Abstract
Cotton-woven fabric-reinforced polyvinyl alcohol-based hydrogel composite was produced by constructing cotton as warp and polyvinyl alcohol/cotton hybrid and polyvinyl alcohol yarns as weft yarns in the fabric structure. As-prepared polyvinyl alcohol/cotton fabrics were treated with different concentrations of aqueous borax and glutaraldehyde crosslinking solutions. Polyvinyl alcohol molecules were transformed to crosslinked gel structure after the treatments. Since cotton yarns kept their yarn structure in the fabrics, woven fabric-reinforced hydrogel composites were obtained. Chemical analysis to investigate crosslinking was conducted by attenuated total reflection–Fourier transform infrared spectroscopy spectra analysis, and the results revealed that a proper crosslinking of polyvinyl alcohol molecules in polyvinyl alcohol yarns occurred by using both borax and glutaraldehyde as crosslinkers. Thermal stabilities of the samples were observed via thermogravimetric analysis measurements. Even though borax crosslinking increased the thermal stability, glutaraldehyde crosslinking did not have a significant effect on the thermal stability of hydrogel composite. Crystalline microstructural analysis was carried out with X-ray diffraction measurement. Tensile properties of the samples by focusing on the crosslinker ratio and water contents in the hydrogel composites were performed. The results revealed that tensile properties of hydrogel composite tremendously increased with fabric reinforcement. Also, breaking force gradually increased when the hydrogel composite structure released the water from its structure at both borax and glutaraldehyde samples. Since the produced fabric-reinforced hydrogel composites have high strength, they are promising candidates as hygroscopic materials for planting and erosion control at inclined terrains.
Introduction
Hydrogels are biofunctional materials which can absorb and hold high amount of water in comparison to their dried weight. Because of their biocompatible properties and resembling characteristics of the living tissue especially due to their soft and flexible characteristics, hydrogels find very broad biomedical applications such as drug delivery systems, artificial organs, superabsorbents, and contact lenses [1,2]. Hydrogels are molecularly networked structures produced from the crosslinking of either small molecules or polymeric macromolecular structures. When hydrogel structures are sunk in water, they are not dissolved because crosslinked structures hold all molecules together and water molecules are filled and accumulated among cross-linked molecules and hydrogels can retain water more than their dried weight [3]. Polyvinyl alcohol (PVA) is considered a biocompatible polymer and can be crosslinked by different crosslinking agents including borax, glutaraldehyde (GA), and maleic anhydride [4–7]. Preparation and various applications of PVA-based hydrogels by using different crosslinkers have been studied and reported in the literature. High-performance hydrogel actuators were produced by Gao et al. [5] via coating borax crosslinked
PVA/GA hydrogels were produced and their rheological analysis was conducted. The addition of GA to PVA increased the viscosity and displayed Newtonian fluid behavior [8]. Chemically crosslinked PVA/GA hydrogels were obtained with various hydrolysis degree in the nanostructured form [9]. The results revealed that increasing hydrolysis degree led to the increase of crystallinity of hydrogel and chemically crosslinking caused a decrease in the swelling ratio. Therefore, pH-sensitivity and swelling reversibility have made them potentially applicable in biomedical fields.
Mechanical properties of hydrogels are low and cause problems such as tearing, breaking, and bursting during the processing, installation, and usage. In order to enhance the mechanical properties of hydrogels, reinforcement of some structures including nanoparticles, fibers, and fabrics is involved to construct a composite hydrogel structure [10–12]. Fabric-reinforced hydrogel composites are the novel method to produce mechanically enhanced integrated hydrogel structures which have high tensile and tearing strength properties. While hydrogels as the matrix undertake main duty in the composite structure, fabric carries the applied load and protects the structure from the mechanical damages. Few studies have been reported in the literature up to now on fiber and fabric-reinforced hydrogel composite structures [11,13]. Mechanically enhanced polypropylene and ultra-high molecular weight polyethylene fiber-reinforced hydrogel composites were produced and analyzed by Holloway et al. [14] for the preparation and application to heal meniscus damage. Polyester fabric-reinforced hydrogel composites were prepared from hydrolyzed polyester fabric [13]. Hydrogel composite reinforced by woven glass fabric was prepared by Huang et al. [11] with enhanced toughness and tensile properties. Arjmandi et al. [15] studied mechanical and tribological properties of three-dimensional woven fabric-reinforced hydrogel composite from acrylamide and sodium alginate for a potential cartilage replacement material.
Crosslinking of alginate-Ca2+/poly(
In this study, high mechanical strength fabric-reinforced hydrogel composites have been fabricated from specifically designed woven fabric which contains cotton and PVA/cotton hybrid yarns. In this regard, woven fabrics were produced and soaked into previously prepared aqueous borax and GA crosslinking solutions. Effects of the crosslinker ratio on chemical structure, thermal stability, and crystalline microstructures were investigated via attenuated total reflection–Fourier transform infrared spectroscopy (ATR–FTIR), thermogravimetric analysis (TGA), and X-ray diffraction analysis by conducting the tests on the dried samples. Mechanical properties were also investigated by observing the dried samples. Effects of the water content in the composite hydrogel structures on the mechanical properties were observed form both borax and GA sample by selecting the crosslinker ratio. Comparing to the previous studies in the literature, generally, liquid hydrogel precursors were injected into fabric or fabrics were coated these precursor and then gelation was carried out. In this study, precursor of the hydrogel was the PVA fibers in solid state, and these were incorporated in the fabric structure during the weaving process. Then, PVA fibers inside the fabric structure were transformed into hydrogel structure by pouring crosslinker solutions (borax/water or glutaraldehyde/water). So, hydrogel homogenously forms everywhere in the final composite structure.
Material and methods
Raw materials and chemicals
Ne 40/1 PVA yarn was obtained from KURABO Marugone Mill, Japan. Ne60/2 open-end cotton yarn was provided from internal market. Borax was purchased from Galenik Ecza ve Kimyevi Maddeler Deposu (Batch no: 1408H0048). Glutaraldehyde (50 wt %) solution was obtained from Amresco. Distilled water was used in all processes. All raw materials were employed without further purification.
Weaving process
Weaving process was conducted to produce the woven fabric samples [25]. Before woven fabric production, two Ne40/1 PVA yarns were doubled and then twisted at 4850 rev/min spindle speed and 300 t/m twist to form Ne40/2 PVA yarns. These doubled and twisted PVA yarns were further doubled with Ne60/2 cotton yarn and twisted together to form a hybrid yarn. The warp of woven fabrics was prepared using Ne60/2 cotton open-end yarn. A reed number of 12 (dents/cm) was employed in weaving with two ends drawn between two red dents. Hence 24 ends/cm warp density was obtained. The hybrid and PVA yarns were inserted as the weft. A weave repeat of 10 warp and 30 weft yarns were obtained as shown in Figure 1 to hold firmly and cover the cotton warp from both bottom and upper surfaces by hybrid and PVA weft yarns, respectively. Fabric samples were woven by using 10 shafts with the plain draft. As seen from the weave repeat, there are three different weft yarn movements. 1., 4., 7., … PVA weft yarns make one up four downs movement, 2., 5., 8., … hybrid weft yarns make one up and one down while 3., 6., 9., … PVA weft yarns make movements of four ups and one down. In this way, the cotton warp was covered by PVA and hybrid yarns from the top and bottom surfaces and embedded in hydrogel after gelation of PVA yarns. Fabric thus designed was woven at 36 picks/cm weft density and 184.7 g/m2 of weight per square meter. This fabric construction includes around 40% cotton and 60% PVA. Drafting and dobby plans are also seen in Figure 1. Fabrics were woven in a sample weaving machine at 400 mm width. Weave repeat, drafting, and dobby plans and the relation between them as well as schematic view, and a short explanation of the weaving process can be found in references 2 and 3 [25,26].
Weave, drafting, and peg plans of neat polyvinyl alcohol/cotton fabric.
Gelation of PVA in cotton fabric-reinforced hydrogel composite
Borax/water solutions were prepared by magnetic stirring process at 50℃ with different concentrations (0.1, 0.3, 0.5, 1, 1.5, and 2 wt %). For aqueous GA solution preparation, a different volume of GA solutions (0.25, 0.5, 1.5, 2.0, and 3.5 ml) were added into 20 ml of water. One milliliter of HCl was added into each solution. Since borax was in the solid state at the beginning, borax/water solutions were prepared in terms of weight percent. The received GA was in the liquid state, and GA solutions were prepared depending on volume (ml). About 5 cm × 10 cm fabric samples were cut in the warp directions and used to prepare fabric-reinforced hydrogel composite samples. Each fabric sample was put on a petri dish, and the prepared borax/water solutions and GA/water solutions (including HCl as the catalyst) were spilled on the fabric samples to transform PVA yarns into hydrogel form by crosslinking PVA molecules in the fabric structure. PVA is a semi-crystalline polymer, and it always requires some heating for an appropriate dissolution when the polymer was used in the granular form [27]. In this study, since PVA was in the form fibers in the yarn and fabric structure, the surface area of PVA was high and the water molecules were very easily approached to PVA molecules and whole PVA fibers were dissolved quickly just after pouring the water. So, with the existence of crosslinker molecules in the water, PVA yarns in the fabric were simultaneously dissolved and crosslinked leading to transformation into hydrogel structure. The samples were kept at ambient conditions during 24 h for further gelation. Then, the excess borax/water and GA/water solutions were removed, and all the samples were washed with distilled water several times to remove the uncrosslinked borax and GA from the hydrogel composite structure. All the hydrogel composites were dried for the performance tests. The thickness of the neat fabric before the gelation was about 0.796 ± 0.02 mm. When the fabric was treated with the crosslinkers and hold in the water in 90 min, the thickness of the composite hydrogels was about 2.3 mm (borax sample) and 1.6 mm (GA sample). When the samples were dried, the thickness decreased but never be less than the thickness of neat fabric.
Chemical analysis of hydrogel
Chemical analysis of neat PVA and PVA/cotton fabrics and composite hydrogel due to crosslinker ratio have been carried out using ATR–FTIR (Bruker Ifs 66). Because dried cotton yarns and hydrogel were not homogeniously distributed on the testing sample which was mounted to ATR–FTIR tip, there were some intensity differences on the peaks.
Thermal characterization of hydrogel
Thermal properties of fabric-reinforced hydrogel composites in dried form were assessed by measuring weight losses with temperature in air environment using a TG analyzer (TA SDT 650) by heating from 50 to 800℃ (heating rate of 10℃ min−1).
Microstructural analysis of hydrogel
Microstructural characterization of neat PVA/cotton fabrics and their borax/water and GA/water treated and dried counterparts was conducted by using Rigaku Ultima IV X-ray diffractometer (2
Mechanical measurements of fabric-reinforced hydrogel composite
Tensile testing was carried out by using Shimadzu tensile tester with AG-X plus model and load cell capacity of 5000 N. Exactly the same sample size was used after the gelation process without cutting them. Gauge length and testing speed were adjusted to 30 and 200 mm/min, respectively. Tensile tests were performed for dried and swollen fabric-reinforced hydrogel composite. Then the samples were waited in the deionized water for 90 min to obtain the swollen samples. After 90 min, the samples considered that they have 100 wt % of water. All the measurements were conducted at both neat fabrics, and their hydrogel composite analogs in the warp direction.
Result and discussion
Concept study of the fabric-reinforced hydrogel composite
Test samples were prepared by cutting the samples as 5 cm × 10 cm fabric in warp direction from as-produced woven fabric. Schematic illustration of neat PVA/cotton fabric and cotton fabric-reinforced hydrogel composite structure after gelation is demonstrated in Figure 2. As seen from the schematic illustration, the warp was made of cotton yarns, but the weft was inserted with the order of two PVA and one cotton yarns. When the fabric samples were treated with aqueous borax or GA solutions, PVA yarns in the fabric structures dissolved and transformed into gel form. Since cotton yarns did not dissolve, they kept their position in the structure and stayed as the reinforcement material in the as-produced hydrogel composites. High magnification photograph images of neat PVA/cotton fabric (Figure 2(a)) and its aqueous GA (Figure 2(b)) and borax (Figure 2(c)) solution-treated analogs are shown in Figure 2. Mirror images of the hydrogel composite samples were given to show how cotton fabric positioned inside the hydrogel matrix (inset images in Figure 2(b) and (c)). As seen from the images, PVA transformation into gel structure when borax was used as a crosslinker is seen more obviously than GA crosslinking at the proposed condition.
Schematic illustration and high magnification photograph images of (a) neat PVA/cotton fabric construction, (b) GA-treated PVA/cotton fabric, and (c) borax-treated PVA/cotton fabric. Insets are the mirror images of the hydrogel composite samples.
Chemical analysis of hydrogel composite via FTIR
In order to investigate the crosslinking of PVA by borax and GA in PVA/cotton fabric structure, ATR–FTIR characterizations were performed for the neat fabric and their different concentration of borax (Figure 3(a)) and GA (Figure 3(b)) treated fabric analogs. Pure fabric (before water or crosslinker solutions treatments) sample exhibiting a broad peak with the highest peak point around 3308 cm−1 corresponded to OH stretching vibration in PVA or cellulose molecules [28]. In Figure 3(A), the reduction of the OH peak with the addition of borax was one of the evidence of crosslinking occurring via OH groups [29]. The reduction of this peak is not so much since cellulose was not dissolved and mostly OH groups on the cotton fiber surfaces complexed with borax, and OH groups in the fibers were mostly conserved. C–H stretching vibration was detected at 2924 cm−1 [29,30]. The peak seen at 1088 cm−1 corresponded to C–O stretching [31]. The new peak detected at 658 with the addition of borax to the structure could be attributed to stretching vibrations of O–B–O bonds [29,31]. With GA treatment in Figure 3(B), OH groups were consumed, and the intensity of OH peak decreased as a result of the possible formation of acetal bridges when crosslinking was formed [9]. Broad OH peak shifted to higher wavenumber at GA sample compared to borax samples. Also, peak intensity of 1716 cm−1 related to C=O carbonyl stretching comes from GA molecules increased with increasing GA ratio, which implied that GA was not completely reacted with the molecules in the fabric structure, and some residuals of GA have still existed after gelation [32].
FTIR spectra of (a) neat PVA/cotton fabric; and PVA/cotton fabric treated in different (A) wt.% of borax/water solutions: (b) 0.3, (c) 0.5, (d) 1, (e) 1.5, and (f) 2; and (B) GA solution contents in water/HCl solution: (b) 0 (just water/HCl), (c) 0.25, (d) 0.5, (e) 1.5, (f) 2, and (g) 3.5 ml.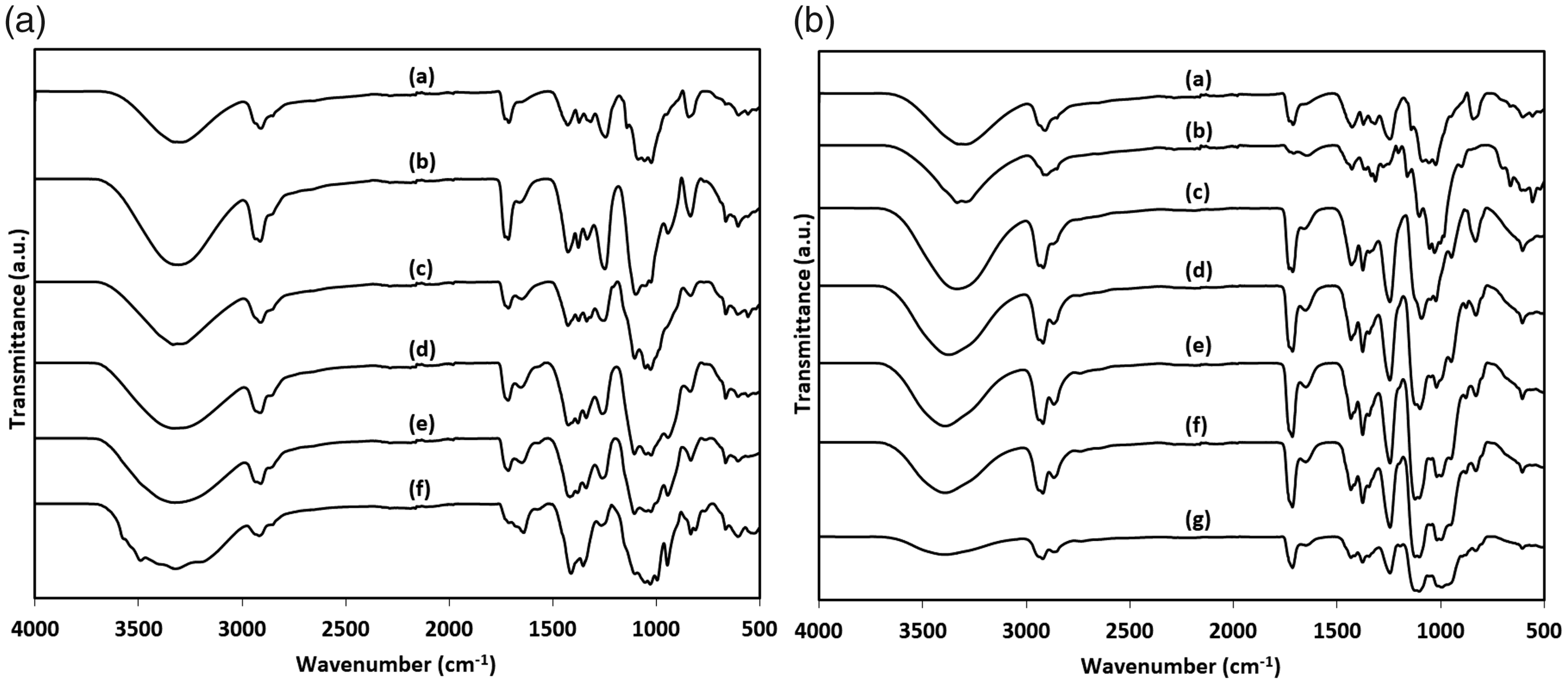
Thermal analysis of hydrogel composite via TGA measurement
TGA of neat PVA/cotton fabric and borax/water and GA-treated PVA/cotton fabric, so-called fabric-reinforced hydrogel composites in the air atmosphere, are shown in Figure 4. Initial weight loss up to around 100℃ corresponded to water evaporation from the samples. Prominent weight loss was observed mainly between 267–389℃ and 389–665℃ for borax-treated samples and between 302–496℃ for GA-treated sample. This can be related to the removal of volatiles as a result of the degradation of the dried hydrogel composite by heating. Prominent weight loss started at a lower temperature in the borax-treated sample compared to neat fabric up to around 400℃, and then increasing temperature made the degradation slower in borax-treated samples. Increasing borax content made the process slower because of the restriction of segmental motion by crosslinking [28]. Residual of the material after the TGA test increased because borax components stayed in the system, and contents were 0 wt % for untreated fabric sample and 1.11, 2.53, 3.89, 8.36, 8.47, and 13.94 wt % for 0.1, 0.3, 0.5, 1, 1.5, and 2 wt % borax/water-treated and dried samples sequentially (Figure 4(a)). GA-treated hydrogel composites were completely degraded up to 800℃ regardless of the GA content (Figure 4(b)). GA treatment did not have a very significant effect on thermal stability of the neat fabric in this study comparing to borax-treated samples, but the degradation continued at higher temperatures with increasing GA content after around 500℃. This can also be explained by the restriction of segmental motion by crosslinking [28].
TGA plots of (a) neat PVA/cotton fabric and PVA/cotton fabric treated in different (A) wt.% of borax/water solutions: (b) 0.1, (c) 0.3, (d) 0.5, (e) 1, (f) 1.5, and (g) 2; and (B) GA solution contents in water/HCl solution: (b) 0 (just water/HCl), (c) 0.25, (d) 0.5, (e) 1.5, (f) 2, and (g) 3.5 ml.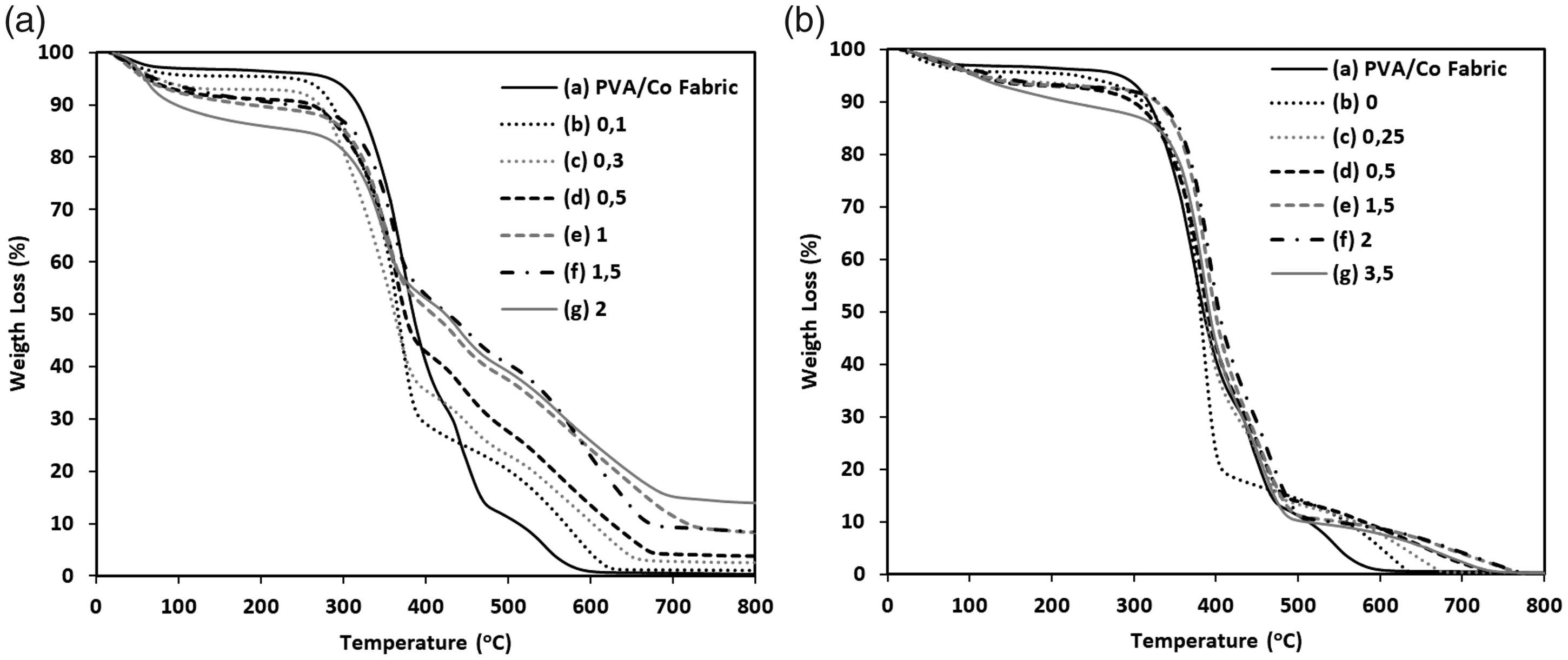
Microstructural analysis of hydrogel composites via X-ray diffraction method
X-ray diffraction measurements were conducted on the dried hydrogel composite samples to investigate how gelation affected the crystalline microstructural changes with both borax and GA treatments, and the XRD plots were demonstrated in Figure 5(A) (borax-treated samples) and (B) (GA-treated samples). Two distinct peaks detected at 19.52° and 22.8° at neat PVA/cotton fabric are related to the orthorhombic lattice structure of semi-crystalline PVA [33,34]. Peak intensity at 19.52° extremely decreased even with a small amount of borax treatment. Peak intensity at 22.8° also decreased with the initial treatment of low concentration of the aqueous borax solution, and increasing borax concentration in the treatment solution caused to reduce this peak even more. The intensity reduction of these peaks could be related to strong interaction and complexation of borax with hydroxide radical of PVA chains that diminished the rearrangement of PVA chains and did not allow a proper crystallization [33]. The overlapping peak at 22.8° can also come from the crystalline structure of cellulose in cotton yarns in the hydrogel composite [33]. Intensity of the peaks coming from cellulose mostly decreased because mostly dried hydrogel matrix was face to the experimental section rather than cotton yarns in the dried composite hydrogel. Pure borax peaks at 35.2° were also seen at high borax concentration treatment sample since excess borax did not go into the crosslinking reaction and crystallized again after drying the composite hydrogel samples [22]. A similar trend was observed at GA-treated samples as the peak intensity at 19.52° tremendously decreased (Figure 5(b)). Crystallinity also decreased with GA treatment as a result of crosslinking of PVA [30]. The decrease in the peak intensity of 19.52° after both borax and GA treatments is another evidence of the crosslinking of PVA chains.
XRD patterns of (a) neat PVA/cotton fabric and PVA/cotton fabric treated in different (A) wt % of borax/water solutions: (b) 0.1, (c) 0.5, (d) 1, (e) 1.5, and (f) 2; and (B) GA solution contents in water/HCl solution: (b) 0 (just water/HCl), (c) 0.25, (d) 0.5, (e) 2, and (f) 3.5 ml.
Mechanical analysis of hydrogel composites due to crosslinker and water contents
The same size of PVA/cotton-woven fabrics were cut at warp directions. The maximum applied force of the neat fabric was measured as 227.32 N. Appropriate ratio of aqueous borax and GA solutions were poured on the fabrics and waited for 24 hours in order to allow a proper crosslinking of the PVA molecules in the fabric structure, and as-obtained woven fabric-reinforced hydrogel composites were rinsed with distilled water several times. Finally, composite structures were left to laboratory condition (50% relative humidity and temperature to 24℃) to release water, and then mechanicals tests were performed. Mechanical properties of the dried woven fabric-reinforced hydrogel composites were demonstrated in Figure 6(A) (borax treated) and (B) (GA treated). As seen from Figure 6(A), the rigidity of dried composite hydrogel increased with the initial addition of borax and then decreased gradually by increasing borax ratio in the aqueous treatment solution. Breaking elongation also decreased by increasing borax ratio. Inset tables in Figure 6(A) and (B) have demonstrated the maximum applied force to failure depending on crosslinker ratios. Breaking force increased first from 301.48 to 340.4 N when borax ratio increased from 0.1 to 0.3 wt % and then it decreased properly to 312.26, 256.71, 254.89, and 165.83 N by increasing the borax ratio to 0.5, 1, 1.5, and 2 wt %, respectively. When the crosslinker concentrations were increased, the amount of crosslinked chains increased and crystallized PVA chains were reduced (this can be seen from XRD data). This is the main reason for the dramatically decrease the mechanical properties. The breaking was observed from the single place as seen in the inset picture of Figure 6(A) as shown with the black arrow. On the contrary, gradual and consecutive breaking occurred at multiple places of the composite at GA-treated sample at high concentrations (Figure 6(B) (c) to (f)). These multiple failing points were shown with black arrows as inset picture in Figure 6(B). Single place breaking was observed at low aqueous GA concentration (Figure 6(B) (a)). Breaking force also first increased from 280.04 to 293.07 N when GA ratio increased from 0 to 0.25 wt %. It then decreased to 228.2, 247.6, 185.1, and 188.04 N not in proper order as in borax-treated samples with the increase in GA ratio to 0.5, 1, 1.5, and 2 wt %. Comparing with borax samples, higher breaking elongations were found with GA samples. This can be attributed to flexibility of GA comparing to crystallizable borax. Optimum crosslinker concentrations for borax and GA samples were determined as 1 wt % for borax and 0.25 ml GA samples. Since most of the PVA chains already crosslinked and no more borax required, after 1 wt % aqueous borax, additional borax crystallized by itself in the composite structure. This can be clearly seen from the XRD spectra Figure 5(A) (e) and (f). About 0.25 ml GA sample was selected for mechanical analysis depending on water contents because consecutive breaking observed at multiple places during the mechanical test after this concentration (Figure 6(B)).
Tensile properties of dried fabric reinforced hydrogel composites: ((A) wt % of borax/water solutions: (a) 0.1, (b) 0.3, (c) 0.5, (d) 1, (e) 1.5, and (f) 2; and (B) GA solution contents in water/HCl solution: (a) 0, (b) 0.25, (c) 0.5, (d) 1.5, (e) 2, and (f) 3.5 ml.
Mechanical properties of fabric-reinforced hydrogel composite depending on water content in the composite structures have been investigated by observing force-elongation plots, and the results were shown in Figure 7(a) (borax treated) and (b) (GA treated). As seen from Figure 7 for borax-treated samples, the rigidity of the fabric-reinforced hydrogel composite dramatically increased by releasing water from the structure. Breaking force gradually increased by releasing water from the structure. Breaking force was 40.8 N when the hydrogel composite contained 100 wt % water and increased to 277 N when it contained 5 wt % water. At the same time, breaking elongation gradually decreased with decreasing water content in the composite structure. At GA-treated samples, a very high breaking elongation was obtained at high ratio GA treatment but it did not change in proper order in all the samples. This could be caused by the organic nature of the molecular structure of the GA. Breaking force first decreased and then gradually increased when the water content in the composite structure increased. Breaking force was 246.4 and 285.9 N when the hydrogel composite contained 70 and 100 wt % water and increased to 325 N when it contained 5 wt % water. PVA yarns in the neat fabric structure were dissolved and transformed into hydrogel structure after pouring the crosslinking aqueous borax and GA solution on the fabric. So, the applied force was mostly withstood by the reinforced cotton yarns in the hydrogel composite. Additionally, in Figure 7, breaking force at borax-based hydrogel composite decreased more by increasing water content comparing to GA-based hydrogel composite.
Tensile properties of fabric reinforced hydrogel composite when contain different content of water: (a) 1 wt % aqueous borax treated samples, (b) 0 and 25 ml GA solution contents in water/HCl solution treated sample, and (c) breaking force comparisons.
Conclusion
Cotton/polyvinyl alcohol-woven fabric-based hydrogel composite structures were successfully obtained by treating as-produced woven fabrics with aqueous borax and GA solutions. PVA yarns were directly transformed to hydrogel matrix structure when the crosslinker solutions were poured on the fabrics. The composite structure became thermally more stable when borax was used as crosslinker comparing to GA samples. Force–elongation tests were performed to investigate the effect of crosslinker and water content in the hydrogel composite structures on mechanical properties. Maximum applied force to failure decreased by increasing crosslinker content for both borax and GA-treated samples. Increasing water content in the structure extensively decreased the breaking strength of the hydrogel composite. The high strength hydrogel composites could be used as hygroscopic materials for planting and erosion control at inclined terrains.
Footnotes
Acknowledgements
The authors thank Uludag University. The study is Ümit Koç’s PhD thesis at the Graduate School of Natural and Applied Sciences, Uludag University. The authors thank Mehmet Tiritoglu for conducting mechanical test and Batmaz Tekstil Company for producing woven fabrics.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received financial support from Scientific Research Project Unit (BAP) of Uludağ University with the project number OUAP(MH)-2018/11 for the research, authorship, and/or publication of this article.
