Abstract
The present research focus is to design a new catalyst by doping polyoxometalate (POM) in conducting polymers. Polypyrrole (PPy)-doped Keggin type POM [PMO12O40]3− with various molar concentrations were synthesized using the coprecipitation method. The doped samples were synthesized and characterized using spectral techniques such as Fourier transform infrared spectra, which show the presence of extended functional groups, and scanning electron microscopic morphology exhibits a square-shaped structure. X-Ray diffraction results reveal crystallite size of around 40 nm. The conductivity of PPy is 3.3 × 10−6 S cm−1, whereas polyacid-doped polypyrrole has resulted in increased conductivity of 7.12 × 10−5 S cm−1. The antimicrobial activity of PPy revealed a better antibacterial activity due to the presence of the NH group and aromatic ring in the structure.
Keywords
Introduction
Polyoxometalate (POM)-based polymers are the most advanced field in today’s research because of its applications as electric and electronic devices. POMs are an exceptional family of inorganic clusters consisting of Mo, W, V, and so on early transition metal ions in their highest oxidation states with structural versatility and a wide range of optical, electronic, and magnetic properties and applications. 1 -3
Polypyrrole (PPy) is highly electrically conductive, and it can be easily deposited through polymerizing on the surface of fiber for wearable devices. 4 In recent years, PPy doped have been widely used in supercapacitors due to their electrochemical stability, high conductivity (10–100 S cm−1), and intrinsic flexibility; PPy also possesses better electronic and electrochemical properties compared with Polyaniline (PANI). 5 Like many conducting polymers, PPy has long-chain conjugated chemical bonds in its molecular structure and can conduct electrons and provides proton transport channel. It is a good material for preparing doping membrane. 6 PPy has been the most widely used polymer in different fields such as electrochemistry, electronic materials, and sensors. 7 Generally, conducting polymers possess low conductivity in the neutral states, and their intrinsic conductivity is attributed to doped charge carriers. However, conducting polymers are able to exhibit a high conducting ability, whose electrical conductivity depends on the doping levels. In addition, conducting polymers have reversible doping ability, excellent optical activity, ease of synthesis, low cost, and outstanding intrinsic flexibility. 8 The conducting polymers possess electrical conductivity which has attracted much of research interest and various applications like batteries, polymer light-emitting diodes, electromagnetic shielding, supercapacitors, and sensors. 9 -16 PPy has charge carriers which are mainly considered to be polarons and bipolarons used for stability of counter ion. 17 In this literature, PPy is synthesized either by oxidative chemical polymerization or by electrochemical oxidation in organic solutions. 18 -20 PPy is synthesized by oxidative chemical polymerization method.
The polyacid (PA) is the metal-oxygen clusters with MO6 units set to bridge the oxygen atoms. 21 -25 PA is an early transition metal, which is used mostly in the different fields. In the field of catalysis, the Keggin type POM has many important applications. Doping of PPy with new materials shows effective results, with an important application such as conductivity, antimicrobial activity, and supercapacitor. A limited number of studies are available in the literature about POM-doped materials and hence we have attempted to design new POM-doped polymer composites. The polymers do not possess catalytic properties but POM-doped polymers are very good catalyst. A new catalyst has been synthesized and characterized using different spectral techniques. In this article, we have discussed the synthesis, characterization, and conductivity studies of Keggin type POM-doped PPy composites. Hereafter, Keggin type POMs is mentioned as PA and polypyrrole as PPy.
Materials and methods
Chemicals used
All common laboratory chemicals and reagents were purchased from Merck. Ammonium molybdate, manganese (II) sulfate, disodium hydrogen phosphate, potassium persulphate, concentrated nitric acid, pyrrole, and ferric chloride were used without further purification.
Synthesis of Mn (IV)-containing PA
The PA was prepared by dissolving 1 g of manganese (II) sulfate, and the oxidant potassium persulfate in 10 ml of distilled water. To this, 4 g of ammonium molybdate which is dissolved in 10 ml of distilled water was added. To this mixture, 1 g of disodium hydrogen phosphate was added, followed by dropwise addition of a few drops of concentrated nitric acid with constant stirring. The formed product was filtered and dried.
Synthesis of PPy
The PPy was prepared by adopting the method in the literature with a slight modification. 26 Pyrrole (3.4 ml (0.3 M)) was dissolved in ethanol and stirred for 10 min. FeCl3 stock solution (25 ml) was added slowly to pyrrole solution with vigorous stirring, a dark blue-colored precipitate of PPy was formed.
Synthesis of PA-doped PPy
To this PPy, PA solution (0.2 M) was added with constant stirring. After ultrasonication (1 h), the stirring was continued for 2 h to form the PA-doped PPy. The formed PA-PPy1 is filtered and dried at room temperature. Likewise, the other two doped compounds (PA-PPy2 and PA-PPy3) were synthesized by adopting the same procedure except varying the weight percentage of (0.3%, 0.2%, and 0.1%) the PA. The same procedure was carried out to prepare the following compounds MnPA-PPy1, MnPA-PPy2, and MnPA-PPy3.
Analytical measurements
The sample was analyzed using analytical techniques. IR spectrum was recorded using a Perkin Elmer spectrometer (USA) in the spectral range of 4000–400 cm−1 to analyze the chemical structure of the composite. The morphology of the composites was investigated using scanning electron microscopy (SEM) (JASCO JSM 6390 SEM) (USA). The X-ray diffraction (XRD) spectrum was recorded using (SHIMAZDU XRD-6000) (Japan) diffraction analysis. The electrical characterization of sample was carried out by impedance analyzer PSM 1735 (UK).
Results and discussion
FTIR spectra
Fourier transform infrared (FTIR) spectra of PA, PPy, PA-PPy1, PA-PPy2, and PA-PPy3 are shown in Figure 1. The IR spectral data of PA, PPy, PA-PPy1, PA-PPy2, and PA-PPy3 are presented in Table 1. The tentative band assignment is based on the literature. 27 The peak at 1547 cm−1 is assigned to the stretching vibration of C–C bond. The observed peak at 1315 cm−1 is due to the in-plane vibrations mode of C–H bond. One at 1045 cm−1 is assigned to the in-plane C–H stretching vibration of pyrrole ring and the other peak at the spectral region 827 cm−1 is wagging (out of plane) vibrational bending mode of C–H bond. The peak at 1643 cm−1 is assigned to intra ring (C=C) vibration. The peak at 1067 cm−1 indicates the absorption of P–O stretching vibration. The peak at 974 cm−1is assigned to Mo–Ot stretching. A peak at 854 cm−1 is stretching frequency of M–O–M bonds. The stretching vibrations of M–O–X is at 704 cm−1 (X=P). The IR spectra of three compounds, PA-PPy exhibits absorption peak at 969, 783, 865, and 1055 cm−1. The IR spectra of MnPA, MnPA-PPy1, MnPA-PPy2, and MnPA-PPy3 are shown in Figure 2. The IR spectral data of MnPA, MnPA-PPy1, MnPA-PPy2, and MnPA-PPy3 are presented in Table 2. The peak at 717 cm−1 indicates the stretching frequency of M–O–X (X=Mn). The IR spectra of three compounds, MnPA-PPy exhibits observed peak at 1061, 980, 944, and 717 cm−1. A smaller change in the frequency occurring (M–Ob–M) is explained by assuming that rearrangements of the molybdic framework have greater capability. An express lowering (1067→1055 and 1045) of ﻻ (P–O) frequency in (PA-PPy1, PA-PPy2, and PA-PPy3) is always happening with an elongation of P–O bonds, 28 due to the protonation of PPy with PA resulting in the downward shift of all three compounds PA-PPy such that the Mo–Ot stretching frequency is varied. The upward shift of 854 cm−1 in all three different percentages of PA-doped PPy is due to the Van der Waals interaction between polymeric chain of PPy and PA, which, in turn, alters the P–O stretching frequency of the PA-doped polymers.

IR spectra of (a) PPy, (b) PA-PPy1, (c) PA-PPy2, and (d) PA-PPy3.
IR spectral data of PPy, PA-PPy1, PA-PPy2, and PA-PPy3.
IR: infrared; PPy: polypyrrole; PA-PPy: polyacid-doped polypyrrole.

IR spectra of (a) MnPA (b) MnPA-PPy1, (c) MnPA-PPy2, and (d) MnPA-PPy3.
IR spectral data of MnPA, MnPA-PPy1, MnPA-PPy2, and MnPA-PPy3.

IR: infrared; MnPA: manganese-containing polyacid; MnPA-PPy: manganese-containing polyacid-doped polypyrrole.
Scanning electron microscopy
The SEM images of PA, PPy, PA-PPy1, PA-PPy2, and PA-PPy3 is shown in Figure 3. The SEM images of MnPA, MnPA-PPy1, MnPA-PPy2, and MnPA-PPy3 are shown in Figure 4. The SEM image of PPy shows the granular structure which is in nanometer range. The SEM image of PA-doped PPy exhibits the morphology of square-shaped structure. The square-shaped morphology is due to network formation between PA and granular PPy. A very high magnification reveals the transformation of highly branched PPy into granular structure of PPy and PA particle is highly agglomerated with the PPy. 29 The increase of weight percentage PA in PPy (0.2, 0.3, and 0.4 wt%) results in increasing the granular size, which, in turn, decreases the porosity of PA-doped polymer. The SEM image of MnPA shows the granular structure, which is in the nanometer range. The diameter of the MnPA-doped PPy exhibits the dopant crystal in chain structure of PPy.

SEM morphology of (a) PPy, (b) PA-PPy1, (c) PA-PPy2, and (d) PA-PPy3.

SEM morphology of (a) MnPA (b) MnPA-PPy1, (c) MnPA-PPy2, and (d) MnPA-PPy3.
X-Ray diffraction
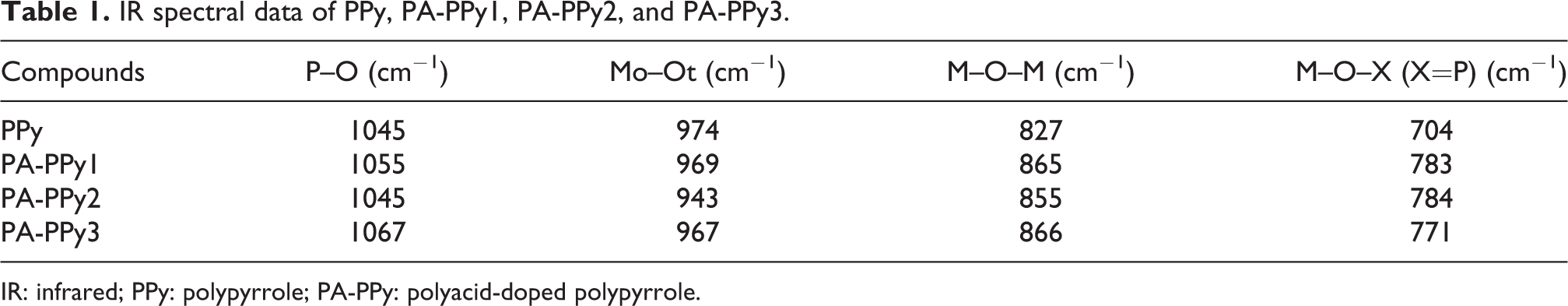
The XRD pattern of PA, PPy, PA-PPy1, PA-PPy2, and PA-PPy3 is shown in Figure 5. These sharp peaks at 23.7°, 43.99°, 64.44°, and 77.60° are characteristic peaks of PPy. PA-PPy at different weight percentages of (0.4%) shows peaks at 10.73°, 26.49°, and 30.68°. The diffraction peak of PA-PPy has reduced peaks, which indicate that PA is attached with PPy. The pattern shows crystalline cubic structure and is in good agreement with Joint Committee on Powder Diffraction Standards (JCPDS) card number (87-0546). 30 The (hkl) values for some of the most prominent peaks are (321), (420), (222), and (311), which corresponds to the interplanar distance (d). The doped compounds of PPy (0.3%) show peaks at 21.33°, 26.33°, and 30.49° and (0.2%) show peaks at 25.98°, 30.16°, and 44.21°. The grain size of MnPA, MnPA-PPy1, MnPA-PPy2, and MnPA-PPy3 is presented in Table 3. The XRD pattern of MnPA-doped PPy, respectively, shown in Figure 6. The sharp peaks at 27.7°, 30.2°, and 40.2° are characteristic peaks of MnPA. The XRD spectra of the complex MnPA-PPy1, MnPA-PPy2, and MnPA-PPy3 at different weight percentages of (0.4%) show peaks at 26.64°, 53.7°, and 60.1, (0.3%) show peaks at 26.2°, 52.1°, and 60.5°, and (0.2%) show peaks at 26.9°, 44.3°, and 53.7° respectively. The pattern shows crystalline monoclinic structure and is in good agreement with JCPDS card number (72-0285). 31 The (hkl) values for some of the most prominent peaks are (311), (204), (311), and (202), which correspond to the interplanar distance. The average crystallite size (D) is estimated from line broadening in XRD using Scherer’s formula, D = kλ/βcosθ where “k” is the phase factor = 0.9, “λ” is the wavelength of X-rays = 1.54 Å, “β” is the full width at half maximum and “θ” is the angle of diffraction. The XRD pattern shows that interaction between PPy and PA has occurred. The grain size and crystallinity of PA, PPy, PA-PPy1, PA-PPy2, and PA-PPy3 are presented in Table 4.
The XRD patterns of (a) PPy, (b) PA-PPy1, (c) PA-PPy2, and (d) PA-PPy3.
XRD pattern of crystalline grain size of MnPA, MnPA-PPy1, MnPA-PPy2, and MnPA-PPy3.

XRD: X-ray diffraction; MnPA: manganese-containing polyacid; MnPA-PPy: manganese-containing polyacid-doped polypyrrole.

The XRD patterns of (a) MnPA (b) MnPA-PPy1, (c) MnPA-PPy2, and (d) MnPA-PPy3.
XRD pattern of crystalline grain size of PPy, PA-PPy1, PA-PPy2, and PA-PPy3.

XRD: X-ray diffraction; PPy: polypyrrole; PA-PPy: polyacid-doped polypyrrole.
Conductivity studies
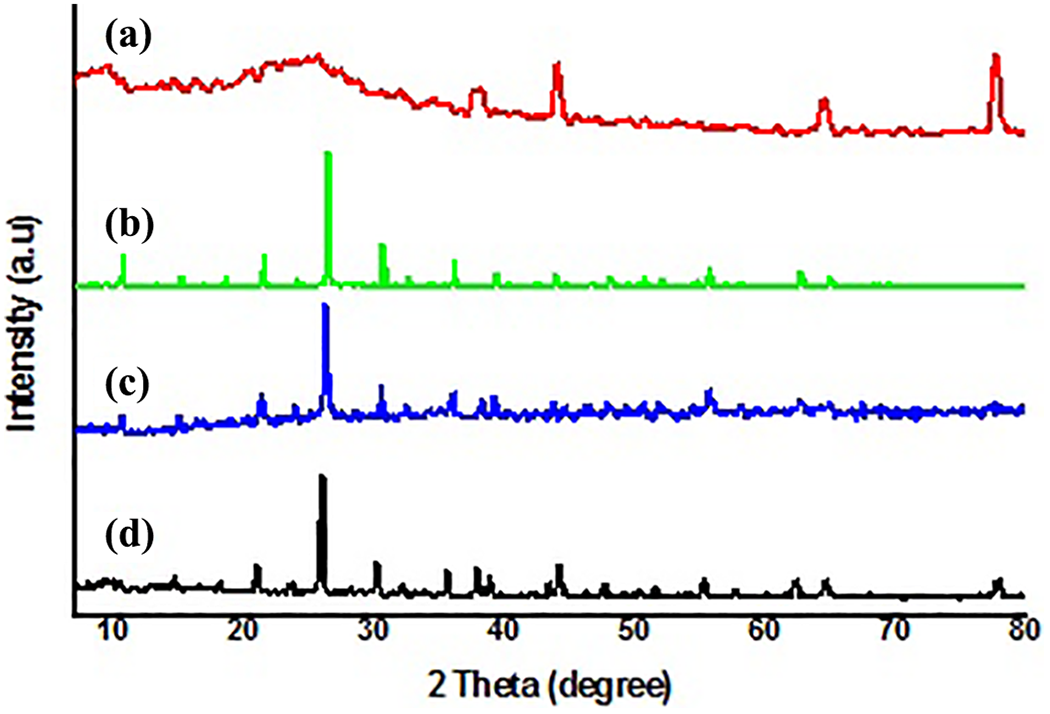
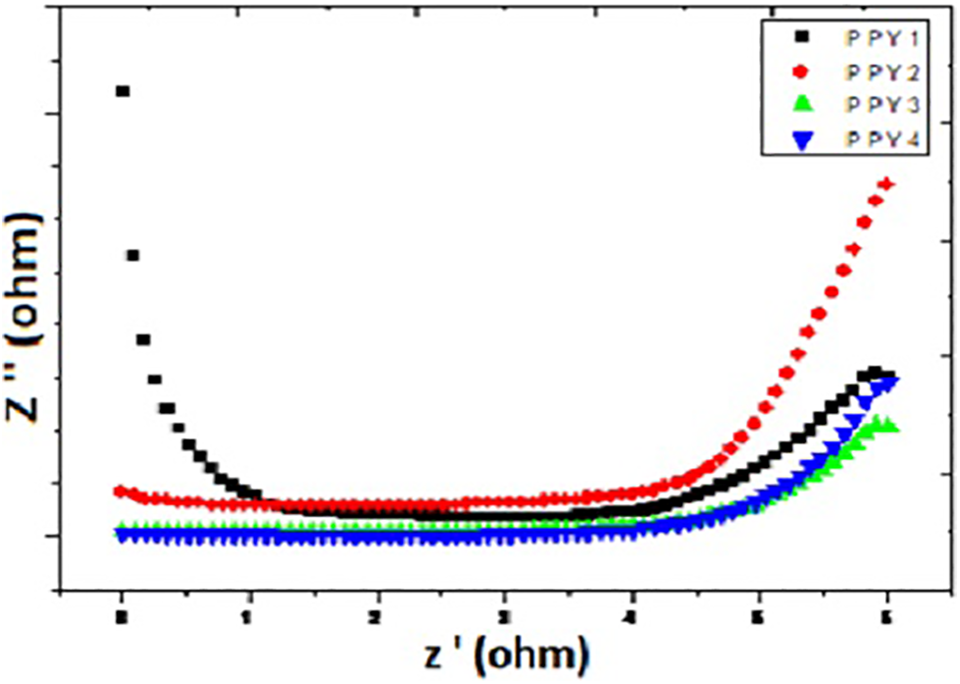
The impedance spectra of PA, PPy, PA-PPy1, PA-PPy2, and PA-PPy3 are shown in Figure 7. The impedance data of PPy, PA-PPy, and MnPA-PPy are given in Table 5. The higher conductivity is due to the molecular interaction between PA and PPy. The conductivity of PPy is 3.3 × 10−6 S cm−1, whereas PA-PPy has resulted in increased conductivity of 7.12 × 10−5 S cm−1. In the case of MnPA–Ppy, the conductivity found to be 7.8 × 10−4 S cm−1 is shown in Figure 8. In low-frequency region, the conductivity increase may be due to the host–guest interaction. The free electronic charge carriers inside the doped material result in higher conductivity. So, the resistivity within the doped material has decreased which is directly proportional to the conductivity. In this structure, oxygen atoms are projected outwards. The oxygen atom present in the POM has free electrons and hence the POM-doped material exhibits higher conductivity at higher frequency. 32
Conductivity studies of (a) PPy, (b) PA-PPy1, (c) PA-PPy2, and (d) PA-PPy3.
Conductivity studies of PPy, PA-PPy, and MnPA-PPy.
PPy: polypyrrole; PA-PPy: polyacid-doped polypyrrole; MnPA-PPy: manganese-containing polyacid-doped polypyrrole.

Conductivity studies of (a) MnPA (b) MnPA-PPy1, (c) MnPA-PPy2, and (d) MnPA-PPy3.
Antibacterial activity
Antimicrobial activity of the compounds was tested in vitro by the well diffusion method 33 against the bacteria Escherichia coli and Staphylococcus aureus. The zone of inhibition values of the investigated compounds against the bacteria is summarized in Tables 6 and 7. The gram-positive and gram-negative activities of PPy, MnPA, PA-PPy, and MnPA-PPy-doped conducting polymer examined by zone of inhibition (mm) are also shown in Figures 9 and 10 with bar chart. The prepared PPy showed a better antibacterial activity due to the presence of the NH group and aromatic ring, thus increasing its partially cationic character. Consequently, the positive charge was strengthened, leading to a better antibacterial activity. 34 PPy showed higher antibacterial activity against E. coli and S. aureus gram-positive bacteria. It causes inhibition zone diameter of ±16 mm and ±18 mm for E. coli and S. aureus, respectively. Since POMs display antibacterial activity toward both gram-negative and gram-positive bacteria, 35 the antimicrobial activities of our PPy were tested against E. coli and compared with that of the corresponding PA in aqueous solution (free PA) to investigate how PA encapsulation affected their antibacterial activity. None of the free PA showed any antibacterial activity up to a concentration of 0.5 mg mL with E. coli. A difference between the antimicrobial activity of the doped material and free PA was observed in the case of PPy. While the free PA showed decreased antimicrobial activity up to a concentration of 0.5 mg mL, polymer based on the corresponding PA presented a significant antibacterial effect; especially those containing PPY, which produced a decrease of 80% viability in the case of PA-PPy.
Antimicrobial activity of PPy, PA-PPy1, PA-PPy2, and PA-PPy3.

PPy: polypyrrole; PA-PPy: polyacid-doped polypyrrole.
Antimicrobial activity of MnPA, MnPA-PPy1, MnPA-PPy2, and MnPA-PPy3.

MnPA: manganese-containing polyacid; MnPA-PPy: manganese-containing polyacid-doped polypyrrole.
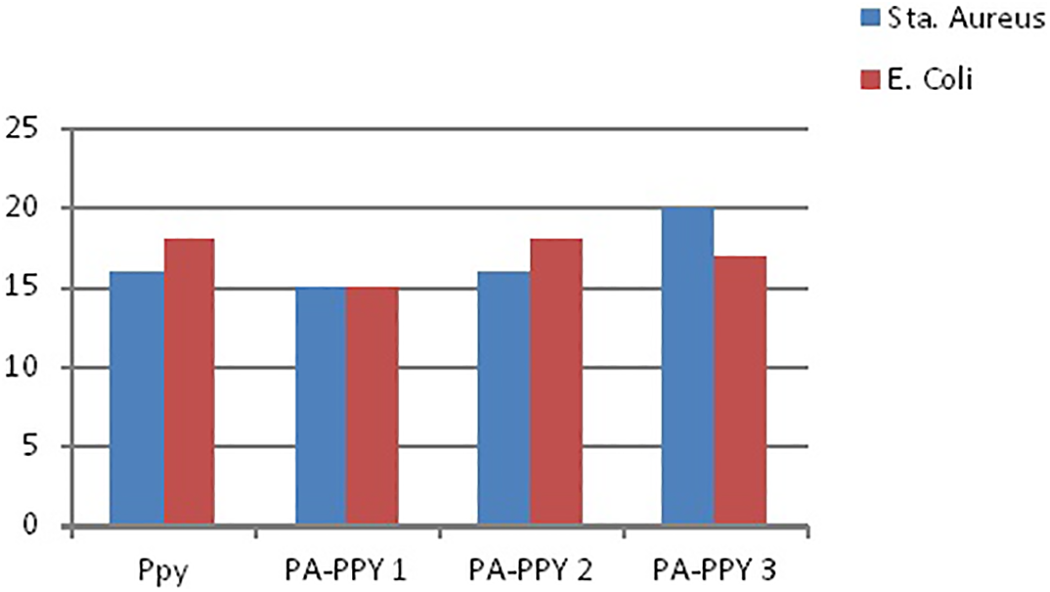
Antimicrobial activity of (a) PPy, (b) PA-PPy1, (c) PA-PPy2, and (d) PA-PPy3.

Antimicrobial activity of (a) MnPA (b) MnPA-PPy1, (c) MnPA-PPy2, and (d) MnPA-PPy3.
Conclusion
PA-PPy is synthesized by different ratios of PA in the PPy matrix to study their electrical and antibacterial activities. Characterization of the doped compounds has been carried out using IR, SEM, and XRD techniques. The IR spectral data show that the characteristic splitting of peaks which are due to the formation of host–guest compound. The SEM morphology of PA-PPy shows square-shaped structure and MnPA-PPy shows granular structure. The XRD studies confirm the crystallite size of PA-PPy and MnPA-PPy. The conductivity was studied in frequency, the free electronic charge carriers inside the doped material result in higher conductivity. The composites exhibit effective antimicrobial effect; especially those containing PPY, which produced a decrease of 80% viability.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported from the Don Bosco Research Grant, Sacred Heart College (Autonomous), Tirupattur, is greatly appreciated.
