Abstract
One-step and rapid preparation of natural fiber woven fabric reinforced hydrogel composites via simultaneous dissolution and crosslinking of polyvinyl alcohol (PVA) yarns in the fabric was reported. In this regards, PVA/Cotton (C), PVA/Flax (F) and PVA/Wool (W) blended woven fabrics were prepared for the manufacturing fabric reinforced hydrogel composites. The hybrid woven fabric reinforced fabrics were treated with different concentrations of borax solutions. Aqueous borax solutions were used to alter the PVA yarns in the fabric into cross-linked structure in order to enhance mechanical performance of the hydrogel composite. Morphological investigation of hydrogel composites in a dried form was carried out by scanning electron microscopy (SEM) imaging. The chemical characterization of aqueous borax treated samples was examined by fourier-transform infrared spectroscopy (FTIR) measurements. Mechanical performances of the hydrogel composites were observed by tensile measurements. Thermogravimetric analysis (TGA) was conducted to characterize thermal stability of hydrogel composites. The results revealed that natural fiber woven fabric reinforcement significantly enhanced the mechanical strength of hydrogel composites, and wool fabric reinforced composite had better mechanical performance than its cotton and flax counterparts. Due to the low mechanical properties of hydrogels in general, the prepared fabric reinforced hydrogel composites could be used in hydrogel applications where mechanical strength is critically important.
Introduction
Hydrogels are physically or chemically cross-linked three-dimensional (3-D) hydrophilic polymer structures which can absorb a significant amount of water and hold it in their structures [1–3]. Hydrogels having very distinct biocompatibility, permeability and hydrophilicity offer potential in various areas such as wound dressings [4], tissue engineering [5,6], drug delivery systems [7,8], healthcare and hygienic products [9] and agriculture applications [10].
PVA is widely used hydrophilic polymeric material which is easily dissolved and forms hydrogel structures by a crosslinked agent such as borax [11], tannic acid [12]. PVA is a non -toxic polymer and crosslinked easily with borax through di-ol complexation [11]. In this regards, when borax and PVA are mixed together, a significant viscosity change is observed in the aqueous PVA solution. This phenomenon would form a complex structure within hydroxyl groups of PVA and borate ions and contributes to a temporary bonding of PVA molecules [13].
The fibers which are manufactured from PVA are suitable to blend with natural fibers such as cotton, flax and wool. This blend is used to form an eco-friendly composite material. This interaction contributes to PVA-natural fibers composites to have a unique properties and good mechanical performance. It also shows a good interaction between reinforcement and matrix materials [14].
Generally the mechanical strength of natural fibers such as cotton and flax obtained from plant source is mostly influenced by so many factors such as moisture content, amount of cellulose, angle of microfibrillar, temperature and amount of defect present on the fibers [15]. The mechanical performance of the cotton and flax fibers are enhanced when the fibers are exposed to wet condition and lose their strength in dry moisture content [16] while the mechanical strength of wool fibers is decreased at wet condition and increased in dry conditions.
Natural fiber-reinforced composites such as flax, jute, hemp, cotton-based offer promising perspectives compared to glass fiber reinforced composites for a sustainable economy in the composite industry [17,18]. Natural fiber's environmentally friendly, biodegradability, low cost, low density properties are significant arguments to use them in the composites [19,20]. In addition to these properties, nanoparticles such as ZnO enhance natural fiber-reinforced composites properties. Shaker et al. [21] produced ZnO nanoparticles by sol-gel method and the ZnO nanoparticles improved the bioactivity flax-woven fabric reinforced composite to prevent against micro-organism attack.
Textile structures such as fabric hydrogel composite membranes which includes cotton fabric [22], glass fabric [23], nano and micro scale fibrous woven fabrics [24] have been combined with hydrogels. In our previous study, as-prepared PVA/cotton fabrics were treated with different concentrations of aqueous borax and glutaraldehyde crosslinking solutions to prepare cotton fabric reinforced hydrogel composite [25]. The results showed that tensile performance of hydrogel composite was dramatically enhanced with fabric reinforcement. Also, the mechanical performance of the composite was gradually increased by releasing the water from its structure at both borax and glutaraldehyde crosslinked hydrogel samples.
This study's purpose is to develop natural fiber woven fabric reinforced hydrogel composite with the high mechanical strength of natural fiber-reinforced materials. Effect of the crosslinker ratio on chemical structure, thermal stability and morphology analysis were investigated via FTIR, TGA and SEM analysis by conducting the tests on the dried samples. Mechanical properties were also examined by conducting tensile strength measurements with the wet samples. Comparing from the previous studies regarding liquid hydrogel composite structure, this study was focused on the investigation and evolution of type of convenient natural fibers such as cotton, flax and wool to enhance the mechanical performance by the formation of hydrogel composites structures with the incorporation of PVA fibers into woven fabric. PVA fibers present in the woven fabrics were transformed into hydrogel form and forming cross linker by borax water solutions. The hydrogel formed by this mechanism was resulted a uniform structures. The hydrogel composite structures formed by this mechanism would have a promising future in agriculture and geotextiles like erosion control.
Material and methods
Materials
Ne 40/1 PVA yarns were purchased from KURABO Marugone Mill, JAPAN. Ne60/2 Open-end cotton yarn (Rotor spinning technique was used producing), Nm33/1 flax yarn, and Nm56/2 wool yarn were used as natural yarns. Borax also known as sodium tetraborate decahydrate (Na2B4O7·10H2O) was received from Galenik Ecza ve Kimyevi Maddeler Deposu (Batch no: 1408H0048). Distilled water was used in all processes. All raw materials were utilized without further purification.
Weaving process
Three types of yarn were prepared for weaving fabrics to produce natural fabric reinforced hydrogel composite. As seen from Figure 1, these are Nm33/1 flax, Ne60/2 open-end cotton and Nm56/2 wool natural fiber yarns, Ne40/2 PVA and doubled and then twisted yarns of Nm33/1 flax+Ne40/2 PVA, Nm56/2 wool+Ne40/2 PVA and Ne60/2 open-end cotton+Ne40/2 PVA hybrid yarns. As a first step, two bobbins of Ne40/1 PVA yarns were doubled and then twisted to produce Ne40/2 PVA yarn. Later, Ne40/2 PVA yarns were further doubled and twisted together with Nm33/1 flax, Ne60/2 open-end cotton and Nm56/2 wool yarns to produce three different hybrid yarns. Both Ne40/2 PVA and hybrid yarns were twisted at 4850 rev/min spindle speed and 300 t/m twist value. Hybrid and Ne40/2 PVA yarns were inserted to the fabric as weft yarns. As shown in Figure 2, warp was prepared from Nm33/1 flax, Nm56/2 wool and Ne60/2 open-end cotton natural fiber yarns respectively for three different fabrics. Hybrid weft with flax yarn component was inserted to the fabric produced with flax warp, hybrid weft with cotton yarn component to the fabric with cotton warp and with the wool component to the fabric with wool warp. Hence, each fabric had only one type of natural fiber (flax, cotton and wool respectively) in addition to PVA yarn. A reed number of 12 (dents/cm) was employed and the warp yarns were prepared at 12 ends/cm warp density for all three fabric types. Weft yarns were inserted at 20 picks/cm for flax warp fabric and 36 picks/cm for both cotton and wool warp fabrics. Fabrics were woven in a sample weaving machine at 400 mm width. Weft yarns were inserted to the fabrics with 2 PVA and 1 hybrid yarns order. In this way, the fabric constructions included around 40% cotton-60% PVA, 40% flax-60% PVA and 40% wool-60% PVA material ratios. After fabrics were woven, they were removed from the loom and made ready for the final step: gelation processing. The weave pattern used in our previous study was also preferred in the production of fabrics for this research [25].

Hybrid weft yarn manufacturing process for the precursor woven fabric.

The manufacturing process of weaving fabric as the hydrogel composite precursor.
Gelation of PVA in cotton, flax and wool reinforced hydrogel composite
Aqueous borax solutions were prepared with the addition of different concentrations (0.5, 1, 1.5, and 2 w/v %) and magnetic stirring process at 50° C. After production of PVA/C, PVA/W, and PVA/F woven fabrics with PVA and hybrid weft and cotton/wool/flax warp yarns, the fabric samples were cut as 5 cm × 10 cm dimensions in the warp and weft directions. Each fabric sample was put on a petridish and the prepared borax/water solutions were spilled on the fabric samples to transform PVA yarns into hydrogel form by crosslinking PVA molecules in the fabric structure. The samples were kept at ambient conditions for 24 hours for the further gelation. Then, the excess borax/water was removed and all the samples were washed with distilled water several times to remove the uncrosslinked borax hydrogel from composite structure. All the hydrogel composites were dried for the performance tests.
Morphology analysis of hydrogel
Scanning Electron Microscopy (SEM) Carl Zeiss Evo 40 was used to analyze the surface and the section morphologies. The samples were sputter coated with a conducting layer for better imaging.
Chemical characterization of hydrogel composites
Chemical analysis of neat PVA/C fabrics, neat cotton yarn, neat PVA/W fabrics, neat wool yarn, neat PVA/F fabrics, neat flax yarn and composite hydrogel due to crosslinker ratio were recorded using Attenuated Total Reflection and Fourier Transform Infrared Spectroscopy (ATR-FTIR, Nicolet iS50) and measurements were performed at 20°C and relative humidity of 65%.
Thermal characterization of hydrogel composites
Thermal properties of fabric reinforced hydrogel composites in dried form were assessed by measuring weight losses in nitrogen environment using a thermogravimetric analyzer (TA Q600) by heating from 50 to 800°C (heating rate of 10°C min−1).
Mechanical measurements of fabric reinforced hydrogel composite
Tensile tests of the hydrogel composites were performed using a Shimadzu tensile tester with AG-X plus model by using 5000 N load cell. The same sample sizes were used after gelation process without cutting them. Gauge length and testing speed were adjusted to 30 mm and 200 mm/min respectively. All the measurements were conducted at both neat fabrics and their hydrogel composites analogs in warp direction and weft direction. Dried hydrogel composites were held in distilled water for 15 min before mechanical testing and then the samples were considered they contain 100% water.
Results and discussion
Concept study result of the gelation of cotton, flax and wool fabric reinforced hydrogel composites
The woven fabric samples were cut in the warp and the weft directions with a dimensions of 5 cm × 10 cm. As seen from Figure 3, when PVA/W, PVA/C, PVA/F fabrics were treated with borax/water solution, PVA yarns totally lost their construction and turned to transparent hydrogel form by simultaneous dissolution and crosslinking of PVA molecules as yarn in the fabrics. Since cotton, wool, flax yarns did not get dissolved by borax-water solution, they kept their position in the structure and stayed as the reinforcement material in the hydrogel composites.

Schematic illustration and optical microscopy images of neat PVA/W fabric and its hydrogel matrix form of aqueous borax treated analog.
Morphology analysis of cotton, flax and wool fabric reinforced hydrogel composites
In order to investigate the interaction between hydrogel matrix and yarns in the fabrics as reinforcement, morphological observation with SEM imaging was carried out. Incorporation of hydrogel into fabric structures varied due to the nature of the reinforced fabric structures. Inspecting SEM images as seen in Figure 4, since the wool has wax materials on the surface structure, borax-water solutions could not penetrate properly into the yarn structures in the fabric. Moreover, during the production of wool yarn, the yarns are treated with oil to eliminate the potential breakage between the fibers and reduction of friction developed between fibers within yarns. This is the reason for having the lower penetration of borax water solutions. The simultaneous dissolution and crosslinking of PVA smoothened the surface of wool fibers by clogging the cuticle cells layers on the fiber surfaces. On the other hand, when observing flax and cotton samples, the borax-water solutions penetrated inside the yarn structure. Comparing wool, flax and cotton samples, hydrogels were mostly collected on the junctions of wool yarns, but the hydrogels penetrated inside of flax and cotton yarns among their fibers due to low amount of wax (as compared with wool fiber) in their physical surface composition.

SEM images aqueous borax treated PVA/W, PVA/C and PVA/F fabrics after hydrogel matrix formation.
Chemical analysis of cotton, flax and wool fabric reinforced hydrogel composites
Chemical investigation of fabric reinforced hydrogel composites with different concentrations of aqueous borax treatment, and their neat fabric counterparts were examined with FTIR spectra and given in Figure 5(A1, A2) for Cotton based, Figure 5(B1, B2) for flax based, and Figure 5(C1, C2) for wool based. For the better comparison and investigation, neat cotton, neat flax and neat wool yarns also treated with the same solution and analyzed with FTIR as shown in the same figures. All the FTIR measurement were conducted with the dried samples. FTIR spectra for PVA/C fabric before and after aqueous borax treatment were presented in Figure 5(A1). The peak at 2930 cm−1 could be assigned to C-H stretching of CH2 group [26]. A broad band showing up between 3033 and 3606 cm−1 with a maximum peak point around 3295 cm−1 belonged to O-H stretching vibration from the hydroxyl groups in PVA and cotton [27]. Decrease of this peak could be attributed to formation of hydrogel structure. The new peak detected at 667 cm−1 with addition to borax to the structure could be attributed to stretching vibration B-O-B bond in the borate network [28]. The peak at 1083 cm−1 assigned to the C–O in stretching mode for PVA [26]. But no significant effect of aqueous borax on cotton yarns was observed (Figure 5(A2)). FTIR spectra of PVA/F fabrics and neat flax yarns before and after aqueous borax treatment were demonstrated in Figure 5(B1) and (5B2) respectively. The FT-IR spectra of yarn treated spectra (Figure 5(B1)) revealed that there is no significant change after borax treatment, so the changes on borax treated PVA/F sample (Figure 5(B1)) could be attributed to changes on PVA molecules as in PVA/C sample. FTIR spectra of PVA/W fabrics and neat wool yarns before and after aqueous borax treatment were shown in Figure 5(C1) and (C2) respectively. A distinct broad parabolic OH peak is seen at aqueous borax treated PVA/W around between 3012 and 3655 cm−1 comparing the neat PVA/W fabric. This is another evidence to formation of PVA based hydrogel structure. As cotton and flax yarn samples, no significant change was not observed at aqueous borax treated neat wool yarns at the applied condition.

FTIR spectra of aqueous borax treated (A1) PVA/C fabrics, (A2) cotton yarn only, (B1). PVA/F fabrics, (B2) flax yarn only, (C1) PVA/W fabrics and (C2) wool yarn only. Treated with different w/v% of borax/water solutions ((A1, B1, C1): (a) without treatment, (b) 0.5, (c) 1.0, (d) 1.5 and (e) 2.0), ((A2, B2, C2): (a) without treatment, (b) 0,1, (c) 0.5, (d) 1, (e) 1.5, (f) 2.0).
Thermal analysis of cotton, flax and wool fabric reinforced hydrogel composites
Thermogravimetric analysis of neat PVA/C fabric, neat PVA/F fabric, neat PVA/W fabric, and their aqueous borax treated counterparts, so-called fabric reinforced hydrogel composites, in dried form were performed under nitrogen atmosphere, and the results are shown in Figure 6 and Table 1. Main water loss of all hydrogel composite samples occurred up to 100°C. The amount of water loss for all sample groups increased after gelation process compared to their neat counterparts. Water loss for cotton samples up to 100°C being higher than flax and wool samples revealed that cotton reinforced hydrogel sample takes more time for fully drying, see Figure 6(A). Treating the neat samples with aqueous borax even at low concentration, reduced thermal stability a little bit at all samples. Increasing the amount of borax concentration after 0.5 w/v % didn’t change the degradation temperatures significantly at PVA/F samples as seen in Figure 6(B). This difference is even smaller at PVA/W hydrogel composite samples as seen in Figure 6(C).

TGA plots of (A) PVA/C, (B) PVA/F and (C) PVA/W fabrics treated in different w/v% of borax/water solutions: (a) neat fabrics, (b) 0.5, (c) 1.0, (d) 1.5 and (e) 2.0.
Data points obtained from TGA plots of PVA/C, PVA/F and PVA/W fabrics treated in different w/v % of borax/water solutions.
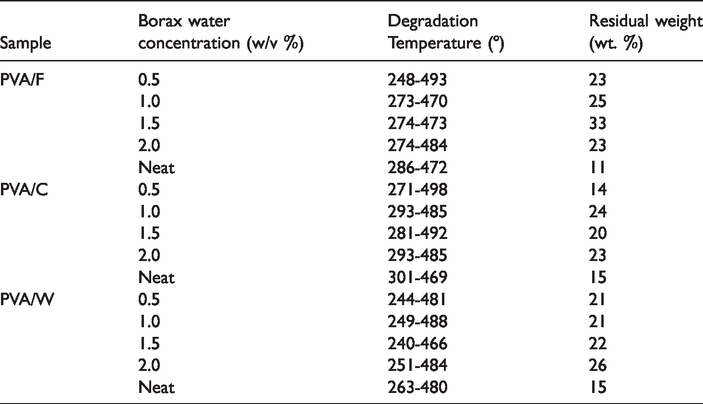
For PVA/C samples, the amount of water loss with respect to borax content is gradually increased due to the percentage of borax concentration as seen in Figure 6(A, (a) to (e)). The untreated PVA/C neat fabric started to degrade thermally at temperatures of 301 to 469°C, whereas the borax-water treated samples started thermal degrade mainly between 271 and 498° C for 0.5 w/v % borax-water concentration and between 293 and 485° C for 1.0 w/v %. For the concentration of 1.5 w/v % and 2.0 w/v % borax-water solutions, thermal degradation was observed at the temperature range between 281 and 492°C and between 293 and 485°C respectively. When PVA/C fabrics were treated with borax-water solution having a concentration between 0.5 and 2.0% w/v, hemicellulose degradation started at temperature of 280°C, while the cellulose structure was degraded at temperature of 490°C and lignin started to decompose at a temperature of 795°C [29]. Residual contents were 15 wt.% for untreated PVA/C fabric sample and 14, 24, 20, 23 wt.% for 0.5, 1.0, 1.5, 2.0 w/v% borax-water treated samples respectively (Figure 6(A)). Inspecting Figure 6(B), the untreated PVA/F neat fabric mainly thermally degraded at the temperature between 286 and 472°C, whereas the borax-water treated samples’ thermal degradation occurred mainly between 248 and 493°C for 0.5 w/v% borax-water concentration and between 273 and 470°C for 1.0 w/v%. For the concentration of 1.5 w/v% and 2.0 w/v% borax-water solutions, thermal degradation was observed at the temperature range between 274 and 473°C and between 274 and 484°C respectively. Like PVA/C fabrics, the PVA/F fabrics have the same fashion because of their similar structural properties. Residual contents after TGA measurements were 11 wt.% for untreated PVA/F fabric sample and 23, 25, 33, 23 wt.%, and for 0.5, 1.0, 1.5, 2.0 w/v% borax/water treated samples sequentially (Figure 6(B)).
In Figure 6(C), the untreated neat PVA/W fabric thermally degraded at the temperatures between 263 and 480°C, whereas the borax-water treated samples’ thermal degradation occurred mainly between 244 and 481°C for 0.5 w/v% borax-water concentration and between 249 and 488°C for 1.0 w/v%. For the concentration of 1.5 and 2.0 w/v% borax-water solutions, thermal degradation was observed at the temperature between 240 and 466°C and between 251 and 484°C respectively. Residual contents were 15 w/v% for untreated PVA/W fabric sample and 21, 21, 22, 26 wt.% for 0.5, 1.0, 1.5, 2.0 w/v% borax/water treated samples sequentially (Figure 6(C)).
Mechanical analysis of cotton, flax and wool fabric reinforced hydrogel composites due to cross linker and water contents
Mechanical properties of the cotton, flax and wool woven fabric reinforced hydrogel composites at both warp and weft direction were investigated via force-elongation curves due to cross linker borax content, and the plots were demonstrated in Figure 7 (Cotton), Figure 8 (Flax), and Figure 9 (Wool). When the cotton samples were examined, the maximum applied force of the wetted (with just water) neat PVA/C fabric was measured as 227 N at warp direction while the maximum applied force of the wetted neat PVA/C fabric was measured as 223 N at weft direction. As seen from the Figure 7(a), the breaking force of the hydrogel composite in the warp direction was 162 N when the neat fabric was treated with 0.5%w/v of borax-water solution, and it was decreased further to 100 N when the borax-water concentration was changed to 1.0% w/v, due to the crosslinking of PVA in the PVA/C blend woven structure. Inter and intra friction developed with in the cotton based woven hydrogel structure could be decreased. The breaking force was increased from 225 to 262 N by changing the borax-water concentration from 1.5 to 2.0 w/v% in the warp direction. Due to the formation of more stable hydrogels within and between the yarns, inter and intra friction of yarns were developed and enhanced the mechanical performance of PVA/C with borax-water treatment. Since the composites were tested in wet form, elongation at the break is higher in this study comparing with our previous study at warp direction [25]. Additionally, breaking strength increase with increasing borax concentration in the treatment solution at wet samples in this study, but breaking point decreased at dried composites except initial increase of borax concentration in the previous study [25]. In another study, it was also confirmed that breaking strength gradually increased when the hydrogel composite released its water content [30]. On the other hand, as seen in Figure 7(B) the breaking force in the weft direction was 108 N when the treatment concentration was 0.5 w/v %. Likewise, the force was gradually decreased from 96 to 84 N when the concentration of borax-water was changed from 1.0 to 1.5 w/v%. The breaking force was 162 N when the aqueous borax treatment concentration was 2.0 w/v%.

Tensile properties of cotton fabric reinforced hydrogel composites ((A) warp and (B) weft directions) after held in water for 15 min: Treatment with different w/v % of borax/water solutions ((a) 0.5, (b) 1.0, (c) 1.5, (d) 2.0).

Tensile properties of flax fabric reinforced hydrogel composites ((A) warp and (B) weft directions) after held in water for 15 min: Treatment with different w/v % of borax/water solutions ((a) 0.5, (b) 1.0, (c) 1.5, (d) 2.0).

Tensile properties of wool fabric reinforced hydrogel composites ((A) warp and (B) weft directions) after held in water for 15 min: Treatment with different w/v % of borax/water solutions ((a) 0.5, (b) 1.0, (c) 1.5, (d) 2.0).
Observing the mechanical performance of flax sample, the maximum applied force of the wetted (with just water) neat PVA/F fabric was measured as 115 N in the warp direction. While, the maximum applied force of the wet neat PVA/F fabric was measured as 95 N in the weft direction. Mechanical properties of wet woven flax-reinforced hydrogel composite were demonstrated in Figure 8(A) at warp direction and in Figure 8(B) weft direction. The breaking force in warp direction was increased from 157 to 239 N proportional with the increasing of borax-water treatment concentration from 0.5 to 1.0% w/v whereas it was gradually decreased from 105 to 101 N, when the concentration borax-water is changed from 1.5 to 2.0% w/v in the warp direction respectively. As seen from Figure 8(B), the breaking forces in the weft direction were 132 N (0.5 w/v %), 88 N (1.0 w/v %), 99 N (1.5 w/v %), 82 N (2.0 w/v %), respectively.
When the mechanical performance of the wool samples were examined, the maximum applied force of the wetted (just water) neat PVA/W fabric was measured as 284 N at warp direction while the maximum applied force of the wet neat PVA/W fabric was 85 N at weft direction. The physic mechanical property of PVA/W fabric was influenced by aqueous borax treatment. The mechanical properties of wool fabric reinforced hydrogel composites have been investigated due to concentration of the cross linker treatment by using the force elongation plots in the warp (Figure 9(A)) and the weft (Figure 9(B)) directions. The breaking force in the warp direction was increased from 290 to 320 N when the used borax-water concentration was increased from 0.5 to 1.5 w/v%, whereas, when the concentration of borax-water was 2.0 w/v%, the force decreased to 313.42 N in warp direction. When the amount of borax concentration was increased it enhanced the crosslinked chains and reduced crystallized PVA chains, this would reduce mechanical performance of the hydrogel composite [31]. As seen from Figure 9(B), the breaking forces in the weft direction were 83 N (0.5 w/v %), 85 N (1.0 w/v %), 92 N (1.5 w/v %), 98 N (2.0 w/v %), respectively.
Conclusion
Natural fiber woven fabric reinforced hydrogel composite formation and the suitability of three different natural fibers for reinforcement were investigated in this research work. Woven fabrics were produced by mixing PVA yarns with three types of natural fiber yarns, namely cotton, flax and wool. PVA fibers in the fabric structure were simultaneously dissolved and PVA molecules were crosslinked when treated with aqueous borax solution. However cotton, flax and wool fibers kept their position in the fabric structure. A natural fiber woven fabric reinforced hydrogel composite structure was obtained. FTIR result revealed that aqueous borax treatment properly converted PVA molecules into crosslinked hydrogel structure, but no significant chemical change was observed at neat fibers when the neat cotton, flax and wool yarns were treated with the same solution. When SEM images were analyzed even though PVA molecules could penetrate inside the flax and cotton yarns and completed their crosslinking processes among the fibers, the penetration didn’t happen inside the wool yarns and hydrogel structures were mostly seen at the yarn interlacing points and on the wool fabric surface. Penetration of hydrogel inside the cotton and flax yarns reduced the inter fiber friction and therefore the strength of fabric reinforced hydrogel composite compared to neat fabric strength with most of borax concentrations but the strength of wool fabric reinforced hydrogel composite remained almost unaffected and stable with all borax concentrations. Overall result suggested that wool fabric reinforcement for PVA based hydrogel composites was better for mechanical enhancement comparing to cotton and flax fabric reinforcements and could be used as a natural reinforcing materials to strengthen hydrogel materials. Despite a serious strength reduction in the fabric reinforced hydrogels with cotton and flax fabric reinforcement compared to neat cotton and flax fabrics, the strength levels of cotton and flax fabric reinforced hydrogel composites are much higher than only hydrogel structures and could be sufficient for many applications.
Footnotes
Acknowledgements
The authors thank to Bursa Uludag University. The study is Ümit Koç's PhD thesis at the Graduate School of Natural and Applied Sciences, Bursa Uludag University. The authors thank Alhayat Getu Temesgen for conducting mechanical test and Batmaz Tekstil Company for producing woven fabrics.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors received financial support from Scientific Research Project Unit (BAP) of Bursa Uludag University with the project number OUAP(MH)-2018/11.
