Abstract
In this study, composite based on polyurethane (PU) containing lemon grass oil (LG) and zinc nitrate (ZnNO3) was fabricated using electrospinning technique. Morphology study revealed the fabricated scaffolds PU/LG and PU/LG/ZnNO3 diameter was lower than polyurethane. LG and ZnNO3 form hydrogen bond with polyurethane as revealed in the infrared analysis. The developed PU/LG composite rendered hydrophobicity while PU/LG/ZnNO3 showed hydrophilic nature than PU. Atomic force microscopy (AFM) depicted the decrease in surface roughness of the nanocomposite compared to polyurethane. The addition of LG and ZnNO3 improved the mechanical strength of the pristine PU as indicated in tensile analysis. Coagulation assay measurements indicated a delay in the activation of clot and also exhibited reduced toxicity for the developed composites PU/LG and PU/LG/ZnNO3. Moreover, the deposition of calcium in the developed composites were found to be higher compared to the PU as noted in the bone mineralization testing. Hence, these developed nanocomposites with desirable properties will translate them as potential candidates for bone tissue engineering.
Introduction
Tissue engineering is a technology which aims to regenerate tissues with the aid of scaffolds. The fabricated scaffold provides cellular, mechanical and molecular signals in order to remodel the damaged tissue [1]. The bone tissue engineering aims to restore a normal physiology or to speed up the bone healing process primarily. It utilizes the bone substitute made of biomaterials that provide an improved musculoskeletal health which aids the repairing the bone defects caused by injuries or deformities. Materials used for bone tissue engineering should act as a carrier or template for implanted bone for inducing the bone formation [2]. It was found that the scaffold based on polymeric nanofibers holds great potential in bone tissue engineering [3].
Electrospinning is one among various manufacturing techniques available for the production of the scaffold. Electrospinning is the process of developing nanofibers with the support of high electric field which makes them versatile and cost effective [4]. Nanofibers were reported to possess high surface area with interconnected pores which could able to resemble the native ECM structure. Further, it have been reported that the nanofibers helps in cell adhesion and proliferation [3]. Several polymers have been utilized for the tissue engineering such as poly(glycolic acid) (PGA), poly(lactic acid) (PLA), polycaprolactone (PCL), polyurethane (PU), poly(lactic-co-glycolic acid) (PLGA), polyanhydride, polyethylene glycol (PEG) and poly(propylene fumarate) [5]. In this study, PU was selected due to their outstanding characteristics such as flexibility, tailorable physical and chemical forms [6]. Further, PU has non-toxic and biodegradable properties which makes their usage widespread in the medical fields [7].
This work aims to develop biocompatible composites via electrospinning process comprising PU and additives. The additives used were lemon grass oil and zinc nitrate. Cymbopogoncitratus, is known as lemon grass and this herb is found in tropical countries, especially in Southeast Asia. The essential oil derived from the plant is used in aromatherapy. The components present in Cymbopogoncitratus are mostly terpenes, alcohols, ketones, aldehyde and esters. Some phyto constituents of this essential oil are citral α, citral β, nerolgeraniol, citronellal, terpinolene, geranyl acetate, myrecene and terpinol methyl heptenone. It is also reported to have flavonoids and phenolic compounds such as luteolin, isoorientin 2’-O-rhamnoside, quercetin, kaempferol and apiginin. Some of its properties like antifungal, anti-inflammatory and antioxidant were reported widely [8]. There have been no reports of using lemon grass in the scaffolds albeit it has beneficial properties. Zinc is a metallic material having atomic number 30. It is hard, brittle and fair conductor of electricity. Zinc is reported to be displaying wide variety of nanostructures possessing unique properties like semiconducting, optical and piezoelectric. Because of this property, it was used in wide range of applications like nano-electronic/nano-optical devices, energy storage, cosmetic products, nano-sensors, etc. [9]. Few literatures have been reported the utilization of zinc in the tissue engineering applications [10,11]. Hence, the motivation of this work is to electrospun polyurethane added with lemon grass oil and zinc nitrate. Further, the behaviour of this polyurethane loaded with these dual additives will be investigated.
Materials and methods
Materials
Tecoflex EG-80A (medical-grade thermoplastic polyurethane) was procured from Lubrizol, USA. N,N-dimethylformamide (DMF) was obtained from Merck Millipore, Germany. The lemon grass oil was purchased locally. Zinc nitrate (ZnNO3) was purchased from Sigma Aldrich, UK. The chemicals, phosphate-buffered saline (PBS, Biotech Grade) and sodium chloride physiological saline (0.9% w/v), were bought from Sigma-Aldrich, Malaysia. The reagents utilized in APTT and PT assay such as rabbit brain activated cephaloplastin, calcium chloride (0.025 M), and thromboplastin (Factor III) were attained from Thermo Fisher Scientific, Selangor, Malaysia.
Preparation of nanocomposite
To prepare the polymer solution, 360 mg of PU beads were dissolved in 4 ml of DMF and stirred for 12 h to obtain at 9 wt%, homogeneous solution. To make lemon grass oil solution at 4 w/v%, 120 µl of lemon grass was mixed with 3 ml of DMF and stirred. Similarly, to make zinc nitrate solution at 4 w/v%, 120 mg of zinc nitrate was mixed with 3 ml of DMF and stirred to obtain a homogenous solution. PU/LG oil and PU/LG/ZnNO3 nanocomposite solution was prepared by slowly adding the homogeneous solution of lemon grass oil and zinc nitrate in PU at a ratio of 8:1 v/v and 8:0.5:0.5 v/v and stirred for 1 h maximum.
Fabrication of PU and nanocomposite scaffolds
The nanofibrous scaffold of pure PU nanofibers, PU/LG oil and PU/LG/ZnNO3 nanocomposite was developed using electrospinning technique. All scaffold were electrospun at a flow rate of 0.2 ml/h, an applied voltage of 10.5 kV and distance of 20 cm. The aluminium foil was used to collect the formed fibers and vacuum dried to remove any residual solvent.
Physicochemical characterization
SEM investigation
The fibres formed in electrospun PU, PU/LG oil and PU/LG/ZnNO3 nanocomposites fibers were studied through Hitachi SU8020. The fabricated scaffolds were gold-coated and imaged at different magnifications. From the captured image, the fiber size was measured using Image J (National Institutes of Health, Bethesda, MD) software.
Fourier transform infrared spectroscopy (ATR-FTIR) analysis
The characteristics peaks present in the electrospun PU, PU/LG oil and PU/LG/ZnNO3 nanocomposite were studied through ATR-FTIR unit. In order to identify the characteristic peaks, a small sample piece was recorded in the frequency range of 600–4000 cm−1 with resolution of 4 cm−1.
Contact angle measurement
The contact angles of electrospun scaffolds was measured through VCA Optima contact angle measurement unit. Initially, PU, PU/LG oil and PU/LG/ZnNO3 scaffolds were cut and a water droplet was placed on its surface. Within few seconds, the static deposition of the water droplet was captured using high-resolution video camera. The manual contact angle was measured from the captured image through computer integrated software.
Thermogravimetric analysis
The thermal investigation of electrospun PU, PU/LG oil and PU/LG/ZnNO3 scaffolds was evaluated through PerkinElmer TGA 4000 unit under dry nitrogen atmosphere. Samples weighing 3 mg was heated in the temperature range 30–1000° C at a rate of 10°C/min.
AFM analysis
To start AFM analysis, PU, PU/LG oil and PU/LG/ZnNO3 scaffolds were cut and scanning was performed. The scanning size was 20 µm×20 µm and the images were captured to obtain the surface roughness.
Mechanical testing
The mechanical testing of the PU, PU/LG oil and PU/LG/ZnNO3 scaffolds was carried out in uniaxial load test machine (Gotech Testing Machines, AI-3000) according to the ASTM D882-10. In this experiment, the membrane with size of 40×15 mm2 was cut and was fixed at its cut ends in the tester machine. The test was done at crosshead speed of 10 mm/min until failure occurs. The average tensile strength was calculated from the machine generated stress–strain curve.
Blood compatibility measurements
APTT, PT and haemolysis assays were carried out to evaluate the blood compatibility nature of the electrospun scaffolds. The procedure for each analysis was done based on research work described earlier [12,13].
Bioactivity test
The bone mineralisation testing was performed by soaking the PU, PU/LG and PU/LG/ZnNO3 scaffolds in a stimulated body fluid (SBF). The nanofibers of 1×1 cm2 was left in 1.5x SBF (pH 7.4; 37°C) for 14 days. After 14 days, the samples were taken out and dried. After drying, the samples were imaged using field emission scanning electron microscopy (FESEM) and energy dispersive X-ray analyser (EDX) to evaluate the amount of calcium deposition [12].
Statistical analysis
All experiments were performed in thrice independently. To measure the statistical significance, one-way ANOVA was carried out and the calculated results are denoted as mean ± SD and for qualitative experiments, a representative of three images is shown.
Result and discussion
SEM investigation
Figure 1 depicts the SEM figures of PU, PU/LG oil and PU/LG/ZnNO3 scaffolds produced using the electrospinning technique. SEM images indicated that the electrospun scaffold consists of randomly oriented smooth nanofibers without any beads. The fiber diameter estimated using Image J reported the average fiber diameter of PU, PU/LG and PU/LG/ZnNO3 were found to be 1513 ± 134 nm, 781 ± 133 nm and 347 ± 178 nm respectively.

FESEM images of (a) PU, (b) PU/LG and (c) PU/LG/ZnNO3.
The diameter of fiber was decreased with the addition of LG and ZnNO3. Further, the corresponding fiber distribution curve was shown in Figure 2. Jaganathan et al. [13] fabricated polyurethane scaffold added papaya and honey. It was observed that the addition of papaya and honey resulted in the reduction of the fiber diameter. It has been reported that the reduction in the PU fiber diameter was because of bio-molecules present in the papaya and honey [14]. In another study, Lakshman et al. [15] made electrospun scaffold comprising polyurethane containing silver nanoparticles for skin tissue engineering. It was observed that the PU scaffold containing silver particles showed reduced fiber diameter compared to the pristine and concluded this might due to the increase in the conductivity of the polyurethane solution on adding silver nanoparticles [15]. In our study, the reduction in the PU fiber diameter was might due to the biomolecules present in the lemon grass and increase in conductivity of the solution on addition of zinc particles. SEM figures revealed the interaction of LG biomolecules with the polyurethane matrix by reduction in the size of fiber diameter which is apparently visible. Further, with the addition of ZnNO3 resulted in the significant decrease in the fibre diameter. Linh and Lee [16] and Prabhakaran et al. [17] reported in their work that the fibrous scaffold with smaller diameter might facilitate the improved osteoblast growth. Our smaller fiber diameter of the developed composites indicating its suitability for the new bone tissue formation. Also, EDX studies revealed the presence of Zn in the composite PU/LG/ZnNO3. PU/LG/ZnNO3 showed some content of Zinc (3.6%) confirming presence of zinc in the polyurethane matrix.

Fiber distribution images of (a) PU, (b) PU/LG and (c) PU/LG/ZnNO3.
IR analysis
The functional groups of the nanofibrous membranes were examined in the IR spectra. Figure 3 indicates the FTIR spectra of the PU, PU/LG, PU/LG/ZnNO3 scaffolds. The pristine PU presented a characteristic broad peak of NH stretch at 3318 cm−1, and its vibrations were visible at 1531 cm−1 and 1597 cm−1, respectively. The CH group can be seen at 2920 cm−1 and 2852 cm−1, and the peak at 1414 cm−1 indicates its vibrations. Furthermore, a twin peak observed at 1701 cm−1 and 1730 cm−1 was assigned to CO group and the CO attributed to alcohol group was seen at 1221 cm−1, 1105 cm−1 and 770 cm−1 respectively [12,13]. In the electrospun nanocomposites, the characteristics bands were same as that of pristine PU. However, there is increase in the intensity of PU peak while adding LG and ZnNO3 respectively. The change in the peak intensity was because of strong hydrogen bond formation [18]. It have been reported that the two different macromolecules would form strong inter-hydrogen bonds compared to the bonds formed because of the molecules in the same polymer [18].Hence, the strong hydrogen bond formation might be due to the combination of macromolecules present in the PU, lemon grass oil and zinc nitrate. The formation of strong hydrogen bond implies that the PU have lemon grass and zinc nitrate. These interactions are greatly in correlation with the observed SEM results described earlier. Further CH peak in PU at 2920 cm−1 was shifted to 2937 cm−1 in PU/LG and 2939 cm−1 in PU/LG/ZnNO3 respectively [19]. The hydrogen bond formation and peak shift was because of combination of molecules of LG and ZnNO3 with the molecules of PU.

FTIR images of PU, PU/LG and PU/LG/ZnNO3.
Contact angle measurements
The wettability and adhesion properties of the developed as-spun membranes were evaluated through contact angles measurement. The contact angle of PU, PU/LG, PU/LG/ZnNO3 membranes were estimated to be 105 ± 3°, 116 ± 2° and 67 ± 4°, respectively. It was found that the angle was increased to 116° while adding LG, signifying hydrophobic behaviour and decreased to 67° upon incorporating ZnNO3 indicating hydrophilic behaviour. It have been reported that the scaffold wettability design is very important that influence the cell attachment and proliferation to a higher extent. It was reported that optimum osteoblast cell adhesion and proliferation occurs when the contact angle is less than 106° [20]. The contact angle of PU/LG (116° ± 2°) was reported to be behind the reported range, which may result in the reduction of the cell attachment and proliferation. Furthermore, when adding ZnNO3 to the PU/LG matrix facilitated the hydrophilic behaviour (67° ± 4°) in which the angle lies in the reported wettability range indicating its suitability for the enhanced cell adhesion and proliferation for new tissue growth. Jaganathan et al. [21] electrospun a bone scaffold utilizing polyurethane added with corn and neem oil. It was reported that the developed PU/corn/neem oil scaffolds displayed hydrophilic behaviour suggesting suitable candidate for the bone tissue engineering [21]. Hence, wettability nature of the fabricated PU/LG/ZnNO3 nanocomposite might be successfully exploited for bone tissue engineering.
Thermal analysis
The thermal stability of PU, PU/LG, and PU/LG/ZnNO3 were evaluated using TGA and their curves are shown in Figure 4. The results of thermal analysis depicted that the thermal stability of the electrospun PU/LG nanocomposite was higher than PU fiber, while PU/LG/ZnNO3 displayed decrease in thermal stability than the pristine PU. The initial decomposition temperature of the pristine PU was 284°C, and for electrospun PU/LG and PU/LG/ZnNO3 it was reported to be 290°C and 262°C, respectively. Hence, the presence of LG enhanced the thermal behaviour of the pristine PU, while the incorporation of ZnNO3 decreased the thermal behaviour and this might be due to the evaporation of water molecules present in the zinc nitrate. Further, DTG curves for the electrospun PU, PU/LG, and PU/LG/ZnNO3 are shown in Figure 5. From the figure, it was observed that the pristine PU showed three weight loss peaks, whereas the electrospun PU/LG and PU/LG/ZnNO3 also displayed three weight loss peaks respectively. However, there was a decrease in the weight loss peak intensity for the fabricated composites indicating reduced weight loss [12]. Moreover, the change in peak intensity confirms the presence of LG and ZnNO3 in the polyurethane matrix.

TGA curve of (a) PU, (b) PU/LG and (c) PU/LG/ZnNO3.

DTG curve of PU, PU/LG and PU/LG/ZnNO3.
Mechanical properties
The tensile properties of the PU, PU/LG and PU/LG/ZnNO3 nanocomposites were evaluated using uniaxial tensile tester and the representation stress-strain plots are shown in in Figure 6. It was found that the mechanical strength of LG and ZnNO3 added nanofibers were higher compared to the pristine PU. The tensile strength of the pristine PU was reported be 6.16 MPa, whereas PU added with LG and LG/ZnNO3 exhibited strength of 8.15 MPa and 11.84 MPa, respectively. Hence, LG and ZnNO3 incorporation into the PU enhanced the ultimate strength of the pristine PU. Jaganathan et al. [22] fabricated an electrospun membrane based on polyurethane incorporated with copper sulphate. It was reported that the addition of copper sulphate displayed the improvement of the mechanical strength which resembles with our findings and attributed this improvement was because of their smaller fiber diameter [22]. Since, the addition of LG and ZnNO3 resulted in the smaller fiber diameter which might have favoured the enhancement of the mechanical strength.

Tensile curves of PU, PU/LG and PU/LG/ZnNO3.
AFM analysis
Additionally, the surface roughness of PU, PU/LG, PU/LG/ZnNO3 nanocomposite fibers were determined through atomic force microscopy and their corresponding 3D AFM images were shown in Figure 7. The reported average surface roughness of the PU was found to be 854 ± 32 nm while the surface roughness of the PU added with LG and ZnNO3 was reported to be 625 ± 166 nm and 369 ± 67 nm, respectively. The addition of LG and ZnNO3 displayed the reduction of the roughness of the PU and their surfaces were smooth. Jaganathan et al. [21] developed PU membranes added with corn and neem oil for bone tissue engineering. It was found that the PU added with corn and neem oil showed smoother surfaces compared to the pristine PU which resembles our observations and suggesting our composites as suitable candidates for bone tissue engineering [21]. Hence, our fabricated composites with reduced roughness might be suitable for bone tissue growth.
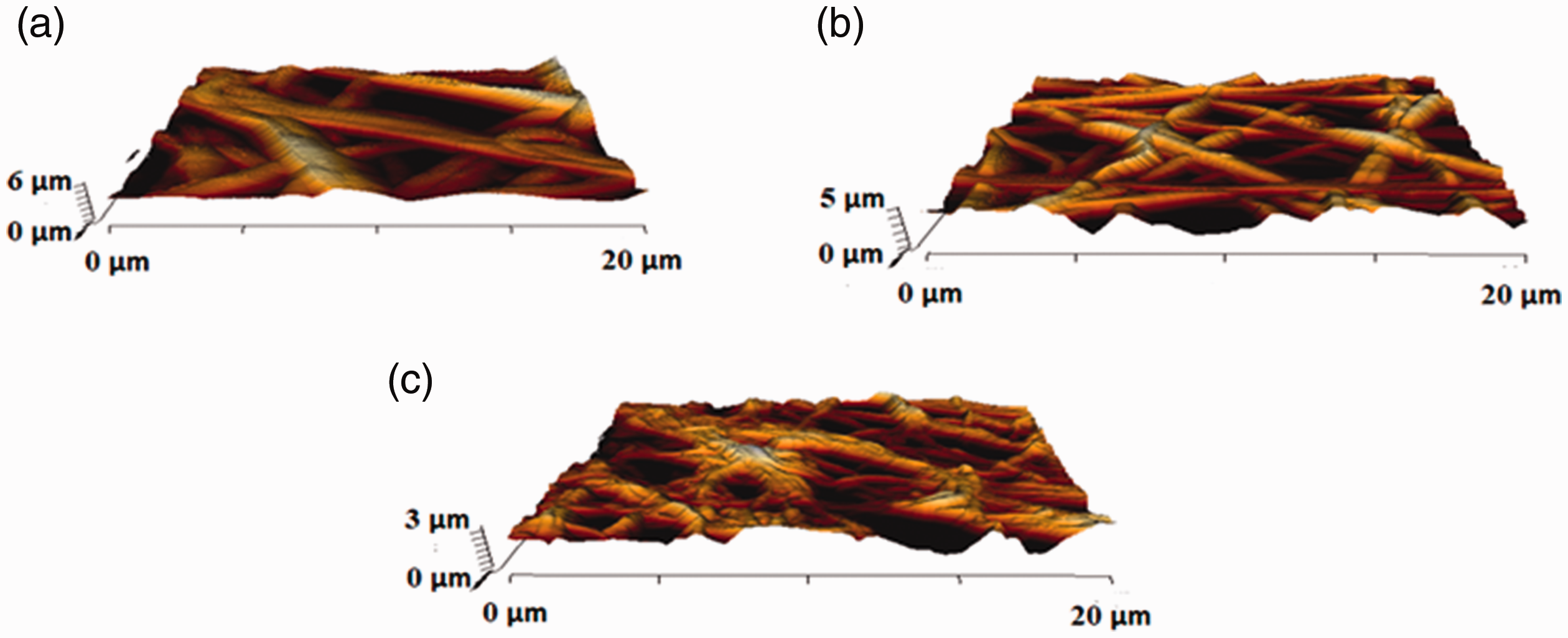
AFM images of (a) PU, (b) PU/LG and (c) PU/LG/ZnNO3.
Coagulation assessments
Figure 8 indicates the coagulation assay assessments for the electrospun PU, PU/LG and PU/LG/ZnNO3 scaffold. The blood clotting time (APTT) for the electrospun PU, PU/LG and PU/LG/ZnNO3 scaffold were estimated to be 176 ± 2 s, 196 ± 3 s and 193 ± 4 s. Similarly, the blood clotting time (PT) for the electrospun PU, PU/LG and PU/LG/ZnNO3 scaffold was estimated to be 94 ± 2 s, 105 ± 2 s, and 103 ± 2 s, respectively. The measurements of APTT and PT clearly showed that the electrospun nanocomposites displayed a prolonged blood clotting times compared to pure polyurethane. This might be because of LG and ZnNO3 in the polyurethane matrix.
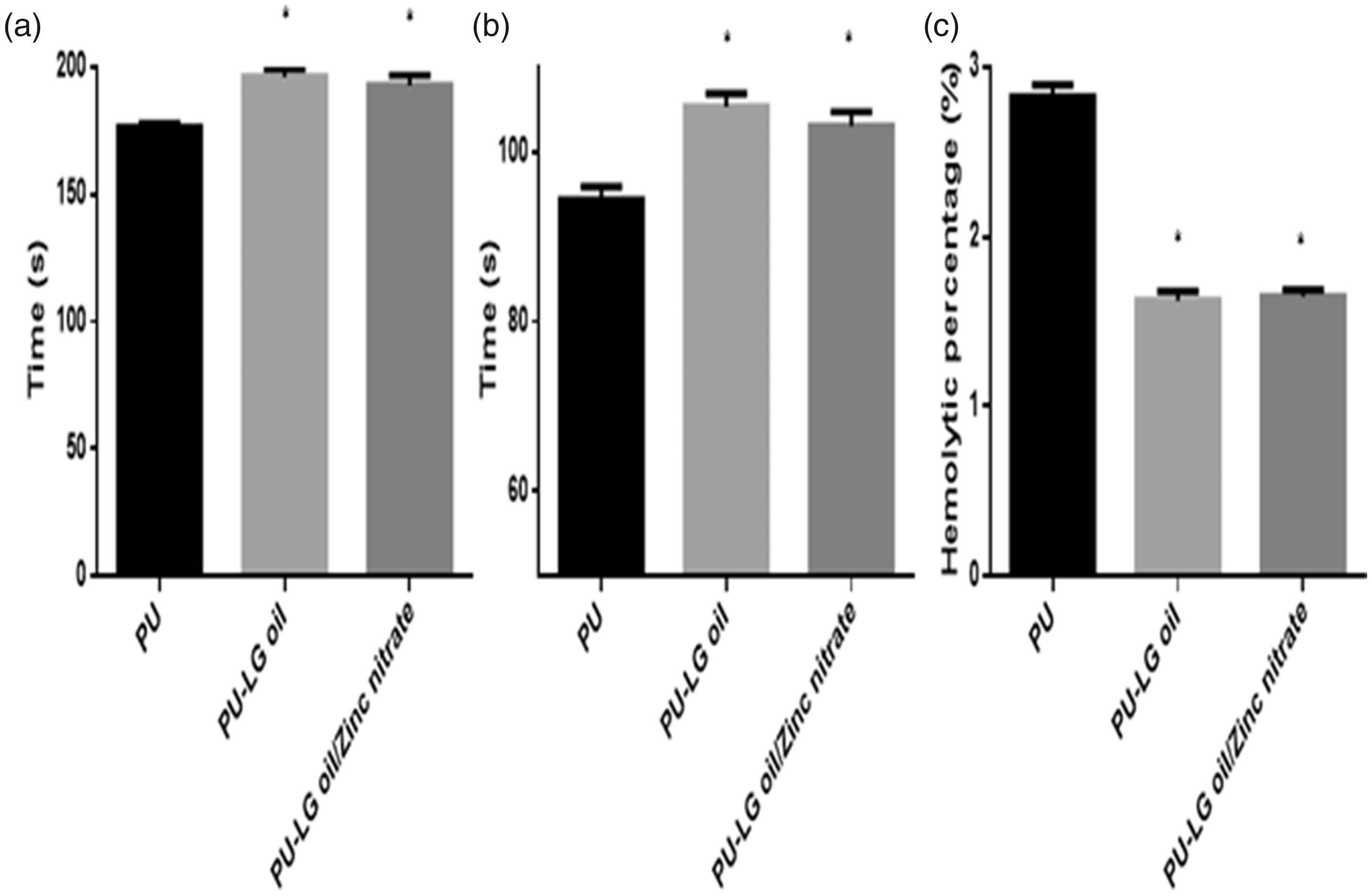
(a) APTT, (b) PT and (c) haemolytic index of PU, PU/LG and PU/LG/ZnNO3.
Furthermore, to determine the release of haemoglobin from the fabricated membranes, the haemolytic percentage was performed. The haemolytic percentage of PU was estimated to be 2.83% and for the electrospun PU added with LG and ZnNO3 scaffold displayed a haemolytic percentages of 1.62% and 1.65%, respectively, representing non-haemolytic materials, conferring to ASTMF756-00 (2000) [12,13]. The electrospun PU/LG showed higher blood compatibility behaviour compared to the pristine PU which might because of its hydrophobic behaviour. In hydrophobic surface, there will be irreversible adhesion of plasma proteins in the blood/surface interaction resulting in the delay in the blood clotting time. However, there was a slight decrease in the blood clotting time while adding ZnNO3 to the PU/LG nanocomposites which might be due to the change in polar and apolar regions in PU/LG/ZnNO3 nanocomposites [23]. Jaganathan et al. [24] developed a polyurethane scaffolds added with canola and neem oil. It was showed that the incorporation of canola and neem oil displayed the enhancement of blood compatibility behaviour compared to the pristine PU. They attributed this behaviour was due to the smaller fiber diameter and hydrophobic nature of the electrospun nanocomposites [24]. Hence, in this study, the smaller fiber diameter (PU/LG and PU/LG/ZnNO3) and hydrophobic behaviour (PU/LG) of the electrospun nanocomposites might have favoured the enhancing the blood compatibility behaviour of the pristine polyurethane.
Bone mineralization testing
Bone mineralization process of the electrospun membranes in SBF were evaluated after 14 days and the corresponding images were shown in Figure 9. The presence of LG and ZnNO3 increased the calcium deposition in the pristine PU. The PU displayed the calcium weight percentage of 2.4%, and for electrospun PU/LG and PU/LG/ZnNO3 it was found to be 5.6% and 10.8%, respectively. Hassan et al fabricated a electrospun scaffold based on polyurethane added with hydroxyapatite for bone tissue engineering. It was found that the addition of hydroxyapatite enhanced the calcium deposition compared to the pure PCL which resembles our findings [25]. Hence, the increased calcium deposition of the fabricated composites might be exploited for bone tissue engineering scaffolds.

Bone mineralization testing (a,c,e) FESEM images of electrospun membranes and (b,d,f) EDX spectrum of electrospun membranes.
Conclusion
PU/LG and PU/LG/ZnNO3 scaffolds exhibited favourable properties which are conducive for bone tissue scaffolds. However, it is important for further examination like cellular compatibility with osteoblast cells which may further promote its candidacy. At the same time, it can be explored for other tissue engineering scaffolds which may further enhance its value in biomedical applications. The usage of LG in the scaffold have yielded promising properties to scaffolds however it would be essential to investigate the allergic nature of the fabricated composite to rule out any side effects induced by the LG.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
