Abstract
Various ratios (30/70%, 70/30%, 50/50% with and without nanosilver) of chitosan (CS; 60–120.000 g/mol) and polyethylene oxide (PEO; 600.000 g/mol) blended nanofibers in the nanofibrous scaffolds were obtained by using electrospinning at ambient atmosphere. Homogenous CS solutions were prepared in 90% aqeous acetic acid. PEO was dissolved in deionized water. Nanosilver dispersion was prepared and added to the 50/50% blend of CS/PEO solution. Properties of all blended solutions were determined by measuring viscosity and conductivity. Differential scanning calorimeter, Fourier transform infrared spectroscopy, scanning electron microscopy analysis and tensile tests were conducted to investigate the characteristics of the final nanofibrous composite structures. Beadless and uniform nanofibers were obtained and the average diameter of the fibers ranged from 63 ± 23 nm to 108 ± 51 nm. The antimicrobial effectiveness of the nanofibrous scaffolds was investigated against
Introduction
Chitosan (CS) [poly-(b-1/4)-2-amino-2-deoxy-
Electrospinning is a term used to describe a class of fiber forming in submicron range by applying electrostatic forces. The process is comprised of several operational components: (i) charging of the fluid, (ii) formation of the cone-jet, (iii) thinning of the instable jet in an electric field and (iv) collection of the jet on a suitable collector [5].
Diameter and morphology of nanofibers fabricated by electrospinning are influenced by several parameters such as solution parameters (viscosity, conductivity, molecular weight, surface tension, polymer concentration and solvent type), process parameters (applied electric field, tip to collector distance and feeding rate) and ambient parameters (humidity and temperature of the surroundings) [6,7].
In the last decade, electrospinning process has gained remarkable popularity in scientific and commercial fields because it provides efficient and simple method for fabricating nanofibrous structures from a wide variety of natural and synthetic polymers [8]. Nanofibrous scaffolds exhibit outstanding characteristics, such as considerably large surface area-to-volume ratio, high degree of porosity with a very small pore size and their 3D structure are similar to extracellular matrix [9]. Thus, electrospun nanofibers are claimed as candidate materials for many biomedical applications, such as wound dressing, drug delivery and scaffolds for tissue engineering [10–12].
In literature, there are numerous studies that have reported CS based nanofibers produced by electrospinning technique. However, the polycationic structure of CS increases the surface tension of the solution dramatically. Consequently, stronger electrical force is needed to produce electrospun CS nanofibers, and beads are often formed during the electrospinning process, which is likely due to the repulsive forces between the ionic groups in the CS backbone in an acidic solution [13].
Since it has been found difficult to electrospin CS individually, CS has been mixed with other synthetic or natural polymers. For instance, polyvinyl alcohol (PVA) [14], polyacrylamide [15] and polyethylene oxide (PEO) [16] have been experimented to electrospin composite fibers.
PEO, a water-soluble and aerobically biodegradable thermoplastic polymer, is extensively studied in electrospinning processes for biomedical applications due to its simple usage individually or with other polymers, and its excellent biocompatibility and very low toxicity. In literature, PEO has been electrospun with a range of natural proteins because it is very difficult to electrospin natural proteins individually in terms of obtaining beadless uniform nanofibers [17].
There are few reports in literature on the use of inorganic ions or nanoparticles as doping agents for producing electrospun fibers [18,19]. Also, CS and other polymers were blended to fabricate nanofibrous scaffolds including silver nanoparticles (AgNPs), for instance; AgNPs/PVA/CS [20], AgNPs/CS/gelatin [21], AgNPs/CS/PEO [22] and AgNPs/
Antimicrobial properties of the CS have been mentioned in many studies. Although showing a broad spectrum of antimicrobial activity, CS posseses different inhibition characteristics against different type of fungi, Gram-positive and Gram-negative bacteria [27].
Silver (Ag) has been used as an antimicrobial agent for a long time. Recently Ag nanoparticles have attracted many researchers as a potent antimicrobial agent because of their large surface area which enable them to fight more efficiently against bacterium and microbes [28].
Qin et al. states that introducing silver into CS significantly can increase the antimicrobial efficiancy of CS [29]. Hence, for the improved antimicrobial activities, researchers have focused on the Ag-loaded CS nanoparticles, CS films and chitin/CS composite scaffolds for wound healing applications [30–32].
The purpose of this study is to develop a nanofibrous scaffolds as a potential wound dressing material composed of CS/PEO based nanofibers with AgNPs. PEO has been used with CS in order to offset CS’s poor solubility and high viscosity in an aqueous solution which might be a serious problem for the continuity of electrospinning process. The impact of AgNPs on the electrospinning process has also been investigated. Nanosilver dispersion in distilled water was directly used in the blended solutions without additional processes such as; heating, microwave irradiation and chemical reduction. Another target of this study is to examine the antimicrobial properties of the varios CS/PEO blended (30/70, 70/30, 50/50 (with and without nanosilver)) electrospun nanofibrous scaffolds against Gram-negative bacteria (
Experimental
Materials
CS (with a deacetylation degree of ≥60 mol%) which has a molecular weight between 60.000 and 120.000 g/mol and PEO (molecular weight is 600.000 g/mol) were purchased from Sigma Aldrich (Germany). Nanosilver dispersion (20 ppm/ml) homogenized in deionized water was acquired from Nanokar (Turkey). Bacterial and fungal suspensions were supplied by the Pharmacology department of Marmara University (Turkey). Plate count agar (PCA) plates for bacteria and dichloran rose bengal chlortetracycline (DRBC) agar plates for fungi were purchased from Diatek (Turkey). All polymers and agents were used as received without any additional chemical processing.
Solution preparation
CS and PEO blend solutions were prepared at ratios of 50/50, 30/70 and 70/30 (w/w) to a final amount of 20 g at room temperature. Total amount of the polymer in solutions was 4 wt.%. First, CS solution was made by mixing the polymer with 90% aqueous acetic acid on the laboratory type magnetic stirrer (Stuart, SB 162) for 24 h. PEO was also dissolved in the deionized water by stirring on the magnetic stirrer overnight. Then, CS and PEO solutions were mixed by stirring for 2 h to obtain homogenous solutions. The 50/50 ratio was used for the fourth solution including nanosilver that was prepared following the same path explained above. Nanosilver dispersion was attenuated with deionized water to yield 10 wt.% concentration of nanosilver dispersion before adding it to the PEO solution. The mixing ratio of PEO solution and newly prepared nanosilver dispersion was 9:1. pH values of each blended homogenous solutions were measured with pH indicator strips (Merck, Germany) and were found as <4. The viscosities of each blended solutions were identified with a Brookfield Digital Viscometer by using s21 type spindle with a rotational speed of 30 r/min. The electrical conductivity of the blended solutions was also measured with a laboratory type conductivity meter (WTW, Cond 3110) under ambient atmosphere.
Electrospinning process
Electrospinning was performed in the laboratory spinning unit (NS24, NanoFMG), which was designed in terms of a vertical working principle. Each solution was placed in a 20 ml syringe and sent to the drum collector (covered with aluminum foil) through a 20 gauge nozzle. The power supply (AC) was set up for a positive voltage of 25 kV. The flow rate of the solution was also determined by setting up the syringe pump at 2 ml/h. The rotational speed of the drum collector was 35 r/min and its distance was set to 10 cm (optimum distance based on preliminary tests) away from the nozzle. At the time of the experiments, relative humidity and temperature values ranged from 35% to 42% RH and 26 to 31 ℃.
Characterization
Electrospun fibers were coated with a thin gold palladium (20/80%) layer using a sputter coater from Polaron (SC7620) and the morphology of the nanofibrous scaffolds was observed by scanning electron microscopy (SEM, JSM-5910 LV from JEOL) at an accelerating voltage of 20 kV. The fiber diameter distribution was calculated over 60 fibers with the Image J software (Image J, 2011) from the SEM images obtained at a magnification of ×20 000.
Transmission electron microscopy (TEM) was performed using FEI Tecnai F20-G2 operating at 200 kV. The samples for TEM were prepared by placing the nanofibrous scaffolds onto a copper grid and then loaded to the equipment.
Thermal analysis of electrospun fibers was performed with a differential scanning calorimeter (DSC; Perkin Elmer Jade). The approximate mass of samples loaded in DSC pans was 4.5 mg. After loading, samples were heated from 20 to 200 ℃ at a heating rate of 10 ℃/min. Results represent an average of three measurements.
The chemical structures of the CS/PEO/NS blended electrospun nanofibrous scaffolds were characterized by Fourier transform infrared (FTIR) spectroscopy (PerkinElmer Spectrum 100). The transmittance of each sample was recorded with five scans at a resolution of 4 cm−1 between 4000 and 380 cm−1.
To determine the mechanical properties (tensile strength and extension rate) of the CS/PEO blended nanofibrous scaffolds (including nanosilver or not), tensile and recovery tests were carried out using an Instron Machine (Instron 4411) in the textile laboratory at Marmara University. The mechanical properties of the blended nanofibrous scaffolds were examined at ambient environment (22 ± 3 ℃ temperature and 50 ± 5% relative humidity). The specimens were cut into approximately 30 mm × 10 mm (length × width) in both machine direction and width direction in order to be loaded into the uniaxial testing machine. During the experiment, 50 N load cell under a cross-head speed of 10 mm/min was applied to the specimens. Three repetitions were taken for each specimens in order to calculate the tensile strength and elongation at break values.
Antimicrobial assessment
Results and discussion
Viscosity and conductivity
Viscosity of the blended solutions variable due to the amount of the polymers in the solutions changed. This can be explained by strong positive deviation behavior with respect to the rule of additivity that proves strong interactions between PEO and CS chains. Hydrogen bonds are expected to be occurred between hydroxyl and amino groups on CS molecules and ether groups in PEO (Figure 1) [34,35]. Although previous works claimed that increase in PEO concentration in the blended CS solution would result in a decrease in viscosity, because of the plasticizer role of PEO by breaking the inter- and intra molecular interactions of CS chains through new interactions with PEO [35,36], our experiments showed that adding PEO to the CS solution increased the viscosity dramatically. This situation could be explained by the size and conformation of the PEO molecules. Large expanded PEO chains in solution can make strong entanglements with CS chains resulting in an opposite affection [37].
Proposed hydrogen bonding interactions between chitosan and PEO molecules [34].
It is very well known that CS possesses strong affinity towards metal ions because of the existence of numerous amine and hydroxyl groups [38]. Nanosilver dispersion was also added to the 50/50 (CS/PEO) blended solution. It is found that the viscosity of the blended solution increased slighltly with addition of nanosilver, comparing with the solution (50/50) without nanosilver. This is attributed to Ag+ ions bound to CS chains by ion–dipole interactions. Because the electron-rich oxygen atoms of polar hydroxyls and ether groups from CS were expected to interact with nanosilver to form CS–Ag+ complex [39].
The solution conductivity is one of the main parameters in the electrospinning process since more charges can be carried at higher solution conductivity. The increase in the conductivity of solution provides bead-free uniform and thinner fibers in the electrospinning process because the polymer solution is subjected to more stretching under the high electrical field [40–42].
Viscosity and conductivity of various CS/PEO blended solutions (30/70, 70/30, 50/50 (with and without nanosilver)) and fiber diameters produced by electrospinning process.

It was found that the conductivity of the blended solutions increased with the increase of CS content because of the –NH3+ groups in the polymer chains of CS. This comes from the conversion of –NH2+ groups to NH3+ when CS is dissolved in acetic acid. Introducing Ag+ to 50/50 blended solution also induced the increase of conductivity as a result of more ionic forms occurred in the polymer chains. On the other hand, addition of PEO decreased the electrical conductivity of the blended solutions because of protonation due to hydrogen bonds formed between amino groups of CS and ether groups of PEO [43].
Size and uniformity of nanofibers
It is confirmed that viscosity and conductivity of the blended solutions influence the morphology and diameter of nanofibers. Polymer solutions with high viscosity provide longer relaxation time for the ejected jet during electrospinning. Longer relaxation time is helpful to prevent the jet from fracturing, and also, could induce the increase of fiber diameter. On the other hand, when the solution conductivity increases, the diameters of electrospun fibers decrease due to high charge density forms on the surface of the jet which travels towards the collector under high elongation forces [44].
SEM images presented in Figure 2 belong to pure CS dissolved in aqueous acetic acid. As mentioned before, pure CS shows very poor electrospinnability at room temperature and only nanobeads and droplets are formed. After addition of appropriate amount of PEO into CS solutions, bead formations morphed into uniform fibers.
SEM image of pure CS (2 wt.%) dissolved in aqeous acetic acid (90%). Instead of uniform fibers, full of bead formation observed. Electrospun process carried out at 20 kV, nozzle to collector distance set to 10 cm and feeding rate was 1.5 ml/h.
According to the SEM images in Figure 3, it is observed that fiber diameters decrease with increasing CS content. For instance, increasing CS/PEO ratio from 30/70 to 70/30 caused a decline in fiber diameter from 108 ± 51 to 63 ± 23 nm at room temperature. This refers to the increase of CS contents in the solutions causes more charge density on the surface of the ejected jet formed during electrospinning process which leads thinner fiber production [17]. On the other hand, it is found that fibers (75 ± 17) produced from 50/50 blended solution including nanosilver were slightly thinner than the fibers (79 ± 25) produced from the same solution ratio without nanosilver. It can be explained by the more ionic forms taken place with the addition of Ag+ to the solution.
SEM images of e-spun fibers of CS/PEO blends; (a) 50/50, (b) 30/70, (c) 70/30 and (d) 50/50 with nanosilver. Uniform nanofibers observed without beads.
Although it is reasonable to comment on the favorable effect of Ag+ ions in terms of electrospinnability, uniform fiber formation and the integrity of fibrous structure may also be negatively influenced by the excessive concentration of Ag+ ions [43]. Figure 4 presents TEM and SEM images of electrospun fibers from 50/50 blend solution containing nanosilver. The diameter of the AgNPs are approximately 50 nm and interaction with fibers were not seemed to cause any structural fragmentation. It was also noticed that the diameter of some particles was above 50 nm which is likely due to the tendency of Ag ions being agglomerated. Findings proved that the nanosilver concentration used in our experiments gave promising results for the process.
(a) SEM and (b) TEM images of 50/50 blend of CS/PEO fibers including nanosilver. Silver nanoparticles are clearly seen on the uniform fibers. Detected size of nanoparticules are approximately 50 nm.
DSC results of nanofibrous scaffolds
Electrospun fibers were subjected to thermal analysis by DSC to determine the influences of blend ratios on fiber properties (e.g. crystallinity). Specimens were heated from room temperature to 200 ℃ and heat flow was measured. Generally, polysaccharides do not melt and degradation is observed above a certain temperature. This is explained by their associations through hydrogen bonding [13]. On the other hand, glass transition temperature is still a complicated matter for CS. The main reason for this is about the polymer properties such as crystallinity, molecular weight, deacetylation degree and the variations about source and/or method of extraction. Therefore, mainly thermal characteristics of PEO in the blended electrospun fibers were to be focused on in our experiments. Endothermic events observed for all blends. According to the DSC thermograms (Figure 5) the peaks of PEO was shifted towards a higher temperature when the PEO content increased in the blends. For instance, the peak for 70/30 (CS/PEO) blend appeared at 54.03 ℃ while it was observed at 60.59 ℃ for 30/70 (CS/PEO). PEO exhibits great tendency for crystallization and its crystals arranged by monoclinic unit cells of crystals with four radially oriented PEO chains. With increasing PEO content, PEO crystals extended and hold each other tightly. However, increasing CS content in the blend destabilized PEO crystallines by interrupting PEO–PEO interactions, therefore, observed peaks shifted to lower temperatures [45].
Thermal diagram of blended nanofibrous scaffolds.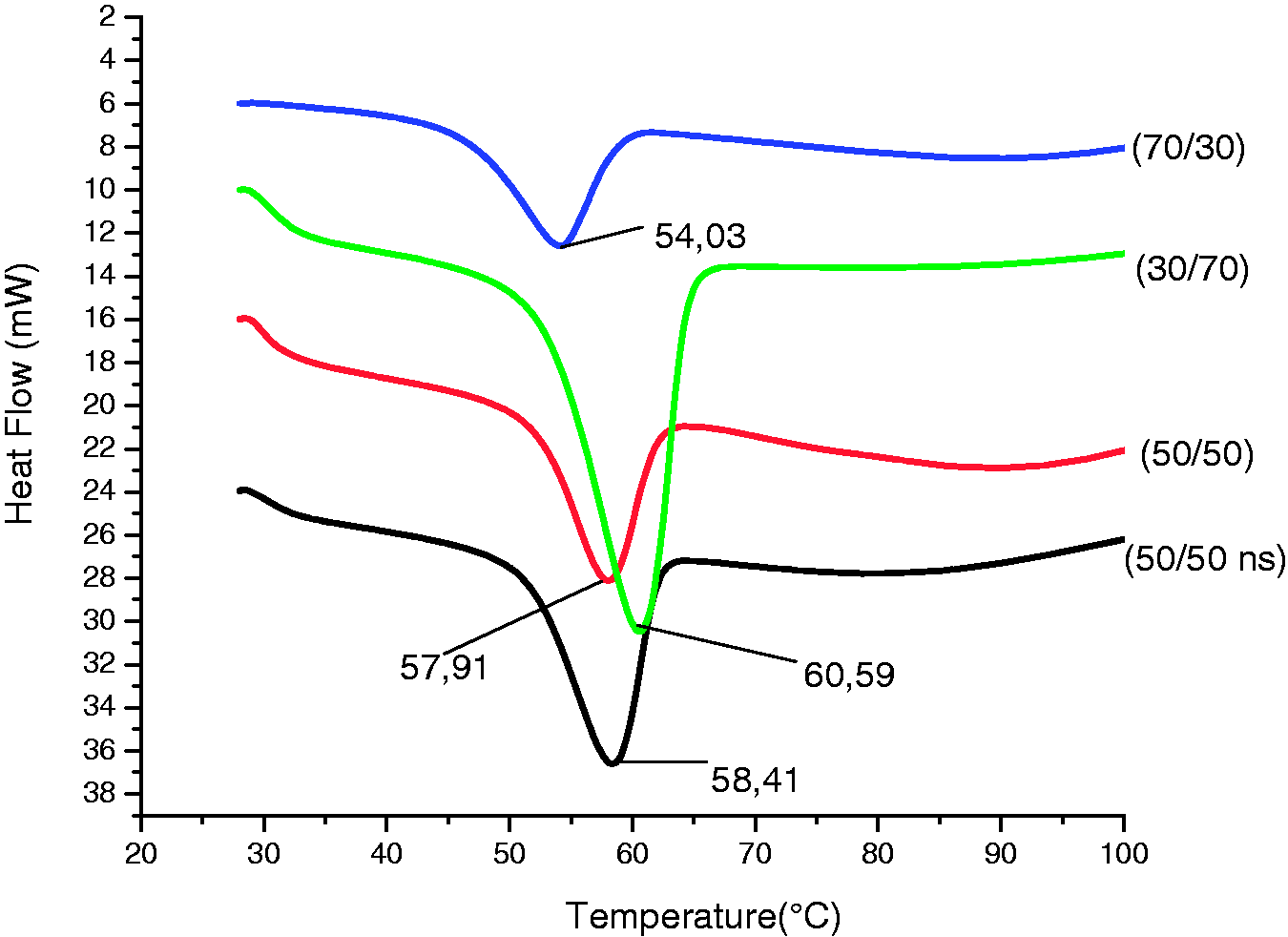
DSC results of blended nanofibrous scaffolds.

On the other hand, there was a slight difference between the peaks of 50/50 blends with and without nanosilver, however, remarkably great variation of Δ
FTIR results of nanofibrous scaffolds
Figure 6 shows the four different IR spectra bands for CS/PEO blends at various ratios (50/50% without nanosilver, 50/50% with nanosilver, 30/70%, 70/30%). The FTIR spectrum of CS shows bands at 892 cm−1 illustrating its saccharide groups [47]. Other bands at 1554, 1639, 2875, 3276 and 3367 cm−1 define the sequence N–H bending of the amino groups, the carbonyl stretching of the amide bands, carboxyl gorups, N–H stretching of the primary amino groups and the (–OH) stretch.
FTIR spectrum of electrospun nanofibrous scaffolds.
Hydrogen bonds occurred among hydroxyl (–OH), carbonyl (C=O–NHR) and amine (NH2) groups in CS [48]. Ether groups (C–O–C) in PEO are also supposed to form hydrogen bonds with CS. Normally, ether groups (C–O–C) in PEO give a characteristic peak at 1112 cm−1 [49]. However in our study, peaks for four different CH/PEO blends, at various contents in ether region, were obtained in lower wavenumbers (around 1095 cm−1) because of the decrease in the amount of ether groups due to the hydrogen bonds occured between the ether groups of PEO and the amine groups of CS. Moreover, it was observed that the absorption band of 70/30% (CH/PEO) shifted to lower wavenumbers (1095 cm−1) in ether region compared with the absorption band of 30/70% (CH/PEO) appeared at 1098 cm−1. The FTIR spectrum exhibited that N–H and O–H strecthing peaks were more distinctive for nanofibers with high content CS (70/30%) compared with low content CS (30/70%).
Tensile properties of nanofibrous scaffolds
The tensile strength of a material accounts for how much stress the material will bear before suffering permanent deformation. In general, tensile strength increases with polymer chain length and cross-linking of polymer chains [50,51].
The tensile strength of nanofibrous scaffolds of various CS/PEO blends (30/70,70/30,50/50 (with and without nanosilver)) were measured. Figures 7 and 8 illustrate the related findings of tensile and elongation values at break of the nanofibrous scaffolds of CS/PEO blends. It is very well known that CS possesses amino groups and hydroxyl groups in its backbone and is a rigid and brittle natural polymer [52]. On the other hand, PEO chains are flexible, so, increasing PEO content was expected to increase the flexibility of the blended nanofibrous scaffolds [53]. As a result of that when CS content increased in the blends, the average value of tensile strength of the materials also increased, however, elongation at break decreased. On the other hand, the tensile strength of the nanofibrous scaffolds was greater in the machine direction comparing with the width direction. This is due to the fibers laid in parallel along with the same rotation direction of the drum collector.
Tensile properties of various CS/PEO blended (50/50 (with and without nanosilver) 30/70/70/30) nanofibrous scaffolds in both machine and width direction. The nanofibrous structures were tested with a 50 N preload at a cross-head speed of 10 mm/min. Elongation at break values (%) of various CS/PEO blended (50/50 (with and without nanosilver),30/70/70/30) nanofibrous scaffolds in both machine and width direction. The nanofibrous structures were tested with a 50 N preload at a cross-head speed of 10 mm/min.

There was no considerable difference between the tensile properties of 50/50 blends of nanofibrous scaffolds including nanosilver and the blends without it. Therefore, it is possible to claim that since amount of AgNPs is very low in the structures, the impact of AgNPs on the tensile property of the nanofibrous scaffolds was negligible.
Although coherent results were obtained through the experiments, in literature, there are critical challenges emphasized for accurate determination of the tensile properties of nanofibers. For example, fiber size distribution in the mats, porosity, individual fiber orientation in the structure, fiber–fiber interaction, entanglement of the fibers, existence of imperfections (beads, etc.) and branching of fibers are important parameters that have great influence on the tensile response of the nanofibrous scaffolds [54]. Moreover, nanofibers of the same polymer electrospun from different solvents often display very different mechanical properties. This is due to the different evaporation rates of the solvents [55].
Antimicrobial properties of nanofibrous scaffolds
The antimicrobial effectiveness of nanofibrous scaffolds has been investigated against After 4 h of incubation colonial growth of Inhibition rate of nanofibrous scaffolds against 

Silver ions and silver compounds have been used as antimicrobial agents for a long time. The inhibitory action of silver ions is assumed that DNA replication ability is interrupted and cellular proteins are inactivated due to Ag+ binds to functional groups of proteins. Since AgNPs are very small in size, antimicrobial impact is stronger because of the high surface area [56].
In our study, 50/50 (CS/PEO) blend nanofibrous scaffolds with nanosilver showed 100% inhibition rate against both
Conclusions
In this study, various blends of CS and PEO nanofibrous scaffolds with/without AgNPs were obtained by electrospinning. PEO was used in order to eliminate CS’s poor electrospinnability. Easy way to integrate nanosilver to the nanofibers was established. Nanosilver dispersion was directly used into the solutions without additional processes, such as heating, microwave irradiation and chemical reduction. Viscosity measurements of blended solutions showed that increasing the amount of PEO in the blended solution also increased the viscosity. This was explained by the size and conformation of the PEO molecules as well as strong entanglements with CS chains. The conductivity of the blended solutions was increased by increasing the CS content as a result of CS’s ability to carry positive charges on the polymer chains. It is also observed that addition of nanosilver increased the conductivity of the solution slightly. FTIR and DSC analysis proved the strong interactions between amine groups of CS and ether groups of PEO. SEM measurements revealed that beadless uniform nanofibers were produced and the diameter of nanofibers in nanofibrous scaffolds decreased from 108 to 63 nm, respectively, by increasing the amount of chitosan in the blends. Addition of AgNPs did not affect the fiber diameter notably. Tensile strength experiments presented that increasing CS content in the blends improved the tensile properties of nanofibrous scaffolds in both machine and width directions. Increasing the CS content in the blended nanofibrous scaffolds promoted the antimicrobial impact on the micro-organisms. All samples exhibited excellent antimicrobial activity. It can be claimed that our experiments showed a way to successfully produce CS-based nanofiber structures (with/without nanosilver) that have potential for use in biomedical applications.
Footnotes
Acknowledgements
The authors would like to thank Mr Mustafa Ilhan for SEM analysis at Material Science Engineering Department of Marmara University, and Mr Omer Faruk Deniz at Gebze Institute of Technology for their kind support about TEM analysis.
Funding
This study was funded by BAPKO (Scientific Research Projects Department of Marmara University, Turkey) with the project code: FEN-C-DRP-090512-0170.
