Abstract
The cardiac patch provides appropriate physicochemical properties and mechanical strength for the regeneration of damaged heart tissues. In this work, for the first-time, beetroot (BR) is blended with cerium oxide (CeO2) to produce nanofibrous polyurethane (PU) composite patch using electrospinning. The objective of this work is to fabricate the composite and examine its feasibility for cardiac patch applications. Morphological analysis revealed a dramatic reduction of fiber diameter of PU/BR (233 ± 175 nm) and PU/BR/CeO2 (331 ± 176 mm) compared to the pristine PU (994 ± 113 mm). Fourier transform infrared analysis (FTIR) analysis indicated functional peak intensities of the newly formed composite PU/BR and PU/BR/CeO2 were not similar to PU. The addition of beetroot rendered PU/BR hydrophilic (86° ± 2), whereas PU/BR/CeO2exhibited hydrophobic nature (99° ± 3). Atomic force microscopy (AFM) analysis depicted the reduced surface roughness of the PU/BR (312 ± 12 nm) and PU/BR/CeO2 (390 ± 125 nm) than the pristine PU (854 ± 32 nm). The incorporation of beetroot and CeO2 into PU enhanced the tensile strength compared with the pristine PU. The blood clotting time of PU/BR (APTT-204 ± 3 s and PT-103 ± 2 s) and PU/BR/CeO2 (APTT-205 ± 3 s and PT-105 ± 2s) were delayed significantly than the pristine PU(APTT-176 ± 2 s and PT-94 ± 2 s) as revealed in the coagulation study. Further, hemolysis assay showed the less toxic nature of the fabricated composites than the pristine PU. Hence, it can be inferred that the advanced physicochemical and blood compatible properties of electrospun PU/BR and PU/BR/CeO2 nanocomposite can be engineered successfully for cardiac patch applications.
Introduction
Myocardial Infarction (MI) and heart failure are the foremost causes for death globally. MI occurs when blood flow to the heart is reduced causing blockage of coronary arteries leading to heart muscle damage. 1 MI is followed by several events like inflammatory responses, cardiomyocyte death, the formation of scar, infracted region expansion, and left ventricular wall thinning.2,3 The heart fails to function along with these events and in the final stages, there will be concomitant arrhythmias and mitral regurgitation resulting in cardiac failure.4,5 Heart transplantation is the golden standard for overcoming cardiac failure, however, the scarcity of donors’ forces physicians and researchers to look for other viable alternative routes. 6 Standard treatments/therapies such as thrombolytic/fibrinolytic therapy, coronary angioplasty, and coronary artery bypass grafting were used in remodeling of the damaged heart muscle. However, these are not efficient in repairing the cardiac tissue.7–9 The usages of a cardiac patch for regenerating damaged tissues is experimented and there is a vast potential for the advanced material to aid this problem. 10 The hourly need for the cardiac patch materials motivated material scientists to collaborate with physicians to develop and experiment with new materials. The key aspect of the search is that the new materials should bio-mimic the properties of the heart extracellular matrix and support the cardiomyocyte adhesion and proliferation for new tissue growth. 11
To achieve this, several ways were utilized to fabricate the cardiac patch materials. One of the versatile and economic modes of fabricating such material is electrospinning. 12 A typical electrospinning apparatus set up mainly consists of three components, namely, high electric field, collector drum, and syringe pump. 13 The electrospinning technique uses high voltage to convert the polymeric solution loaded in the syringe pump into fibers which are collected at the collector drum. 14 Fibers produced through the electrospinning technique possess tremendous advantages such as large specific surface area and porous structure grossly mimicking the extracellular matrix (ECM) which makes them a widely accepted choice for tissue engineering applications.15–17 Recently, polymeric scaffolds play a vital role in tissue engineering applications. 18 The characteristics and properties of polyurethane are ease of processing, flexibility, biocompatibility, biodegradable, haemocompatibility, and tailorable chemical and physical forms. 19 Few works highlighted the usage of electrospun polyurethane fibers for tissue engineering applications.20,21 Polyurethane has been blended with various molecules in order to improve its bioactivity behavior for tissue engineering applications. Few literatures reported the usage of nanoparticles incorporated PU scaffold for tissue engineering applications.22,23 Cerium oxide is reported to be beneficial in reactive oxygen species scavenging 24 and also possesses antioxidant behavior. 25 Cerium oxide-loaded scaffolds holds potential in the biomedical field 26 and their application is limited due to toxicity. 27 Fruits and vegetables contain wide sources of minerals, nutrients, vitamins, folic acid, and dietary fiber. Some components of fruits and vegetables (phytochemicals) possesses strong antioxidants behavior. Further, the phytochemicals present in the fruits and vegetables have a complementary mechanism which include antibacterial and antiviral effects, scavenging of free radicals, promotes cell adhesion, proliferation, and differentiation and hormone metabolism regulation. 28 We hypothesize by blending with natural root vegetables like beetroot would be beneficial in overcoming this drawback of cerium induced toxicity to the tissue engineering scaffolds. Hence, this work for the first time attempts to combine cerium oxide with beetroot to develop a cardiac patch using electrospinning.
Beetroot (Beta vulgaris L.) is a vegetable plant belonging to the family Amaranthaceae. 29 Raw beetroot contains carbohydrates, protein, and fat. It provides 43 calories with a rich source of folate and a moderate source of manganese along with other nutrients. 30 It is used in cooking foods, food coloring, and also as a medicinal plant. 31 Beetroot is reported to be rich in nitrates which help in reducing the blood pressure thereby exerting benefits in protecting the cardiovascular system. Additionally, the consumption of beetroot juice exhibits cardioprotective properties because of the presence of betalains and antioxidant properties of its components.32–34 Recent reports indicated the various biological properties of Beta vulgaris extracts such as antioxidant, anti-inflammatory, antihypertensive, hypoglycemic, and hepatoprotective activities. 29 However, its potential is still untapped in the field of regenerative medicine. Cerium is a rare earth metal having anatomic number 58. It belongs to the lanthanide series and occurs in two oxidation states, that is, +3 and +4. Since it belongs to the lanthanide series, it is used as a catalyst, ultraviolet absorber, gas sensors, and polishing agent. It is also used as electrode material in gas sensors because of their very good oxide ion conductors. The cerium particles were reported to possess antioxidant properties. 35 Further, few studies have been reported the low toxicity nature of cerium particles which do not induce any cytotoxicity or inflammation.36–38 The application of cerium nanoparticles in biomedical applications is widely documented in areas such as redox modulation, drug delivery agent, pharmaceutical, treating bone cancer, and tissue engineering scaffolds.39–41 This study aims to develop a cardiac patch based on polyurethane added with beetroot and cerium oxide using electrospinning technique and explore its physicochemical and blood coagulation properties for promoting PU/BR and PU/BR/CeO2in cardiac patch applications.
Materials and methodology
Materials
Tecoflex EG-80A polyurethane ((molecular weight = 1000 g/mol)) was procured from Lubrizol and the solvent dimethylformamide (DMF) was purchased from Sigma Aldrich, UK. PU is a segmented polymer that has methylene bis(cyclohexyl) diisocyanate (hydrogenated MDI, H12MDI) as a hard segment and polytetramethylene oxide as a soft segment having a molecular weight of 1000 g/mol. It also contains 1,4 butane diols as a chain extender. Beetroot was obtained from local vendor. CeO2 was procured from Sigma Aldrich, UK. Phosphate buffered saline (PBS) and sodium chloride (NaCl) physiological saline (0.9% w/v) were procured from Sigma-Aldrich, Kuala Lumpur, Malaysia. The blood clotting assay reagents such as rabbit brain-activated cephaloplastin, calcium chloride (CaCl2) (0.025 M), and thromboplastin (Factor III) were purchased from Thermo Fisher Scientific, Selangor, Malaysia. All chemicals used in this work are of analytical grade.
Preparation of beetroot extract
Initially, the surface of the beetroot was peeled off fully and cleaned using distilled water. Using a common kitchen blender, it was grinded to obtain extracts from it. Finally, the filtered extract was stored in the refrigerator for further use.
Preparation of solution
Initially, PU was prepared at a concentration of 9 wt%. It was done by dissolving 360 mg in 4 ml of DMF and stirred for 12 h to attain a homogeneous solution. For BR homogeneous solution, 4 w/v% was used which was prepared by adding 120 μl of BRin 3 ml of DMF and stirred for 1 h. Similarly, CeO2 homogeneous solution was prepared at 4 wt% by adding 120 mg of CeO2 in 3 ml of DMF and stirred for 1 h. Finally, PU/BR and PU/BR/CeO2 composite solutions were prepared by mixing BR and CeO2 homogeneous solution into PU homogeneous solution at a ratio of 8:1 (v/v%) and 8:0.5:0.5 (v/v%) and stirred for 1 h maximum. The schematic representation of preparation of solution is indicated in Figure 1. Schematic representation of preparation of solution.
Electrospinning process
The electrospun technique was used to make the cardiac patch from the prepared PU,PU/BR, and PU/BR/CeO2 composite solutions. In this study, the applied parameters were voltage of 10.5 kV, a flow rate of 0.2 ml/h, and the collector distance was set at 20 cm. The collected nanofibers in the aluminium foil were detached carefully and vacuum dried at room temperature.
Physicochemical characterizations
Field Emission Scanning electron microscopy (FESEM)
FESEM unit (Hitachi SU8020) was utilized to investigate the morphology of the PU, PU/BR, and PU/BR/CeO2. Before attaining photomicrographs, samples cut into small pieces were gold plated at 20 mA for 2 min. Then, the images were captured at different magnifications with an accelerated voltage of 10 kV. The fiber diameter was calculated from the captured image using Image J by choosing 30 locations randomly. The cerium oxide present in the electrospun nanofibers were determined using energy dispersive X-ray (EDX) spectrometer attached to the FESEM unit.
Fourier transform infrared (FTIR) analysis
The characteristic bands present in the PU, PU/BR, and PU/BR/CeO2 were recorded using the Thermo Nicolet FTIR unit. ATR crystal fixed in the NICOLET IS5 spectrometer was zinc selenium (ZnSe). The sample spectra were recorded between the wavelength of 600 and 4000 cm−1 at 32 scans per minute with a resolution of 4 cm−1.
Contact angle measurements
The surface wettability of PU, PU/BR, and PU/BR/CeO2was determined using a video contact angle measurement unit. A water droplet image was deposited on the small cut mesh of the electrospun membrane and an image of the droplet laid was captured with the help of a high-resolution camera within a few seconds. The angle between the water droplet and the surface was calculated from the captured image using computer integrated software provided by the manufacturer.
Mechanical properties test
According to ASTM standard (ASTM D882-10), the tensile strength of PU, PU/BR, and PU/BR/CeO2 were investigated. For measuring this, samples with a rectangle size of 40 × 15mm2 were cut and fixed in the grips of the uniaxial tester. The test was carried out at a strain rate of 10 mm/min and pulled until failure occurs. The average tensile strength was obtained from the machine generated stress–strain curve.
Thermogravimetric analysis (TGA)
TGA unit (PerkinElmer TGA 4000 unit) was used to evaluate the thermal stability of the PU, PU/BR, and PU/BR/CeO2. All analysis was performed in a dry nitrogen atmosphere. A small piece weighing 3 mg was heated at a rate of 10°C/min with a flow rate of 50 mL/min at a temperature range of 30–1000°C.
Atomic force analysis (AFM)
AFM was performed using AFM apparatus (Nanowizard, JPK instruments) to analyze the surface properties of the PU, PU/BR, and PU/BR/CeO2. The sample surface area was scanned in a size of 20 × 20 μm area in a normal atmosphere at room temperature. The 3D digital images with 256 × 256 pixels were captured using JPJSPM software.
Blood compatibility measurements
Activated Partial Thromboplastin Time (APTT) Assay
The cut samples of electrospun PU and its composites scaffolds were incubated with 50 μL of obtained PPP for 1 min at 37°C and then added with 50 μL of rabbit brain cephaloplastin reagent for 3 min at 37°C. Finally, the mixture was added with 50 μL of CaCl2 which activates the blood clot. The time taken for the clot formation was measured using a chronometer.42,43
Prothrombin time (PT) assay
The cut samples of electrospun PU and its composites scaffolds were incubated with 50 μL of obtained PPP for 1 min at 37°C followed by adding 50 μL of NaCl–thromboplastin reagent (Factor III) which triggers the blood clot. The time taken for the formation of the clot is noted as PT.42,43
Hemolysis assay
To start this assay, the cut samples of electrospun PU and its composites scaffolds were cleaned in physiological saline (0.9% w/v) at 37°C for 30 min. After the samples were exposed to the citrated blood and diluted, saline mixture was prepared at 4:5 for 1 h at 37°C. Then, the samples were retrieved and centrifuged at 3000 r/min for 15 min. Finally, optical density (OD) was recorded for the aspirated supernatant which measures the damage of red blood cells (RBCs). The hemolytic percentage was calculated using the following formula as reported previously.42,43
Statistical analysis
All experiments were performed thrice independently. To measure the statistical significance, one-way ANOVA was carried out in Graph pas prism and the calculated results are denoted as mean ± SD and for qualitative experiments, a representative of three images is shown.
Result and discussion
FESEM investigation
Figure 2 depicts the FESEM images of nanofibers made from PU and PU, PU/BR, and PU/BR/CeO2. All three samples had nanofibers with smooth and randomly oriented fibers without any beads. ImageJ analysis depicted the average diameter of the pure PU measured was found to be 994 ± 113 nm, while PU/BR and PU/BR/CeO2 displayed average diameter of 233 ± 175 nm and 331 ± 176 nm, respectively (p < 0.05). FESEM images of (a) PU, (b) PU/BR, and (c) PU/BR/CeO2 (arrow mark indicates the intercalation of reinforcements within the polyurethane).
EDX table of electrospun membranes.

FTIR analysis
FT-IR spectra of PU, PU/BR, and PU/BR/CeO2 displayed in Figure 3 and Table 2 denotes the characteristics peak names.42,43 FTIR images of PU, PU/BR, and PU/BR/CeO2. Characteristic peaks of PU.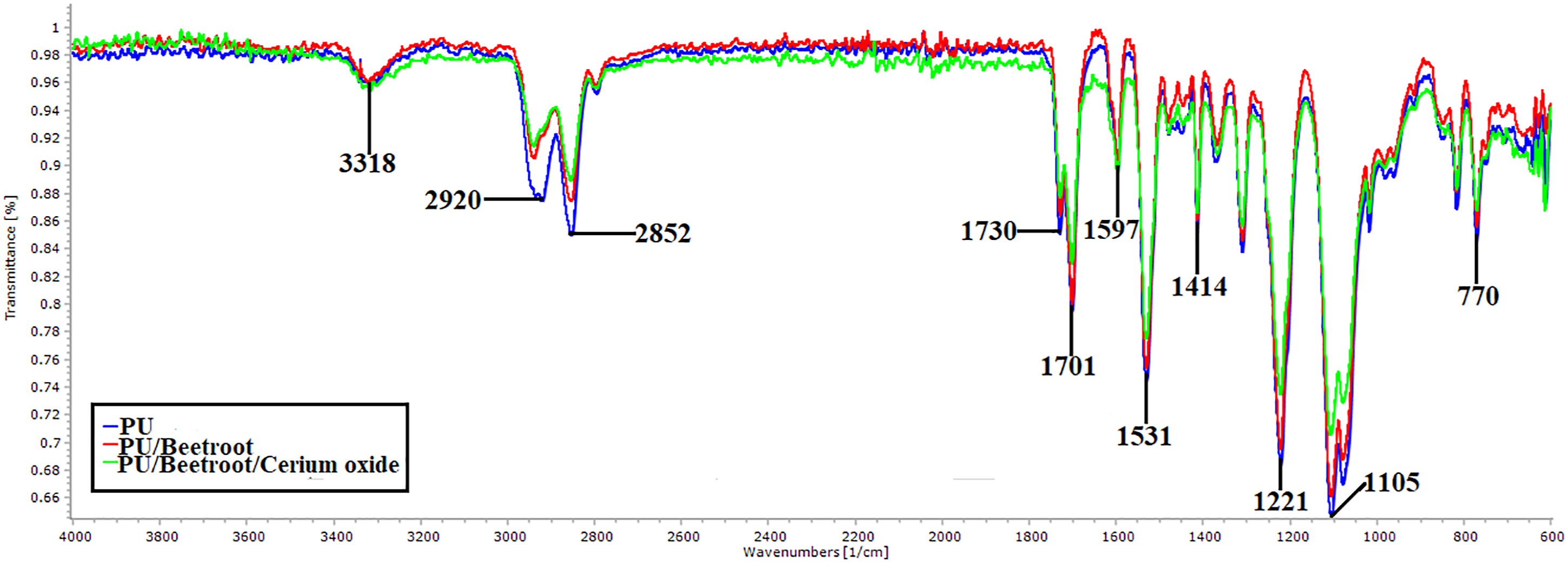

PU/BR and PU/BR/CeO2 displayed similar peaks like that of PU with no new peak formation. However, the addition of BR and CeO2 decreased the intensity of PU owing to strong hydrogen bond formation. 21 Further, the change in peak shift is observed while adding BR and CeO2 in the polyurethane matrix. 47 The CH peak at 2920 cm−1 was shifted to 2938 cm−1 in PU/BR and 2940 cm−1 in PU/BR/CeO2, respectively. The formation of hydrogen bonding and change in peak shift portrays the existence of BR and CeO2 in the matrix of PU.
Contact angle measurements
The contact angle measurements revealed the angle for the pristine PU was 105° ± 3, while the angle for the PU/BR and PU/BR/CeO2 was found to be 86° ± 2 and 99° ± 3, respectively (p < 0.05). The wettability measurements observed the hydrophobic nature of the polyurethane and it was decreased on adding BR indicating hydrophilic and the angle was increased on adding CeO2indicating hydrophobic. The most interesting observation is the addition of BR imparted PU as hydrophilic and making hydrophobic on adding CeO2. Norahan et al. fabricated electroactive graphene oxide (GO) containing collagen (Col) for cardiac tissue engineering. The contact angle of Col, Col–GO-90, and Col–rGO-90 were reported to be 113.3 ± 2.16, 70.06 ± 3.51, and 94.46 ± 2.78, respectively. It was found that the cell viability of both Col–GO-90 and Col–rGO-90 were reported to be higher.
48
The contact angle of our fabricated composites is found to be overlapping within the reported range and suggesting its feasibility for cardiac tissue growth. Jaganathan et al. developed a cardiac patch using an electrospinning technique based on a polyurethane scaffold added with nickel oxide. They had observed an increase in the hydrophobicity of the PU with the addition of nickel oxide and suggested it as a potential candidate for cardiac tissue engineering.
43
In another recent study, Wang et al. reported the fabrication of PU/poly(ethylene glycol) methacrylate
Thermal behavior
The thermal behavior of PU, PU/BR, and PU/BR/CeO2is presented in Figure 4. It is observed that PU displayed an initial onset degradation temperature at 284°C and for the electrospun PU/BR and PU/BR/CeO2, it was estimated to be 289°C and 281°C, respectively. The thermal performance of PU was increased on incorporating BR and on adding CeO2 it is found to have been decreased owing to the degradation of oxide group present in it. Further, the weight loss peaks of the electrospun PU, PU/BR, and PU/BR/CeO2were presented in Figure 5. The degradation in PU occurs in three stages, namely, the first loss observed at 220–360°C, the second loss between 360 and 472°C, and the third loss occurs at 472–732°C, respectively. These three losses were because of the decomposition of rigid and flexible segments of the PU. In the electrospun PU/BR composite, it showed four weight losses in which the first loss is observed at 227–331°C, the second loss around 331–367°C, the third loss at 367–492°C, and the fourth loss at 492–817°C.It was noted that in PU/BR composite, the number of weight loss peaks and their peak intensity is increased compared to PU membrane indicating enhanced weight loss. BR containing water molecules which might have resulted in the enhancement of the weight loss. In the case of electrospun PU/BR/CeO2composite, the weight loss occurs in four stages, namely, first weight loss occurs at 236–374°C, second loss at 374–428°C, third weight loss at 428–489°C, and final loss at 489–654°C, respectively. With the addition of cerium oxide, the numbers of weight loss peaks were the same as that of PU/BR. However, the peak intensity is increased compared to the pristine PU indicating increased weight loss. The change in weight loss peak intensity confirms the presence of BR and CeO2 in the polyurethane matrix. TGA curve of (a) PU, PU/BR, and PU/BR/CeO2. DTG curve of PU, PU/BR, and PU/BR/CeO2.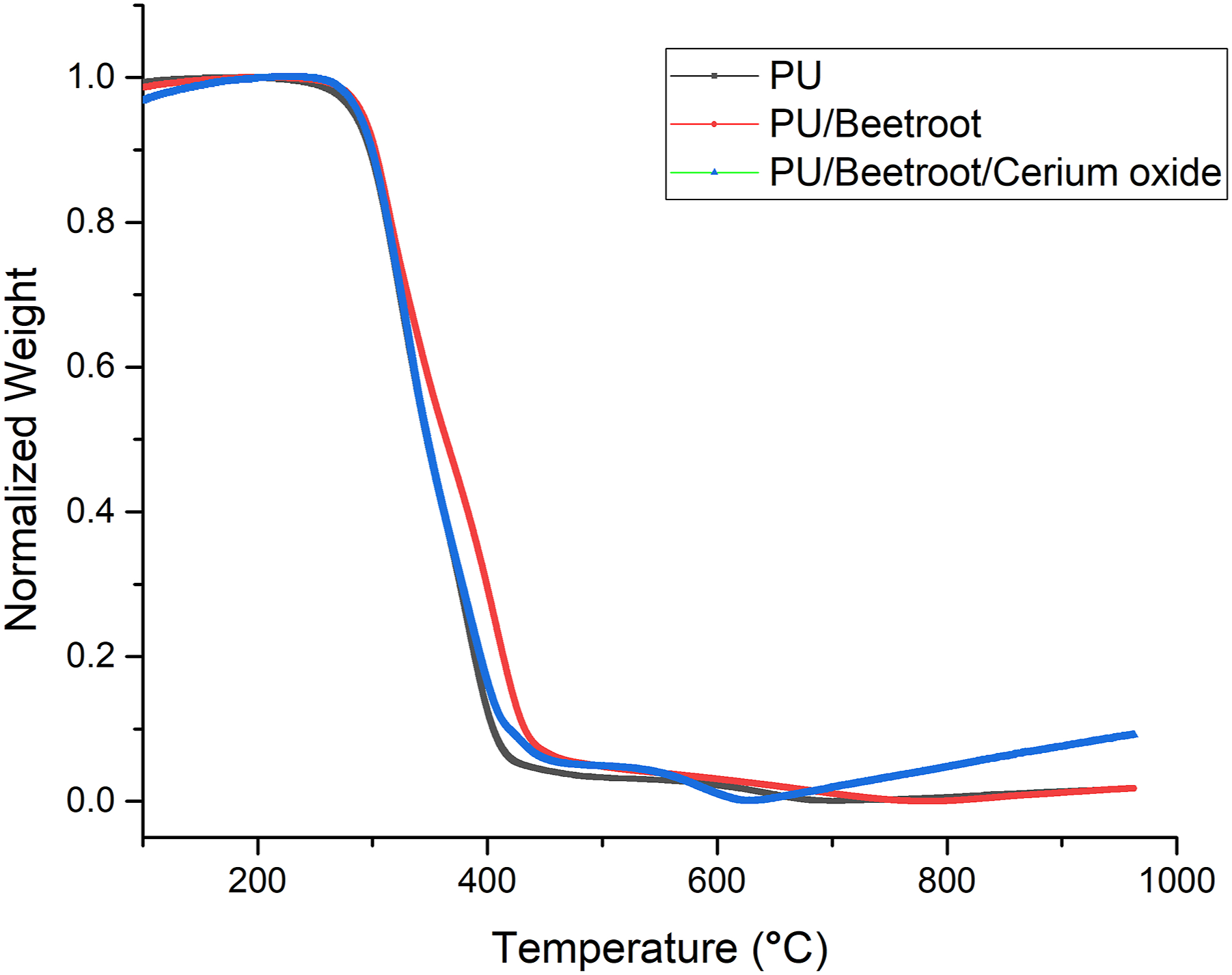

Tensile testing
Figure 6 denotes the stress–strain curves of the fabricated patches PU, PU/BR, and PU/BR/CeO2. PU displayed a tensile strength of 6.16 MPa, while in the case of PU added with BR and BR/CeO2, the strength was found to be 7.57 MPa and 10.92 MPa, respectively (p < 0.05). Further, their mean and standard deviations were given in Table 3. Tensile curves of PU, PU/BR, and PU/BR/CeO2. Mean and standard deviation of tensile strength for electrospun membranes.

It is observed that the tensile strength of the PU is improved by adding BR and CeO2. The added components might have intercalated with the polyurethane. This might have resulted in bonding between the fibers making them stronger in resisting the fibers break. Chen et al. fabricated polyurethane scaffold added with cellulose for cardiac tissue engineering. It was observed that the addition of cellulose increased the tensile strength of the pristine PU which resembles our findings. The reported tensile strength of PU, P9C1_M, and P9C1_L were reported to be 4.6 ± 1.08, 12.8 ± 3.27, and 8.1 ± 0.67, respectively. 50 Our reported tensile strength was closely matching the reported range indicating its suitability for cardiac tissue engineering. Seikh et al. fabricated an electrospun scaffold based on PU/silver nanoparticles and it is found that the addition of additives improved the mechanical strength of PU which resonates with our findings and conclude this might because of their smaller fiber diameter. 51 In this study, the addition of BR and cerium oxide in the pristine PU showed reduced fiber diameter which might have resulted in the enhanced tensile strength.
AFM analysis
AFM images of electrospun PU, PU/BR, and PU/BR/CeO2wereshown in Figure 7. The measured Ra for the electrospun PU, PU/BR, and PU/BR/CeO2 was found to be 854 ± 32, 312 ± 12, and 390 ± 125 nm, respectively (p < 0.05). Hence, PU surfaces were found to be rougher and become smoother on adding BR and CeO2. The decrease in the surface roughness can be owed to the interaction of molecules present in BR and CeO2 with the molecules of pristine PU.As reported in the FTIR study, the interaction of the molecules might cause hydrogen bonding resulting in a decrease in surface roughness. Saporito et al. reported that the scaffold based on gelatin and chondroitin sulfate displayed enhanced adhesion and proliferation of fibroblast cells suggesting it as a potential candidate for cardiac tissue growth. In another study, Jang et al. also reported that the electrospun scaffold comprising poly(L-lactide-co-ε-caprolactone) (PLCL) exhibited improved fibroblast cell attachment and proliferation. Hence, the fibroblast cells play a key role in repairing the damaged cardiac tissue.52,53 Kim et al. investigated the effect of the fiber diameter on the surface roughness of electrospun poly(ε–caprolactone) scaffolds. It had been found that a scaffold with a smaller fiber diameter showed a smoother morphology and became rougher when the diameter became larger.
54
Hence, our fabricated composites showed smaller fiber morphology which might favor the smoother surfaces. It has been reported that the smooth surfaces might favor the enhanced adhesion and proliferation of fibroblast cells.
55
Hence, the smoother surfaces of the developed composites might be used for the new cardiac tissue growth. AFM images of (a) PU, (b) PU/BR, and (c) PU/BR/CeO2.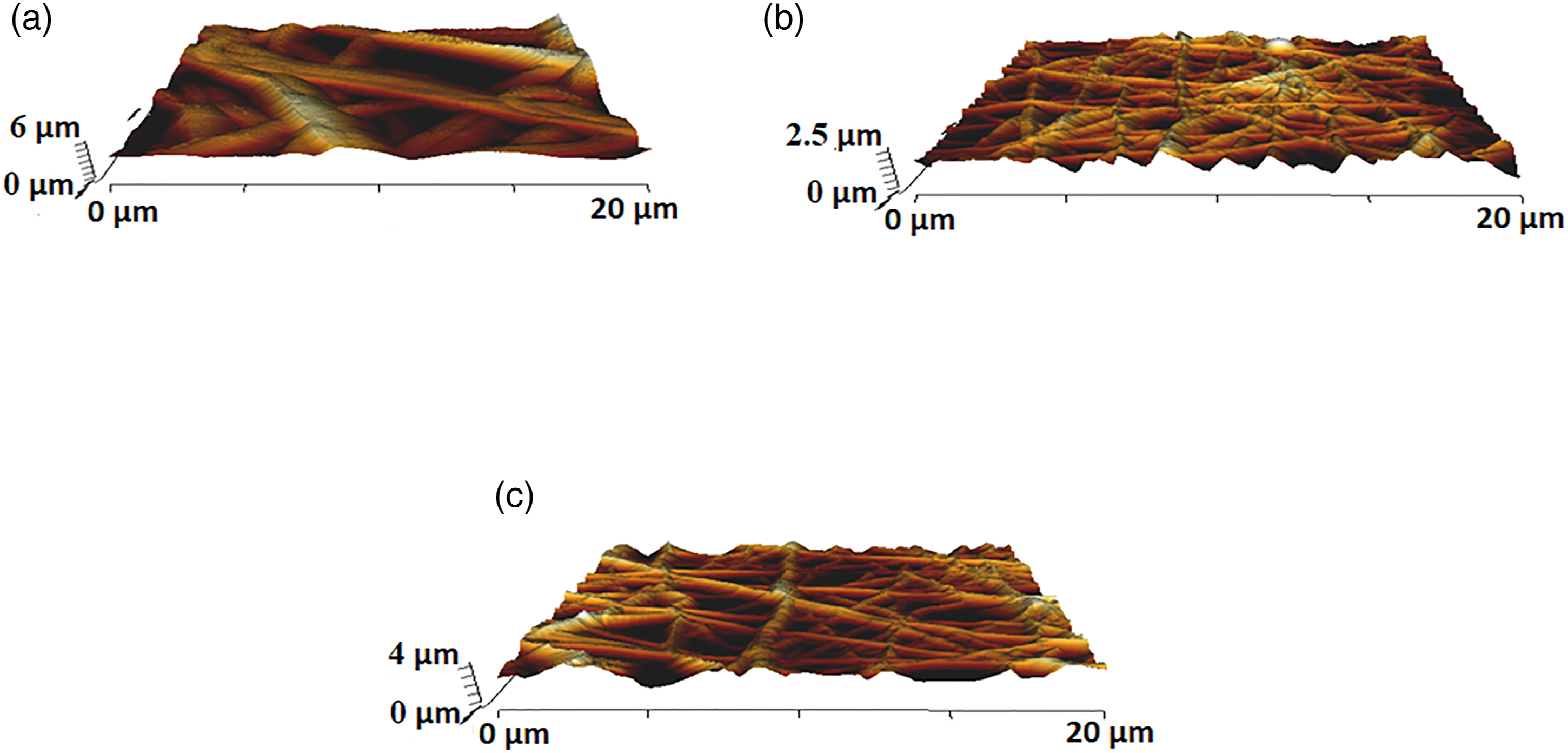
Blood compatibility assessments
The anticoagulant behavior of the electrospun membranes is determined through APTT, PT, and hemolytic index and the corresponding images were denoted in Figures 8–10. APTT assay displayed the clotting time for PU is about 176 ± 2 s and for PU added with BR and BR/CeO2 it is estimated to be 204 ± 3 s, and 205 ± 3 s (p < 0.05). Similarly, PT assay displayed the clotting time for PU is about 94 ± 2 s and for PU added with BR and BR/CeO2, it is found to be 103 ± 2 s, and 105 ± 2 s (p < 0.05). Hence, the coagulation assay has depicted the increase in the blood clotting time of the PU on adding BR and CeO2. APTT measurements of PU, PU/BR, and PU/BR/CeO2. PT measurements of PU, PU/BR, and PU/BR/CeO2. Hemolytic index of PU, PU/BR, and PU/BR/CeO2.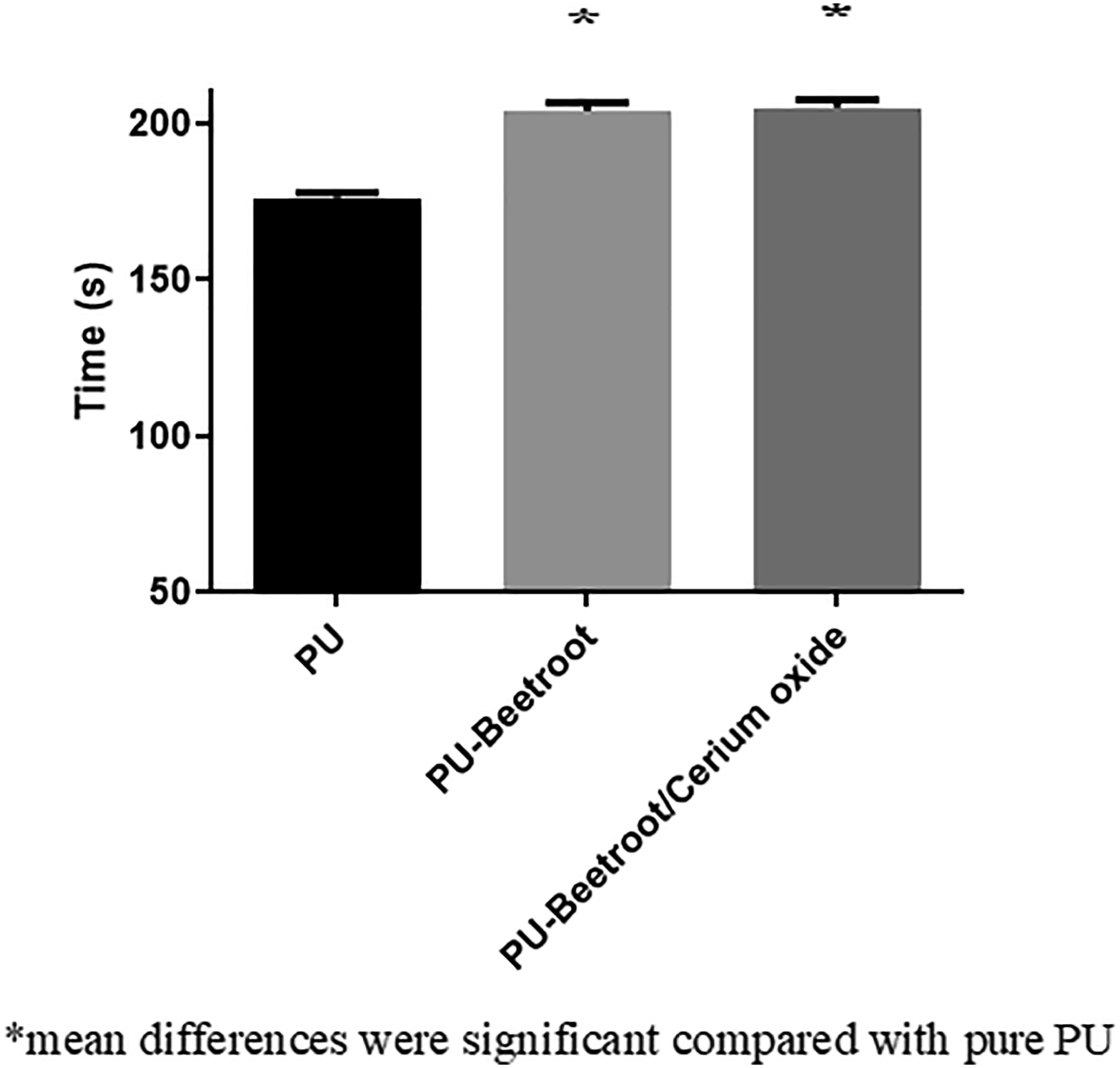


Further, the hemolytic assay was done to evaluate the hemoglobin release from the electrospun patches. The hemolytic assay measurements depicted the PU membranes added with BR and PU/BR/CeO2 patches showed less toxic behavior to the RBC’s. The hemolytic index of the pristine PU was found to be 2.83%, while the PU having BR and BR/CeO2 nanofibers showed hemolytic index of 1.633% and 1.74%, respectively (p < 0.05). Hence, the composite materials were considered as non-hemolytic nature (index below 2%) according to ASTMF756-00(2000).42,43 Jaganathan et al. prepared an electrospun scaffold based on polyurethane added with Alternanthera sessilis oil (AS). The addition of AS oil improved the blood clotting time of the PU which resembles our findings. They concluded this behavior was owing to their smaller fiber diameter and hydrophobic behavior. 42 In our study, the electrospun nanocomposites displayed smaller fiber diameter (PU/BR and PU/BR/CeO2) and hydrophobic (PU/BR/CeO2) behavior which might have favored the enhanced blood compatibility.
Conclusion
The cardiac patch requires materials with tailor-made properties to suit its function. This work developed PU/BR and PU/BR/CeO2 patches with adequate properties mimicking the extracellular matrix of the cardiac tissue via: Nanofibrous morphology (Decrement percentage was 77% for PU/BT and 67% for PU/BT/CeO2 compared to the pristine PU) and altered wettability of the fabricated composites compared to the pristine PU. Reduced surface roughness (Decrement percentage was 63% for PU/BR and 54% for PU/BR/CeO2) and enhanced tensile strength (Percentage increment was 23% for PU/BR and 77% for PU/BR/CeO2 compared to the pristine PU) of the electrospun composites compared to the pristine PU. Blood coagulation and hemolysis assay showed its antithrombogenic (Percentage increment was 16% for both PU/BT and PU/BT/CeO2in APTT assay and in PT assay, the percentage increment was 10% for PU/BT and 12% for PU/BT/CeO2) and less toxic nature of the fabricated composites than the pristine PU. Hence, the electrospun PU/BR and PU/BR/CeO2 nanocomposite with tailor-made characteristics indicating its potential benefits for cardiac patch applications.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Ahmad Athif Mohd Fauzi would like to acknowledge the grant (vot.no QJ130000.21A6.00P13) funded by Ministry of Higher Education, Malaysia.
