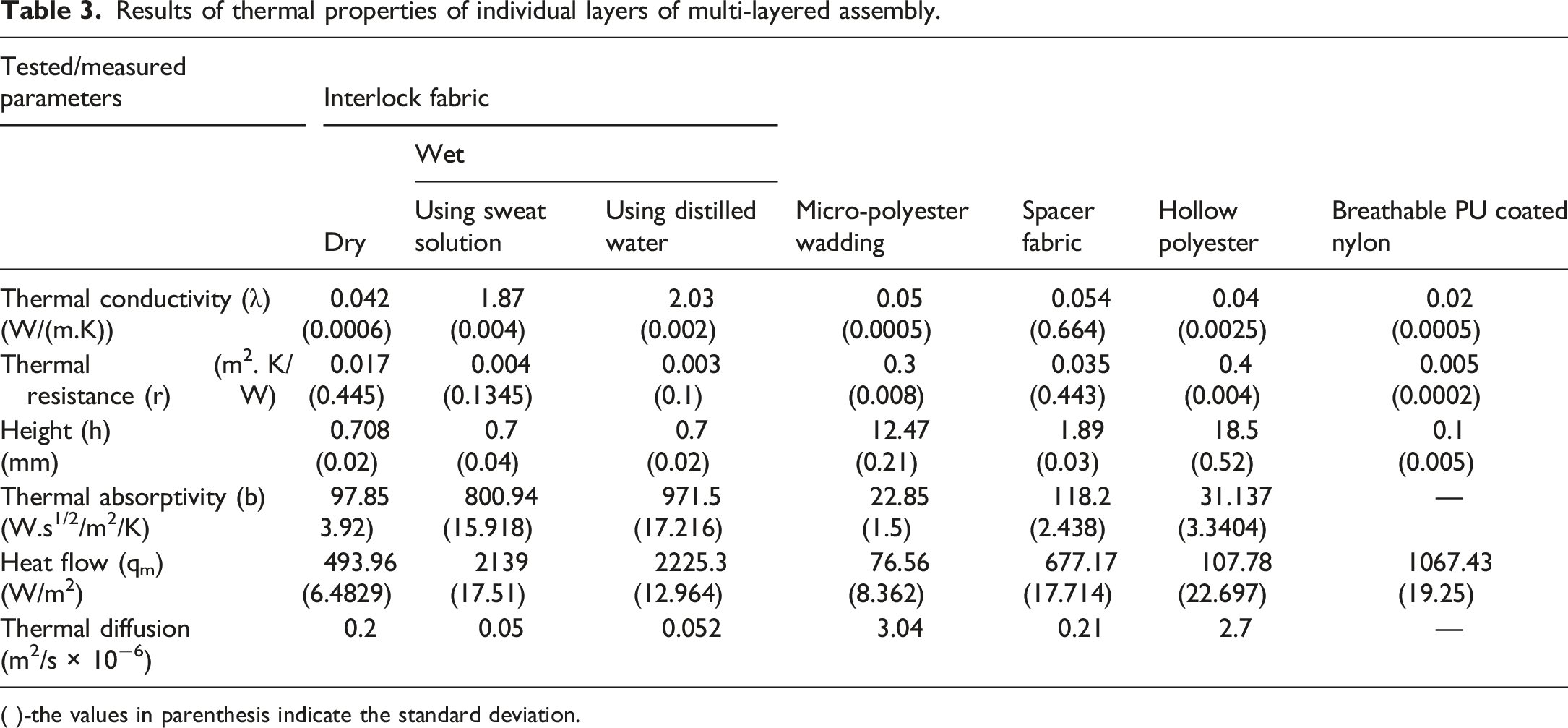
Results of thermal properties of individual layers of multi-layered assembly.
( )-the values in parenthesis indicate the standard deviation.
Thermal conductivity
The thermal conductivity of individual layers (three different types of middle layers, one outer layer and one inner layer) is shown in Table 3. Among the middle layer fabric types, it is observed that spacer fabric shows highest thermal conductivity in comparison to hollow polyester wadding and micro polyester wadding. This is due to the low porosity of spacer fabric than the other two types of middle layers. Inner layer, i.e., interlock fabric shows thermal conductivity value 0.042 W/m.K but after being wet with sweat solution and distilled water it is seen that there is increase in the thermal conductivity of the inner layer i.e., 1.87 and 2.03 W/m.K respectively. With the introduction of water in the fabric structure of interlock fabric the swelling of fabric takes place which changes its volume. The change in volume of hydroscopic fibers reduces the air void in the yarn which causes decrease in the thermal resistance. With this the value of thermal conductivity increases when fabric is wet. Further, it is also observed that interlock fabric wetted with sweat solution shows low thermal conductivity, as compared to when wetted with distilled water. This is due to the difference in the thermal conductivity of distilled water and saline water. While preparing the sweat solution 20 g/L of NaCl is added as per ISO 3160/2. The addition of an electrolyte such as the ions Na + and CI− has an effect on the structure of water which changes the value of thermal conductivity. 34 With the presence of NaCl the sweat solution became saline in nature and it is stated in literature that saline water has low thermal conductivity in comparison to distilled water.35–37
Thermal resistance
Table 3 shows the thermal resistance of individual layers of multi-layered fabric. The hollow polyester wadding shows highest thermal resistance followed by micro polyester wadding, spacer fabric, interlock fabric, and breathable PU coated nylon as shown in Table 3. Both fibrous waddings material shows comparatively higher thermal resistance than spacer fabric. Heat resistance of spacer fabric is lower due to the lower fabric thickness as stated in equation (3) thickness is directly proportional to thermal resistance. The interlock fabric in wet states, have low thermal resistance. As the water/sweat gradually occupies the intra-yarn and inter-yarn air voids of the fabric, the fabric’s intrinsic thermal insulation will be diminished and, hence, contribute to the loss of total insulation. In case of sweat solution, the thermal resistance is more in comparison to distilled water.
Thermal diffusion
When it comes to comfort testing, thermal diffusion (a) is critical. It’s a property of heat movement through the air in a fabric construction; the greater the thermal diffusion, the quicker the heat spreads. The results obtained for individual layers show that thermal diffusion of hollow polyester and micro polyester wadding is higher in comparison to other fabrics (Table 3). This is due to the bulky structure with high thickness and hollow cross section of fibers, retaining large amount of air in the structure, which facilitates thermal diffusion. For interlock fabric in the normal state, the thermal diffusion value is measured to be 0.2 × 10−6 m2/s and when wet this value became 0.054 × 10−6 m2/s (with distilled water) and 0.05 × 10−6 m2/s (with sweat solution). While comparing the sweat solution and distilled water for thermal diffusion no significant difference is observed as the
Thermal absorptivity
In terms of ‘cool/warm’ sensation, thermal absorption is a surface characteristic that enables a fabric’s character to be evaluated when the human skin contacts an item, such as textile material. Thermal conductivity, specific heat, and density are the variables considered when calculating thermal absorptivity. It is seen from Table 3, that spacer fabric has the highest thermal absorptivity followed by interlock fabric. Higher thermal absorptivity values resulted in a cooler sensation at the start of skin contact. Thermal absorptivity is mostly determined by surface qualities like smoothness. A smoother fabric surface improves the surface area of contact and heat transfer, resulting in a cooler sensation. For breathable PU-coated nylon thermal absorptivity is very low to be recorded by the Alambeta instrument. The interlock fabric in the normal state shows thermal absorptivity of 97.85 Ws1/2/m2K and when the fabric is wet its thermal absorptivity value increases as water is having high thermal conductivity and thermal capacity ρc [J/m3]. The interlock fabric with sweat solution has a thermal absorptivity value of 806.58 Ws1/2/m2/K and the interlock fabric with distilled water has a thermal absorptivity value of 996.66 Ws1/2/m2/K. As the thermal conductivity of water is more as compared to sweat solution, therefore, the interlock fabric saturated with sweat solution has lower thermal absorptivity. Also, the sweat solution is saline in nature, the salinity of the sweat decreases the heat capacity of the solution. So, it is concluded that distilled water makes fabric cooler when human skin comes into contact with the fabric surface.
Peak heat flow
The feel of fabric can also be evaluated by peak heat flow (qmax). This value appears when the cool surface of the fabric comes in contact with the hot plate of the Alambeta instrument. Here from Table 3, the qmax for interlock fabric in an unwet state is 493.96 W/m2, when wet became 2131.07 W/m2 (for sweat solution) and 2226.28 W/m2 (for distilled water). The increase in qmax value is because when water is present in the interstices of the fabric it will draw more heat from the surface it contacts. The higher qmax of distilled water means the feel of the fabric is cool. Therefore, from both thermal absorptivity and qmax it is stated that with distilled water fabric feel is becoming cool. For individual layers (Table 3), the qmax value for breathable PU coated nylon is found to be highest followed by spacer fabric and interlock fabric. The spacer fabric has the same areal density as micro-polyester wadding and hollow polyester wadding shows a high qmax value. Because a densely packed fabric has a smooth surface, which increases the amount of skin contact and heat from the body may easily be transferred to the environment, resulting in a cooler sensation when touched.
Thermal behaviour of multi-layered fabric assembly in the dry and wet states
The amount of water in the fabric and the amount of air in the fabric both contribute to its thermal comfort. The presence of water in textiles increases thermal conductivity and specific heat, as well as affects their density and thickness.38,39 In the present experiment, the thermal properties of multi-layered assemblies were analyzed after saturating the inner layer to its maximum water absorbency capacity. The effect of time on the thermal behaviour of the wet fabric assembly in a steady-state is measured. The results of the thermal properties measurements in the dry and wet states are displayed in Figure 2. Effect of distilled water and simulated sweat solution on thermal properties of multilayer fabric assembly: (a) Thermal conductivity; (b) Thermal resistance; (c) Thermal diffusion; (d) Thermal absorptivity; (e) Peak heat flow.
Results of thermal properties of multi-layered assemblies in the dry state.

( )-the values in parenthesis indicate the standard deviation.
When the multi-layered fabric is initially wetted, there is a sharp increase in thermal conductivity as seen in Figure 2(a). This rise is due to an increase in the amount of water in the fabric structure, which tends to squeeze out the air. After wetting the samples, the thermal conductivity was assessed consecutively every 2 min, and it was found that IMP and IHP damped with distilled water exhibited a sharp decline in thermal conductivity after a time interval of 46 and 54 min respectively. The decline is observed due to the decrease in water content from the samples. Whereas in the case of wet ISP, the decrease in thermal conductivity with distilled water occurs after 20 min which is much earlier than that of the other two types of multi-layered fabrics. This may be because of a lower thickness of ISP, the water travels faster to the outer layer and, with a decrease in water content from the fabric assembly, the thermal conductivity decreases. While in the case of IMP and IHP, higher thickness causes a slow transfer of water from the inner layer to the outer layer. As shown in Figure 3, the outer layer for IMP and IHP takes a longer time to wet and once the water comes out of the assembly, its thermal conductivity starts falling rapidly. Water absorbed by the outer layer of multilayer assembly: (a) IMP after 42 min; (b) IHP after 54 min.
It is observed from Figure 2(b), that there is the decrease in thermal resistance of fabrics after wetting and increases with time. In addition, for both wet and unwet states, the thermal resistance of ISP is found to be the lowest when compared to the other two multi-layered assemblies. IHP is having thermal resistance of 0.43 m2. K/W and thermal diffusion value 2.818 × 10−6 m2/s, which is highest among the three fabric assemblies. IHP consists of hollow polyester fibres in the middle layer, which has higher thickness than the other fabrics. Higher thermal resistance of IHP fabric is due to higher fibre volume and therefore, higher amount of air trapped inside the structure. Whereas IMP have less thermal resistance than IHP due to lower thickness. The thickness of IMP after the application of 200 Pa pressure (corresponding to the measuring head of Alambeta) is 0.015 m and that of IHP is 0.021 m.
As shown in Figure 2(a) and (b), the thermal conductivity and thermal resistance of multi-layered assemblies treated with sweat solution is quite different for all fabric type. It can be seen that for all the multi-layered samples with sweat solution, there is a constant decrease in the thermal conductivity and constant increase in thermal resistance. This may be due to the difference in density of distilled water and sweat solution. The sweat solution has a density of 1040 kg/m3, whereas distilled water has a density of 990 kg/m3. This density difference influences the flow of the liquid from the inner layer to the outer layer. Lower density liquid will take a longer time to come down in a multi-layered assembly. The increase in thermal conductivity causes a decrease in thermal resistance. It also affects thermal diffusion and thermal absorptivity of the fabric assembly, as seen in Figure 2(c) and (d). The thermal absorptivity of sample IHP is 27.28 Ws1/2/m2/K, ISP is 121.81 Ws1/2/m2/K and IMP is 33.87 W.s1/2/m2/K. It is seen that IHP has the lowest value of thermal absorptivity in comparison to the other two fabric assemblies. As the thermal conductivity is low and thermal resistance is high for IHP. Therefore, IHP gives a warmer feeling as compared to the other two multi-layered fabric assemblies. As the thermal conductivity and thermal capacity of water are higher than that of air, it causes the thermal absorptivity of the fabric to increase. When wetted, ISP has a thermal absorptivity of 537.7 Ws1/2/m2/K, whereas IMP and IHP have less than 250 Ws1/2/m2/K. The fabric with a higher thermal absorptivity value gives a cooler feeling. Thermal absorptivity increases with the introduction of water/sweat solution, but as time passes, the accumulated water is transferred to the outer layer, which results in a decline in thermal absorptivity. Thermal diffusion is the ability related to heat flow through air in the fabric structure. The thermal diffusion value for IHP is found to be highest, i.e., 2.82 ×10−6 m2/s (unwet) and 0.76×10−6 m2/s (wet) and lowest for ISP, i.e., 0.2 ×10−6 m2/s (unwet) and 0.13 ×10−6 m2/s (wet). This is because of the hollow polyester present in the middle layer of IHP. The hollow cross-section of the fiber provides more space for air inside the structure. It can be seen that with time, the thermal diffusion of the IHP sample with distilled water shows a random increasing and decreasing trend. This may be due to the transfer of water to the outer layer, creating more room for the air to enter the IHP sample.
The results of the peak value of heat flux (qmax) are presented in Figure 2(e). Based on these results, it is seen that ISP has the highest qmax value in both unwet and wet state, i.e., 473.29 W/m2 and 1918.05 W/m2, respectively whereas IHP has the lowest qmax value of 132 W/m2 and 1399.1 W/m2. The lower thickness and densely packed fabric structure of ISP both contributes to give higher qmax. It is seen that on wetting the fabric, either with distilled water or sweat solution, the qmax increases. The increase in moisture content will lead to an increase in the qmax of the fabric, which affects the fabric feel. When inner layer of multi-layered structure is saturated (i.e., 314% moisture content) it shows highest value of qmax, with time this moisture content of assembly starts decreasing leading to decrease in qmax. It shows that higher water content and low thickness of the assembly facilitate increases skin cooling which leads to cool feeling of fabric. Therefore, the ISP has a cool feel.
It is also observed from Figure 2, that when the amount of moisture from the samples is decreasing, the thermal values of the sweat solution take a longer time to come closer to the unwet thermal value range. In the end, when the samples dry, the thermal conductivity value remains high for samples with sweat solution in comparison to distilled water. A similar trend is seen in the case of all the other thermal properties analysed here. This is due to the presence of various chemicals used in sweat solutions. As the solution penetrates the fabric structure, residuals of the chemicals remain stuck on the fabric. According to the literature, when saline water enters the porous medium, crystals form, increasing the thermal conductivity of the wetted porous structures. 29 As a result of this experiment, a distinct pattern may be seen for both sweat solution and distilled water. Because of this, it is advised to use sweat solution for thermal behaviour analysis in a wet state for fabrics to get results that are closer to reality.
Conclusion
Protective and sports apparel are often wet during high level of activity by the wearer, which affects the comfort levels. Various researchers studied the comfort behaviour of textiles in wet state using distilled water or NaCl aqueous solution. However, human sweat is a very complex aqueous mixture of chemicals comprising of salts, lipids, urea, lactic acid, carbohydrates, and minerals. Therefore, in this study, the effect of simulated sweat solution on the thermal behaviour of multi-layered fabric assembly is studied and compared with that of distilled water. It is observed that the type and structure of the middle layer of the multi-layered fabric has significant influence on their thermal behaviour in dry state. Amongst all the combination, the one with hollow polyester wadding as middle layer shows highest thermal insulation value of 0.43 m2. K/W. Whereas fabric combination with spacer fabric as middle layer has highest qmax and thermal absorptivity value, making the fabric feel cool. When evaluated in wet state, it is seen that the thermal insulation behaviour of the examined multi-layered fabric assemblies significantly decreases. This is because of the higher heat conductivity of water than air, which replaces the air in the pores of the fabric. The thermal resistance value of interlock fabric when wet by distilled water or simulated sweat is low as compared to the dry fabric.
The difference in composition of sweat and distilled water also affects the thermal behaviour of multi-layered fabric assembly. From the experimental results, it is observed that the thermal conductivity of fabric assemblies when wetted with sweat solution is higher than those wet with distilled water. Interlock fabric has high thermal conductivity and thermal absorptivity with distilled water as compared to sweat solution. The thermal conductivity value with distilled water remains the same up to a certain time but after that, it starts to decrease rapidly whereas the multilayer fabric wetted with sweat solution has shown a constant decline in thermal conductivity. However, fabrics wet with sweat solution take a longer time to evaporate the water and reach a thermal state equivalent to that in dry state. These effects are critical to consider when designing sports or outdoor gear for cold weather.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
