Abstract
Cotton gauzes coated with chitosan have been tested as water filter for biological disinfection against Gram-positive bacteria. The present study develops the previous ones, aiming to the stability evaluation of ultraviolet-cured chitosan coating of cotton gauzes in order to optimize the chitosan amount applied to the substrate, lowering the add-on percentage as much as possible: 10%, 5% and 2% wt were here considered. Antibacterial activity and permanence of this property after water flow were tested, showing good results, in particular as regards 10% wt and 5% wt chitosan coated cotton gauze. Moreover, chitosan release was studied in both static and dynamic conditions, with the result that water flux causes a reduction of the amount of chitosan, which remains bounded to the fibres. This reduction depends on the duration of the flux and on the original amount of chitosan on the filter.
Keywords
Introduction
Communicable diseases can be transmitted by contaminated water. In many countries, an inadequate management of urban, industrial or agricultural wastewater exposes millions of people to dangerous levels of biological contaminants [1]. For this reason, water decontamination processes aimed to eliminate microorganisms are fundamental. Moreover, it is well known that increasing water demand, deterioration of raw water qualities and stringent regulations of drinking water raise a demand for new treatment processes with better performance, smaller footprint, lower operating cost and highly reliable than the conventional process [2]. For this purpose, previous works regarded a water treatment system utilizing floating media filter [3] or fixed bed filtration system [4].
Chitosan, a d-glucosamine and N-acetyl-d-glucosamine linked by β (1–4) glycosidic bond polymer, is a deacetylated form of chitin, an abundant polysaccharide present in crustacean shells chitosan with degree of acetylation in the range 0.10–0.30 is industrially produced from marine chitin and it is widely available as by-product of the food industry [5]. The chemical structure of chitin is similar to cellulose, having one hydroxyl group on each monomer substituted with ad acetylamine group. In its extracted crude form, chitin has a highly ordered crystalline structure, it is translucent, resilient and quite tough. It has, however, poor solubility and low reactivity. The chitin structure can be modified by removing the acetyl groups by means of a chemical hydrolysis in concentrated alkaline solution at elevated temperature to produce a deacetylated form. When the fraction of acetylated amine groups is reduced to 40–35%, the resulted co-polymer is then referred to as chitosan [6].
Regenerated chitin, chitosan and their chemically modified derivatives combine in themselves biological significance, convenient physicochemical characteristics, in-vivo biodegradability, biocompatibility and antimicrobial action. Chitosan promotes aggregation of bacterial cells and disorganization of the bacterial cell wall and cytoplasmic membrane, thus giving place to the loss of cellular fluids [5].
These structural changes may result in bacterial death, depending on the strains examined, therefore chitosan has been proposed as a textile finishing agent [7,8]. For same reasons, chitosan has been largely used for antitumor applications or as wound dressing material [9].
Besides this wide spectrum of biological activities, chitosan is a promising adsorbent for the removal of heavy metal ions, because of the presence in chitosan chains of many hydroxyl and amino groups, which can act as coordination sites for heavy metal ions, such as copper and chromium ions [10].
All these properties make chitosan an attractive material to coat gauzes for water filtering in different applications: biological disinfection, the removal of dyes from wastewater and in chemical and biochemical field, the adsorption of metal ions. In our previous works, chitosan was applied on cotton, polyester, polyamide, silk and wool gauzes by padding, and grafted by radical ultraviolet (UV)-curing; treated gauzes were than tested as antibacterial and adsorbent medium, with promising results in both cases [7,8,11–14].
In the present work, the same techniques of padding and UV-curing were used to coat cotton gauzes with chitosan. Cotton is composed by cellulose fibres, which can be easily functionalized by radical grafting; for this reason, in addition to its low cost and availability, it was chosen as pristine substrate. The aim of the present study, developing the previous ones and focusing on antibacterial properties, is the stability evaluation of UV-cured chitosan coating on cotton gauzes, in order to optimize the chitosan amount applied to the substrate, lowering the add-on percentage as much as possible (10%, 5% and 2% wt were here considered). For this purpose, the release of chitosan under continuous water flow was investigated, in order to establish the saturation of OH groups of the cellulosic substrate involved in the grafting process. Antimicrobial activity was tested on
Experimental
Materials
A pure cotton gauze fabric, 49 g m−2 with hexagonal holes 2 mm opening, already used in the previous studies on water treatments [7,8], as chosen as substrate.
Chitosan was the pure low viscous product (Sigma Aldrich), 75–85% deacetylation degree, 20–200 cps viscosity value of 1% wt solution in 1% vol acetic acid. It was radically cured adding Darocur 1173 (Ciba Specialty Chemicals) 2% wt as photoinitiator. Chitosan was diluted at 2% wt or 5% wt in a 2% vol acetic acid (Sigma Aldrich) solution, by ripening for 24 h followed by magnetic stirring at ambient temperature for further 24 h. The final pH of the chitosan solutions was about 4.
The bacterium used for antibacterial test was
Samples preparation
A proper amount of the chitosan solution was spread on the gauze with 12-h impregnation time by pouring and the same was dried for about 20 min at 80–100℃. Then the coated fabric was exposed for 60 s to UV radiation under a medium pressure mercury lamp, with irradiance on the fabric of about 60 mW cm−2, at a distance of about 20 cm in a small box equipped with a quartz window flushed by nitrogen (oxygen content lower than 20 ppm). To assure the complete curing of the chitosan on the fabric, it was radiated on both sides.
Three series of samples were prepared: with 2%, 5% and 10% weight gain (2CH, 5CH and 10CH, respectively, hereafter). The weight gain of fabrics was calculated as in equation (1)
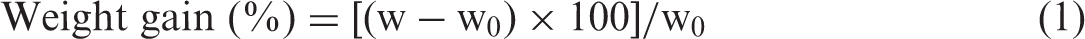
Chitosan coating stability was tested both in static and in dynamic conditions with milli-Q and deionized water, respectively. Considering dynamic conditions, deionized water was continuously flowed through the filter several times, in order to assess the chitosan loss due to water filtration. A scheme of the system was reported in a previous work [7]. It consists of a Pellicon® peristaltic pump (by Millipore), sterile plastic filter holder (25 mm internal diameter) and Materflex® Tygon® LFL autoclavable tubing. The gauzes were cut in 25-mm disks. Three layers of the same fabric were placed in the filter holder. Water volumes of 4.8 ml, 48 ml and 144 ml, for filtering times of 1, 10 and 30 min, respectively, were pumped at 4.8 ml min−1 flow rate in the system. The present test conditions were selected in order to validate the results obtained in a previous work [7]. The samples were stored for at least 24 h at controlled conditions (20℃ and 65% relative humidity) before further testing.
Gauze characterization
The gauzes coated with chitosan were characterized by scanning electron microscopy (SEM), atomic force microscopy (AFM), Fourier-transform infrared (FTIR)-attenuated total reflection (ATR) and TGA. Characterizations were carried out on untreated gauze and on chitosan-coated gauze, before and after water filtration or immersion in water.
The surface morphology of chitosan-treated fabrics was examined by SEM with a Leica (Cambridge, UK) Electron Optics 435 VP scanning electron microscope with an acceleration voltage of 15 kV, a current probe of 400 pA and a working distance of 24 mm. The samples were mounted on aluminium specimen stubs with double sided adhesive tape and sputter-coated with gold in rarefied Argon using an Emitech K550 Sputter Coater with a current of 20 mA for 180 s.
The surface morphology of the samples was examined by an AFM Nano-R2TM Pacific Nanotechnology (USA), equipped with a Si cantilever. Analyses were performed in close-contact (noncontact) mode; the attractive forces acting between the tip and the specimen were measured and topographic images were constructed by scanning the tip above the surface at a distance of 50–150 Å. Scanned images were processed with NanoRuleTM software by Pacific Nanotechnology. Roughness values were obtained as average on at least three different points of samples.
A Nicolet FTIR 5700 spectrophotometer, equipped with a single reflection, monolithic diamond ATR accessory, was used for FTIR-ATR measurements: each spectrum was collected by cumulating 128 scans, at 4 cm−1 resolution and gain 8, in the wavelength range 4000–600 cm−1.
Thermogravimetric analysis (TGA) were carried out on samples, before and after the water fluxing, with a Mettler Toledo TGA-DSC 1 instrument under nitrogen flux (75 ml min−1), temperature range from 30 to 500℃, with heat rate of 10℃ min−1.
As regards static conditions, a functionalized gauze was immersed in milli-Q water (pH 5.6) for at least 24 h, under stirring, with a liquor ratio of 0.2:10 and 1:10 g ml−1. The drying was carried out at 50℃ for 24 h and finally the samples were stored for at least 24 h at controlled conditions (20℃ and 65% relative humidity) before further testing. In order to solubilize the chitosan present in solution, acetic acid was added at a final concentration of 1% v/v; the resulting solution was stirred for at least 12 h.
Chitosan release test on water
Viscosity of water after contact with chitosan-coated fabrics was measured using a cone-on-plate Anton Paar Physica MCR 301 rotational rheometer (Austria).
About 2 ml of solutions were placed on the surface of the plate using a calibrated micropipette; the surface of the cone-plate system was maintained at the temperature of 25℃ using a heat-exchanger Heater Unit H-PTD 200 (Anton Paar, Austria). The spindle of the cone-plate system was equipped with a cone model CP 75-1 with a diameter of 75 mm and an angle of 1°.
Viscosity of solutions was assessed in a range of shear rate from 100 to 1000 s−1 (Figure 1(a)). The calibration curve (Figure 1(b)) was prepared with different concentrations of chitosan (0.01, 0.05, 0.10, 0.25, 0.10, 0.25 and 0.50% wt) in aqueous solutions of acetic acid at 1% v/v. It shows a linear trend in the considered range of concentrations.
Viscosity measurements of chitosan solutions with different concentrations of chitosan (0.01, 0.05, 0.10, 0.25, 0.10, 0.25 and 0.50% wt) in aqueous solutions of acetic acid at 1%v/v (a) and calibration curve (b).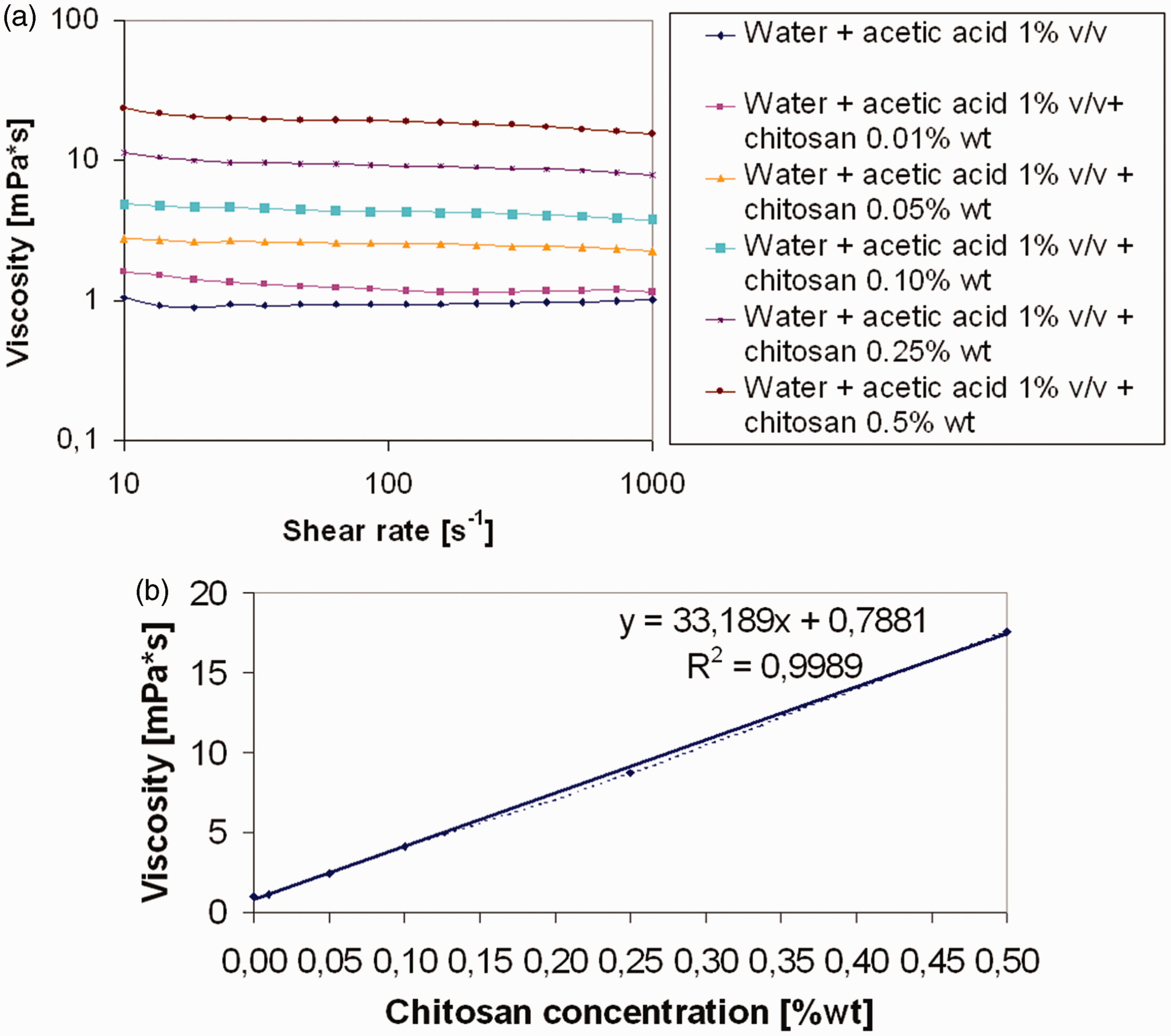
Antibacterial activity
Antibacterial activity was evaluated before and after water filtration following the antibacterial standard AATCC Test Method 100–2012, which is a quantitative method for the assessment of antibacterial finished textile materials.
The procedure is briefly described in here. Test bacterium culture grown for 24 h in a suitable nutrient broth was diluted to give a concentration of 1.5–3.0 × 105 CFU ml−1 (inoculum). A specimen of each fabric (about 1 g) was transferred to a flask. 1.0 ± 0.1 ml of the inoculum were added on each specimen. All flasks were incubated at 37℃ for 1 h (contact time). After incubation, 100 ± 1 ml of a buffer solution at pH 7 were added in each flask and shaken vigorously for 1 min. Total of 1.0 ± 0.1 ml of the solution was diluted 1:10 in the same buffer solution. In all, 1.0 ± 0.1 ml of diluted solution was plated in nutrient agar. In this way, a maximum of 150–300 CFU were transferred to each plate. The inoculated plates were incubated at 37℃ for at least 24 h and surviving colonies were counted. The tests were performed in duplicate. The results are the average of two separate counting.
The antibacterial activity is expressed as percentage reduction of the micro-organisms after contact with the test specimen (A) compared to the number of bacterial cells originally present in the dilute inoculums solution contacted with each specimen, the so-called ‘0’ contact time (B), according equation (2)

Results and discussion
Morphological analysis
In Figure 2, SEM images of untreated (Figure 2(a)) and chitosan coated (Figure 2(b) to (d)) samples are reported. The formation of macroscopic aggregates, ascribable to chitosan deposition, can be observed. In addition, these aggregates do not present a homogeneous distribution on the cotton fibre. On the other hand, after immersion in water, SEM images show that some aggregates are still present on the gauze, even after 30 min of water flowing (Figure 3). Hence, in the adopted experimental conditions, they were not completely removed by the contact with water.
SEM images of untreated cotton gauze (a), 2% (b), 5% (c) and 10% (d) chitosan add-ons, freshly prepared. SEM: scanning electron microscopy. SEM images of 2% (a) (b), 5% (c) and 10% (d) chitosan add-ons, after 30 min of water flowing. SEM: scanning electron microscopy.

In Figure 4, AFM analyses, carried out on 4.0 µm2 area, are reported. They show how the surface morphology of cotton fibres changes when chitosan is deposited. The functionalization process forms a thin layer that cannot be observed by SEM; it indicates that aggregation is not the only form of chitosan deposition present during the process. The results of average roughness are showed in Table 1. The average roughness decreased with the growth of chitosan amount from ∼600 nm to ∼50 nm, for untreated cotton and 10CH, respectively. As a result, by increasing the content of chitosan, a homogenous coating is formed on the cotton fibre, in fact the most uniform values are represented by 5CH and 10CH, with the lowest standard deviation.
AFM images of untreated cotton gauze (a), 2% (b), 5% (c) and 10% (d) chitosan add-ons, freshly prepared. AFM: atomic force microscopy. Average roughness from AFM analyses. AFM: atomic force microscopy.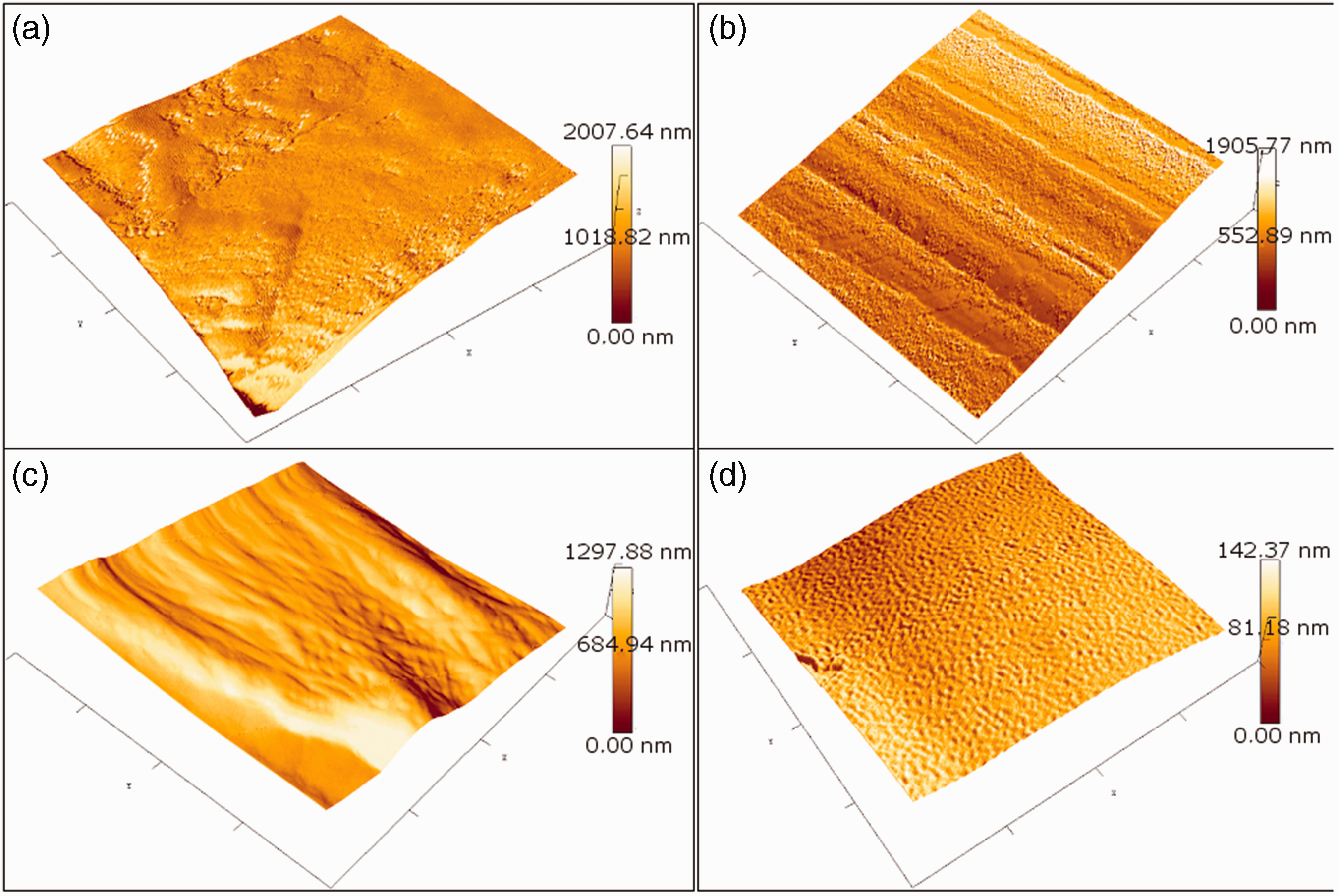

In addition, results of water immersion in dynamic conditions, for 10CH, 5CH and 2CH, are also reported in Table 1. The average roughness of 10CH grows after tests until a value close to 5CH treated sample, while the roughness of 5CH and 2CH rise to higher values near the one of the untreated sample. It means that a certain amount of chitosan that coated the samples was removed by water, but 10CH maintains a higher residual amount of chitosan.
The result of water immersion of 10CH sample, in static conditions for 24 h, shows that a longer time is necessary to remove chitosan from the surface of cotton, in comparison with dynamic conditions.
Infrared spectroscopy
The FTIR-ATR analysis was first carried out on freshly prepared samples, with different chitosan add-ons, and compared with untreated substrate to find peaks identifying chitosan (Figure 5).
FTIR-ATR analysis on freshly prepared samples, with different chitosan add-ons, and untreated substrate (a); magnification in the wavelength range useful to find peaks identifying chitosan (b). FTIR: Fourier-transform infrared; ATR: attenuated total reflection.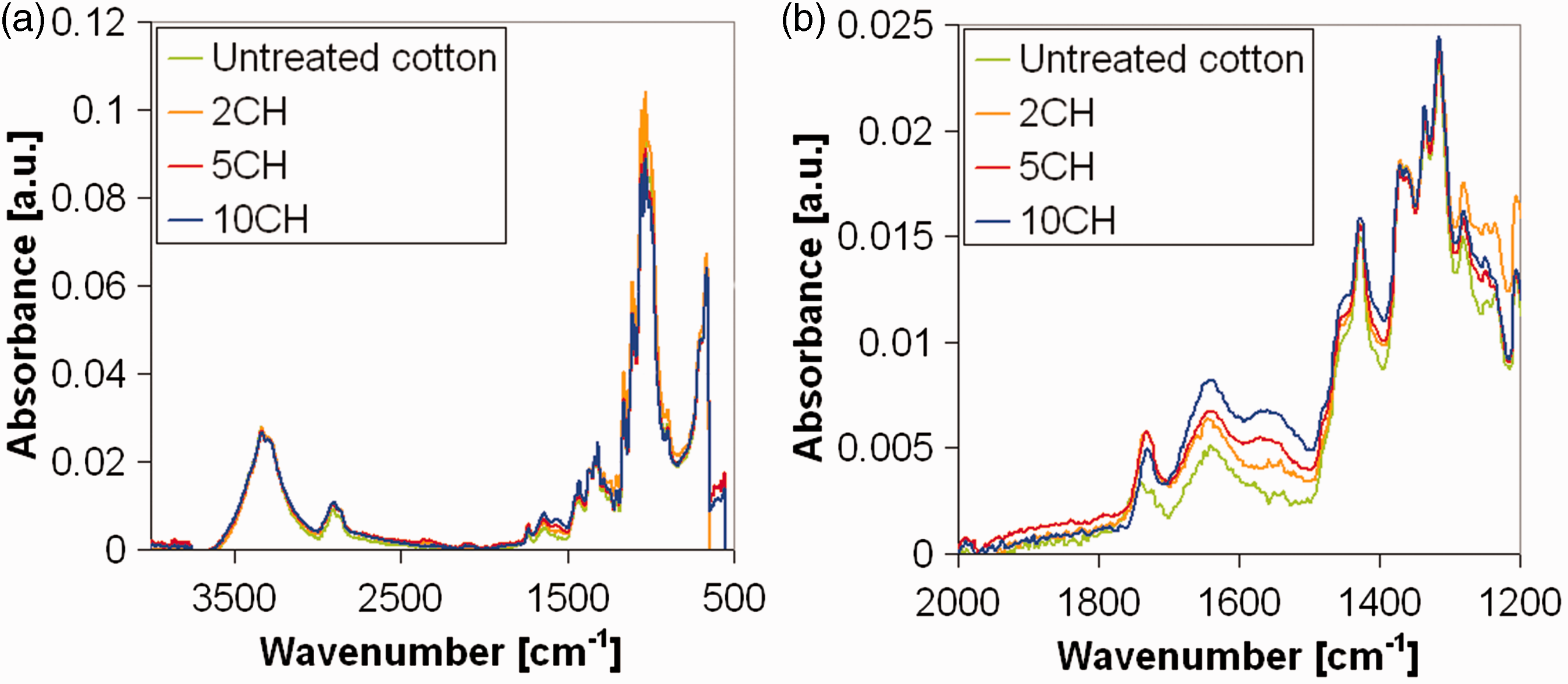
The comparison of FTIR-ATR spectra of untreated and 10CH weighted cotton filter shows the evidence, in the spectrum of the treated fabric, of chitosan peak at 1560 cm−1 attributed to the bending of N–H amide group, in accordance with results previously reported [7,8]. The other peaks can be due both to cotton and chitosan component of the sample, while the band at 1644 cm−1, corresponding to C=O vibration of the acetylated units [16], was due to the adsorbed water [17].
The chitosan peak is still evident on the spectrum of the sample 5CH, while the spectra of untreated cotton and 2CH are comparable, since the chitosan amount is likely too low to be detected by FTIR-ATR analysis. It means that the efficiency of the method is limited in detecting the chitosan presence on the treated filter: add-ons lower than 5% cannot be revealed by this kind of analysis.
On 10CH sample, FTIR-ATR was carried out after water fluxing through the filter for 1, 10 and 30 min. In this way, if chitosan molecules are not strongly bound to the fibres, they are physically removed by water flux. The peak of chitosan at 1560 cm−1 is reduced after 1 min of water flux (Figure 6). It means that water removed just some surface chitosan, not chemically grafted to the cellulosic substrate.
FTIR-ATR on 10CH sample after water fluxing through the filter for 1, 10 and 30 min or static water immersion for 24 h. FTIR: Fourier-transform infrared; ATR: attenuated total reflection.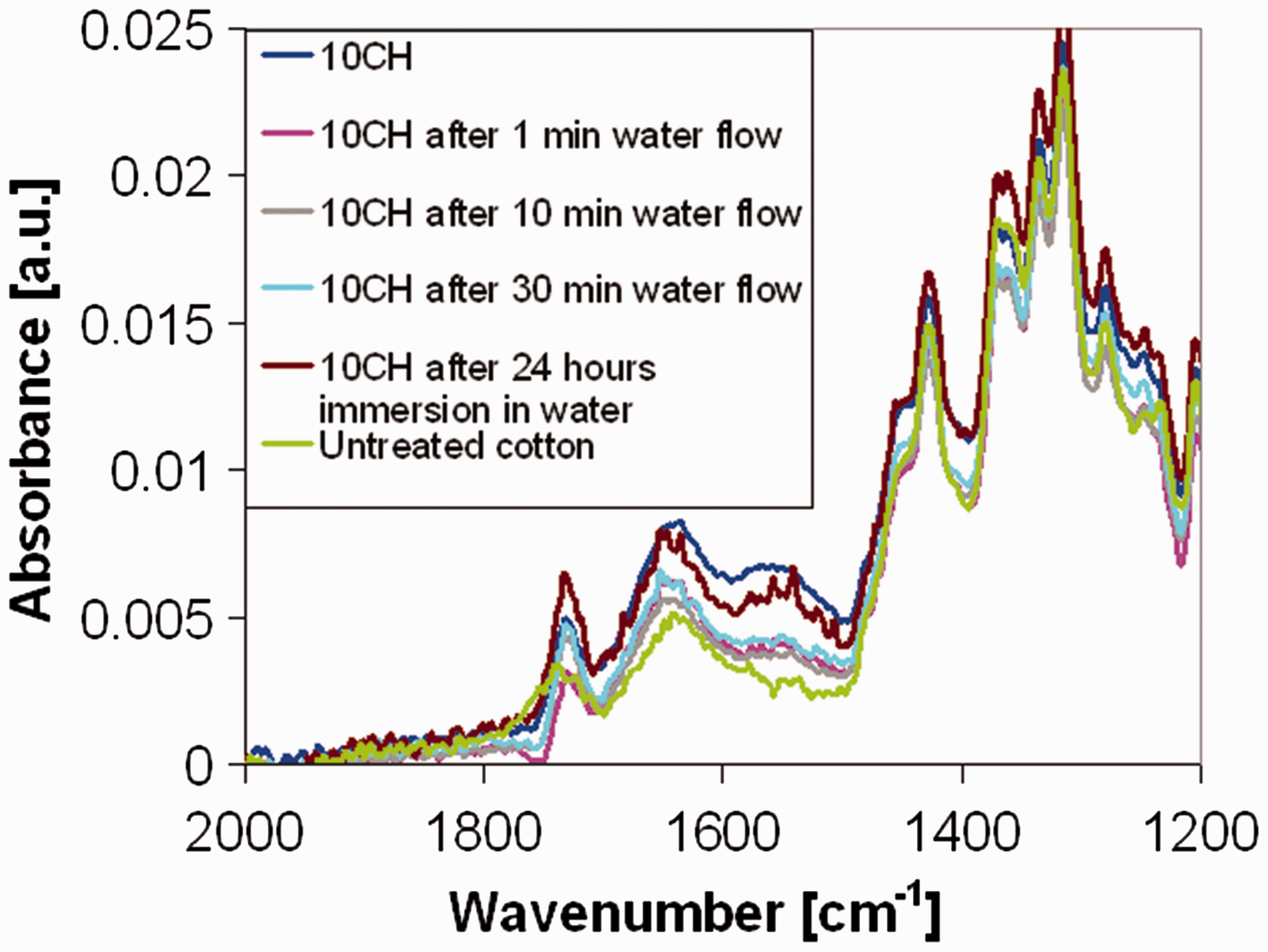
FTIR-ATR analysis was carried out also after 24 h immersion in milli-Q water. It causes a reduction of the intensity of the characteristic peak of chitosan at a lower extent than the water flux. As expected, compared with the only immersion in water, the dynamic water flux has a more intense effect on weakly bound chitosan molecules. In Figure 6, the spectra related to 10% chitosan add on are reported but, with respect to dynamic water flux, a similar effect was found also on 5% treated samples.
Thermogravimetry
The presence of chitosan on treated filters was assessed by residual weight after thermal analysis at 500℃. For the sake of brevity, pictures of the thermograms have not been reported, but the results are described in the following paragraph.
The residual percentage evaluated by TGA on 5CH and 10CH samples is in good agreement with the applied add-ons. Both untreated cotton and 2CH have a residue close to 10%, whereas 5CH and 10CH have 16 and 19%, respectively.
TGA analysis was repeated on 5CH and 10CH samples after 30 min of flowing since data on residues are consistent with the theoretical add-ons. 10CH sample reported a residual weight percentage of 16% after 30 min of water flowing. Then, the residual percentage of samples decreased probably when the presence of chitosan fixed to the fibres is reduced. The residual weight percentage of 5CH was 15% after 30 min of water flowing (close to the value before flowing).
Since the residual weight of 10CH after 30 min of water flowing is close to the residual weight of 5CH, it can be supposed that there is a limit value of chitosan add-on (i.e. 5CH), over which macromolecules cannot be well fixed on the fibres, enough to resist fluid force under dynamic conditions.
Viscosity
As previously reported, in order to determine the amount of chitosan not strongly bounded to the fibres, the viscosity of the water after the immersion of the 10CH sample was measured (Figure 1). It can be related to the chitosan amount solubilized, that is, the amount released during the sample immersion.
Viscosity measurements showed that the release of chitosan is negligible (weight loss <1% wt on the chitosan add-on), in good agreement with thermogravimetric results.
Antibacterial activity
Antibacterial activity of cotton filters treated with greater percentage of chitosan add-ons has been proved in previous works [7,8]. In this work, the preservation of biocidal effect of 2CH, 5CH and 10CH samples, before and after water flowing, was examined following the AATCC 100 Test Method against
Antibacterial activity of 2CH, 5CH and 10CH cotton filter before and after water flowing.

Conclusions
A cotton gauze was coated with chitosan, by UV-curing, at 2%, 5% and 10% of chitosan add-ons and tested as antibacterial medium for water filtration. All the samples showed good antibacterial activity against Gram positive microorganisms; in particular, the two latter samples showed bacterial reduction of 100%.
The persistence of the antimicrobial activity of the samples after 30 min of water flowing was also evaluated, obtaining good results: 95, 98 and 99% of bacterial reduction for, respectively, 2%, 5% and 10% chitosan-treated samples.
In view of a possible real application of these filters, the effects of batch and dynamic conditions on treated samples were studied: while the immersion in water is not able to remove the chitosan not strongly bounded to the fibres, water flux causes a reduction of the amount of chitosan on the fibres. This reduction depends on the original amount of chitosan on the fibres, as resulted from FTIR-ATR and TGA analysis, but it does not affect significantly the antibacterial properties of samples, as demonstrated by AATCC 100 test, carried out with
Footnotes
Acknowledgement
The authors thank Prof. Franco Ferrero (Politecnico di Torino, DISAT) for the fruitful discussion on obtained results.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
