Abstract
Electrospun polyurethane (PU) scaffolds were developed containing cerium oxide (CeO2). Photomicrograph of the composites revealed the diameter of the PU/CeO2 (264 ± 169 nm) was smaller than the polyurethane scaffold (994 ± 113 nm). The fabricated PU/CeO2 (110° ± 1) scaffold displayed a more hydrophobic nature as depicted by increasing contact angle compared to the pristine PU (105° ± 3). Fourier transform infrared spectroscopy (FTIR) results presented evidence for the cerium oxide presence in the PU matrix through the formation of the hydrogen bond. The surface roughness of PU/CeO2 (301 ± 52 nm) was reduced in comparison with pristine PU (854 ± 32 nm) as estimated in the atomic force microscopy (AFM) analysis. Cerium oxide enhanced the thermal and tensile behaviour of the pristine PU. Coagulation assays indicated delayed clotting time and a less toxic nature to red blood cells of PU/CeO2 than pristine PU. Further, the calcium deposition in the nanocomposites (10.5%) was higher compared to pure PU (2.4%) as showed in bone mineralization testing. Hence with these potent properties, PU/CeO2 holds as a promising candidate for bone regeneration.
Background
Bone tissue engineering occupies the largest share of the global regenerative medicine market. The global market for orthopaedics, musculoskeletal, and spine applications is estimated to be US$13.5 billion in 2018 and is expected to rise to US$34.7 billion by 2024 [1]. The human bone is a complex tissue that executes diverse functions like organ protection, facilitates locomotion, and storing nutrients [2]. Bone defects refer to the bone tissue damage that occurs due to illness or accident. The major issue due to accident damage of bone is that it results in huge bone defects if it was not treated immediately [3]. The traditional methods like autograft or allografts were the usual respite for repairing the damaged bone tissues. Although these methods are pronounced useful, they have several disadvantages like limited donor supply, immunogenicity risk, and infections which restrict their utility in clinical applications [4]. Various strategies have been found by the researchers to remodel the damaged bone tissue. Among these strategies, bone tissue engineering holds as a promising strategy compared to the traditional methods. Cells, scaffolds, and biochemical cues are the three major components of bone tissue engineering. Out of these three, the scaffold plays a vital role in supporting the new tissue growth via enhancing cell adherence and proliferation by mimicking the native structure of the extracellular matrix (ECM) [5].
The designing of the scaffold mimicking the ECM structure is a significant challenge in bone tissue engineering applications. The human ECM is a three-dimensional structure that contains nanofibrous proteins [6]. Designing scaffolds for bone tissue engineering is an onerous task and scaffold must be non-toxic, non-allergic, biocompatible, and providing required mechanical support. Further, they should support the cells to adhere and proliferate more in order to facilitate the new bone tissue growth [7]. Recently, the nanofibrous scaffold mimicking the ECM fabricated via electrospinning has gained a cynosure in tissue engineering applications. The electrospinning technique is a facile and cost-effective method that develops fibres ranging from micrometre to nanometre possessing huge surface area with interconnected pores [8]. Owing to its outstanding properties, they will enable the transport of nutrients and also cellular response for new tissue growth [9]. Literature reported that the scaffold has been fabricated from several types of biomaterials ranging from natural and synthetic polymers. The scaffold-based on polymeric materials have been widespread in bone tissue engineering applications [10]. In this research, the PU was used because it has desirable properties like biocompatibility, biodegradability, and flexibility [11]. Polyurethane (PU) belongs to synthetic elastomers which are tough (soft segment) and also having high strength (hard segment). It possesses unique chemistry that makes them versatile. It can be easily processed using various manufacturing techniques like extrusion, injection molding, blowing, and compression [12]. It was widely used in biomedical fields because of its excellent tailor-made mechanical and biocompatible properties. In the form of a nanofibrous scaffold, polyurethane holds great potential in regenerative medicine by influencing cell adhesion, proliferation, and differentiation [13]. Among various polymers, polyurethane has gained a widespread reputation in manufacturing scaffolds for bone tissue engineering [14]. Jing et al. fabricated a scaffold based on polyurethane added with winter green oil and titanium dioxide. The polyurethane scaffold added with additives was reported to be good cell adhesion and increased apatite formation [15]. Ali et al. fabricated a polyurethane scaffold for bone tissue engineering added with nylon and gelatin. It was found that the fabricated composite displayed good bioactivity and high osteoblast cell proliferation [16]. In another study, Jaganathan et al. electrospun scaffold utilizing polyurethane added with rosemary oil and copper sulphate. The reported nanocomposite showed improved blood and cytocompatibility properties [17].
The metallic nanoparticles are added into scaffolds to increasing the mechanical strength, cell adhesion and proliferation, and apatite forming capacity. Literature reported the use of various metallic materials such as titanium, gold, copper, zinc, and iron for tissue engineering and regenerative medicine [18]. Scaffold laden with the metallic nanoparticles was reported to be enhancing collagen synthesis, alkaline phosphate activity, calcium deposition, and tensile strength. In this research, cerium oxide was incorporated into the PU matrix. Few literatures have been reported on the usage of cerium oxide in tissue engineering applications. Augustine et al. incorporated cerium oxide nanoparticles into the electrospun poly (3- hydroxybutyrate-co-3-hydroxyvalerate, PHBV) and experimented for diabetic wound healing applications. The results showed that CeO2 nanoparticles incorporated PHBV membranes promoted cell adhesion and proliferation and also have enhanced wound healing potential [19]. Dai et al. encapsulated polyethylene glycol coated cerium oxide nanoparticles (PEG-CeO2) and vascular endothelial growth factor (VEGF) into polyurethane (PU) scaffolds via electrospinning. It has been found that the polyurethane containing PEG-CeO2 scaffold facilitated the endothelialization resulting in the formation of small diameter artificial blood vessels [20]. In a recent study, Xiang et al. incorporated CeO2 into the poly (L-lactic acid) (PLLA) scaffold and investigated its effects on the growth and paracrine behavior of mesenchymal stem cells (MSCs). The results showed that CeO2 nanoparticles incorporated within PLLA scaffold activated calcium channel of mesenchymal stem cells (MSC’s) resulting in enhanced angiogenic factor (VEGF) expression and also showed the new formation of bone by accelerating blood vessel formation with the inhibition of apoptosis [21]. Purohit et al. fabricated a scaffold using a freeze drying process by mixing with nanoceria coupled with gelatin and this scaffold was found to assist the differentiation of MSCs to osteoblast [22]. Similarly, Lu et al. showed incorporation of cerium oxide in hollow mesoporous bioglass scaffolds resulting in enhanced bone regeneration by the activation of the extracellular signal-regulated kinases (ERK) signalling pathway [23]. Though these works revealed the potential of cerium oxide loaded scaffolds and there is no single work highlighting the morphological, mechanical, and blood compatible properties of the polyurethane loaded with cerium oxide for bone tissue engineering applications. This work will fill those identified gaps by deciphering the physicochemical and blood compatible properties of the PU/CeO2 in comparison with the pristine polyurethane for further pivoting its role in bone tissue engineering applications. Generally, the metallic nanoparticles render toxic behaviour. A recent work done by Babu et al. reported their synthesized cerium oxide particles showed non-toxic behaviour against RAW 264.7 cells [24]. Few literatures have been shown that the toxicity of cerium oxide depends on its nanoparticle dose and exposure time. The cell viability decreases with increases in nanoparticles dosage and time exposure [25]. Hence, it is safe to use a lower dosage of cerium oxide for manufacturing scaffolds.
Materials and methodology
Chemicals
In this work, Tecoflex EG-80A PU was purchased from Lubrizol, USA. N, N-dimethylformamide (DMF) was supplied from Merck Millipore, Germany. CeO2 was purchased from Sigma Aldrich, UK. Phosphate buffered saline (PBS) and sodium chloride (NaCl) physiological saline (0.9% w/v) was procured from Sigma-Aldrich, Malaysia.
Optimization of PU polymer and PU/cerium oxide composite
To obtain beadles PU polymeric nanofibers, polymer concentration was varied by keeping the distance and voltage fixed. The weight concentration investigated in this study are 3 wt%, 6 wt% and 9 wt% and the voltage was set as 5 kV, 10 kV and 15 kV with fixed distance of 10 cm and 20 cm. The fine formation of fibers without any beads was obtained for 9 wt% with the voltage and distance of 10 kV and 20 cm respectively and the optimization study results were shown in Tables 1 and 2. Employing the same configuration, PU/Cerium oxide was electrospun at three volume ratios namely 8:1 v/v, 7:2 v/v and 6:3 v/v before arriving 8:1 v/v. The other ratios (7:2 v/v and 6:3 v/v) either yielded beaded fibers or spitting of the polymer/cerium oxide solution. The chosen 8:1 v/v displayed the uniform formation of nanofibers without beads and spitting of the composite solution during manufacturing.
Optimization of PU polymer for various weight concentrations and voltages with distance 10 cm as constant.

Optimization of PU polymer for various weight concentrations and voltages with distance 20 cm as constant.

Preparation of nanocomposite
9% (w/v) the solution of polyurethane was made by adding 360 mg of PU beads dissolving in 4 ml of DMF and mixed for 12 h in a magnetic stirrer. 120 mg of cerium oxide was mixed with 3 ml of DMF (4%) and magnetically stirred for a minimum 2 h. Cerium oxide solution was mixed into the PU at a ratio of 8:1 v/v to make PU/CeO2 and magnetically stirred for 1 h.
Fabrication of PU/cerium oxide nanocomposite scaffold
The electrospinning technique (Progene Link Sdn Bhd, Selangor, Malaysia) was utilized to fabricate the PU and PU/CeO2 composites and carried out at room temperature with humidity of 55%. Both solutions were spun at a flow rate of 0.2 ml/h and applied the voltage of 10.5 kV and distance of 20 cm. Fibres collected using aluminium foil were dried in a vacuum at 60 °C to make sure the removal of any remaining DMF present in the PU and PU/CeO2 scaffolds [26,27].
Physicochemical characterization
Field emission scanning electron microscopy (FESEM)
FESEM unit (Hitachi SU8020, Tokyo, Japan) was utilized to image the fibres formed in the electrospun PU and the PU/CeO2. Before analysis, both scaffolds were cut into small pieces and it was gold plated to improve the image quality captured at various magnifications. The fibre diameter was calculated using Image J software from the captured image by choosing 30 locations randomly.
Fourier transform infrared spectroscopy (FTIR) analysis
The ATR-FTIR unit (Thermo Fischer Scientific, Waltham, MA, USA) was used to analyse the functional groups of PU and PU/CeO2. A small piece of the sample was cut and placed in the unit for recording in the frequency range of 600–4000 cm−1 at 32 scans per minute.
Contact angle measurement
The video contact angle (VCA) measurement unit (AST products, Inc., Billerica, MA, USA) was employed for measuring the contact angle of electrospun PU and PU/CeO2 scaffolds. Using the software within the system, the contact angle was measured from the captured image.
Thermogravimetric analysis (TGA)
Perkin Elmer TGA 4000 unit (Perkin Elmer, Waltham, MA, USA) was utilized to investigate the thermal stability of electrospun PU and the PU/CeO2. The temperature was set between 30–1500°C at an ascending rate of 10 °C/min.
Atomic force microscopy (AFM) analysis
To evaluate the surface roughness of the electrospun membranes, atomic force microscopy (AFM, Nano Wizard®, JPK Instruments, Berlin, Germany) was used. To do AFM analysis, a small piece of the sample was cut and scanned in the region of the size of 20 × 20 μm. Ra was obtained using the machine software.
Mechanical testing
Tensile testing of PU and PU/CeO2 was evaluated using the uniaxial tester (Gotech Testing Machines, AI-3000, Taichung City, Taiwan) according to the standard ASTM D882–10. The utilized gauge length and strain rate for this study was 10 mm/min and 500 N respectively. From the stress-strain plot, mechanical strength was calculated.
Blood compatibility measurements
The protocol for the blood compatibility study was approved by the Chairman, Ethical and Medical Researcher Committee, Universiti Tekonologi Malaysia, with the ref no UTM.J.45.01/25.10/3Jld.2(3).
Activated partial thromboplastin time (APTT)
Activated partial thromboplastin time is used to evaluate the time taken for the activation of the blood clot. Initially, the electrospun samples were cut into small sizes and were pre-incubated with PPP (100 µl at 37 °C). Then, the mixture was added with rabbit brain cephalin (100 µl) for 3 min and then added with calcium chloride (0.025 M). The mixture was stirred using a sterile steel needle and the time taken for the blood clot formation was noted as APTT [28].
Prothrombin time (PT)
For PT assay, the cut samples were pre-incubated with PPP (100 µl) at room temperature followed by adding 0.9% NaCl–thromboplastin containing Ca2+ ions. Then, the mixture was stirred using a sterile steel needle and the time taken for the blood clot formation was noted as PT [28].
Hemolysis assay
The red blood cells (RBCs) damage that occurs in the electrospun membranes was determined through hemolysis assay. Firstly, the cut samples of electrospun membranes were soaked in physiologic saline (0.9% w/v; 37 °C, 30 min) and then it was exposed to the mixture of 3 ml aliquots of citrated blood diluted with saline. The blood was added with distilled water at a ratio of 4:5 to make complete hemolysis and in the physiological saline constitute the positive and negative control. The incubated samples were retrieved and centrifuged at 3000 rpm for 15 min to obtain a clear supernatant. The resultant supernatant was pipette and measured at 542 nm to evaluate the absorbance value which represents the damaged red blood cells (RBC’s) [28].
Bioactivity test
Bone mineralization is an important parameter of bone formation. In this work, developed scaffolds of PU and PU/CeO2 of dimension 1 X 1 cm2 were soaked in 1.5x simulated body fluid (SBF). The nanofibers were left in SBF (pH 7.4; 37 °C) for 14 days. After 14 days, PU and PU/CeO2 scaffolds were removed and subjected to imaging using FESEM. An energy dispersive X-Ray analyser (EDX) was performed to estimate the calcium deposition [28].
Statistical analysis
All experiments were carried out three times independently. The unpaired t-test in Graph pad prism was performed to analyse the statistical significance and the observed results were expressed as mean ± SD and for qualitative experiments, a representative of three images is shown.
Result and discussion
Photomicrograph analysis
The morphology of pure PU and PU/CeO2 was depicted in the FESEM image as shown in Figure 1 respectively. FESEM image depicted that the obtained fibres were smooth and randomly oriented. Fiber diameter estimation using ImageJ analysis revealed the reduced fiber diameter of PU/CeO2 than the pristine PU. The fiber diameter of PU/CeO2 was found in the range of 264 ± 169 nm, where the pristine PU showed in the range of 994 ± 113 nm. There is an almost five-fold reduction of fibre diameter due to the addition of cerium oxide. Lakshman et al. fabricated a wound dressing based on polyurethane added with silver nanoparticles. It was found that the addition of silver nanoparticles resulted in the reduction of the fiber diameter and concluded this attribute might be because of the increased conductivity on adding silver nanoparticles [29]. Hence, in our study, the reduced fiber diameter might be due to increased conductivity while adding cerium oxide into the polyurethane. Further, a viscosity of the composite solutions is also affected due to the addition of cerium oxide which may also have a putative role in fibre diameter reduction [30]. Jaganathan et al. electrospun polyurethane scaffold utilizing olive oil, honey, and propolis for bone tissue engineering. It has been reported in their study that the addition of olive oil, honey, and propolis resulted in the reduction of fiber diameter in PU which correlates with our findings [31]. In another study, Chao et al. incorporated grape seed oil and honey/propolis into PU for bone tissue engineering. It was found that PU added with grape seed oil and honey/propolis displayed reduced fiber diameter which was in agreement with our findings [32]. Hence, our smaller fiber diameter of the fabricated nanocomposites might be suitable for the bone tissue growth through enhancing the cell attachment and proliferation. Further, the EDX study confirmed the presence of cerium in the polyurethane matrix as indicated in Table 3. PU spectrum showed only carbon and oxygen while PU/CeO2 showed cerium (0.2%) in addition to carbon and oxygen.

FESEM images of (a) PU and (b) PU/CeO2.
EDX table of electrospun membranes.

IR analysis
Figure 2 shows the FTIR spectrum of pristine PU and the fabricated PU/CeO2 composites. In the FTIR spectrum of pristine PU, NH peak was the band at 3318 cm−1 and their vibrations were observed at the peak 1597 cm−1 and 1531 cm−1 respectively. The band at 2920 cm−1 represents the CH2 asymmetric stretch while the band at 2852 cm−1 is attributed to the CH3 symmetric stretch and their vibrations were seen at 1441 cm−1. The twin peak seen at the frequency range of 1730 cm−1 and 1701 cm−1 indicate the C = O stretch. The other peak at 1221 cm−1, 1105 cm−1 and 770 cm−1 indicates the C–O group corresponding to alcohol [31,32]. The IR spectra of PU/CeO2 showed similar peaks like that of PU with no new peaks formation but the peak intensity was altered (intensity of NH peak increased and CH peak decreased) due to the formation of hydrogen bond [33]. Further, there was also observed the change in CH peak shift while adding cerium oxide to the PU [34]. CH peak at 2920 cm−1 in PU was shifted to 2941 cm−1 in PU/CeO2 respectively. This hydrogen bond formation and CH peak shift indicate the interaction between the PU and cerium oxide.

FTIR images of PU and PU/CeO2.
Contact angle measurements
The contact angle measurements of electrospun membranes were indicated in Table 4. It was shown that the contact angle of PU/CeO2 composites was slightly higher than the pristine PU indicating its hydrophobic nature.
Contact angle measurements of electrospun membranes.

Cui et al. electrospun scaffold based on poly (D, L-lactide) (PLA) and poly (ethylene glycol) fibers. It was found that the scaffold with a smaller fiber diameter exhibited hydrophobic nature [35]. Further in another study, a scaffold based on polystyrene (PS) and polyvinyl chloride (PVC) was fabricated through electrospinning. They found increased contact angles when the fibre diameters are reduced [36]. Hence, the smaller fibre diameter of the fabricated composite might have favoured increasing the contact angle of the PU/CeO2. Ajith et al. developed a membrane for bone tissue engineering based on polyoxymethylene (POM) membrane and hydroxyapatite (nHA) nanoparticles. The prepared composite showed hydrophobic behaviour and exhibits enhanced protein adsorption suitable for the new bone formation [37]. Hence, the hydrophobic behaviour of the fabricated nanocomposites might adsorb number proteins for the new bone formation.
Thermal analysis
The initial decomposition temperature of pristine PU was observed at 284 °C and for fabricated PU/CeO2 composites, it was found to be 291 °C respectively (Figure 3). The presence of cerium oxide improved the thermal behaviour of the pristine PU. Li et al. prepared electrospun polystyrene-graphene nanofibers and investigated their thermal and electrical properties. The prepared composites showed higher thermal stability than the polystyrene which resembles our findings [38]. Similarly, Chen et al. also reported the improved thermal stability of polybutylene succinate (PBS) composites with the addition of calcium carbonate (CaCO3) [39]. Peaks of derivative thermogravimetric (DTG) (Figure 4) indicated the mass loss from the samples. In this case, polyurethane expressed a degradation rate of 0.18 mg/min followed by PU/CeO2 (0.155 mg/min). This further evidences that the slower degradation properties of the PU/CeO2. Further, based on the change in peak intensity between the pristine polyurethane and the PU/CeO2 we could infer the presence of CeO2 in the polyurethane backbone. In a recent research, the addition of metallic particles has increased the thermal behaviour [40] and it may be due to the association of the crystalline phase in the composite.

TGA curve of PU and PU/CeO2.

DTG curve of PU and PU/CeO2.
Surface roughness measurements
The average surface roughness of pristine PU was estimated to be 854 ± 32 nm but for electrospun PU/CeO2 composite, the surface roughness was decreased to 301 ± 52 nm which was due to the presence of cerium oxide in the PU polymer matrix (Figure 5). Kim et al. fabricated a scaffold based on poly (ε–caprolactone) (PCL) scaffolds via electrospinning technique and investigated the effect of the fibre diameter on the surface roughness. It was found that the scaffold with a smaller diameter depicted a smoother surface compared to the larger fiber diameter [41]. Further, the interaction between the polyurethane and cerium oxide would have altered its surface chemistry which would also result in reduced roughness. In a recent research, the addition of cobalt particles into the polyurethane matrix had resulted in the reduction of surface roughness of the composite scaffold [42]. Hence, in our study, the reduced surface roughness of the electrospun nanocomposite might be because of their smaller fibre diameter and its altered surface chemistry of the PU/CeO2. Recent studies reported the scaffold with decreased surface roughness will be suitable for bone tissue engineering [31,32] and our smoother surfaces of the fabricated nanocomposites might be conducive for bone tissue engineering.
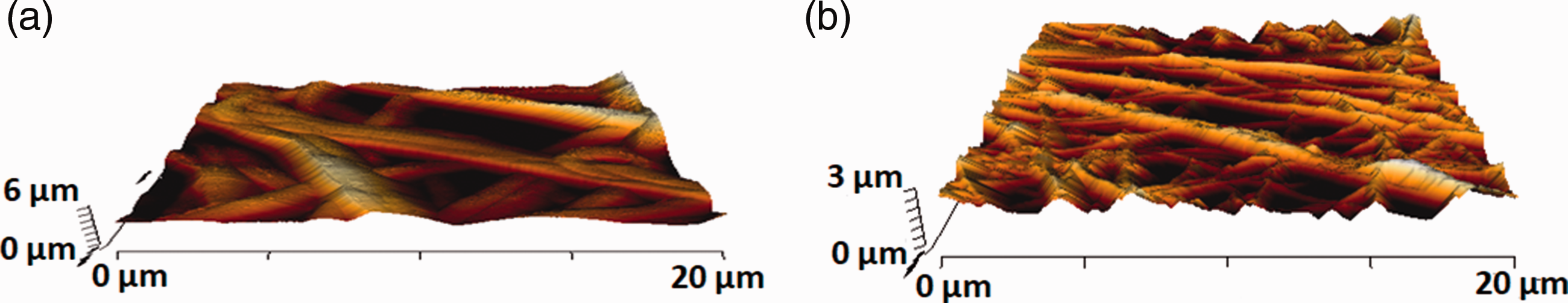
AFM images of (a) PU and (b) PU/CeO2.
Tensile analysis
The tensile strength of pristine PU was observed to be 6.16 MPa and for electrospun PU/CeO2 composites, it was found to be 10.89 MPa respectively (Figure 6). Further, Table 5 indicates the mean and standard deviation of tensile strength and elastic modulus for the electrospun membranes. The presence of CeO2 enhanced the tensile strength of the pristine PU. Form the literature, it was reported the scaffold with the strength of 4 Mega Pascal’s is suitable for bone tissue regeneration [43]. Our tensile value of the prepared composite was better than those reported values and might be suitable for supporting the new bone tissue growth. It has been reported that the smaller fiber diameter favouring the enhanced mechanical strength [44]. Hence, the smaller diameter of fabricated PU/CeO2 might have a putative effect in higher strength compared to polyurethane scaffolds.
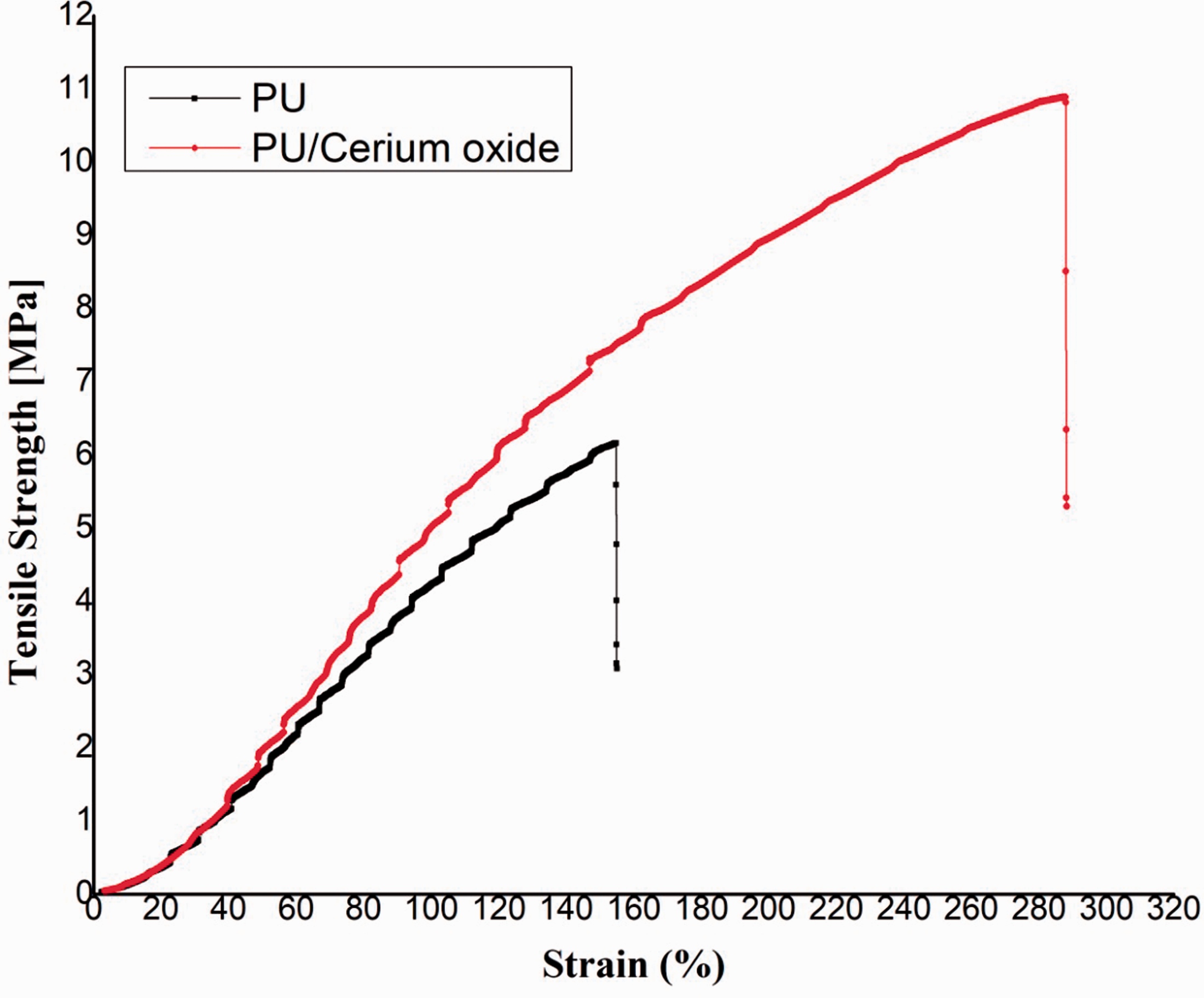
Tensile curves of PU and PU/CeO2.
Mean and standard deviation of tensile strength and elastic modulus for the electrospun membranes.

Blood compatibility analysis
PT and APTT results depicted the increase in the blood clotting time with the addition of cerium oxide into the polyurethane matrix. The mean APTT value of PU was reported to be 176 ± 2 s whereas, the fabricated PU/CeO2 nanocomposites showed a clotting time of about 190 ± 2 s as shown in Figure 7(a). Similarly, the mean PT value of pristine polyurethane was reported to be 94 ± 2 s whereas, the fabricated PU/CeO2 nanocomposites showed a clotting time of about 106 ± 1 s as shown in Figure7(b). The increase in blood clotting may be due to the presence of cerium oxide in the polyurethane matrix. Finally, the hemolysis test was carried out to analyse the release of hemoglobin from the surface of PU and PU/CeO2 composites. The hemolytic value of pristine PU was reported to be 2.83%, while for fabricated PU/CeO2 composites, it was estimated to be 1.42% respectively as indicated in Figure 7(c). According to ASTMF756-00(2000) standard, the fabricated composite was non-hemolytic material since their hemolytic value was below 2% [28]. It has been reported that the scaffold with smaller fiber diameter and hydrophobic behaviour might influenced the blood compatibility behaviour [31,32]. Hence, it is concluded that the smaller fiber diameter and hydrophobic behaviour of the fabricated nanocomposites due to the addition of cerium oxide might have caused the increase in the blood compatibility behaviour.
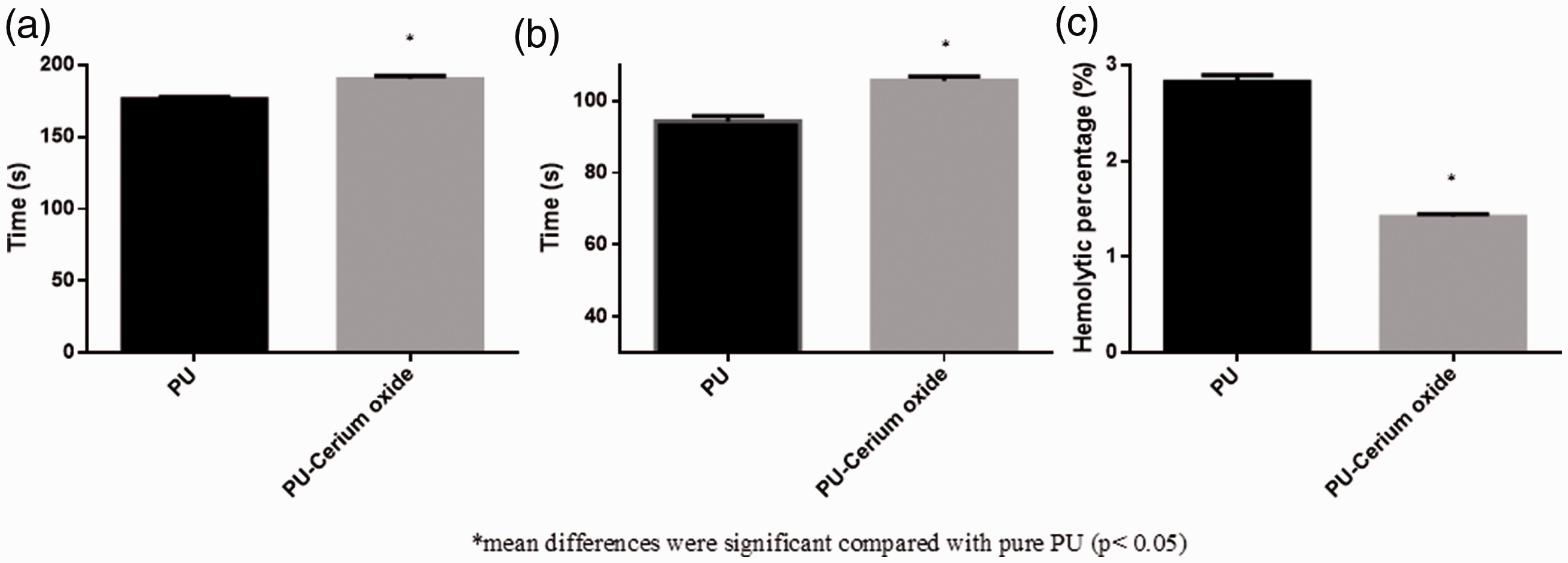
(a) APTT, (b) PT and (c) hemolytic index of PU and PU/CeO2.
Bone mineralization testing
Bone mineralisation testing determined the amount of calcium deposition for the electrospun scaffolds were shown in Figure 8. PU/CeO2 showed enhanced deposition of calcium compared to the pristine PU. The calcium deposition weight percentage for the pristine PU was found to be 2.4% and for electrospun PU/CeO2, it was reported to be 10.5% respectively. Andric et al. fabricated electrospun poly (L-lactide) (PLLA) scaffolds and studied the bone mineralisation for the membranes of pre-treatment with sodium hydroxide (NaOH) and gelatin added PLLA membranes. The treatment membranes and gelatine added membranes exhibited a higher degree of calcium phosphate deposition compared to PLLA and concluded the potential candidate for bone tissue engineering [45]. Hence, the addition of cerium oxide accelerating the calcium deposition will be beneficial for bone tissue engineering.

Bone mineralization testing: (a, c) FESEM images of electrospun membranes and (b, d) EDX spectrum of electrospun membranes.
Conclusion
For the first time, cerium oxide was integrated with in a polyurethane matrix through a facile electrospinning process for bone tissue engineering applications. PU/CeO2 exhibited reduced fibre diameter compared to the polyurethane scaffold. The presence of cerium oxide in the polyurethane matrix was inferred through hydrogen bond formation and increased thermal stability compared to the polyurethane matrix. Further, scaffolds exhibited enhanced mechanical strength due to the addition of cerium oxide. Coagulation and hemolysis assays of PU/CeO2 revealed the antithrombogenic and safety against red blood cell lysis. Enhanced calcium deposition was observed in the PU/CeO2 scaffolds insinuating its suitability for bone tissue engineering. However, further experimentation of the developed scaffold using osteoblast cells will reinforce PU/CeO2 application in bone tissue engineering. Further, these conducive physicochemical and blood compatible properties could be explored for several tissue engineering applications like skin, cardiac, or nerve tissue engineering etc.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
