Abstract
This study offers a new wound dressing by immobilization of amino acid residues on partially carboxymethylated cotton nonwoven fabrics (CM-CN). To improve the absence of capacity to protect the open wound from infection, firstly, a usual cotton nonwoven fabric was chemically modified by utilizing a pad-dry-cure technique including a carboxymethylation procedure. Subsequently, the chicken feather wastes as a natural source were used for the cationization of CM-CN by the same method. Keratin successfully was extracted from chicken feathers using both chemical and enzymatic processes. Physicochemical and biological properties of the prepared samples were evaluated by FTIR, SEM, mechanical properties such as tensile, bending test and wrinkle recovery, biodegradation, permeability (air and water vapor), and MTT assay for cell viability and proliferation. The tensile strength increased to 200%, the air permeability almost doubled, and the greatest cell growth was observed for modified samples. According to the obtained results, enzymatic extraction was influential and the samples coated with enzyme-extracted keratin showed enhanced properties suitable for wound dressing applications.
Keywords
Introduction
Cotton is a common material for wound dressing for its effective properties [1]. Nevertheless, a lack of functionality due to the inert surface could lead to some limitations in it. To this aim, several functional groups (e.g., carboxyl, hydroxyl, and amine) have been reported to modify the surface of cotton fabrics to achieve applicable functionalities [2–4]. The finishing industry also used some of these modifications for the production of commercial products [5]. Although the cotton adhesion to the wound site, not having an active role in the healing process, and the possibility of bacterial growth are main concerns regarding their use as wound dressing [6–8].
Provision of a suitable healing environment [9,10], protecting the wound from bacterial infection, and biocompatibility are the main properties of an ideal wound dressing material [11–13].
Until recently, one of the challenging issues in the treatment of burn injuries, especially in developing countries, was the effective treatment by relatively expensive biomaterials for wound dressing [14]. Hence, there is a need to develop inexpensive and efficient wound dressing materials from available natural sources [15,16]. Natural biomaterials such as chitin [17], chitosan [18], alginate [19], silk fibroin [20] have been grafted onto cotton fabric to accelerate the healing process and providing active wound dressing.
The small molecules such as peptides and proteins have a perfect effect on dressing features and cell behaviors. The functional groups of the small molecules have a high influence on surface properties like wettability that can affect the ability of protein adsorbance or cell behaviors. Some functional groups such –NH2 and –OH on wound dressing or other biomaterials have more benefits compared to other groups such –COOH or –CH3 for cell behaviors [21].
Keratin as a source of functional groups can hydrolyze [22] and then the hydrolysates, amino acids, enzymes, and other products have been obtained. Proteins and peptides derived from chicken feathers keratins can be added to the cotton surface. Keratin consists of cysteine-rich proteins derived from nails, hooves, horns, feathers, wool, and human hair [23], and notably, human hair keratins and feather keratins are the two most appreciated options. Howbeit, feather keratin is more available (around three billion pounds of chicken feathers are produced in a year), and has easy quality control. As a result of exceptional properties such as natural abundance, biodegradability, biocompatibility, and bioactivity [24], keratins have been widely utilized in wound healing and hemostasis applications [25,26].
Recently, Wang et al. [27] compared the properties of feather keratin-based wound dressing with human hair keratin
The present work aims are to utilize the inexpensive and optimal textile finishing processes to produce modern active wound dressing material. On the other hand, the widely used cotton gauze suffers from some disadvantages namely the lack of ability for open wound protection from infection. The carboxymethylation process as a chemical method for modifying the cotton gauze is a way to solve this shortcoming [28]. In this study, the cotton nonwoven fabric was partially carboxymethylated and then was coated by keratin using a pad-dray-cure method. Keratin was extracted from chicken feather by chemical and enzymatic approach. Their characteristics were compared in terms of microstructure, physical, mechanical, antibacterial, and biocompatibility properties.
Materials and methods
Materials
Chicken feathers used in this study were supplied from a local poultry processing center. Spun lace cotton nonwoven fabric (Cotton 100%) having 110 g/m2 was obtained from the Niko Industrial group, Abhar, Iran. Reducing agents including borate, acetic acid, ethanol, sodium carbonate, sodium dodecyl sulfate (SDS), and sodium sulfite with analytical grade were purchased from Merck Co (Germany). Savinase 16.0LEX (EC.3.4.21.14), an alkaline serine protease enzyme, was supplied from Novozymes A/S, Denmark.
Keratin extraction from chicken feathers
Feathers were washed in 1% aqueous Diadavin EWN01, a non-ionic surfactant (Saveh Resin, Iran), and 1% sodium carbonate at 60°C for 30 min. The removal of lipids from feathers was carried out by 80% ethanol for 2 h. Subsequently, the drying of the degreased feathers was done at 100°C, and then feathers were chopped to 2–5 mm pieces [29].
Sulfitolysis method
Keratin was extracted from chicken feathers by industrial sodium sulfide. The reaction of sodium sulfide with feather keratin is complex, but in water solution, the hydrosulfide and hydroxide ions have formed and then the protein disulfide bond have been reduced (Figure 1) According to a study conducted by Poole et al. [30], after 24 h, a maximum dissolution yield of 62% was achieved, at a concentration of 50 g/L of Na2S. Although the obtained yield amount was lower than what was reported by using the MEC (75%), the authors utilized a lower level of thiol, suggesting that the ideal concentration of Na2S for keratin extraction from feathers is 10 g/L. Therefore, around 0.5 g of dried feathers were immersed in 10 g/L Na2S and stirred at 70°C for 6 h. Thus, the keratin solution was filtered to separate residual feathers, and then the filtered solution was centrifuged to precipitate keratin and washed using acetone. The acetone was removed, and the remaining keratin was dried in an oven at 100°C for an hour [31].

The formation of hydrosulfide ion from Na2S and the reduction of S-S bonds in cysteine by it.
Enzymatic hydrolysis method
Keratin is also extracted by enzymatic hydrolysis by sodium sulfite reducing agent, surfactant sodium dodecyl sulfate, and an alkaline protease. The incubation of the processed feathers was carried out with Savinase in a borate buffer solution with a 50 mM concentration and pH equal to 8.5, including sodium sulfite as a reductant and 1 g/L sodium dodecyl sulfate (SDS). The samples were incubated at a temperature of 55°C for 1.5, 3, and 6 h. For enzyme deactivation, after the hydrolysis process, 5 ml of acetic acid (0.5 M) was added to the samples (pH = 4), and then the temperature reached 75°C and maintained at this condition for 15 min. Subsequently, residual waste feathers were then filtered. Lastly, a cellulose tube with a molecular weight cutoff of 3.5 KD was used to dialyze the obtained keratin solution against distilled water. The dialyzing process was done for 2 days, and water was frequently replaced during the process. Bovine serum albumin was used as a standard according to the Hartree-Lowry method to calculate the amounts of the released protein in each effluent and optimize the enzyme hydrolysis parameters [31]. The studied variables were extraction time, the concentration of the enzyme, and the reductant.
Moreover, the following equation was used to calculate the weight loss of feather:
In equation (1), the initial weight and the weight after enzymatic treatment are represented by w1 and w2, respectively [32,33].
Modification of cotton nonwoven fabric using carboxymethylation treatment
To ensure removing machinery additives or oils, the cotton nonwoven fabrics were placed in a bath with a detergent concentration of 1 g/L and L:R of 1:10 at 60°C for 30 min. After the removal of the specimens, the rinsed fabrics were washed three times with distilled water, dried in an oven at 105°C for 1 h, and immediately placed in a desiccator for 1 day.
For the carboxymethylation process, samples with 20 × 20 cm2 dimensions were immersed in an aqueous 15% sodium hydroxide solution at 30°C for 15 minutes and kept at room temperature for 30 min. The alkaline cellulose samples were then immersed in the chloroacetate solution (30%) neutralized by sodium hydroxide for 15 min. The samples were then cured at 70°C for 3 h, then neutralized with 0.2 M acetic acid and washed three times in isopropanol-water solution (1:4 volume ratio) and finally dried in an oven at 90°C [14].
Grafting of keratin
The carboxymethylated cotton nonwoven fabric was soaked in the prepared keratin solution (50 g/L) for 1 h and padded with 100% pickup via pad-dry-cure technique at room temperature. The padded samples were then placed in an oven at 80°C for 15 min and then cured at 130˚C for 3 min. Specimens coding is listed in Table 1.
Samples coding.

Characterization of prepared dressings
Fourier transform infrared spectroscopy (FTIR) analysis
The FTIR spectra of the prepared dressing materials were obtained using an infrared spectrometer (870 ESP FT-IR), Thermo Nicolet Nexus (USA) equipped with the Omnic data processing software by KBr pellets with a resolution of 8 cm−1 in the range of 400–4000 cm−1. The FTIR analysis was carried out at 65% humidity and 20°C temperature. The absorbance vs. wavenumber was recorded.
Scanning electron microscopy (SEM) analysis
Scanning electron microscopy (SEM) (XL30, Philips, Netherlands) was utilized to study the microstructure and morphology of the specimens. Before observations, the surface of the prepared fabrics was coated by a thin layer of gold by sputtering. The surface scanning was performed at 15 kV.
Mechanical properties
To measure the tensile properties of the samples according to ASTM D5035, Universal Testing Machine (Instron 5566, United States) was employed. The respective samples were taken in triplicates. To this aim, the samples were cut in 180 × 25 mm2 sections, at the angles of 0° and 90°. The vertically clamped specimens were tested with a gauge length, a trigger load, and the test speed of 50 mm, 10 N, and 100 mm/min, respectively, and were stretched until failure. Bending strength was evaluated by Shirley stiffness tester (SDL Atlas, UK). This test followed ASTM D1388-08 Standard Test Method for Stiffness of Fabric.
Pore size and porosity analysis
For assessment of the porosity and pore size of specimens, many instrumentations and PC tools could also be used. Some various soft wares like SemAfore or ImageJ can be used to analyze SEM images to measure pore sizes and porosity. From the SEM images, an estimation of the cross-sectional area, interconnectivity, and wall thickness can even be obtained. During this study, we've used the ImageJ package that technique permits fast measurement of porosity from thin section images and it is easy to use and does not need specialist training [34,35].
Water uptake
For bulk hydrophilicity evaluation, the dried specimens were weighed (Wdry) and then soaked in PBS (Phosphate-Buffered Saline) (pH = 7.4) incubated for 5 h at 37°C. In time intervals (1, 2, and 3 days), the soaked samples were taken out from the solution, the excess PBS on the surface removed using a filter paper and finally reweighed (Wwet). The water uptake ΔW was obtained by equation (2):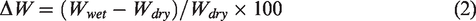
Air permeability and water vapor transmission rate
Air permeability was measured using Shirley air permittivity tester as per ASTM D7337-04. The airflow direction was perpendicular to the surface of the specimens and the change of airflow volume in a determined time was reported as air permeability.
The water vapor transmission rate (WVTR) was measured according to ISO 11,092 using PERMETEST. Heat flux sensing is the principle used in the instrument. The measuring head is kept at room temperature to maintain isothermal conditions. The water flow results in some heat loss in the measuring head, measured with and without the fabric covering to determine the water vapor permittivity. Measuring the ratio of heat loss value from the measuring head with fabric covering (u1) and the bare head without fabric covering (u0) can calculate the water vapor permeability of the dressing materials, presented in equation (3).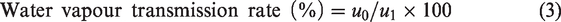
Cell morphology study and cell viability assay
The biological activity of the samples was investigated through cell morphology and viability using the interaction of mouse L929 fibroblast cells on the fabrics. In this study, Roswell Park Memorial Institute (RPMI)-1640 growth medium (for fibroblast cells) and Dulbecco modified Eagle medium (DMEM)-Ham's F12 (for HUVECs) are used as culture medium.
The cell morphology study was performed using mouse L929 fibroblast cells. For this purpose, the sterilized samples by ultraviolet (UV) irradiation were placed at the bottom of each well of a 12-well plate. Then, cells were seeded onto surfaces of membranes at a density of 1 × 104 cells per well and cultured in an expansion medium composed of RPMI-1640 growth medium supplemented with 10% (v/v) FBS and 1% antibiotic penicillin/streptomycin in a humidified atmosphere incubator at 37°C and 5% CO2. The culture medium was changed every two days. After 24 h of cell seeding, the cell-containing samples were washed with PBS twice, followed by removing the culture medium, then fixed with a 2.5% GTA solution. To observe cell attachment and morphology on the surfaces of the samples by SEM, the fixed cells were dehydrated in the ethanol and sputter-coated with gold.
To evaluate the cell viability, the L929 fibroblast cell line was used. The cultured cells undergone three passages and then were seeded on the samples with 1 × 105 cells/well density. The fabricated dressing specimens were incubated for 3 h with 5% CO2 at 37°C. Dulbecco Modified Eagle Medium (DMEM; Sigma Aldrich) was added and incubated with 5% CO2 at 37°C. The medium was composed of streptomycin (50 unit/ml), penicillin (50 unit/ml), and 10% fetal bovine serum (FBS; Invitrogen) and was changed every 2 days. Cell viability was determined after 24, 48, and 72 h using 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT; Sigma Aldrich). This assay quantitatively determines the cell viability based on the color change resulting from the MTT to MTT formazan conversion by viable cells’ mitochondrial metabolism. The extract of samples, dressing materials, were prepared and then added to MTT solution following by the absorbance measurement at the wavelength of 540 nm on a micro plate reader (Biotek; USA).
Statistical analysis
A variance analysis was employed for the statistical analysis. A mean and standard deviation (mean±SD) were obtained by using triplicate results of each sample (n = 3), and p < 0.05 was considered statistically significant.
Results and discussion
Enzymatic hydrolysis optimization for keratin extraction
As mentioned, the extraction of keratin from chicken feather was performed using an alkaline protease for enzymatic hydrolysis. To determine the optimal enzymatic condition, the effects of hydrolysis time, the concentration of the reducing agent, and enzyme loading on the hydrolysis yield were studied; the results are listed in Table 2. According to the results, using 10 g/L the reducing agent, 2% enzyme, and 6 h hydrolysis time, the maximum feather fiber weight loss was obtained. It is expected that the degradation of protein has improved by the hydrolysis time. However, the 1.5 h hydrolysis time yielded the highest protein content because the free enzyme in the reaction mixture breaks down the released proteins. The influence of reducing agent concentration was studied at an enzyme concentration of 2% and a hydrolysis time of 1.5 h. As seen, with the increase in the reducing agent concentration, the liberated soluble proteins increased. The reductant concentration increment leads to the breakage of the disulfide bonds in the polypeptide chain and consequently, easier hydrolysis by the protease enzyme [36].
Effect of different variables on the released amount of protein from chicken feather.

The influence of the enzyme loading was studied at the optimal reductant concentration of 15 g/L and the hydrolysis time of 1.5 h. The results showed that when enzyme loading increased from 0.5 to 2%, both weight loss and released protein increased, and the further increase led to a significant reduction. The reduction is the factor of inhibiting the action of free protease molecules for proteolytic attacks, which disables the enzyme action. Adsorption saturation for hydrolysis for a fixed substrate is achieved by a certain amount of enzyme [36]. Hence, for obtaining the maximum yield, enzyme concentration should be an optimum value. Consequently, 15 g/L the reducing agent, 2% enzyme and 1.5 h hydrolysis time were selected for optimal enzymatic hydrolysis.
FTIR analysis
FTIR spectra of the samples have been shown in Figure 2. As could be seen, the peaks at 2871 and 2931 cm−1 are related to C-H vibrations, and the broad peak appeared at 3402 cm−1 is related to -OH vibration. Furthermore, the peaks at 1120 cm−1, 1235 cm−1, and 1735 cm−1 are associated with C-O-C stretching, C-O ester bonds, and carbonyl ester. The peaks also appeared at 1602 cm−1, 1424 cm−1, and 1326 cm−1 are related to carboxylate. The existence of C-O and C = O ester with COO- carboxylate bond in CMC structure is the carboxymethylation confirmation of the samples [14]. After keratin coating, the peaks appeared at 1655 cm−1 and 1542 cm−1, representing the C = O stretching vibrations related to amide I bonds and N-H bendings of amide II bonds. The successful immobilization of keratin is determined by the presence of these peaks [29].

FTIR spectra of the samples.
Mechanical properties
Skin is continuously subjected to a little stress and acts as a viscoelastic material. The tensile strength (TS) of skin changes with the stress direction and depends on the loading rate. Furthermore, it varies from the place, age, and gender and is reported to be around 1.8 MPa. The TS of a wound alters during the different wound healing stages and is generally weaker than normal skin [9]. TS and elongation at break (E) of the prepared samples are shown in Table 3.
Tensile properties of the samples.

Bending test results of the samples.

Compared with other samples, untreated cotton nonwoven fabric showed a high elongation at break with lower tensile strength. The tensile strength of N-SK and N-CM-SK did not change significantly, while an increase in the tensile strength and elongation was observed for N-CM-AK. The existence of enzymes leads to the random reaction of the cellulose chains, the formation of open chains ends, and disruption of local crystallinity of cellulose structure, which results in the provision of more active places available for keratin grafting and improvement of mechanical properties.
One of the critical drawbacks of cotton fabrics is wrinkling. When cotton fabrics absorb water, movements of internal polymer chains can be facilitated, which results in the removal of hydrogen bonds, presenting in configurations of cellulosic. Marked wrinkles could be created on the surface of fabric due to the new hydrogen bonds forming in new positions. The obtained results showed that the wrinkle recovery angle was enhanced by treating cotton fabrics with CM and keratin. Glucose rings joined together to form cellulose macromolecules to provide reactive sites for crosslinking by projecting hydroxyl groups. Improvement of elastic recovery and higher resistance to deformation is caused by crosslinking reactions between available hydroxyl groups of cellulose [10]. The enzyme’s role in the crosslinking mechanisms can be related to the fact that it can act as a catalyst and an accelerator in grafting keratin to cellulose fibers.
Basic information on the mechanical properties of wound dressing materials is incredibly necessary while these materials require similar durability, resilience, bending, and elastic properties to the skin. Bending strength is the resistance of a fabric to bending when a force is applied along one edge while the opposite edge is held in a fixed position. When the cotton nonwoven fabric is used as a dressing, owing to the superior flexibility, it can be easily contoured over parts such as the toes, knees, sacrum, heel, etc. [37]. This will improve the efficiency of treating deep and irregular shaped wounds. As shown in Table 4, In terms of flexural strength, all specimens including N, N-CK, N-EK, and N-CM-EK were very flexible and N-CM-CK wasn't close to the commercial wound dress (VG).
Pore size and porosity and water uptake analysis
The pore size of the samples and surface porosity was determined based on the SEM images with the Image J software. The surface porosity of the specimen is given in Table 5. As can be seen, both pore size and porosity are in the near amount.
Surface porosity and mean pore size of samples.

Water absorption capacity is related to the interaction of the fiber molecules and the water molecules. The water uptake results of the samples are demonstrated in Figure 3 and the water uptake after 70 h are shown in Table 5. The up taken water contains the water that fiber molecules absorb and the moisture fills up in the inter-yarn and inter-fiber pores. The process of water uptake massively depends on the porosity and hydrophilic groups/segments.

Water uptake of the samples.
The presence of some functional hydrophilic groups such –NH2, -OH or –COOH increase water uptake, and some functional hydrophobic groups like –CH3 decrease it. As seen, the presence of functional groups and porosity influence water uptake. Both carboxymethylated samples (N-CM-CK) and (N-CM-EK) that have hydrophilic group carboxyl groups have shown more water uptake during the test, than non-carboxymethylated samples (N-CK) and (N-EK). With the increment in the number of hydrophilic groups of biomaterials, the water absorption by fibers has enhanced. By the increase of the porosity, water entrapment is also increased.
Air permeability
The air permeability results of the tested samples are demonstrated in Table 6. In comparison with the cotton nonwoven fabric, air permeability increased in coated samples. Moreover, the air-permeable value was mainly reduced in the N-CK fabric compared with other samples. Hence, fabric coating changes the pore amount in the specimens [38]. The proper air permeability and presence of oxygen can accelerate wound healing. However, all samples possess a sufficient amount of air permeability in comparison to commercial dressing material (Table 6).
The air permeability of the samples.

The ideal dressing would manage the evaporative water loss from a wound at an optimal rate. The water vapor permeability of a wound dressing should prevent both excessive dehydration and buildup of exudate [9]. As been shown in Figure 4, the WVTR of all samples was near the commercial wound dressing (VG) and these specimens would supply an adequate level of moisture without risking wound dehydration.

WVTR for the samples (p < 0.05).
Scanning electron microscopy (SEM) analysis and cell morphology
As mentioned through the carboxymethylation, the fabric first becomes alkaline, altering the molecular order, structure, and degree of crystallinity of the fibers. If normal cellulose is considered a weak acid, alkaline cellulose I will be produced after processing with the alkaline solution. Cellulose I converts to cellulose II by subsequent washing and neutralization [39,40]. Evidently, after carboxymethylation, the natural twists of the cotton fibers were opened, and the cross-section of the fibers changed from bean shape to cylinder. Based on Figure 5(a) and (b), after surface modification, the swelling of the fibers and changing morphology (from ribbon to circle) were observed. This phenomenon correlated with the covalent bonding between monochloroacetic acid and cellulose, which leads to an increase in the distance among cellulose polymer chains and swelling. Due to the carboxymethylation, the thickness of the fibers is enhanced compared to the untreated cotton that creates a smaller volume between fibers. Carboxymethylation disrupts the bonds between fibrils and fibers during swelling and the Surface accessibility for chemical bonding increases, leading to higher fluid absorption. Based on Figure 5(c) and (d), in the N-CK sample, the fibers’ surface is not only rough but also, the keratin distribution is uneven. In contrast, the surface of fibers in N-EK is smooth and homogeneous with minor twisted fibers. The adhesion of fibers and formation of holes and cracks could be observed in Figure 5(e), although such structure is not visible in Figure 5(f).

SEM images of (a) the cotton nonwoven fabric before carboxymethylation (N), (b) the cotton nonwoven fabric after carboxymethylation, (c) N-CK, (d) N-EK, (e) N-CM-CK, and (f) N-CM-EK.
Adhesion and growth of cells on cotton nonwoven fabrics were evaluated using mouse fibroblast cells (L-929). The behavior or response of cells when exposed to a scaffold, which includes cell adhesion, filopodia, or spreading of cells, is an important factor in determining their biocompatibility [39]. SEM images indicate that the cells are present on the surface of all samples indicating the biocompatibility of the used cellulose. Comparing the images of Figure 5(a) and (b) and (c) and (d), it appears that both modifications (carboxymethylation and keratin grafting) enhance the cell supporting capacity of the modified cotton.
Cell viability assay
The cell viability evaluation was carried out on the samples for 3 days. As seen in Figure 6, the dressing fabrics were non-toxic and could support cell proliferation and growth. It can be observed that all samples’ cell viability increased over time, revealing their capability in cell proliferation. In previous studies, suitable cell proliferation and cell growth on scaffold consisting of keratin and chitosan for tissue engineering applications were reported by Balaji et al. [41]. Furthermore, a keratin scaffold coated by calcium phosphate for bone tissue engineering represented high cell growth and proliferation [33,42]. The maximum cell growth is observed for N-CM-EK and N-EK, which illustrates that the presence of enzymes can positively affect cell activity, adhesion, and growth.

Cell viability of the dressing materials in 3 days. Results are expressed as a percentage of control (N) (*p < 0.05, **p < 0.01, ***p < 0.001).
Conclusion
In this research, the cotton nonwoven fabric was partially carboxymethylated and then was treated with feather keratin extracted by both chemical and enzyme for achieving modern active wound dressing properties. Carboxymethylation and grafting of keratin were successfully carried out by the pad-dry-cure process. According to the results, partial carboxymethylation can enhance keratin coating due to more active sites on the cellulose surface. Moreover, enzyme treating showed better microstructural, physical and, mechanical properties as well as a biological activity because of the improvement of keratin coating on the fabric and activation in the biological environment offering this material as a promising fabric for active wound dressing.
The result of this research indicates that the surface modification through grafting feather keratin onto carboxymethylated cotton improved the air and water vapor permeability. The extraction method of the keratin could play the key role in the result. The air permeability was increased by 87.9% and dressing can facilitate air circulation when we use the feather keratin that extracts enzymatically. The WVTR were 16.3% higher than the untreated specimen and near to commercial wound dressing. Thus, when used for a wound, the dressing can absorb exudate more. Also, while using recycling material such as keratin, the cost of wound dressing fabrication was much lower than using other materials. The small molecules like proteins and peptides derived from keratin have an excellent influence on both dressing properties and cell behavior.
Footnotes
Acknowledgements
The authors would like to thank Miss Mina Aleemardani and Miss Sara Sadeghi of Amirkabir University of Technology for their help and efforts in completing this research.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The article is the result of a PhD thesis and the authors are supported by Amirkabir University of Technology as a state university.
