Abstract
Polyvinyl alcohol (PVA)/cotton (Co) woven fabrics were produced by constructing Co as warp yarns and PVA as weft yarns in the fabric structure. As-prepared PVA/Co fabrics were treated with borax/water solution. Because of the simultaneous dissolution and gelation of PVA yarn in the fabric and transformation of PVA molecules into cross-linked gel structures, gel penetrated among the yarns in the matrix form and hence Co yarn-reinforced composite hydrogels were obtained. The retention time of water by composite hydrogels was first increased and then decreased by increasing borax concentration in the cross-linker solution. With yarn reinforcing, the tensile strength of hydrogel structure significantly increased. Mechanical properties of hydrogel composites were very variable depending on water content in the structure and tensile strength tremendously increased when water evaporated from the structure. Thermal and chemical characterizations of yarn-reinforced hydrogel composites were conducted in addition to swelling and mechanical analysis to investigate the performance of the hydrogel composites.
Introduction
Hydrogels are biocompatible materials that contain a large amount of water and used for a variety of applications including artificial organs, contact lenses, drug delivery systems, and superabsorbent areas. 1,2 Due to their flexible and soft characteristics, hydrogels resemble living tissues. 1 Hydrogels are cross-linked molecules and they are not soluble in any solvent. 3 Since there are abundant amounts of hydrophilic groups in the structures and water molecules are accumulated between cross-linked molecules through hydrophilic groups, hydrogels can retain water more than their weight. 4
Polyvinyl alcohol (PVA), which is a hydrophilic polymer, can be converted into hydrogel structure by using different cross-linkers such as borax, suberic acid, and glutaraldehyde. 5,6 Effect of the shape of the cross-linker on the PVA hydrogels was studied by Sonker et al. 6 Suberic acid as aliphatic and terephthalic acid as aromatic cross-linkers were used and cross-linking of PVA by suberic acid was reported to be better than terephthalic acid by considering mechanical and thermal test results. 6 Mansur et al. 7 produced pH-sensitive PVA–based hydrogels using glutaraldehyde as the cross-linker for drug delivery applications with various degrees of hydrolysis of PVA.
On the other hand, PVA-based hydrogels can gain self-healing characteristics by applying different methods. For instance, PVA-based hydrogel was produced via freezing–thawing method without using any chemical cross-linker or other reinforcing agents. 8 The crystallization of PVA conducted at low temperatures (−15°C to −10°C) via freezing–thawing method resulted in physical cross-linking and the formation of self-repairing hydrogels was achieved. 9,10 PVA-based self-healable hydrogels were also produced by using nanoparticles such as iron oxide and silver. 11
Hydrogels are mechanically low-grade materials and are being produced in composite form with various reinforcement materials to improve their mechanical properties. 12,13 Fiber or fabric reinforcement in the structure of composite hydrogels is defined as the part carrying the load. Holloway et al. 14 produced ultra-high-molecular-weight polyethylene and polypropylene fiber–reinforced PVA-based strong hydrogel composite to heal meniscus damage. Production of fabric-reinforced hydrogel composite structures with enhanced mechanical properties is important for biomedical applications. When studies in the literature have been investigated, the number of studies on fabric-reinforced hydrogel composite structures seems to be limited. 13,15 Hydrogel-treated polyester fabric was produced by previously hydrolyzed polyester fabric with PVA, tragacanth, and silver nitrate mixture along with gelation and in situ synthesis of nano silver through reduction of silver nitrate. 15 Woven glass fabric–reinforced and high energy-dissipative polyampholyte matrix hydrogel composite as strong structural biomaterial with high toughness and tensile properties were produced by Huang et al. 13 Cotton (Co) fabric was treated with as-synthesized chitosan hydrogel for pH sensitivity and antimicrobial properties. 16 The surface of fabric was modified with hydrogel via covalent bonding for wound dressing application that the hydrogel structure was isolated from protein. 17 Konjac glucomannan–based hydrogel was prepared and coated on glass fabrics for oil–water separation in harsh environments. 18 On the other hand, facile preparation of yarn-reinforced hydrogel composite from a woven fabric has not been reported in the literature.
In this study, yarn-reinforced hydrogel composites have been produced from PVA/Co woven fabrics. In this regard, PVA/Co fabrics were produced and then dipped into previously prepared borax/water solution in order to obtain a proper yarn-reinforced composite structure by simultaneous dissolution and gelation of PVA in the fabric. Then, an optimum borax ratio in the solution was determined by conducting the swelling test and material properties produced with optimum borax ratio were investigated by applying mechanical, thermal, and chemical analyses. Because PVA 19 and cellulose 20 are considered biocompatible and environmentally friendly materials, and Co yarn reinforcement provides additional strength to the hydrogel structure, the prepared hydrogel composite structure can be used as planting element at inclined terrains for erosion control.
Materials and methods
Raw materials and chemicals
Ne 40/1 PVA yarns were obtained from KURABO Marugone Mill, Japan. 60/2 Open-end Co yarns were received from Batmaz Tekstil company (Kestel OSB, Bursa, Turkey). Ne: Number of hangs (840 yards) in one pound of yarn. Borax was purchased from Galenik Ecza ve Kimyevi Maddeler (ITOB OSB, Izmir, Turkey) Deposu (batch no: 1408H0048). Distilled water was used in all processes. All raw materials were used without further purification.
Weaving process
Before production of the woven fabrics, two sets of PVA yarns of Ne 40/1 were doubled (combined) into one single yarn at 4850 rev/min spindle speed and 300 t/m twist. Warp was prepared using Ne 60/2 Co open-end yarn. A reed number of 12 (dents cm−1) was employed with two ends drawn between two reed dents. Hence 24 ends cm−1 warp density was obtained. Ne 40/2 PVA weft was inserted. Fabric samples were woven by using 10 shafts with the plain draft. A weave repeat of 10 warps and 30 wefts were obtained to hold firmly and cover the Co warp from both bottom and upper surfaces by PVA weft yarns. In this way, the Co warp was covered by PVA yarns from top to bottom sides and embedded in hydrogel after gelation of PVA yarns. Fabrics were woven in a sample weaving machine at 400 mm width. A picture of woven fabric surface is shown in Figure 1(a).

Schematic illustration and photograph images of (a) PVA/Co fabric construction (before borax treatment), (b) yarn-reinforced hydrogel (after borax treatment), (c) (c1) dried yarn-reinforced hydrogel and (c2) swollen yarn-reinforced hydrogel with water, and (d) mirror images of yarn-reinforced hydrogel composite. PVA: polyvinyl alcohol; Co: cotton.
Gelation of PVA in Co yarn-reinforced hydrogel composite
Borax/water solutions were prepared by the magnetic stirring process at 50°C with different concentrations (0.1, 0.3, 0.5, 1, 1.5, and 2 wt%). Then, 5 × 10 cm2 fabric samples were cut in both warp and weft directions. Each fabric sample was put on a Petri dish and the prepared borax/water solutions were poured on the fabric samples to transform PVA yarns into hydrogel by cross-linking PVA molecules in the fabric structure. The samples were kept at ambient conditions for 24 h for further gelation. Then, excess borax/water solution was removed and all the samples were washed with distilled water at least five times to remove the uncross-linked borax from hydrogel composite structure. All the hydrogel composites were dried for the performance tests.
Chemical analysis of hydrogel
Chemical analysis of neat PVA and PVA/Co fabrics and composite hydrogel due to cross-linker ratio has been carried out using attenuated total reflection and Fourier-transform infrared spectroscopy (ATR-FTIR; Bruker IFS 66, Middle East Techinical University Central Laboratory, Ankara, Turkey).
Thermal characterization of hydrogel
Thermal properties of yarn-reinforced hydrogel composites in dried form were evaluated by measuring weight losses with temperature in air environment using a thermogravimetric analyzer (TA SDT 650, Middle East Techinical University Central Laboratory, Ankara, Turkey), heating from 50°C to 800°C (heating rate of 10°C min−1).
Microstructural analysis of hydrogel
Microstructural characterization of neat PVA/Co fabrics and their borax/water-treated and dried counterparts were conducted using Rigaku Ultima IV X-ray diffractometer (Middle East Techinical University Central Laboratory, Ankara, Turkey).
Swelling and water removal test
Each dried sample was immersed in a distilled water pool in Petri dishes and held for 90 min. Samples absorbed the water and swelled. After the swollen samples were taken from the water, excess water was removed with paper tissues and then yarn-reinforced hydrogel composite samples were weighed. It was assumed that the first measurement of the sample contained 100% of water. Then, the samples were kept at ambient conditions to allow water evaporation. Weight measurements were repeated after every 15 min to observe water removal by time. This process was also conducted for neat fabric containing no hydrogel. The degree of swelling was calculated with the following equation in percentage 21
where w s is the weight of hydrogel composite with water and w d is the weight of dried hydrogel composite.
Mechanical measurements of fabric-reinforced hydrogel composite
Tensile testing was carried out using Shimadzu tensile tester with AG-X plus model (Bursa Uludag University Textile Engineering Test and Analysis Laboratory, Bursa, Turkey) and load cell capacity of 5000 N. Exactly the same sample size was used after the gelation process without cutting them. Gauge length and testing speed were adjusted to 30 mm and 200 mm min−1, respectively. Tensile tests were performed for dried and swollen fabric-reinforced hydrogel composite. Elongation (%) was given with respect to gauge length.
Results and discussion
Concept study of the fabric-reinforced hydrogel composite
Schematic illustration and high magnification photograph images of PVA/Co fabric construction (before borax treatment), yarn-reinforced hydrogel composite (after borax treatment), and the dried and swollen forms along with mirror images of yarn-reinforced hydrogel composite are shown in Figure 1.
After production of PVA/Co woven fabric with PVA weft and Co warp yarns, fabric samples were prepared by cutting woven fabric as 5 × 10 cm2 dimensions in the warp and weft directions. Samples were treated with previously prepared borax/water solution as explained in the “Materials and methods” section. As seen from Figure 1, when PVA/Co fabrics were treated with borax/water solution, PVA yarns (Figure 1(a)) totally lost their structures and turned to transparent gel form by simultaneous dissolution and gelation of PVA in the fabric. In this way, PVA forms matrix structures of Co yarn-reinforced hydrogel composite (Figure 1(b)). Aligned Co yarns in the hydrogel composite structure can be clearly seen in dried (Figure 1(c), c1) and wet forms (Figure 1(c), c2). Since the matrix is a transparent hydrogel, yarns in the composite can be seen from both sides as in the mirror image (Figure 1(d)).
When the previously prepared borax/water solutions were poured on the fabric samples (10 × 5 cm2), fabrics were extended in the Co yarn direction (warp) and shrunk in the weft direction; 10 cm (warp) × 5 cm (weft) and 5 cm (warp) × 10 cm (weft) cut samples turned to 11 cm (warp) × 2 cm (weft) and 8 cm (warp) × 7 cm (weft) size, respectively. This extension and shrinkage can be explained with the cross-linking of PVA molecules and partial opening of the crimp in Co yarns in fabric structure. PVA yarns lost their structure with borax/water treatment and as a result of the simultaneous dissolution of PVA molecules and cross-linking of these dissolved molecules.
Chemical analysis of hydrogel composite via FTIR
In order to investigate the chemical change of the fabric samples when treated with different ratio of borax/water solutions, ATR-FTIR measurements were carried out with dried samples and the results are presented in Figure 2. Cellulose molecules at Co yarn have plenty of –OH groups and can form many hydrogen bonds with the hydrogel matrix on the surface of the Co yarn. 22,23 So, investigation of the transformation of PVA to hydrogel structures with borax cross-linking was carried out with FTIR analysis. All samples exhibited broad characteristic OH stretching vibration peak between 2971 cm−1 and 3637 cm−1 with the highest peak point around 3300 cm−1. 24 This peak corresponds to hydrophilic hydroxyl groups in PVA and Co, and also with increasing borax content in borax/water solution, the intensity of this peak gradually decreases because of the complexation (cross-linking) of borax and hydroxyl groups. 5 Treatment of samples with borax leads to a new peak detected at 662 cm−1. This corresponds to the stretching vibrations of O–B–O bonds. 5 The peak at 2924 cm−1 can be attributed to C–H stretching vibration in CH2 groups. 5 The peak at 1088 cm−1 assigned to the C–O in stretching mode for PVA 22 and this peak shifts to 1107 cm−1 after formation of hydrogel structure. Both types of complexes as trigonal (around 1425 cm−1) and tetrahedral (around 1335 cm−1) were formed between PVA and borax molecules. 5 The ratios of these peaks are not distinctively changed by increasing borax ratio. It can be assessed that the ratios of these complexes are not changed by increasing borax ratio. Free hydroxyl groups on trigonal complexes can also allow more hydrogen bonding between PVA and Co molecular chains. 5

FTIR spectra of (a) PVA fabric, (b) neat PVA/cellulose fabric; PVA/cellulose fabric treated in different wt% of borax/water solutions: (c) 0.5%, (d) 1%, (e) 1.5%, and (f) 2%. PVA: polyvinyl alcohol; FTIR: Fourier-transform infrared.
Thermal analysis of hydrogel composite via thermogravimetric analysis measurement
Thermogravimetric analysis (TGA) of neat PVA/Co fabric and different concentration of borax/water-treated PVA/Co fabric, so-called yarn-reinforced hydrogel composites in the air atmosphere, is shown in Figure 3. Initial weight loss up to around 100°C arises from water evaporation from the sample. The following sharp weight loss results from the removal of volatiles because of the degradation of materials. By increasing borax/water concentration, the thermal stability of yarn-reinforced hydrogel composites increases and thermal degradation temperatures shift to higher degrees as a result of the restriction of segmental motion originated from cross-linking of PVA molecules. 24 So, the enhancement of thermal stability is obvious. Residual material content increases by increasing borax content in the structures after TGA measurement. As seen from the inset figure in Figure 3, residual contents were 3.95 wt% for untreated fabric sample and 3.22, 2.66, 4.43, 8.58, 9.01, and 11.17 wt% for 0.1, 0.3, 0.5, 1, 1.5, and 2 wt% borax/water-treated and dried samples, respectively.
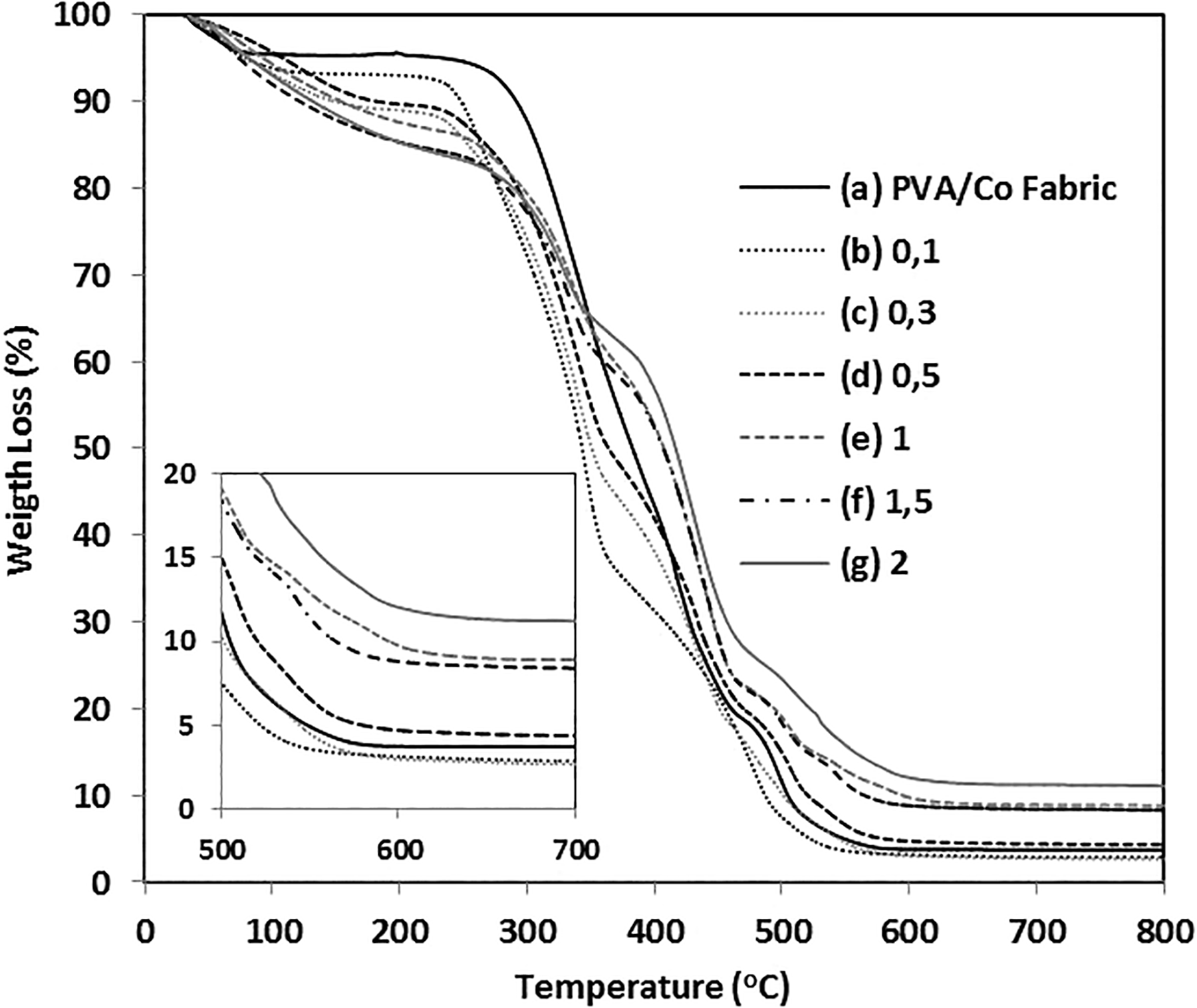
TGA plots of (a) neat PVA/Co fabric; PVA/Co fabric treated in different wt% of borax/water solutions: (b) 0.1, (c) 0.3, (d) 0.5, (e) 1, (f) 1.5, and (g) 2. PVA: polyvinyl alcohol; Co: cotton; TGA: thermogravimetric analysis.
Microstructural analysis of hydrogel composites via X-ray diffraction method
X-ray diffraction measurements were conducted to study crystal formation and transformation after the borax/water treatment of the PVA/Co fabric samples (Figure 4). Neat PVA fabric exhibited two obvious diffraction peaks at 19.45° and 22.69°, which corresponded to the orthorhombic lattice structure of semicrystalline PVA. 25,26 The peak at 19.45° shifts to 20.2° when the sample was treated with 0.1 wt% of borax/water solution and then dried. Increasing borax concentration up to 2 wt% fluctuated the location of this peak between 20.2° and 20.46°. On the other hand, increasing borax concentration decreases the intensity of the peaks at 19.45° and 22.69° as a result of the strong interaction of borax with hydroxyl group of PVA and complexation between them. 25 These don’t allow organized arrangement of PVA chains and hence reduce crystallization. A sharp peak at 22.6° and the peaks at 15.33° and 16.9° are caused by the crystalline structure of cellulose of Co in the fabric. 25 Intensities of these peaks do not change homogeneously depending on borax concentration since the density of the materials is not homogeneous at a specific thickness.

X-ray diffraction patterns of (a) neat PVA/Co fabric; PVA/Co fabric treated in different wt% of borax/water solutions: (b) 0.1, (c) 0.3, (d) 0.5, (e) 1, (f) 1.5, and (g) 2. Gray dotted line is neat PVA fabric. PVA: polyvinyl alcohol; Co: cotton.
Water loss and swelling percentages of yarn-reinforced hydrogel composites
After keeping the dried yarn-reinforced hydrogel composites in water for 90 min, samples were weighed for every 15 min to investigate weight loss of the samples depending on water evaporation and the results are demonstrated in Figure 5. As seen from the figure, weight loss speed of borax-free neat PVA/Co fabric is higher than borax-treated fabric samples. Increasing borax concentration up to 1 wt% decreases water evaporation rate, because partial cross-linking occurred up to 1 wt%, then continuously increasing borax concentration increases the water evaporation rate from the hydrogel composites as chains are mostly cross-linked after this point.
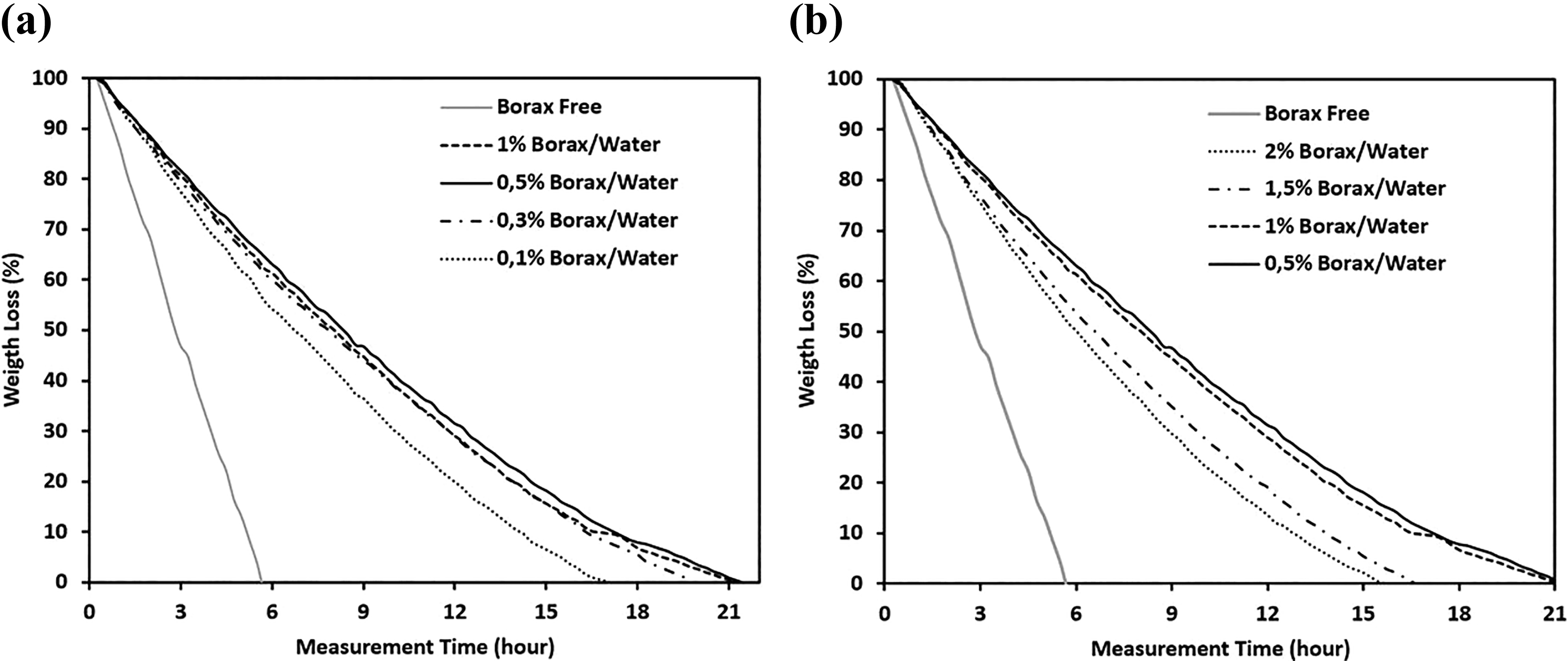
(a) and (b) Water loss percentage by time of different borax ratio–treated yarn-reinforced hydrogel composites.
Swelling percentage depending on cross-linker borax/water concentration and water evaporation from the hydrogel composites by time was calculated from equation (1) as given in the “Materials and methods” section, and the plots are presented in Figure 6. As seen from the figure, the swelling percentage decreases by time at any borax concentration applied to the samples. On the other hand, measuring the different borax concentration–treated samples at the same time frames and depending on the water removal from the hydrogel composites, swelling percentage continuously increases up to 1 wt% and then decreases with increasing borax concentration. Swelling of PVA-based hydrogel depends on the amount of hydroxyl group and the chain mobility in the structure. 6 When PVA molecules are cross-linked, chain mobility and the number of hydroxyl group decrease. Amount of hydroxyl group decreases (can be seen from FTIR results) by increasing cross-linker concentration, but since partial cross-linking occurs up to 1 wt% still chain mobilities are not restricted tremendously and therefore swelling increases. On the other hand, when cross-linker concentrations continuously increased after 1 wt% borax concentration, the swelling percentage decreased as a result of the restriction of chain mobilities and mostly cross-linking of PVA molecules.
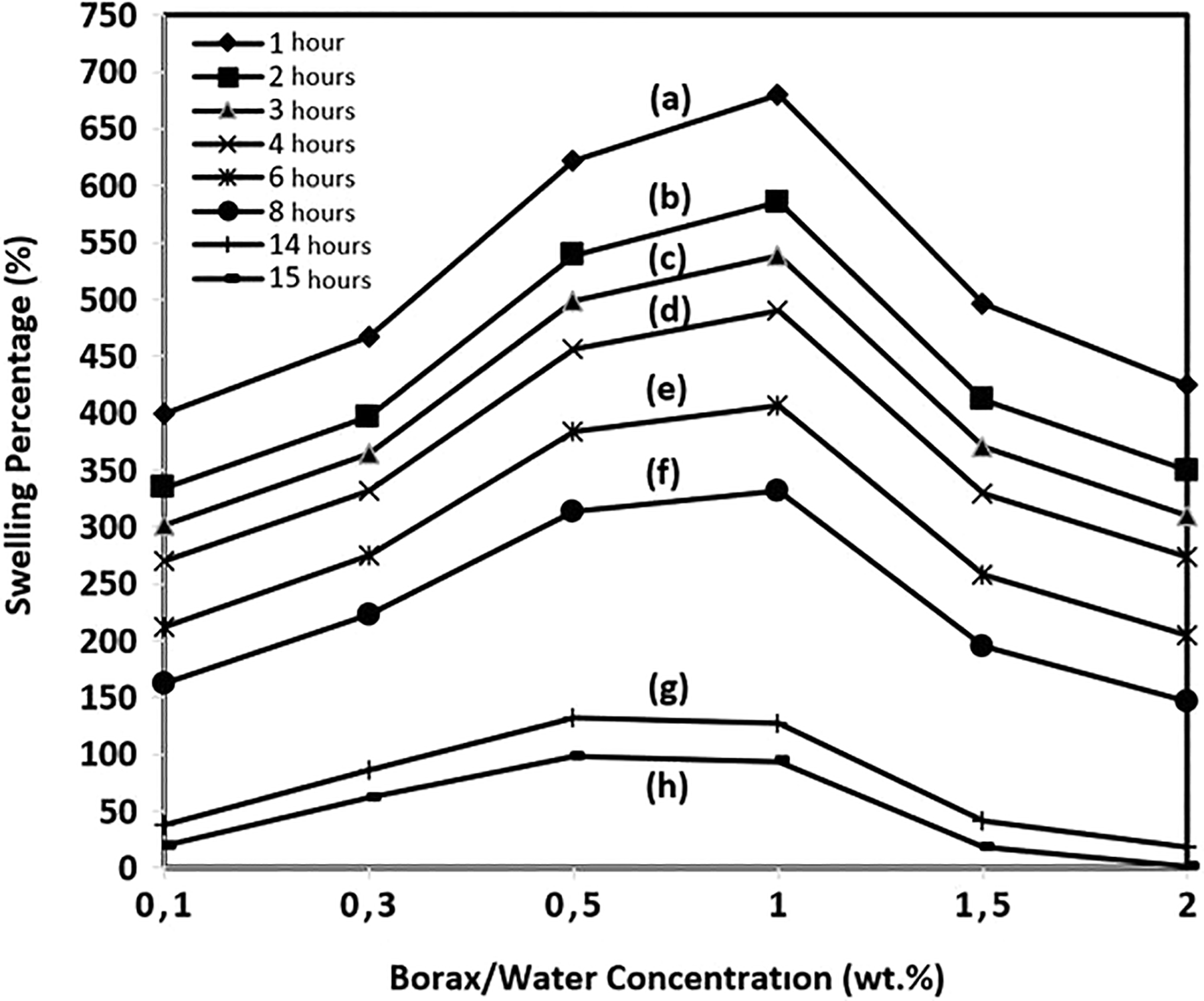
Swelling percentage of the yarn-reinforced hydrogel composites due to borax/water treatment concentration: PVA/Co fabric treated in different wt% of borax/water solutions: (b) 0.1, (c) 0.3, (d) 0.5, (e) 1, (f) 1.5, and (g) 2. PVA: polyvinyl alcohol; Co: cotton.
Mechanical analysis of hydrogel composites due to water contents
Mechanical properties of yarn-reinforced hydrogel composites have been investigated due to the water content of the samples by measuring force-elongation plots in the warp (Figure 7(a)) and weft (Figure 7(b)) directions. As seen from the plots, breaking force tremendously increases when the water content decreases in the samples in both warp and weft directions. Water molecules are inserted among the cross-linked molecular chains and diverged them from each other by swelling the hydrogel structure. On the other hand, when the hydrogel structure releases water, molecules come close to each other and tend to crystallize at certain points. So, breaking forces for the dried samples are higher than the wet samples. Mechanical properties are much higher in the warp direction than in the weft direction since Co yarns are aligned in the warp direction and reinforced the hydrogel composite structure. In order to compare the breaking point depending on water content in both warp and weft directions, the results are demonstrated in the same plots as given in Figure 8. Breaking force in the warp direction was 26.8 N when the hydrogel composite contained 90% of water and increased to 366.3 N when it was fully dried. Breaking force in the weft direction was 1.22 N when the hydrogel composite contained 90% of water and increased to 225.9 N when it was fully dried.

Tensile properties of yarn-reinforced hydrogel composite (1 wt% borax/water treated) containing different content of water at (a) warp and (b) weft directions.

Breaking force comparison of the hydrogel composite sample (1 wt% borax/water treated) in the warp and weft direction due to water content. Inset figure: (a) cotton yarn in the neat fabric before gelation and (b) cotton yarn in hydrogel composite.
Breaking force of PVA/Co fabric in the warp and weft directions are 212.7 and 1055.9 N, respectively. Co yarns withstood against breaking when the measurements were conducted in the warp direction, and PVA yarns withstood against breaking when the measurements were conducted in the weft direction. Since perfectly spun PVA yarns were totally dissolved and then transformed to hydrogel structure after borax/water treatment, a tremendous mechanical loss in the weft direction was observed. Since also some hydrogels were formed among the Co fibers in the Co yarns (inset Figure 8(a) and (b)), Co fibers have been moved away from each other and Co yarn resembles a roving. Because friction decreases between Co fibers and fibers can slide easily in the hydrogel, mechanical properties dramatically decrease with increasing water content in hydrogel composite in the warp direction. Dried hydrogel composites show maximum breaking force in both warp and weft directions since hydrogel turned to a brittle rigid structure in the dry form.
Conclusion
Co yarn-reinforced hydrogel composites were prepared from previously produced PVA/Co fabrics by simultaneous dissolution and cross-linking of PVA with borax/water solution. With increasing borax concentration up to 1 wt%, water evaporation rate decreases, then increases with continuously increasing borax concentration in the hydrogel composites. Breaking force of PVA/Co fabric in the warp and weft directions are 212.7 and 1055.9 N, respectively. Breaking force tremendously increases when water content decreases in the samples in both warp and weft directions. During the drying processes, there was a geometric increase in the breaking force after warp direction sample released 90% of its water content. The geometric increase started for the weft direction sample after it released 60% of its water content. Breaking force in the warp direction was 26.8 N when the hydrogel composite contained 90% of water and increased to 366.3 N when it was fully dried. Breaking force in the weft direction was 1.22 N when the hydrogel composite contained 90% of water and increased to 225.9 N when it was fully dried. Because hydrogels also formed among the Co fibers in the Co yarns in the hydrogel composite, Co fibers have been moved away from each other and Co yarn resembles a roving. As a result, friction between Co fibers is reduced and fibers can slide easily in the hydrogel and therefore mechanical properties dramatically deteriorate with increasing water content in hydrogel composite in the warp direction. Maximum breaking force in both warp and weft directions were observed at dried hydrogel composites since hydrogel turned to a brittle rigid structure in the dry form.
Footnotes
Authors’ note
The study is Umit Koc’s PhD thesis at the Graduate School of Natural and Applied Sciences, Bursa Uludag University.
Acknowledgement
The authors thank Mehmet Tiritoglu for conducting the mechanical test and Batmaz Tekstil Company for providing woven fabrics.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Scientific Research Project Unit (BAP) of Bursa Uludag University with the project number OUAP(MH)-2018/11.

