Abstract
In order to prepare flexible electrode with excellent performance, cotton fabric was used as the substrate in this study. The surface of cotton fabric was modified by polydopamine(PDA). The cotton fabric was then carbonized. Finally, a three-dimensional porous flexible electrode for carbonized cotton fabric was prepared. The maximum stress of the flexible electrode is 0.5 MPa. With the increase of carbonization temperature, the specific capacity of flexible electrode increases gradually. The specific capacity of flexible electrode reaches 282 mAh.g−1 at 0.1c charge/discharge rate. The specific capacity of flexible electrode reaches 200 mAh.g−1 at 5C charge/discharge rate. The specific capacity retention rate of the flexible electrode is above 95% when the cycle is 100 weeks at 0.1 c and 600 weeks at 5C. CV test results show that with the decrease of scanning rate, the more stable the cyclic performance of flexible electrode is, the better the reversibility of Li+ stripping/embedding is. The conductivity of the flexible electrode increases with the increase of carbonization temperature. The surface resistance and AC impedance of the flexible electrode are reduced. When the carbonization temperature is 1000°C, the surface resistance of the flexible electrode is 2 Ω.−1, and the AC impedance is 5 Ω.
Introduction
In recent years, With the development of science and technology and the renewal of electronic products. Portable wearable electronics are also emerging. The potential demand for flexible electronic equipment is increasing, such as folding flexible screens in the bendable area, implantable electronic devices, ultra-thin mobile phones and multimedia wearable systems. This has promoted the research and development of multifunctional flexible and deformable energy storage devices. Flexible electronic devices have made great progress, but traditional energy storage devices cannot meet the rapid development of wearable devices due to their poor mechanical properties. 1 Therefore, it is urgent to develop energy storage devices with excellent electrochemical performance, flexibility and mechanical properties.
Among various emerging energy storage devices, lithium-ion batteries have the advantages of long cycle life, non-toxicity, low self-discharge efficiency and high quality/volume energy density. It is widely used in various energy storage devices. A traditional lithium-ion battery is composed of a casing, a current collector in the form of a metal foil, a battery separator, an electrolyte, and positive and negative electrodes. 2 Flexible foldable optoelectronic devices are one of the most promising research in the next generation. However, the current coating and assembly process of lithium-ion batteries determines that traditional lithium-ion batteries cannot be bent and folded. The bending process will cause the electrode material to fall off from the current collector. This will affect the performance of the battery, and may even pierce the diaphragm. At the same time, this will short-circuit the battery and affect the battery life. 3 In the current research on flexible lithium-ion batteries, flexible substrates are very important. Flexible electrode substrates are divided into conductive substrates and non-conductive substrates The conductive substrates may include conductive fibers, carbon fibers, carbon cloth, and other conductive substrates. They are light in weight and have excellent electrical conductivity and mechanical properties. The non-conductive substrates include some non-conductive substrates made of cellulose. These flexible electrodes have poor conductivity but excellent mechanical properties and flexibility. 4 In recent years, researchers have not stopped their research on flexible electrodes. M.Rana et al. adsorbed Fe2O3 nanoparticles on a bacterial cellulose nanofiber with a 3D grid structure. Carbonized at the optimal temperature of 600°C to obtain three-dimensional carbon nano Fe3O4 carbon fiber aerogel. The aerogel is highly flexible and can be directly used as a working electrode. The specific capacity of the aerogel after 80 cycles is still higher than 750 mAh.g−15; C.Sun et al. obtained graphene paper by filtering the graphene dispersion. The V2O5 cathode was grown on graphene paper using pulsed laser deposition in a vacuum chamber. The first discharge capacity is 806 mAh.g−1 at a rate of 0.5 C 6 ; L.Zhang et al. succeeded in growing a neat array of Ca2Ge7O16 nanowires on carbon paper by a simple, large-scale hydrothermal method, and formed a porous 3D structure. The specific capacity remains at 900–1100 mAh.g−1 after 100 cycles at 300 mA.g−17; E.Kasaw et al. used a highly scalable solid-state reaction method to prepare LiNi0.5Mn1.5O4 particles, and used a vacuum impregnation method to compound LiNi0.5Mn1.5O4 on carbon paper directly as a cathode material. This method avoids the use of current collectors and adhesives, and reduces the mass of the traditional electrode by 55% 8 ; S.Ullah et al. prepared flexible ZnCo2O4 nanofilament array/CFRP anode material by hydrothermal method. The specific capacity of the electrode remains at 1200 mAh.g−1 after 160 cycles at a rate of 0.2 C. 9
Cotton fabric has the three-dimensional porous structure. It has the advantages of good mechanical elasticity, high corrosion resistance, low cost, light weight and easy processing. It is an ideal base material for the preparation of flexible lithium ion electrodes. 10 Before this research, our research team has used nanofibrillated cellulose to prepare flexible lithium ion electrodes. Its specific capacity reached 180 mAh.g−1. It had excellent mechanical properties, but there were still some problems such as cyclic instability and low electronic conductivity. In order to solve these problems, cotton fabric was used as the base of flexible electrode in this study. The prepared flexible electrode has excellent mechanical and electrochemical properties. PDA is a surface modifier. It is widely used in the field of material functionalization and surface modification. 11 In this study, cotton fabric was modified with PDA. It effectively improved the mechanical strength and electrochemical properties of carbonized cotton fabric. The results showed that the three-dimensional porous cotton fabric can store more lithium ions. This greatly increased the capacity of flexible lithium-ion batteries. It is of great significance to use carbonized cotton fabric as electrode material for flexible lithium ion battery.
Preparation of flexible electrode sheet
Configuration of PDA Modification Solution
0.2 g of dopamine hydrochloride solid was weighed and dispersed in 100 mL distilled water. 1 mL tris-hydrochloric acid buffer was added to the dispersion and the solution PH was adjusted to 8.5.
Preparation of carbonized samples of cotton fabric
The cotton fabric used in this study was from The Two houses of Cloth. It had excellent mechanical properties. The Cotton fabric was cut into 5 * 8 cm specifications of the samples. The cotton fabric was placed in polydopamine modified solution, and the reaction was rotated for 24 h. The cotton fabric was taken out and dried in a vacuum oven at 60°C.Samples of cotton fabric modified by PDA were named cotton fabric@PDA. The dried cotton fabric@PDA was carbonized in a vacuum tube furnace. The carbonized cotton fabric@PDA were named cotton fabric@CPDA. The carbonization steps were as follows: Passed nitrogen into the tubular furnace. First kept it warm for 1 h at 300°C, and then heated it up to 800°C, 900°C and 1000°C respectively at the rate of 5°C/min. Kept it warm at three different temperatures for 1h, and took it out after cooling to room temperature. The Carbonization process of cotton fabric@PDA is shown in Figure 1. Carbonization of cotton fabric.
Preparation of flexible electrodes
The LFP electrode active material, conductive carbon black and binder (PVDF) were dispersed in N-methylpyrrolidone (NMP) at a ratio of 8:1:1. We immersed the carbonized sample of cotton fabric@CPDA in the dispersed electrode slurry. The soaking concentration of LFP were 0.08 g.ml−1 and 0.16 g.ml−1. After immersion, the sheet was dried in a vacuum oven at 100°C for 10 h. The flexible electrode were successfully prepared. Afterwards, the flexible electrod with different LFP qualities were obtained, which were left for later preparation of half-cells.
Results and discussion
Figure 2 shows the infrared spectrum of each sample. It can be seen from the Figure 2 that cotton fabric and cotton fabric@PDA have obvious characteristic peaks around 3300 cm−1, 2894 cm−1, 2118 cm−1, 1621 cm−1, 1330 cm−1, and 1030 cm−1. Near 3300 cm−1 is the stretching vibration peak of -OH. Near 2894 cm−1 is the stretching vibration peak of CH. Near 1030 cm−1 are the stretching vibration peaks of C-O-C and C-C. Near 1330 cm−1 is the flexural vibration peak of CH. Some of the other peaks may be impurity peaks. The results show that the peak strength of the corresponding functional groups decreases or disappears after the cotton fabric was carbonized. This is because during the carbonization process of cotton fabric, oxygen-containing functional groups react to produce gas.
12
This shows that the carbonization degree of cotton fabric is relatively complete.13,14 Infrared spectrum of each sample
Figure 3(a) shows the Raman spectra of cotton fabric and carbonized cotton fabric. It can be seen from the figure that the cotton fabric has a broad peak near 1500 cm−1. After carbonization of the cotton fabric, peaks D and G appear around 1360 cm−1 and 1600 cm−1, respectively. The D peak is used to characterize the movement of sp3 hybrid bonds in the graphite structure defects of carbon materials. The G peak is used to characterize the movement of sp2 hybrid bonds related to the integrity of the structure in carbon materials. The ratio of peak D to peak G can reflect the integrity of the carbon material and the degree of graphitization. The smaller the ratio of D peak to G peak, the higher the degree of carbonization and graphitization. The ratio of D peak to G peak of carbonized cotton fabric is 0.9.It shows that the degree of carbonization and graphitization of the cotton fabric is relatively high.15,16 (a) Raman spectrum of cotton fabric and cotton fabric@CPDA ; (b) XRD spectrum of cotton fabric and cotton fabric@CPDA
Figure 3(b) shows the X-ray diffraction analysis chart of cotton fabric. It can be seen from the Figure3(b) that the cotton fabric shows the peaks of four unique organic functional groups of the fiber fabric. The corresponding crystal planes are 101, 101-, 002 and 040 crystal planes respectively. The corresponding angles are 14.9°, 16.7°, 22.8°, 34.5°. After the cotton fabric was carbonized, the strong diffraction peak of cotton fabric disappeared. This shows that part of the oxygen-containing functional groups were reacted at high temperature during the carbonization process and could not survive. It is worth noting that a broad diffraction peak appears near 23.4°, which corresponds to the typical steamed bun peak of carbon materials. At the same time, it corresponds to the 002 crystal plane of carbon material. The appearance of this peak indicates that the carbonized cotton fabric has the properties of graphite crystals.17,18
Figure 4 shows the SEM image of each sample. It can be seen from Figure 4(a) that the fibers of the cotton fabric are arranged neatly and tightly and have high strength before carbonization. Figure 4(b) and (c) shows that after the cotton fabric was carbonized,, the fibers became twisted and dry. This is because the water vapor in the cotton fabric reacted with carbon during the carbonization process to produce H2 and CO2. After carbonization, only carbon remained in the cotton fabric. Figure 4(c) shows that the PDA particles adhered to the surface of the fiber, which improved the electron mobility on the surface of the cotton fabric. It can be seen from Figure 4(d) and (e) that after the LFP was coated on the surface of the carbonized cotton fabric, the pores on the surface of the cotton fabric were covered. This further improved the electron mobility of the flexible electrode. Figure 4(f) shows that the LFP particles have regular morphology and the crystal morphology is intact. It can be seen from Figure 4(g) and (h) that the surface of the flexible electrode is flat and has good bending ability.19,20 (a) SEM image of Cotton fabric; (b) SEM of cotton fabric@CPDA(900°C and 200 times); (c) SEM of cotton fabric@CPDA(900°C and 1000 times); (d) SEM of Flexible electrode(900°C and 200 times); (e) SEM of Flexible electrode(900°C and1000 times); (f)SEM of LFP; (g) Physical Picture of Flexible Electrode; (h) Flexible electrode bending diagram.
Figure 5(a) (a) shows the stress-strain curve of the cotton fabric raw material. Figure 5(b) shows the stress-strain curve of the flexible electrode. The integral area of the curve is the break work of the membrane material. It can be seen from Figure 5(b) that as the carbonization temperature increased, the area enclosed by the stress-strain curve of the flexible electrode decreased. The fracture work done by the flexible electrode was reduced, and its strength was reduced. This is because as the carbonization temperature increased, the mechanical strength of the cotton fabric decreased. As shown in Table 1, as the load of lithium iron phosphate increased, the elastic modulus and the rigidity of the flexible electrode increased. When the carbonization temperature was 800°C and the LFP immersion concentration was 0.08 g.ml−1, the elastic modulus of the flexible electrode was 10.21 MPa. When the LFP immersion concentration was 0.16 g.ml−1, the elastic modulus of the flexible electrode was 17.35 MPa. It can be seen from Figure 6 that the mechanical strength of cotton fabric was higher. The tensile strength of carbonized cotton fabric was about 0.5 MPa. As the loading of lithium iron phosphate increased, the mechanical strength of the flexible electrode increased.21–23 (a) The stress-strain curve of cotton fabric; (b) The stress-strain curve of flexible electrode. Elastic modulus of flexible electrode. Cycle rate curve of flexible electrode(a) LFP 0.08 g.ml−1; (b) LFP 0.16 g.ml−1. Charge and discharge platform of flexible electrode(c) 800°C/LFP 0.08 g.ml−1; (d)800°C/LFP 0.16 g.ml−1; (e)900°C/LFP 0.08 g.ml−1; (f) 900°C/LFP 0.16 g.ml−1; (g) 1000°C/LFP 0.08 g.ml−1; (h)1000°C/LFP 0.16 g.ml−1.

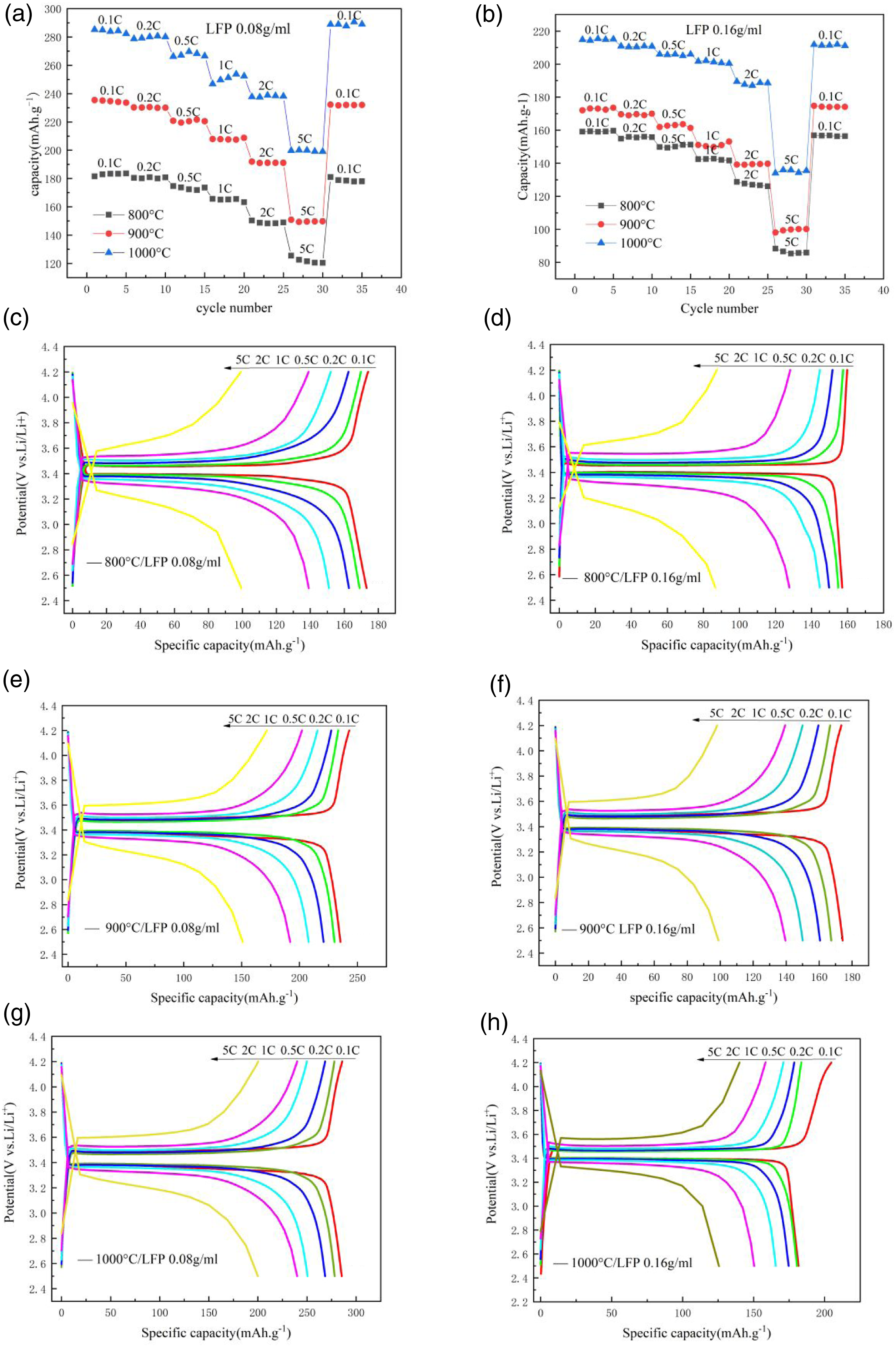
The battery test system was used to test the rate performance of half-cells. The half-cells are subjected to cyclic charging and discharging performance tests at six rates of 0.1 C, 0.2 C, 0.5 C, 1C, 2C, and 5C. Figure 6 showed the charge and discharge rate curve of the flexible electrode. The test results were as follows. It can be seen from Figure 6(a) and (b) that when the discharge rate increasesd from 0.1 C to 5C, the specific discharge capacity of the battery gradually decreased. When the discharge rate returns to 0.1 C, the specific discharge capacity of the battery remained basically unchanged. Taking the LFP 0.08 g.ml−1 immersion concentration as an example, when the carbonization temperature was 800°C and the discharge rate was 0.1 C, the discharge specific capacity of the flexible electrode was 182 mAh.g−1. When the discharge rate rised to 5C, the discharge specific capacity dropped to 120 mAh.g−1. When the discharge rate returns to 0.1 C, the discharge specific capacity was 180 mAh.g−1, and the capacity retention rate was 99%. It showed that the stability of the flexible electrode was excellent. As the carbonization temperature increased, the specific discharge capacity increased. When the LFP immersion concentration was 0.08 g.ml−1 and the carbonization temperature was 1000°C, the discharge specific capacity of the flexible electrode reached up to 282 mAh.g−1. It showed that the conductivity and the specific capacity of the flexible electrode increased with the increase of carbonization temperature. When the LFP immersion concentration was 0.16 g.ml−1 and the carbonization temperature was 1000°C, the discharge specific capacity of the flexible electrode was 215 mAh.g−1. It showed that as the load of LFP increaseed, the specific capacity of the flexible electrode would decrease.24-26
It can be seen from Figure 6(c) to (h)that the smaller the charge-discharge rate was, the longer the charge-discharge platform lasted, and the higher the platform voltage would be. This was because the greater the charge-discharge rate, the greater the capacity loss of the battery during cycling. The voltage platform would be shortened. Under the same charge-discharge rate, the electrode carbonized at 1000°C had a longer voltage plateau than the charge-discharge voltage at 800°C. The platform voltage was also higher. This was because the higher the carbonization temperature, the greater the electron conduction rate of the lithium-ion battery electrode and the higher its plateau voltage. 27
Figure 7(a) and (b) was the charge and discharge curve of the flexible electrode at a rate of 0.1 C for 100 weeks and Figure 7(c) and (d) of 5C for 600 weeks. It can be seen from the figure that the capacity remained basically unchanged. The capacity retention rate was over 95%. When the load of LFP increaseed, the cycle curve of the flexible electrode fluctuated instead. This was due to the increase in active materials, the internal reaction of the battery became more complicated. (a) Cycle curve of flexible electrode at 0.1 C magnification(LFP 0.08 g.ml−1); (b) Cycle curve of flexible electrode at 5C magnification(LFP 0.16 g.ml−1); (c) Cycle curve of flexible electrode at 5C magnification(LFP 0.08 g.ml−1); (d) Cycle curve of flexible electrode at 5C magnification(LFP 0.16 g.ml−1).
The electrochemical workstation was used to test cyclic voltammetry. Cyclic voltammetry was performed on half-cells at scan rates of 10 mv.s−1, 20 mv.s−1and 50 mv.s−1. It can be seen from Figure 8 that all samples had a pair of redox peaks. Corresponding to the redox reaction of Fe2+ and Fe3+.This was a typical charge and discharge characteristic of lithium iron phosphate. The voltage difference between the oxidation peak and the reduction peak could reflect the polarization of the electrode. The smaller the voltage difference between the redox peaks, the smaller the polarization, and the better the reversibility of Li+ extraction/intercalation. The greater the scan rate, the greater the voltage difference between the redox peaks. This showed that the higher the scan rate, the more unstable the battery interior and the more serious the polarization phenomenon.
28
(a) Cyclic voltammetry curve(800°C/LFP 0.08 g.ml−1); (b) Cyclic voltammetry curve(800°C/LFP 0.16 g.ml−1); (c) Cyclic voltammetry curve(900°C/LFP 0.08 g.ml−1); (d) Cyclic voltammetry curve(900°C/LFP 0.16 g.ml−1); (c) Cyclic voltammetry curve(1000°C/LFP 0.08 g.ml−1); (d) Cyclic voltammetry curve(1000°C/LFP 0.16 g.ml−1).
Surface resistance of flexible electrodes.

Figure 9 showed the AC impedance spectrum of the half-cell. The AC impedance spectrum is composed of an approximate semicircle in the high frequency region and a diagonal line in the low frequency region. The diameter of the semicircle is the macroscopic expression of the charge transfer resistance (Rct) of the material. The smaller the semicircle diameter, the smaller the Rct of the material. The semicircle in the high frequency region represents the surface resistance of lithium ions passing through the interface, and the oblique line in the low frequency region represents the diffusion resistance of lithium ions.29,30 It can be seen from the Figure 9 that as the carbonization temperature increased, the AC impedance of the flexible electrode decreased. When the carbonization temperature rised to 1000°C, the AC impedance of the flexible electrode reduced to 5 Ω. This is because with the increase of carbonization temperature, the better the carbonization effect of cotton fabric and the higher the conductivity. (a) AC impedance spectra of flexible electrodes(800°C/LFP 0.08 g.ml−1); (b) AC impedance spectra of flexible electrodes(800°C/LFP 0.16 g.ml−1); (c) AC impedance spectra of flexible electrodes(1000°C/LFP 0.08 g.ml−1); (d) AC impedance spectra of flexible electrodes(1000°C/LFP 0.16 g.ml−1).
Conclusion
In this study, The cotton fabrics@PDA was carbonized to prepare flexible elctrode. PDA can enhance the electronic conductivity and mechanical strength of the carbonized cotton fabric surface. Through the physical structure characterization and electrochemical characterization of the electrode, the following conclusions can be drawn: 1. The elastic modulus of the flexible electrode gradually increases with the increase of the load of LFP. The elastic modulus of the flexible electrode is the lowest 10.21 MPa and the highest 29.33 MPa. The tensile strength of the flexible electrode is about 0.5 MPa. 2. As the charge and discharge rate decreases, the specific capacity of the battery gradually increases. When the charge-discharge rate changes 5C back to 0.1 C, the capacity retention rate is 99%. The specific discharge capacity increased with the carbonization temperature increased. The specific capacity of the battery is up to 282 mAh.g−1 with the carbonization temperature of 1000°C and the the charge-discharge rate of 0.1 C. 3. Cycle 100 weeks at the charge-discharge rate of 0.1 C and 600 weeks of 5C.The capacity retention rate is over 95%. 4. The CV tests shows that the smaller the scan rate, the smaller the voltage difference between the redox peaks and the better the reversibility of Li+ extraction/embedding. 5. The surface resistance of the flexible electrode decreases with the increase of the carbonization temperature. The surface resistance of the flexible electrode is 17 Ω.¨−1 at the carbonization temperature of 800°C, 6 Ω.¨−1 of 900°C and .2 Ω.¨−1 of 1000°C. 6. The AC impedance of the flexible electrode decreases with the carbonization temperature increases. The AC impedance of the flexible electrode is at least 8 Ω at the carbonization temperature of 800°C. When the carbonization temperature rise to 900°C and 1000°C, the AC impedance of the flexible electrode decrease to 9 Ω and 5 Ω.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
