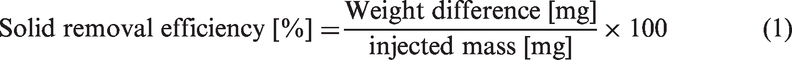Abstract
As development of industrialization grows constantly, the purification of hazardous solid particles and ions is one of the most important topics in environment and ecosystem. In this report, we designed and developed a novel and advanced type of filter media for the removal of both solid particles and hazardous ions, we choose Cs+ and Ca2+ here, by enclosing zeolite in wet-laid nonwoven media. The performance of the prepared filter media was evaluated by continuous sorption experiments, which were followed by solid particles and ion-removal efficiency studies. The prepared filter media showed excellent uniformity. The prepared filter media exhibited a solid removal efficiency rate which ranged from 80 to 82%, and the initial removal efficiency of ions exceeded 99%. These values are in effect as the main layer in the completed liquid filter media and for the next step to prepare the completed multi-layered liquid filter units. The results here suggest that this novel filter media can be used in high-efficiency and multi-functional liquid filter units for residential and industrial engineering.
Introduction
Water purification is a crucial aspect in all parts of the world, and it can have considerable and direct effects on humans. The primary danger in the food chain is water; hence, removing very hazardous materials from water, such as solid particles, hazard ions from natural sources, and cesium ions from nuclear reactors, for instance, is highly desirable. Furthermore, the heavy metallic pollution in the water ecosystems mainly from the discharge of industrial (artificial) sewage has become a severe issue because of the increasing damage to humanity and other living system [1,2]. On the other hand, the major issue is fouling in machinery operation and it occurs due to the adsorption, accumulation, and growth of various foulants in the inner surface pipeline, which eventually deteriorates the machine quality and efficiency [3,4]. Thus far, the removal of extremely hazardous ions has remained a complex problem, so many research groups are trying to use facile method [5–11]. The methods by which water can be purified include filtration, settling, and microbial treatments (self-purification of water) and others [5–7]. Filtration is the most common form due to its high efficiency [5,8,9]. At present, reverse osmotic (R/O) membranes have become a complete system for water purification [10,11]. However, the loss of high water pressures (lower mechanical efficiency), the higher cost, the creation of wastewater, and the need for an additional system, to make higher pressure and recover waste water [12], are negative points. Therefore, simple and high-capacity filtration systems have been a focus of the water-treatment industry as an alternate to R/O membranes. Wet-laid system, a type of non-woven media, can be an alternative to R/O membranes due to their low cost, ease of fabrication, controllable pore structure, and suitability for a wide variety of applications [12–14]. However, simply using wet-laid media, based on short cut fibers without ionic exchanger, does not guarantee any ionic functional properties, implying that these systems can capture only particle-phase materials using their pore structures but not ionic hazards [14].
Zeolite is a unique inorganic ion exchanger and is thus widely used in gas and water treatments [15]. Zeolite is a type of hydrated aluminosilicate of alkali and alkaline earth metals so, SiO4 and AlO4 are the primary building units, forming the framework [16]. The presence of aluminum results in a negatively charged framework, which is compensated for by cations, mostly sodium ions. Cationic radioisotopes such as positive ions can easily adsorb onto zeolite by replacing sodium ions from the crystals, thus imparting significant ion-exchange properties to zeolites. Recently, many efforts have been made to develop novel ion exchangers for hazardous ion adsorption [17,18–21]; however, applying a filter to a liquid purification system is very rare. A spread method of using zeolite in contaminated water has not been developed in solution form due to the small particle sizes and the need to collect the materials again. The collection of the adsorbent after the purification of ionic contaminants is another troublesome step. Additionally, high pressure is required for conventional column type purification systems, because of unwanted self-aggregation of the adsorbent materials. Such highly agglomerated active material causes flow channel blockage, hence low pressure operable filtration systems are highly demanding. Therefore, the use of a filtration system is a novel approach due to its pore structure, efficiency, ease of use, and safety.
The wet-filtration is classified by surface filtration, depth filtration, and cake formation [14]. Surface filtration means that all solid particles are bigger than the pores of filter media, which are captured on the filter surface. Depth filtration system can capture solid particles that are small to be sieved out as in flat filtration. Solid particles penetrate into the fibrous structure media. Filtrated particles are captured in terms of the filtration mechanism. This type of filtration phenomena is important for most of the filtration systems. The cake formation is formed by substances that are retained on a filter media. The cake grows in the course of filtration and becomes thicker as particulate matter is retained.
Many researchers have studied the zeolite-based hybrid adsorptive nonwoven webs to make the ideal structure and performance such as zeolite-coated fibers [23,24], hollow zeolite fiber [25,26], zeolitic wood structures [27], analcime zeolite fiber by hydrothermal reaction [28], and zeolite-based electrospun [29,30]. However, long processing time, complex fabrication, low stability, and high cost are the limitations for the commercial applicability of such previous methods at the market scale.
Wet-laid nonwoven media are capable of uniform immobilization of adsorbent particles sized 1 to 80 microns [31]. When using fibrillated fibers, more adsorbent particles can be immobilized due to mechanical interlocking action of fibrillated fibers [31,32]. The most competitive advantage of the wet-laid process is the absence of adhesives or melt binding compared to the previous process, resulting in ideal (not shielded) pore structure of the additive adsorbents [31]. Unit per hour tends to be as short as that of papermaking processes and the absence of various chemicals makes it environment friendly [33].
Herein, a facile wet-laid technique was developed for efficient removal of solid particles and heavy metal ions from waste water, using zeolite and short-cut fibers composite. The prepared media was characterized based on their structure and evaluated based on the elimination of contaminants. The prepared media was found to be highly stable and showed good removal capabilities in water. This represented the first attempt, short-cut fiber and zeolite composite using wet-laid processing for the liquid filtration system, and the results suggested that the proposed wet-laid filter media can be used in all liquid filtration systems, increasing their water-treatment performance without compromising the quality of the water.
Materials and methods
Chemicals and materials
Zeolite (powder type, Ca2+ ion exchange capacity; 311 mg CaCO3/g) was purchased from Taesung Environmental Research Institute, ROK. Glass staple fiber (0.3, 0.6 µm × 2 mm) and low-melting-point PET fiber (LM PET, 1.1 De × 5 mm, low melt at 150℃) were purchased from Clean and Science Co., ROK. Cesium chloride (CsCl, 99.9%) and calcium chloride dihydrate (CaCl2·H2O, 95%) were prepared from Sigma-Aldrich Co. Standard fine solid particles (ISO 12103-1 A2) was purchased from Jinsol Dust Co., ROK.
Preparation of the zeolite-based wet-laid filter media
The zeolite-based filter media were prepared using a wet-laid method, as noted in the literature [34]. Glass staple fiber 0.45 g (0.1 g of 0.3 µm and 0.35 g of 0.6 µm), LM-PET fiber 0.05 g, and zeolite 0.5 g were placed in a PTFE-covered vessel containing 1000 mL of H2O which was then dispersed in a solution under stirring at 5000 r/min for 10 min. After stirring, 4000 mL of H2O was added to the vessel under continuous stirring at same r/min and time. Subsequently, the prepared slurry was removed from the vessel and then transferred to a lab-scale wet-laid system (Figure 1). The glass staples were allowed to settle (1 bottle = 100 g/m2), and the supernatant was removed from the wet-laid system (100 cm2 scaled). The mixed precursor was then placed on commercial filter paper on a 5-mm diameter holed PTFE plate indiscriminately. After drying below 70℃ for overnight, highly uniform zeolite-based non-woven filter media was produced. The filter media were prepared in three weight types of 100, 200, and 300 g/m2. To improve the mechanical strength and uniformity, the non-woven fabric was calendared (pressed between cylinders) under 1.0 kg/cm2 of pressure at 120℃. In this sequence, LM-PET play an important role in wet-laid nonwoven as binding materials for increasing their mechanical strength and binding between zeolite and glass staple fiber. Before experiment, to remove the moisture, all samples were allowed to air dry for at least 30 min at 70℃ in forced convection oven. Above recipe of optimized filter media, with 50 wt% zeolite concentration revealed outstanding stability and performance, however, exceeding this limit, stability of wet-laid filter media was compromised. Hence, filter media with such high concentration of zeolite is not recommendable.
Schematic illustration of the lab-scale wet-laid system (left) and an actual photograph of the system (right).
Filtration condition and studies
Figure 2 shows a schematic layout of the filtration system containing assemblies with the prepared filter media. This system consists of a constant-delivery pump (JIHPUMP-153Yx), a flexible tube (JIHPUMP-17#), a filter jig (Advantec-47 mm), and a glass flask. The active area of the media has a diameter of 47 mm. Contaminated water samples were prepared with Cs+ at 10 PPM, CaCl2 at 500 mg/L (modified standard KS I 3206), and ISO 12103-1 A2 medium solid particles at an amount of 0.1 g/1 L (modified RS K 0004). The particle-removal efficiency was calculated by comparing the weight before and after supplying 1 L of contaminated water (equation (1)). To validate the efficiency, filtered water was measured by solid concentrations using liquid boil off at 70℃ for overnight. Five units in total were tested and averaged with CV% respectively. This system is designed to supply contaminated water using a constant-delivery pump and supply it uniformly to the media mounted on the jig. The flow rate is 10 mL/min and check line efficiency at 10, 30, and 50 min (100, 300 and 500 mL). The ion concentration was measured using ICP-MS (for Cs+) and AES (for Ca2+). Using equation (2), ion removing efficiency was calculated. Batch experimental data are often difficult to apply directly to the fixed bed sorption column because isotherms are unable to give accurate data for scale-up since a flow in the stack is not at equilibrium. Fixed stack sorption experiments were carried out to study the removing efficiency. The fixed stack (100, 200, and 300 g/m2) operation allows more efficient utilization of the removing efficiency than the batch process. The shape of the breakthrough curve and the time for the breakthrough appearance are the predominant factors for determining the operation and the dynamic response of the wet-laid stack. The general position of the breakthrough curve along the volume/time axis depends on the capacity of the column with respect to bed height, the feed concentration, and flow rate. So, prepared system can replace the bed height to wet-laid media thickness (depends on their weight) [35–37].
Schematic illustration of the filtration test system with the wet-laid filter media.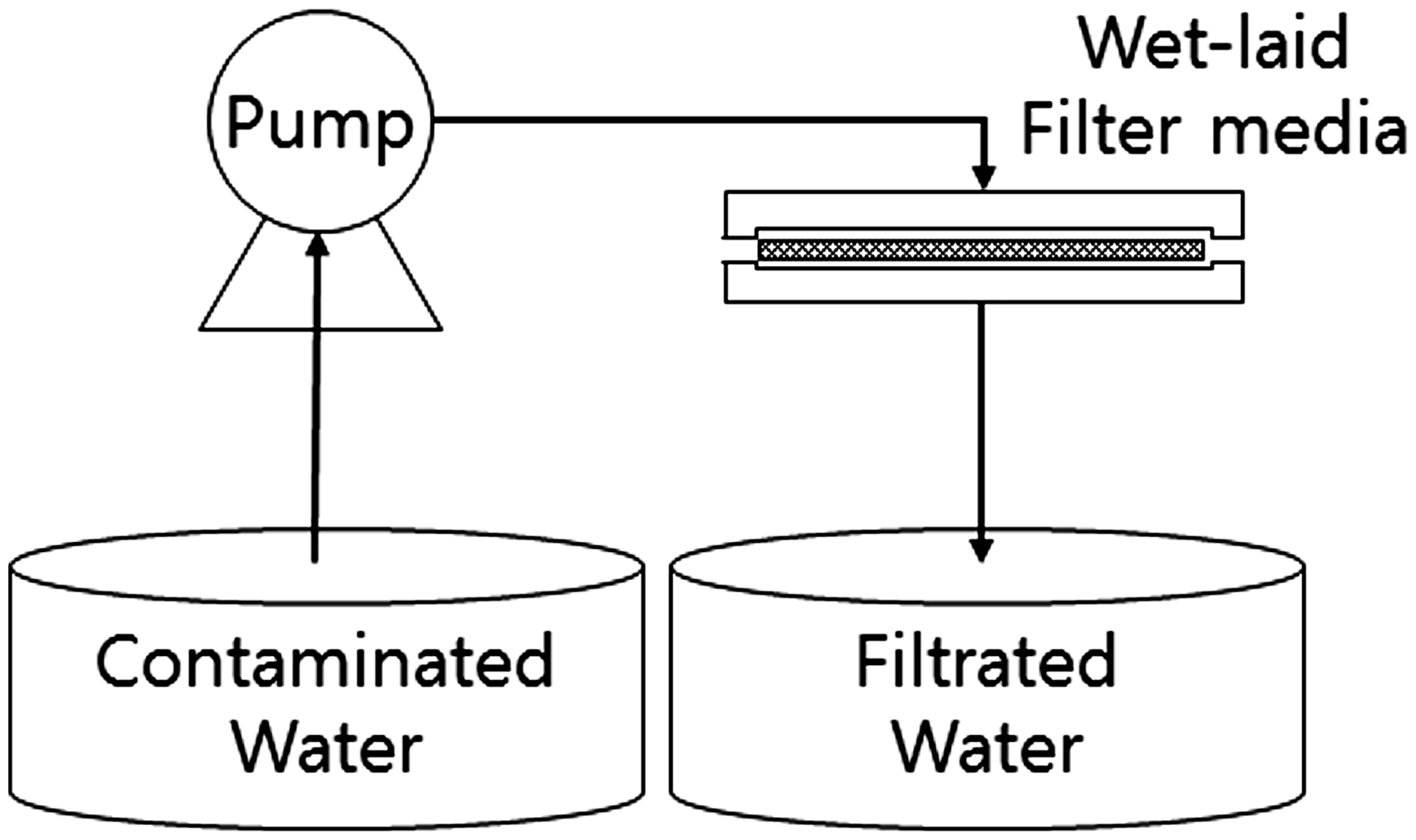
Other characterization
The morphology and structure of the materials were investigated by field emission-scanning electron microscopy (FE-SEM, JEOL JSM-6700F, 15 kV accelerating voltage). All particles were analyzed and calculated by LS particle size analyzer (Beckman Coulter LS 13 320, dry powder mode, 0.375 to 2,000 µm). The elements in detail were investigated by energy-dispersive spectroscopy (EDS, Oxford-6587 with Window-ATW2, all elements analyzed mode, 0 to 10 KeV). The weight and thickness were measured following suitable test standard of KS K 0514 (standard analytical balance of WBA-220) and ASTM D645 (vernier calipers of Mitutoyo 547-401). Air permeability was measured by FX3300 (Textest Co.) at 125 Pa (ASTM D 737). Porosity and pore size were measured using an AUTOPORE V through Mercury porosimetry (Micromeritics Co.) and a CFP-1200-AEL through capillary flow porometry (PMI Co.).
Results and discussion
Physical characterization of the filter media
An SEM image of the zeolite is shown in Figure 3, where the morphology of the crystals is clearly visible. The shape of the given zeolite has an angulated structure. The sizes of the zeolite particles are listed in Table 1 and illustrated in Figure 4 with statistical analysis. Given zeolite power show average 4.287 µm with 59.1 C.V.%. To check the elements in detail, the energy dispersive spectrometry results of the prepared zeolite are listed in Table 2, mainly showing oxygen, sodium, aluminium, and silicon. This typical combination of elements confirms that the prepared particles are zeolite.
Field emission-scanning electron microscopy (FE-SEM) images of the prepared zeolite powder (a) and a corresponding single crystal (b). Particle size distribution of the prepared zeolite particles. Particle size of the prepared zeolite powder with statistical analysis results. Results of energy dispersive spectrometry of the prepared zeolite.



Figure 5 shows an FE-SEM image of the prepared wet-laid filter media. Basically, the media are highly porous and have a completely interconnected 3D network structure. Figure 5(a) is a control sample (without zeolite) and (b) is zeolite-based wet-laid media. As a result of the image analysis, it was confirmed that the filter media observed the contain fiber and zeolite are combined, with the zeolite uniformly distributed and located in the pore of the prepared media and apparently bonded together well. On the other hand, the zeolites were collected by the fiber and pore structure of the wet-laid filter media. Usually the zeolites (particles) and glass fibers (hairs) are attached to the LM-PET (rod) due to the sticky-type surface of LM-PET.
Field emission-scanning electron microscopy (FE-SEM) images of the prepared wet-laid media; glass (hairs) and LM-PET (rods) composite (a) and zeolite (particles) added composite (b).
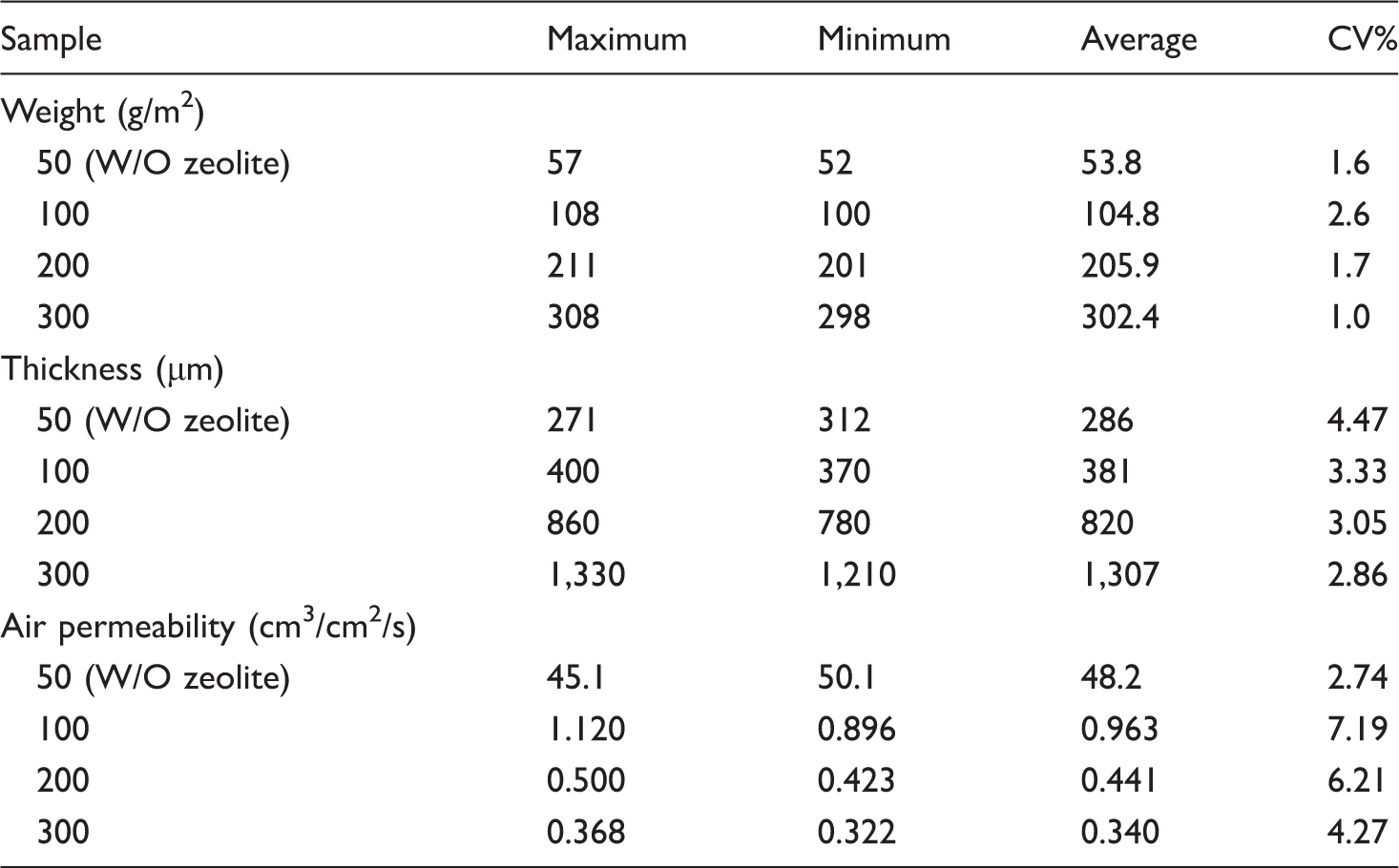
Weight, thickness, and air permeability of the prepared wet-laid media with statistical analysis.
Impurities from a liquid fluid can be removed by using particle and microfiltration by passing liquid through a porous media. In a typical microfiltration filter media, pore size ranges from 0.1 to 10 microns. It is not required to apply additional pressure in filtration system [14]. The pore sizes of the prepared wet-laid media were measured and the results are listed in Table 4 and Figure 6. The mean pore size of each product is as follows. The 50 g/m2 without zeolite sample has mean pore size of 20.2289 µm. The 100, 200, and 300 g/m2 with zeolite sample has a mean pore size of 1.6934, 2.8288, and 3.0356 µm. In terms of the diameter at the maximum pore size distribution, the 50 g/m2 without zeolite sample shows a value of 18.9714 µm. The 100, 200, and 300 g/m2 with zeolite sample shows a value of 1.6789, 1.4642, and 1.3360 µm. Porosity shows same trend from 59.5171% of 100 g/m2 to 76.7977% of 300 g/m2. Essentially required pore size and porosity was achieved by optimum addition (50 wt%) of zeolite in glass filter media. Direct relationship of porosity and sample weight is clear from Table 3 and 4. Such increase of porosity is induced due to the densification limit of calendaring pressure (1.0 kg/m2 of calendaring for all samples).
Pore-size distribution of the prepared wet-laid media. Pore sizes and porosity of the prepared wet-laid media.

Filtration performance of the filter media
Stability study in liquid flow
In order to prove the stability of the prepared filter media under liquid flow, the prepared filter media was subjected to water flow (1,000 mL). After the liquid is passed, wet-laid media show no weight differences and loss of zeolite and short-cut fibers were never found after centrifugation in purified water (Figure 7). By the ICP test of filtration, no Al and Si concentration were found, as shown in Table 7. That is the basis for the stability that zeolite and glass fibers cannot be detected in the fluid phase. Used sheet show similar external structure compared to original sheet.
D.I. water passed by wet-laid media (a) and their purified and centrifuged water (b). Single particle size of the prepared standard solid particles with statistical analysis results. Solid-removal efficiency of the prepared wet-laid filter media. ICP analysis of cesium and calcium ion removal efficiency and aluminum and silicon stability with various samples.



Solid particle removal efficiency study
A preliminary comparative solid particle removal efficiency investigation using different weights was conducted. The sizes of the A2 fine solid particles are shown in Figure 8 and listed in Table 5 with statistical analysis and pictured in Figure 9. Figure 10 shows images of the particles-loading assessment both before and after. An amount of approximately 100 mg of solid particles was added to 1000 mL of pure water. The solid particle removal efficiency was then calculated, as listed in Table 6. Zeolite-based samples showed an efficiency range of 80 to 82% and pure glass media show the efficiency from 63.15 to 67.83%. To discover their phenomena of filtration, surface and cross image of filter media and surface image of supported media (cellulose based 0.4 micron pore size and 0.5 µm absolute grade) were achieved (Figure 11). According to the Figure 11, tested pure glass media show depth filtration phenomena due to the penetrated zeolite particles in the structure, also its supported media show ultra-fine particles in their surface. It means ultra-fine particles cannot capture and collect using pure glass media due to their bigger pore structure. Normally, particle filtration is the separation of particles having size above 10 microns. These can be filtrated out easily without any usage of micro-porous membrane [14]. Solid particles were separated on the surface of tested zeolite filter media, in the form of surface cake (filtrate) due to the microfiltration phenomena. Filtrated liquid samples were measured by the solid concentration to prove their balance exactly. Every samples show the concentration about 20 ± 1 mg/L. This result might be that purified liquid includes under 0.4 µm-sized particles and this amount is about 20%. Prepared composite media show equal purified performance with 0.5 µm grade absolute filter media. The prepared media acts as a filter that allows water to flow through it, removing suspended solid particles and other substances [14].
Field emission-scanning electron microscopy (FE-SEM) image of the prepared A2 fine solid particles (5000×). Size distribution of A2 fine solid particles. Original image of the solid-removing tested wet-laid media (left) and supported absolute filter media (right), (a) control, (b) 100, (c) 200, and (d) 300 g/m2. Field emission-scanning electron microscopy (FE-SEM) image of the solid-removing tested wet-laid media, (a and b) W/O zeolite media’s surface and cross cut, (c) control media’s supported media, (d and e) zeolite media’s surface and cross cut, and (f) zeolite media’s supported media.



Ion-removal efficiency study
In this study, contaminated water samples with Cs+ and Ca2+ were prepared without solid particles individually and injected into the filter media at a rate of 10 mL/min. The results of the ion-removal efficiency with respect to the passing time are shown in Table 7. To prove the role of zeolite in ion-removal, basic filter without zeolite was prepared and compared. Pure glass filter media, no zeolite, shows zero efficiency. Zeolite in the wet-laid media plays a significant role in ion sorption. In this case, the area between the contaminated water and the zeolite contributes to the ion-removal efficiency. For the cesium ion sorption assessment, the initial concentration was about 10 PPM. The more the weight increased, the greater the removal efficiency of ions became due to the increased ion exchanger. Every 100 mL level showed nearly an absolute grade of the removal efficiency, but in the cases of 300 mL at 100 g/m2 and 500 mL at 200 g/m2, sharp declines in the efficiency were noted. For calcium ion sorption, the initial concentration was close to 130 PPM. In this case, 100 mL at 200 and 300 g/m2 showed nearly an absolute grade, but 100 mL at 100 g/m2 showed a slightly decreased efficiency rate. As the weight of the media increases, the efficiency improves. This is attributed to the similar adsorption principle relative to that in previous work. However, the efficiency here was much lower than that of cesium, which appears to be due to the difference in the concentration of the contaminated water. This is due to the fact that a high metal ion concentration easily saturates the column bed, thereby decreasing the breakthrough time. The main driving force for the sorption process is the concentration difference between the metal ion in the solution and in the sorbent [15,44]. To check desorption stable by lean water, tested filter media, shown saturated and 0% of removing efficiency, was dipped in D.I. water and rested for 1 h. After an hour, treated water showed no concentration of Cs+ and Ca2+.
Conclusion and outlook
In conclusion, highly uniform, efficient, and easily fabricated zeolite-based media was successfully prepared through a scalable wet-laid process, commonly known in the non-woven textile industry. This novel media was used as filter media for solid particles and hazardous ion removal with a lab-scale filtration system. The prepared filter media has shown that it is actually not a depth filtration but a cake filtration and it exhibited initial removing efficiency of solid particles rate which ranged from 80 to 82%, and the initial removal efficiency of ions exceeded 99%. These values are in effect as the main layer in the completed liquid filter media and for the next step to prepare the completed multi-layered liquid filter units. With regard to the solid removal efficiency, the weight of the materials is not a contributing factor, but it contributes to the ion-removal efficiency due to the use of an ion adsorbent, zeolite. This indicates that solid particles can be loaded on the structures due to the cake filtration, but ions were not included in it. If it is possible to prepare zeolite with a higher ionic capacity or with an alternative having deactivated in liquid water, new ion exchanger, more useful information can be provided in relation to this study. Conclusively, proposed composite structure can pave a way for future industrially scalable, low cost, and highly efficient filter media structures for wastewater treatment systems.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Trade, Industry and Energy of the Republic of Korea (10067689).