Abstract
In this study, an attempt was made to enhance the oil sorption capacity of nettle fibers by grafting of butyl acrylate. Box–Behnken experimental design was used to study the effect of parameters such as reaction time, reaction temperature and monomer concentration-to-fiber ratio on graft add-on (%) and oil sorption capacity. At 4-h reaction time, 70℃ temperature and 2% monomer concentration-to-fiber ratio, highest graft add-on (%) and oil sorption were attained. Maximum oil sorption capacity of grafted nettle was 36.60 g/g and 25.56 g/g against crude oil and vegetable oil, respectively. Grafted nettle fibers were also subjected to characterization by Fourier-transform infrared spectroscopy, scanning electron microscopy and contact angle tests. Reusability test results showed that grafted nettle exhibited better oil sorption capacity than unmodified nettle even after seven sorption–desorption cycles. It is also observed that the oil sorption capacity of grafted nettle was higher than that of commercial polypropylene material. Based on these results, it is concluded that functionalized nettle prepared by grafting technique can be a potential material for oil spill treatment.
Introduction
One of the naturally available substances is oil. It has been a component of the natural surroundings for million years. Once oil is spilled on sea, it extends for kilometers around and causes major environmental harms. So, it is necessary to clean the oil spill immediately. Oil spill cleanup products normally used to remove the oil from water include dispersants, skimmers, gelling agents and sorbents. Among different products, sorbent materials are generally considered as the most efficient for cleanup of spilled oil due to its uniqueness [1].
Sorbents may be classified into three different materials such as synthetic organic polymers, inorganic mineral materials and organic natural materials. Polypropylene, polyacrylate and polyethylene are popular synthetic organic sorbent materials used for oil spill removal. Examples of inorganic mineral materials include graphite, silica, fly ash organic clay, zeolites, vermiculite and perlite. Organic natural sorbents include different fibrous materials such as cotton, kapok, milkweed, cattail, jute, coir, cotton, kapok, banana, nettle, luffa, etc. [2]. Sorbents prepared from fibrous webs contain small pores to assist the passage of liquids into the sorbent materials and hold the liquids subsequent to sorption [3].
The ideal oil sorbents are likely to possess the features such as (i) high sorption capacity, (ii) hydrophobicity, (iii) high sorption rate, (iv) high retention capacity, (v) mechanical properties, (vi) reusability and (vii) sustainability and biodegradability. However, it is still a challenge to get ideal oil sorbents that could meet all the above criteria [4].
Nettle is a cellulosic plant fiber that exists in tropical wasteland areas around the globe. It grows on rich soils and up to 1.20 m high [5]. Several researchers studied the characteristic features of nettle fibers for textile and technical textile product development. One-hundred percent of natural sustainable fabrics for apparel applications were developed from nettle, nettle–cotton, nettle–ramie, nettle–wool and nettle–flax combinations. Summer apparels and winter apparels with closed and open weave structures were also developed by some researchers [6]. Nettle fibers were also used to develop fashionable and luxurious products, namely wall hangings, hand bags, door chain, flower vase, decorative items for rooms, etc. There is enormous potential to develop different technical textile products using nettle fibers [7–9].
In recent years, considering the advantages of natural fibers, including low cost, biodegradability and reusability, the fibers such as kapok [4,10], cattail [11], cotton [12], banana [13], nettle [7–9], milk weed [14], jute [15], coir [16], poplar seed fiber [17], chitosan [18] and other various vegetable fibers have been investigated for the oil spill cleanup applications. It was also recommended that natural fibers of low cellulosic content cannot be simply degraded by cellulose degrading microbes [4]. Even though natural fibrous sorbents are cost-effective than most synthetic sorbents, one of the main drawbacks associated with these materials is their low oil sorption capacity. Several approaches such as acetylation [19], grafting with oleic acid [20], thermal carbonization [21], coating with poly-n-butylmethacrylate (PBMA)/SiO2 [22], thermal bonding [23] and polymerization [24] are normally employed to improve the oil sorption capacity of natural fibers. Amongst the abovementioned methods, polymerization is a simple and effective method to graft monomers onto fiber surface [24]. This method has been used by researchers for different materials such as polypropylene [24], banana [13] and coir [16]. Previous studies showed that the maximum oil sorption capacity of grafted coir is only 13.45 g/g [16] and grafted banana is only 14.45 g/g [13].
It is also noted that no extensive work has been carried out with respect to the grafting of nettle fiber and its influence on graft add-on (%) and oil sorption behavior. In this study, nettle fibers are grafted with butyl acrylate monomer and Box–Behnken experimental design has been used to study the effect of various preparation parameters such as time, temperature and monomer concentration (%) on graft add-on (%) and oil sorption. The aim of this study was to enhance the oil sorption capacity of nettle fiber through the surface grafting and evaluate the effects of preparation variables on the oil sorption and characterization.
Materials and methods
Characteristics of nettle fiber.

Properties of oils.

Graft copolymerization of butyl acrylate on to nettle fiber
As reported earlier, the grafting reaction was performed in a three-neck round bottom flask [16].
In order to control the reaction temperature, the flask was positioned in a thermo stated heating mantle. The required weight of nettle fibers and butyl acrylate monomer was placed in the flask and agitated for 10 min. Subsequently, cross-linking agent N,N-methylene-bis-acrylamide and initiator potassium persulfate were added to the flask. The reaction was performed under nitrogen atmosphere and the mixture was agitated continuously as per the experiment conditions. Homopolymer generated through the graft-copolymerization reaction was separated out from the grafted copolymer by stirring for 12 h in ethyl alcohol for entire elimination of homopolymer [16]. The nettle fiber samples were dried in oven at 50℃ and weighed.
Graft add-on (%) is calculated as follows

Measurement of oil sorption capacity
Measurement of oil sorption capacity of raw and grafted nettle fibers was carried out in a batch system as per the existing literature. One gram of nettle fiber was immersed into crude oil and vegetable oil for 15 min and then wet samples were taken out and hung in free air for 1 min to drip out the surplus oil absorbed by the samples. Subsequently, the weight (Ww) of wet nettle samples was measured [7]. The oil sorption capacity was calculated using the relationship given below
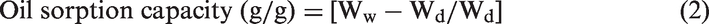
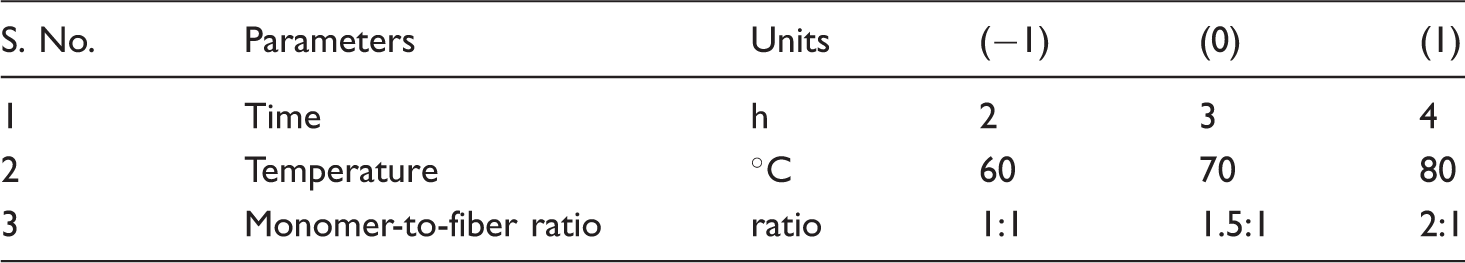
Experimental design
Particulars of process parameters.
Reusability test
As per the method described earlier, reusability of the nettle fibers was carried out [25]. Initially, the oil sorption capacities of nettle fibers were measured and then samples were squeezed between two rollers at a pressure of 10 kgf/cm and loss in weight of the sample was measured. The squeezed fibers were again dipped in the oil for 15 min and oil sorption capacity was measured [25]. The efficiency of nettle fiber reusability was determined by oil sorption capacity of fibers after repeated sorption and desorption cycles.
Fourier-transform infrared spectroscopy
The infrared (IR) spectra of unmodified and grafted nettle fiber samples were recorded using Fourier-transform infrared spectroscopy (FTIR) spectrophotometer (Shimadzu 8400s, Japan) using Attenuated Total Reflection (ATR) sampling technique by recording 45 scans in % T mode in the range of 4000–600 cm−1.
Scanning electron microscopy
Analysis of the morphology of untreated and modified sample was carried out using field emission scanning electron microscope (SWIZZ, Turkey), from PSGTECHS COE INDUTECH, Coimbatore. The nettle fibers were sputter coated with gold layers and figures were recorded using scanning electron microscopy (SEM).
Results and discussion
Table 4 shows the experimental runs of Box–Behnken design on graft add-on (%) and oil sorption of grafted nettle fiber. The surface response curves (Figure 1) were drawn to understand the individual and interaction effect of reaction time, temperature and monomer-to-fiber ratio on graft add-on (%) and oil sorption of nettle fiber using standard statistical software.
(a) Effect of time and temperature on graft add-on (%) on monomer-to-fiber ratio 1:1. (b) Effect of time and temperature on graft add-on (%) on monomer-to-fiber ratio 1.5:1. (c) Effect of time and temperature on graft add-on (%) on monomer-to-fiber ratio 2:1. Experimental runs of Box–Behnken design on graft add-on (%) and oil sorption of grafted nettle fiber.

Effect of preparation parameters on graft add-on (%)
The effects of reaction time and temperature on graft add-on at different monomer concentration-to-fiber ratio 1:1, 1.5:1 and 2:1 are shown in Figure 1(a) to (c), respectively. It is observed from Figure 1(a) that the graft add-on increases with increase in reaction time and temperature. At monomer concentration-to-fiber ratio 1:1, with the increase in reaction time from 2 h to 4 h, the graft add-on increases from 10.74% to 12.37%, respectively. The positive dependence of grafting on time may be attributed to the increase in the number of grafting sites, owing to the higher amount of initiator involving in the arrangement of reactive sites on the backbone of cellulose structure [13]. The rate of reaction also depends on the accessibility of the cellulose which takes part in the reaction. Therefore, on grafting, the modified nettle is expected to provide high graft add-on (%). Our results are in line with the findings of Teli and Valia [13]; they performed grafting of banana fibers for oil spill cleanup applications. Similar trend is observed at monomer concentration-to-fiber ratio 1.5:1 (Figure 1(b)) and 2:1 (Figure 1(c)).
It is also observed from Figure 1(a) that, at 1:1 level of monomer concentration-to-fiber ratio, with the increase in reaction temperature from 60℃ to 80℃, the graft add-on (%) increases from 10.49% to 12.34%, respectively. The increase in graft add-on with temperature is mainly due to the higher rate of initiator dissociation, in addition to the higher diffusion and mobility of the monomer from aqueous bulk phase to cellulosic fiber phase [16]. Our results are correlated with previous published data [13,16]. Similar trend is also observed at 1.5:1 (Figure 1(b)) and 2:1 (Figure 1(c)) ratio levels of monomer-to-fiber ratio.
The effect of monomer concentration and reaction temperature on graft add-on at reaction times 2, 3 and 4 h was also studied. It is observed that the graft add-on (%) increases with increase in monomer concentration and reaction temperature. At 2-h reaction time, with the increase in monomer concentration-to-fiber ratio from 1:1 to 2:1, the graft add-on increases from 10.13% to 12.85%, respectively. The reason for increase in graft add-on may be attributed to the higher availability of the monomer with respect to fixed quantity of fiber in the reaction bath for grafting [13]. Similar findings are observed at 3-h reaction time and 4-h reaction time. Further, it is noted that with the increase in reaction temperature from 60℃ to 80℃, the graft add-on (%) increases. The reason for the same has been explained earlier.
The effect of reaction time and monomer concentration on graft add-on (%) at reaction temperatures 60, 70 and 80℃ is also studied. It is observed that the graft add-on increases with increase in reaction time and monomer concentration at all levels of reaction time and monomer concentration. The reason for the same has been explained in the earlier section.
Characterization
FTIR spectra
The FTIR spectrum of unmodified and grafted nettle is presented in Figure 2. FTIR spectra grafted nettle fibers show a peak at 1735 cm−1; this can be attributed to the presence of carbonyl group (C=O) or ester groups, whereas the peak at 1162 cm−1 can be due to the saturated esters, which were absent in raw fibers, confirming the grafting of butyl acrylate onto the nettle fibers through covalent bonds [26]. These findings clearly indicate that the nettle fiber undergoes modification by means of grafted reaction. The esterification enhances the hydrophobicity and oleophilicity of the grafted nettle fibers.
FTIR spectra of raw and grafted nettle fiber.
SEM analysis
From SEM images (Figure 3(a) and (b)), it can be seen that there is a significant variation in the exterior surface structure of the unmodified and modified nettle fibers. This indicates that the grafting process altered the physical arrangement of the nettle fiber. An undulant and corrugated structure can be seen on the grafted nettle fiber. The undulant and corrugated structure surface increases the surface area of the material, which can prevent absorbed oil escaping from the surface [24]. Heterogeneous grafting of butyl acrylate onto the fiber has made the fiber hydrophobic and oleophilic, and thus in turn makes it a good material for absorbing oil. It may be possible that the high amount of oil can be trapped into the interior porous structure of the fiber (Figure 4) exhibiting the improved oil sorption capacity. The porous interior of the fiber improves the penetration of oil through the fiber and enhances the sorption properties of fiber. Hence, two key factors that synergize in improving the sorption behavior are hydrophobic/oleophilic rough surface arrangement along with the porous nature of interior fiber. Oil is initially adsorbed on undulant and corrugated surface structure and then fills up the porous interior of the sorbent. Accordingly, the sorption mechanism depends on both adsorption and absorption.
SEM images: (a) raw nettle fibers and (b) grafted nettle fibers. Hollow structure of a nettle fiber.

Contact angle measurements
Contact angles of raw and grafted nettle fibers.

Oil sorption behavior of grafted nettle
The effect of graft add-on (%) on oil sorption of nettle fiber is shown in Figure 5. From Figure 5, it is observed that the oil sorption capacity of nettle fiber increases with graft add-on (%) against both crude oil and vegetable oil. This could be due to the hydrophobicity and oleophilicity imparted to the nettle fiber as a result of surface modification with butyl acrylate monomer and ester formation. From Figure 5, it is also observed that the oil sorption capacity of grafted nettle fibers are higher for crude oil than vegetable oil, which might be due to the inherent density difference along with the possible polarity of asphaltenic compounds such as resins, asphaltenes, etc. [27]. Further, the crude oil was more viscous and the rate at which it was coming out from the fibrous structure was low. Microscope images of crude oil and vegetable oil absorbed nettle fiber bundle are shown in Figure 6.
Effect of graft add-on (%) on oil sorption. Oil absorbed nettle fiber bundle: (a) crude oil and (b) vegetable oil.

The f-values and p-values for different response variables.

aValues which are not significant.
The “p” values and “f” values for different response variables are shown in Table 6. From Table 6, it is observed that individual effects of parameters such as time, temperature and monomer concentration on graft add-on (%), crude oil sorption and vegetable oil sorption were found to be significant, whereas the interaction effects such as time vs. temperature, temperature vs. monomer concentration and monomer concentration vs. time were not significant.
Regression equation for graft add-on (%), crude oil and vegetable oil sorption.

Reusability
Reusability is a key indicator for assessing the capacity of oil sorbent material to recover the oil absorbed. Superior recycling performance of oil sorbent material can reduce the price of oil pollution treatment. The oil sorption capacity of grafted nettle fiber for both vegetable oil and crude oil after seven sorption–desorption cycles is illustrated in Figure 7. It can be seen that the oil sorption capacity continuously decreases during seven cycles. The reduction in oil absorbency after each sorption/desorption cycle could be due to the factors such as irreversible deformation of partial fiber structure, the reduction of fiber interspace and the presence of residual oil in fiber bundles [28]. After seven cycles, the losses in oil sorption capacity for vegetable and crude oil are 12.29 and 9.12, respectively. In addition, after seven cycles, the resulting grafted nettle still exhibits better oil sorption capacity than unmodified nettle for both oils. The results indicate that grafted nettle fiber shows excellent reusability capability.
Reusability of grafted nettle fiber.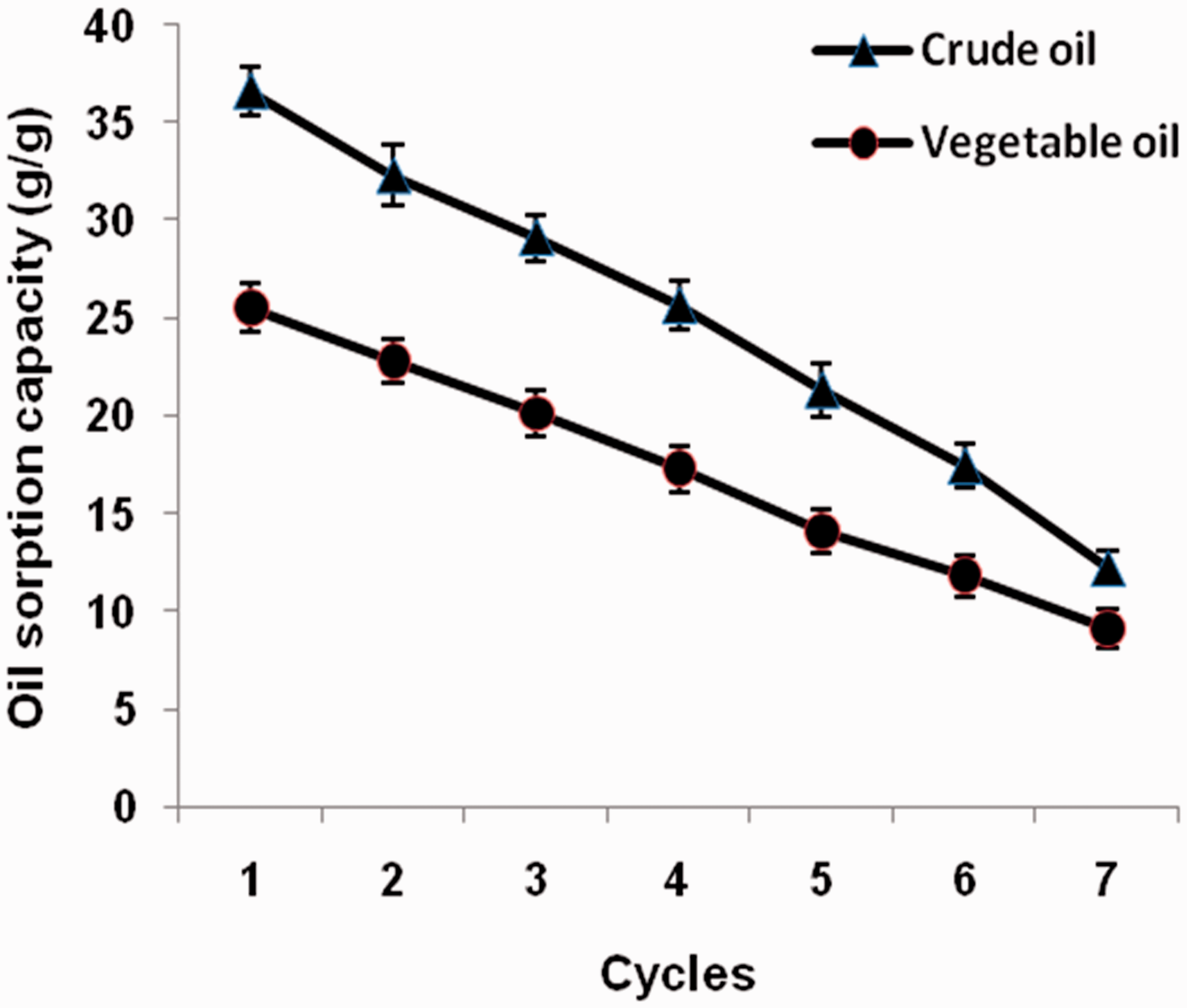
Comparison between nettle and commercial sorbents
Oil sorption capacity of raw nettle, grafted nettle and commercial polypropylene.

Conclusions
The grafting process played a significant role in deciding the oil sorption capacity of nettle fibers. Highest graft add-on (%) and oil sorption were achieved at 4-h reaction time, 70℃ temperature and 2% monomer concentration-to-fiber ratio. Maximum oil sorption capacity of grafted nettle was 36.60 g/g and 25.56 g/g against crude oil and vegetable oil, respectively. Reusability test results showed that grafted nettle exhibited better oil sorption capacity than unmodified nettle even after seven sorption–desorption cycles. It is also noted that the oil sorption capacity of grafted nettle was higher than that of commercial polypropylene material. Based on these results, it is concluded that functionalized nettle prepared by grafting technique could be a potential material for oil spill treatment.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding received from University Grant Commission (UGC), Government of India under Project Number (F.No-43-158/2014) for carrying out this research work is gratefully acknowledged.
