Abstract
Development of a gel-free bio-potential electrode for the wearable health monitoring applications is a challenging goal. A conductive fabric electrode can replace the traditional conductive gel electrode. This paper describes the development of a conductive fabric electrode with regard to its potential use for electrocardiogram (ECG) acquisition. Since direct contact between the conductive fabric and human skin will be involved, an investigation on the effect of perspiration on the electrical conductivity of fabric is critical. Hence, the developed electrode was treated with alkaline (pH=8.0) and acidic (pH=4.3) perspiration for 3, 8 and 40 h to study the effect of perspiration on the conductivity and surface morphology. The acquired ECG signals were analysed with respect to morphology and frequency distribution.
Conductivity tests were carried out on the perspiration-treated test electrodes by two probe method and surface resistivity meter. The ECG signals of volunteers were also recorded. The results showed a slight decrease in conductivity but without affecting the morphology and the quality of ECG signal. Leached silver content in the acid perspiration-treated solution was found to be 0.117 ppm as determined by Atomic absorption spectroscopy. The result shows that soft conducting textile materials can indeed be used as an electrode for ECG acquisition. This is a novel type of gel-free fabric electrode for long term wearable health monitoring applications including space application.
Introduction
 Most commonly used bio-potential electrode in clinical fields is the gel type silver/silver chloride (Ag/AgCl), which can be found both in reusable and disposable form. This electrode is classified as a wet electrode,
1
since it requires the application of an electrolyte gel to promote the skin-electrode impedance reduction. This is crucial to establish an electrical contact between the skin and the electrode. The subject preparation time, itchiness after drying of gel and skin impedance is higher in the conventional electrodes which limit their usage in long-term monitoring tasks. These limitations prevent the use of wet electrodes in wearable applications, leading to the development of gel-free bio-potential electrodes.
2
Gel-free bio-potential electrode is defined as a dry electrode. The development of gel-free bio-potential electrode is one of the most important elements of designing a ubiquitous wearable health monitoring system. These components are responsible for establishing an interface between the skin surface and measurement apparatus. A revolutionary approach for the electrophysiological monitoring of military pilots, other defence and security personnel have emerged from textile technology.
3
Most commonly used bio-potential electrode in clinical fields is the gel type silver/silver chloride (Ag/AgCl), which can be found both in reusable and disposable form. This electrode is classified as a wet electrode,
1
since it requires the application of an electrolyte gel to promote the skin-electrode impedance reduction. This is crucial to establish an electrical contact between the skin and the electrode. The subject preparation time, itchiness after drying of gel and skin impedance is higher in the conventional electrodes which limit their usage in long-term monitoring tasks. These limitations prevent the use of wet electrodes in wearable applications, leading to the development of gel-free bio-potential electrodes.
2
Gel-free bio-potential electrode is defined as a dry electrode. The development of gel-free bio-potential electrode is one of the most important elements of designing a ubiquitous wearable health monitoring system. These components are responsible for establishing an interface between the skin surface and measurement apparatus. A revolutionary approach for the electrophysiological monitoring of military pilots, other defence and security personnel have emerged from textile technology.
3
Textile electrode, popularly referred as ‘textrode’, is a sensor developed by textile processing to sense the surface bioelectricity of human body. Currently, various structures of textile electrodes, such as, woven, knitted, non-woven and embroidery have been developed.4–9 Fabric electrodes belong to the dry electrode category and soft surfaces can be bonded to the body surface so as to reduce the artefact and noise generated by the motion between the electrode and skin. A fabric electrode is more favourable since it is foldable and washable, 5 and hence the cardiovascular and respiratory parameters can be routinely monitored when an individual is in need of intensive care.10,11 Here the textile-structured electrodes and wearable electronics have enabled wearable continuous monitoring of the bio-potential signals and long term usage.5,12
Intelligent biomedical clothes and wearable ambulatory health-monitoring systems can act as key enablers for lifelong continuous monitoring of health for all individuals. Non-invasive sensors have a huge potential for disease prevention in medicine and for diagnosis. Integration of such sensors into clothing can, therefore, enhance home healthcare, citizen medicine and disease prevention at an early stage. 13 Minimal electrode acquisition systems play an important role in the wearable health monitoring applications. 14 Long-term ECG recording is a standard procedure in monitoring cardiac patients. In the case of ECG recording during daily life activity, washable and comfortable textile electrodes can be woven at a precise target location in the wearable garments and hence make the ECG recording less complicated concerning electrode handling and adjustments on the body unlike the conventional gelled electrodes. 15
The conductive fabrics are often fabricated by coating a thin layer of highly conductive materials like silver over non conducting polymeric fabric materials.16–18 These materials are susceptible to erosion caused by the corrosive reaction of gas, vapours and liquids they are exposed to. Since the conductive surface is in close contact with the body surface, the effect of perspiration on the structural stability of the conductive layer has to be thoroughly studied because it can directly affect the conductivity of the electrode and the quality of the acquired signal. 19 In the present study, a fabric electrode was developed for bio-potential signal ECG acquisition. The developed electrode was treated with alkaline and acidic perspiration [AATCC-15–2002] separately for different time durations to determine the effect of perspiration on the conductivity and surface morphology. Conductivity tests were carried out by two probe method for bulk conductivity and surface resistivity meter for surface conductivity on treated and untreated test electrodes. ECG of volunteers was recorded using various electrodes to establish the effect of perspiration on the quality of the ECG signals.
Materials and methods
Chemicals
Sodium chloride, ammonium carbonate, anhydrous dibasic sodium phosphate, lactic acid and l-histidine monohydrochloride (Reagent Grade Sigma Aldrich Chemicals, USA).
Apparatus
Keithly Source Meter (Series 2600A), Surface resistivity meter (Z 203), Titronic Titrator, VEGA3, Tescan, Scanning Electron Microscope and Atomic Absorption Spectrophotometer (iCE TM 3500AAS, Thermo Fischer Scientific, USA, in air-acetylene mode using SOLAARTM Software)
Fabric sample
Conductive knit fabric Medtex P 180 was used as a textile electrode. This fabric is a silver-plated nylon that is stretchable in one direction. It is highly conductive with a surface resistivity of <1 Ω/sq. The fabric is plated with pure silver of 99.9%. This fabric can be used as a wound care, antimicrobial garment. The total thickness of the fabric is 0.55 mm and weight is 224 g/m2. A sample of conductive fabric is shown in Figure 1. Conductive fabric sample.
Design and fabrication of fabric electrode
The specific structure of the fabric electrode is shown in Figure 2. The conductive cloth is wrapped with filler (3 mm high density polyurethane fibre) to ensure the complete contact with human skin. The filler and the support pad (2 mm soft PVC) of electrode will provide enhanced support for the clothing from the external compression. The conductive fabric, filler and support pad are encapsulated using polyester: cotton (80:20) fabric which houses the complete sensing electrode. The wire to the signal input measurement unit of ECG conditioning circuit was passed through the surface of the electrically conductive fabric. Novelty lies in the preparation of the sensing textrode with optimal size and area to get a faithful ECG. The material is cut into the shape and interfaced with the electronics where the impedance match between the skin and the electrode is adjusted by providing matching impedance from electronics side. The developed test electrode is as shown in Figure 3. Development of fabric electrode. Test electrode.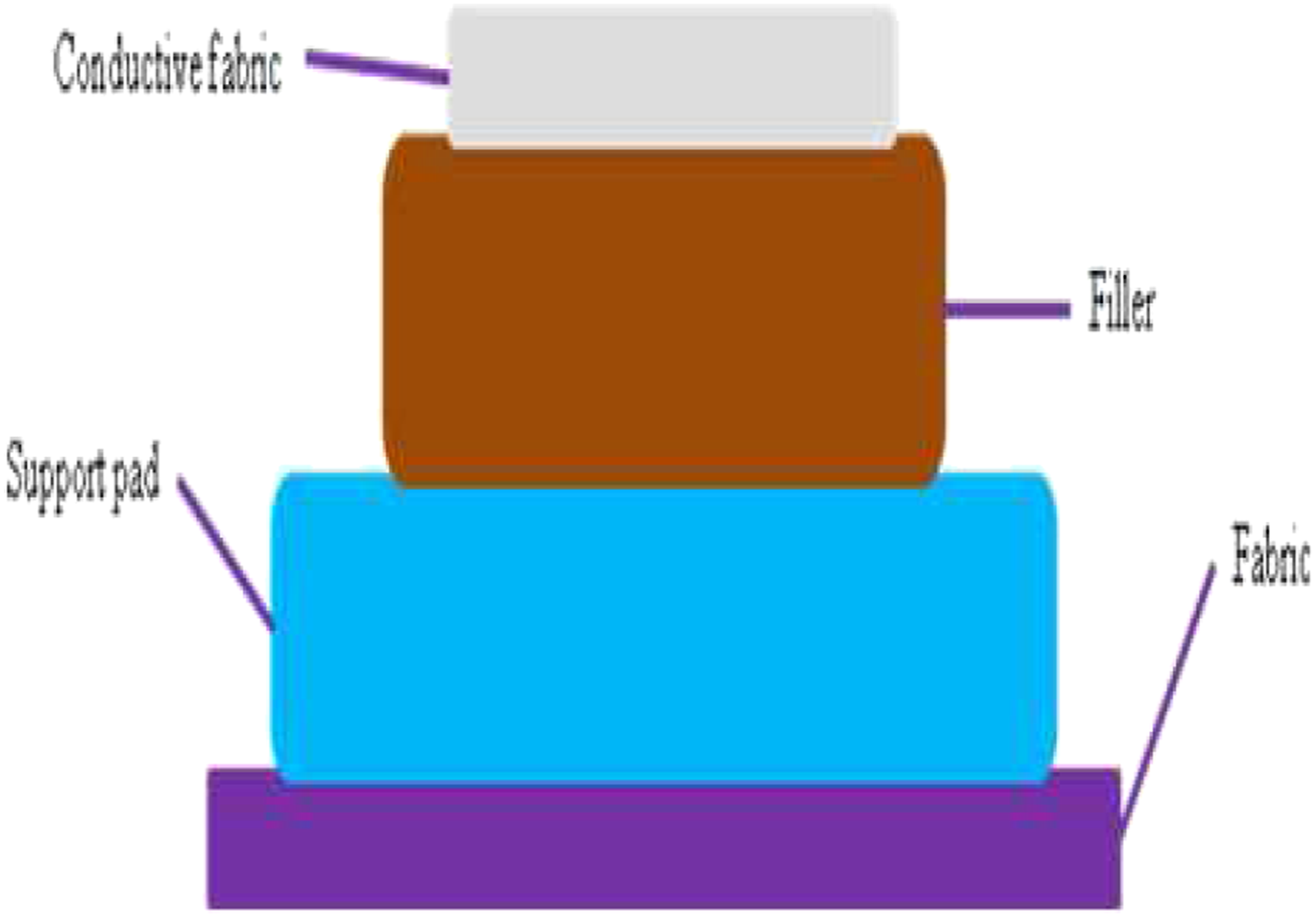

Preparation of acidic and alkaline perspiration AATCC 15–2002. 20
Preparation of acidic (pH=4.3±0.2) perspiration
10 ± 0.01 g sodium chloride, 1 ± 0.01 g lactic acid, 1 ± 0.01 g anhydrous dibasic sodium phosphate (Na2HPO4) and 0.25 ± 0.001 g histidine monohydrochloride were dissolved in 1 L distilled water. The pH of the prepared solution was 4.3 ± 0.2.
Preparation of alkaline (pH=8) perspiration
10 ± 0.01 g sodium chloride, 4 ± 0.01 g ammonium carbonate, 1 ± 0.01 g anhydrous dibasic sodium phosphate (Na2HPO4) and 0.25 ± 0.001 g histidine monohydrochloride were dissolved in 1 L distilled water. The pH of the prepared solution was 8.0 ± 0.1.
Perspiration treatment
Conductive fabric sample of the size 2.5 × 6 cm was treated with acidic and alkaline perspiration solution for 1h, 3h, 8h and 40h, respectively, at a flow rate of 2L/hr.21,22
Conductivity/Surface Resistivity measurement
Two probe method
Two point probing method was used to carry out the conductivity test on the perspiration treated samples. Resistivity is the characteristic measurement of a material and the process control parameter in the semiconductor manufacturing process. Electrical current was passed between the two probes and the voltage created was measured between the same two probes. The resistivity was calculated from the measured values of current and voltage. 23
Methodology
A basic property of a conductive material is its electrical resistivity. Electrical resistivity was measured using two point probe method; current and voltage were measured in the same wire. The experiment was carried out using Keithly Source Meter (Series 2600A). Uniform probe spacing was adopted in order to reduce the error caused by non-uniformity of probe spacing.
Figure 4 shows a thin conducting sheet with current passing through the ends of the sheet from surface 1 to surface 2. The sheet has a length ‘L’ and a cross sectional area ‘A’. The current generates a voltage difference between surface 1 and surface 2. Basic setup for two probe measurement.
The average resistance R = V/I -------------------------------- (1)
R can also be defined as R= (Resistivity x Length)/A ------------- (2)
where resistivity is an electrical property of the material in ohm-cm.
A=Thickness (t) X Width (w) --------------------------------- (3)
R= Resistivity x Length/(t x w) --------------------------------- (4)
The resistance of one square (length=w) of the thin sheet,
RS = Resistivity/t. --------------------------------- (5)
Sheet resistance is measured by this formula for square sheet
Rs = 4.53 * V/I (Ohm/Square) --------------------------------- (6)
Surface Resistivity Meter
Surface resistance, Rs, is defined as the ratio of a DC voltage U to the current, Is flowing between two electrodes of specified configuration that are in contact with the same side of a material under test as shown in Figure 5. Basic setup for surface resistance and surface resistivity measurement.
Rs = U/Is Surface resistivity ρs, is defined as the ratio of DC voltage U drop per unit length L to the surface current Is per unit width D.
ρs =(U/L)/(Is/D)
Surface resistivity is a property of a material and it remains constant regardless of the method and configuration of the electrodes used for the surface resistivity measurement. A result of the surface resistance measurement depends on both the material and the geometry of the electrodes used in the measurement. Though the physical unit of surface resistivity is Ohm (Ω), surface resistivity is often expressed in Ohm/square (Ω/sq.), which is not a valid unit from the dimensional analysis point of view. 24
Scanning Electron Microscopy Analysis
Scanning electron microscopy (SEM) analysis was carried out for control and perspiration treated fabrics using a Vega3 Tescan SEM in low vacuum mode humid atmosphere at a pressure of 60 Pa. The distance from the scanning pin was 15 mm with a scanning speed of 0.1 ns per pixel at a scanning depth of 3–4 μm. A tension of 12 kV was applied on the Wolfram filament and a large field detector was used for collecting the image. 25
Atomic absorption spectroscopy Analysis
Standard silver stock solutions for the calibration curve (0.5, 1.0, 1.5, 2.0 and 2.5 ppm), treated samples solutions and the blank samples were introduced into the air-acetylene flame and the silver measured at 328.1 nm with a slit of 0.7 nm.
ECG Acquisition
ECG readings were recorded with the test electrodes and the results were analysed for its morphology. Textile electrodes to detect ECG signals were usually integrated in a wearable system which gives a more complete description of the patient’s health status. The electrode was sutured to the fabric of the shirt and sponge of appropriate size was placed inside, in order to increase the reliability of the electrode being in contact with the skin. The signals were acquired using the two electrodes featured in the front end of the ECG acquisition system and the difference between the two signals was filtered and amplified.
The common mode voltage on the body cause interference in the output signal due to the finite common mode rejection ratio (CMRR) of the instrumentation amplifier and hence to be suppressed. A so-called ‘driven-right-leg’ circuit with an additional reference electrode was used for this reason, which is shown in Figure 6. The sum of the electrode output signals was fed back via an inverting amplifier to the body. The ECG signal acquisition system consists of an instrumentation amplifier, a second order Butterworth high pass filter, a second order Butterworth low pass filter, a gain amplifier, a 50 Hz notch filter and finally a buffer circuit. INA333 was used as the instrumentation amplifier in the present deign, which has a CMRR of 100 dB when the amplifier gain was 10. The cut off frequency of the high pass filter and low pass filter were 0.5 Hz and 100 Hz, respectively. The overall gain of the acquisition system was 1000. The system was designed to work with a single supply (+5 V) to minimize power consumption. Three electrode ECG (Lead-I) acquisition system.
Results and Discussion
The ECG monitoring should be robust and there should be minimal inconvenience to the subject wearing the electrodes. Gel-based conventional electrodes cannot be used for wearable long time monitoring since the gel dries out, causes irritation and can cause change in contact impedance very often. Hence the conductive fabric Medtex P 180 was chosen for making an effective electrode for ECG monitoring. Medtex 180 Ag Nylon was of single directional stretch. This works similar to a dry silver electrode and due to its flexible shape it sits on the skin matching the shape and providing minimal artefacts. It picks up the potential on the surface of human body due to ionic current and converts it into electronic current.
The evaporation of sweat from the body surface is the most important thermoregulatory mechanism for dispersing excess heat from the body. Fire-fighters and Military personnel exposed to hot environments and/or strenuous operational conditions can sweat up to 2 L/hr for several hours.21,22 The conductive layer of the electrode can be affected by corrosion and leaching due to exposure to perspiration. Human perspiration is found to be moderately acidic to neutral, typically the pH between 4.5 and 7.0. 26 Hence, a study has to be conducted in order to evaluate the perspiration effect on the conductivity of the electrode. Therefore, a conductive fabric sample was treated with acidic and alkaline perspiration separately for 1 h, 3 h, 8 h and 40 h at a flow rate of 2 L/hr for the determination of the effect of perspiration on the conductivity and surface morphology of the developed fabric electrode. The flow rate was chosen based on the average perspiration rate of a person engaged in activity as outlined above.
Conductivity of the perspiration-treated and control fabric electrode.

Average of five determinations (±SD.).
Surface resistivity of the perspiration-treated and control fabric electrode.

Average of five determinations (±SD.).
The surface resistivity of all the samples was found to be within the required limits of a good conductor [ANSI/EIA-541–1988]. A material is considered to be conductive if its surface resistivity is below 1.0 × 105 Ohms/square according to electrostatic discharge (ESD) standard ANSI/EIA-541–1988. 27 Results shown in Table 1 and Table 2 confirm the absence of any adverse effect of perspiration on the conductivity of the fabric.
Effect of perspiration treatment on the ECG signal
ECG readings were recorded from the young volunteers using a Tektronix MSO 4104 oscilloscope. The morphology of the acquired ECG signal was not altered even though the bulk conductivity of the perspiration-treated samples was decreased as observed in the Table 1. The surface conductivity of the fabric, which is the critical property that decides the acquisition of the ECG signal from the body, remained within the limits of a good conductor, even after 40 h of treatment (Table 2) and gives rise to good quality ECG signal as shown in Figure 7. Results shown in Figure 7 confirm the absence of adverse effect of perspiration for the acquisition of ECG signal. ECG recording with the control and perspiration-treated samples.
Effect of perspiration treatment on the morphology of the fabric
The effect of perspiration treatment on the surface morphology of the fabrics was determined by SEM analysis. The SEM pictures of the untreated, acidic perspiration-treated and alkaline perspiration-treated fabrics are shown in Figure 8(a), Figure 8(b) and Figure 8(c), respectively. There was no opening of the silver layer and exposure of the base fabric even after 40 h of perspiration treatment as shown in Figure 8. (a) SEM picture of Control fabric. (b) Acidic Perspiration-treated fabric. (c) Alkaline Perspiration-treated fabric.
This can be attributed as the reason for the fabric to remain as a good surface conductor even after the exhaustive perspiration treatment. A decrease in the brightness on the fabric surface of the 40 h-treated samples was observed as shown in Figures 8(b) and (c) in comparison with untreated control fabric sample (Figure 8(a)).
Atomic absorption spectroscopy
Concentration of the silver content in both the treated perspiration solutions was measured by Atomic absorption spectroscopy analysis. Leached sliver content found in the solution treated for 40 h with acidic and alkaline perspiration solutions were found to be 0.117 ppm and 0.769 ppm, respectively. It is relevant to mention here that the permissible limit of silver content in the drinking water is 0.1 ppm as per Indian standard. 28 This value is almost equivalent to silver content found in the acidic perspiration-treated solution. Whereas alkaline perspiration treatment has shown significant leaching of silver content from the fabric which is much beyond the permissible limit as per Indian standard for drinking water. Although the alkaline test has been eliminated, there may be certain instances in foreign trade or special end uses that require alkaline test. 20 Therefore, in this study additional data on the effect of alkaline perspiration is also reported.
Conclusions
Perspiration treated fabric electrode displayed a decreasing trend in conductivity up to 8h treatment after which there was no considerable change in the bulk conductivity of the fabric. The surface resistivity of the samples even after the 40 h sweat treatment was found to be within the required limits of below 1.0 × 105 Ω/square as a good conductor. The morphology of the acquired ECG signal was not altered even though the bulk conductivity of the sweat-treated samples was slightly decreased. Leached sliver content found in the solution treated for 40 h with acidic perspiration solution was found to be 0.117 ppm. It is almost equivalent to permissible limit of silver content in the drinking water as per Indian standard.
The study shows that the textile electrodes integrated in a wearable system to detect ECG signals can withstand the exposure to sweat for sufficient duration. The developed gel-free textile electrodes will be highly useful in the wearable health monitoring system for continuous measurement of ECG signals. There is a bright scope to explore this electrode in space application for continuous measurement of ECG signals.
Footnotes
Acknowledgments
Authors are thankful to Director, DEBEL, Bengaluru for the constant support and encouragement.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
