Abstract
Modifying cotton fabrics to obtain significant new properties is of relevance to creating multifunctional textiles that could address challenges across different sectors. One of the critical challenges associated with textiles is hospital-acquired infections, which could be prevented through the utilization of antimicrobial fabrics. Titanium dioxide (TiO2) nanoparticles (NPs) have been introduced in literature for their photocatalytic antibacterial applications against prevalent microorganisms, such as
Introduction
Hospital-acquired infections constitute a major problem for hospital administrations and health authorities worldwide. Cotton-based hospital linens and healthcare providers clothing are major sources for nosocomial infections, which could be prevented through the utilization of antimicrobial fabrics. Several technologies have been investigated to introduce antimicrobial capabilities to textiles such as coating with triclosan, 1 phenolic derivatives 2 and silver. 3 However, coating durability has been a challenge and the release of such antimicrobial agents during the use of these fabrics may affect the safety of patients and the environment. 4 One of the promising methods towards achieving effective and robust antimicrobial cotton fabric is through the introduction of nanoparticles (NPs) to the surface of the fabric. 5 The intrinsic properties of cotton such as wettability, flexibility, porosity, absorbance, and layered surface, enables the introduction and attachment of NPs while benefitting from its various functionality.4–7
TiO2 NPs in particular has been an attractive additive to textiles due to its non-toxicity, cost-effectiveness, heat-resistance, and its outstanding photocatalytic antimicrobial activity that leads to the decomposition of bacterial outer membranes and hence cell death.7–12 Photocatalytic materials (including TiO2 NPs) are extremely effective against microbes such as Illustration of the UV effect on photocatalytic TiO2 NPs, forming reactive oxygen species that act against microbial cells and lead to cell death.
TiO2 coating techniques have been utilized in several studies to prepare superhydrophobic coating on cotton fabric,
24
prepare fabrics with temperature regulating properties,
25
and functionalizing cotton fabric for self-cleaning and antibacterial property enhancement.26–28 It has been proven that cotton fabrics treated with TiO2 can achieve significant removal of
In this work, a newly developed and scalable TiO2 NPs coating process is introduced for the realization of cotton textiles with durable and highly effective antibacterial properties. The process in the present study utilizes 25 nm in diameter TiO2 NPs with anatase crystal structure as the main additive to be dispersed homogeneously on the fabric. The particle size was selected to benefit from the high surface-to-volume ratio which has direct impact on the photocatalytic activity of the NPs as well as the affinity of the NPs to adhere to the surface of the fabric during the coating process.34–36 The coating process contains three distinct phases which includes chemical modification for the surface of the fabric as a pre-treatment step, followed by the introduction of NPs dispersed in a carrier solution, and then ends with fabric drying and UV radiation exposure for nanoparticle activation to obtain the desired antimicrobial characteristics.
The key differentiator lies in the utilization of TiO2 NPs attached to the fabric directly through chemical bonding with eliminating the need of using binding additives. The pre-treatment step enhances the presence of negatively charged OH* groups on the cotton fiber (typically ionized in aqueous phase). The positively charged TiO2 NPs are firmly attached to the fiber through hydrogen bonding and van der Waals forces through the interactions between the negatively charged hydroxyl groups on cellulose fiber and the positively charged TiO2 NPs surface.
TiO2 coating durability has been investigated in previous studies which proves the potential for TiO2 NPs to robustly adhere to textile surfaces while showing significant effect against various microbes such as Gram-positive and Gram-negative bacteria as well as fungi.37,38 Investigations were carried out for the durability of the TiO2 coating on the cotton fabric, which was investigated by performing washing cycles following internationally recognized protocols, and then conducting thorough qualitative and antibacterial analysis.
Thorough characterization was conducted on the treated cotton fabric to investigate the coating quality. The morphology and quality of the coating was evaluated using scanning electron microscopy (SEM), and energy dispersive X-rays spectroscopy (EDS). The adherence and the robustness of TiO2 NPs on the fabric were verified as well by SEM. The antimicrobial effect of TiO2 coating was examined against the most important pathogenic bacteria, such as
Materials and methods
Chemicals and materials
Commercial titanium dioxide nanoparticles (TiO2 NPs), P25 were obtained from Sigma-Aldrich (average particle size: 25 nm, purity ≥99.5% trace metals basis, crystalline phase: 80% anatase 20% + rutile).
All other chemicals used in the study were commercially available high analytical grade reagents (Glacial Acetic acid with ≥99.8% purity, Sodium hydroxide pellets, and ≥99.8% purity Methanol).
Preparation of TiO2 NPs suspension and coating process
In this work, the newly developed coating process allows direct attachment of TiO2 NPs to the cotton fabric without relying on a binding agent. TiO2 NPs tend to have a high affinity towards organic acids, hydroxyl groups, and alcohols; therefore, introducing higher amounts of these stabilizing and fixative chemicals as an organic matrix can lead to a higher adsorption rate of TiO2 on the surface of cotton fabric. The good adhesion mechanism between TiO2 nanoparticles and the selected cotton fabric is due to the internal hydroxyl (-OH) group presented in the chemical formula of the cotton and in the acid-alcohol medium. Thus, TiO2 NPs may easily bind to hydroxyl groups through hydrogen bonding.
Cotton fabrics (100% cotton, with weight of 125 gsm (g/m2), and thread count of ∼46 thread/cm2) was utilized in this work. To eliminate any contaminants or organic residue that could affect the adhesion of NPs, the fabric was first cleaned with a commercial detergent at 100°C for 15 min, then rinsed with distilled water twice at room temperature (25°C) for 30 min. The coating process was conducted on cotton fabric samples weighing 5 ± 0.1 g.
A fabric pre-treatment chemical modification step was carried out using inorganic base solution (4M NaOH) for 20 min at room temperature using dip coating method. The purpose of the surface pre-treatment is to avail the hydroxyl radicals on cotton which helps the TiO2 NPs adhesion to the fabric. When NaOH is added to cellulose (cotton), it causes the cellulose fibers to swell and become more accessible to chemical reactions. 40 A 10-s distilled water rinse step is then applied to wash away excess NaOH residue from the fabric which could act as a barrier that partially insulates the surface of the fabric from the NPs suspensions and hence affect the coating quality.
The main coating step to introduce the TiO2 NPs to the pre-treated fabric utilized a mixture of 300 ppm by wt.% Anatase TiO2 NPs diluted in 1000 mL colloidal solution. The solution contains deionized water, and methanol: glacial acetic acid (a ratio of 3:1 by volume). Solution mixing was carried out in an ultra-sonication water bath to ensure proper mixing for 15 min. The fabric is then dip-coated in the solution and sonicated for 10 min to reduce agglomerates and introduce the NPs to the pre-treated fabric. During the neutralization step, pH was adjusted using HNO3 to a pH of 6.8-7.2. Fabric drying was then conducted at 100°C–110°C in a dry oven to ensure thorough evaporation of solvents, leaving the TiO2 NPs only on the fabric. Finally, the samples were subjected to ultraviolet light (UV light; λ = 365–405 nm (UV-A); I = 5000 mW cm−2)) for 15 min at a distance of 10 cm from the textile to activate the photocatalytic TiO2 NPs and enhance the antimicrobial effect. The treated cotton fabrics were then ready for characterization and antibacterial performance testing. The coating process for preparing the fabrics is demonstrated in Figure 2. Preparation steps for TiO2 NPs coated antibacterial cotton textile. (1) The surface of the cotton fabric is first modified with an inorganic base solution to obtain the hydroxyl (-OH) group on the fabric which helps the TiO2 NPs adhesion to fabric through hydrogen bonding. (2) A distilled water rinse step to wash away excess NaOH from fabric. (3) A colloidal solution containing Anatase TiO2 NPs was prepared using glacial acetic acid and methanol as carrier solutions mixed in distilled water. The fabric is dip-coated in the solution and sonicated to reduce agglomerates and introduce the NPs to the pre-treated fabric. (4) pH adjustment and Acid neutralization using HNO3 [pH: 6.8-7.2]. (5) Fabric drying at 100°C to ensure evaporation of solvents, leaving the TiO2 NPs only on the textile. (6) UV activation of the photocatalytic TiO2 NPs (to maximize antimicrobial effect).
Morphological, structural, and chemical analysis
Scanning electron microscopy (SEM) (Model SEM: TESCAN VEGA 3, Czech Republic, operated at 20 kV) equipped with energy dispersive X-ray spectroscopy (EDS) detector, was carried out to evaluate the surface morphology and structure of the TiO2 coated fabrics, and the distribution as well as density of the NPs clusters on the fabric. SEM imaging was carried out on raw cotton samples as well as coated samples with a 300 ppm TiO2 solution concentration for direct comparison. All specimens were analyzed after sputter coating with 5 nm of gold to minimize the charging effect of the non-conducting nature of the fabrics and hence increase the quality of the analysis and imaging. Prior to preparing the coating solution, the average size, shape, and structure of the TiO2 nanoparticles was determined by transmission electron microscopy (TEM) (TEM Model: Morgagni 268, FEI, Czech Republic, operated at 80 keV).
Antibacterial performance testing
Antibacterial testing following ISO 20743:2013 protocols (Determination of antibacterial activity of textile products) was performed against Gram-positive and Gram-negative bacteria to understand the antibacterial efficacy of fabric treated with antibacterial agent. This is conducted by directly exposing the bacteria to the treated surface of the fabric and then counting the colonies to estimate the number of surviving bacterial cells. The preparation process is illustrated in Figure 3. Antibacterial testing procedure for cotton fabric. The antibacterial concentration is obtained using the colony forming units (CFU) and the respective dilution rates.
Bacterial Inoculum were prepared by growing a fresh 18-24 h culture in sterile nutrient broth for each series of samples. This is followed by diluting the culture with a sterile phosphate buffer solution (pH 7.4) until the solution has 1 × 10
The fabric pieces are obtained with a mass of 0.40 g ± 0.05 g and cut to suitable size. Six test samples of control fabric and six test samples of antibacterial treated fabrics are used. 0.2 mL of the inoculum is pipetted to the vial containing the test sample. Just after the inoculation (0-h), 20 mL of Soya Casein Digest Lecithin Polysorbate Broth (SCDLP) media is added to each of the vials and incubated at 37 ± 2°C for 18-24 h 20 mL of SCDLP media is added to each vial after incubation. 1 mL of each dilution is added to two sterilized petri dishes. Agar media is added and incubated for 24-48 h. The bacterial concentration, is obtained using the following formula:
The number of colonies in the serially diluted petri dish are counted for the reference sample at 0-h and 18-h, as well as the number of colonies for the treated sample after 18 h. The number of bacteria obtained by the counts of colonies is measured according to the following formula:
Growth value would be estimated by comparing the 0-h count to the 18-h count for the reference sample. The growth value obtained according to the formula shall be ≥ 1.0 in the plate count method.
The antibacterial activity value would be obtained by comparing the 18-h reference count to the 18-h treated sample count using the following formula:
Coating durability analysis
The stability and the adherence of the NPs coatings were evaluated through washing the specimen for different cycles at 10x, 20x, and 40x washing cycles. Washing was conducted as per ISO 6330:2021 protocols: Domestic washing and drying procedures for textile testing. The procedures are applicable to textile fabrics which are subject to combinations of domestic washing and drying procedures. The test is a commonly used standard within the textile testing sector that defines test materials and specifies standardized washing and drying conditions as well as the reference detergents. In this study, the washing temperatures are fixed at 70°C with normal washing strength and with applying line air drying method (LAD). SEM imaging as well as antibacterial testing were conducted after the washing cycles to test the performance and assess the NPs durability on the fabric in reference to the unwashed coated fabrics.
Results
Morphological and structural analyses of TiO2 NPs
Before preparing the coating solution, the structure and morphology of the TiO2 NPs were analyzed by TEM using drops of NPs suspension in carrier solution dispensed on the TEM microgrids. As shown in Figure 4, TEM images revealed spherical shaped particles with some extent of aggregation due to the nature of the nanoparticles. It was observed that the size of the NPs varied between 10 and 70 nm, and the average size was calculated as 25 nm which is consistent with the average size reported by the manufacturer. TiO2 NPs exhibited a polycrystalline structure when analyzed by electron diffraction recorded in the TEM. The main characteristics planes of the TiO2 NPs were labeled in the pattern. Furthermore, roughly 50-70 nanoparticles were clearly counted in 500 nm2 area, which helped to estimate the number of particles on the surface of the coated cotton fibers. Transmission electron microscopy (TEM) images (A-B) along with corresponding (C) electron diffraction patterns showing the anatase crystalline structure of the TiO2 NPs, and (D) size histogram. The average diameter of the particle was found ∼25 nm.
NPs coating analysis
SEM was used to analyze the presence, morphology, and quality of TiO2 NPs coating on cotton fibers. The qualitative assessment could be considered as an underestimation for the coating quality and NPs presence due to the fact that only NPs clusters larger than 200 nm could be detected easily with SEM considering the applied settings and respective field of view. The SEM results of the coated and uncoated (control) cotton fabrics can be found in Figure 5. The raw cotton was examined under different magnifications in order to evaluate the morphology, estimate the diameter of the individual fibers of the cotton and obtain visual comparison between the uncoated and coated fabrics. SEM micrographs reveal a clear and smooth morphology of the cotton fibers with fiber thickness in the range of 10-15 µm (Figure 5(a)). (Figure 5(b)) shows clearly the NPs coating on the fabric as brighter particles. Overall, a uniform distribution of TiO2 NPs and a complete coverage on the fibers was observed with clusters smaller than 2 µm in diameter found on the fibers, confirming the successful preparation of the coated cotton fibers. It was also observed that no other chemical residue is visible on the fabric which could happen from having poor rinsing steps that leads to the formation of a film that prevents the NPs from interfacing with the fabric. SEM images of uncoated (a) and coated (b) cotton fabrics at different magnifications. Coated fabric shows clearly theTiO2 NPs clusters coverage and distribution.
To estimate the added mass of NPs to the coated fabric and the potential effect on density and flexibility, the SEM images for the coated fabric were analyzed using ImageJ software to estimate the coverage area of NPs on the fabric. 41 ImageJ allows estimating the surface area of the NPs, where the mass of the NPs was estimated by calculating the total volume of the NPs and using the density of TiO2 NPs that is equivalent to 3.9 g/cm3. The analysis shows 14% area coverage of NPs on each side of the fabric, which results in a total NPs mass of 1.6 µg per cm2 of coated fabric. This contributes to less than 0.012% of the total weight of the fabric.
The durability of the coating would be subject to the strength of the TiO2 NPs bonding to the fabric since the NPs in this work are attached to the fabric directly through chemical bonding without using binding additives. The TiO2 NPs are believed to be firmly attached to the fiber through hydrogen bonding and van der Waals forces as a result of the interactions between the negatively charged hydroxyl groups on cellulose fiber and the positively charged TiO2 NPs surface. The durability of the TiO2 NPs coating was assessed by washing the coated samples for 10x, 20x and 40x following ISO 6330:2021 protocol (Domestic washing and drying procedures for textile testing) and capturing the SEM images for the samples after the washing periods to compare the existence and density of the TiO2 NPs to the as coated samples. The results can be found in Figure 6. It was observed that a significant amount of the TiO2 NPs remained attached to the cotton fibers even after washing 40 times, confirming the durability of the coating. It was also observed that most of the lost NPs after washing were the large clusters which highlights the importance of clusters size reduction on the coated fabric for further durability enhancement. It is believed that the non-visualized individual and small cluster NPs are strongly attached to the fabric even after the washing cycles, providing significant contribution to the durability of the antimicrobial performance. SEM imaging of TiO2 coated fabrics as prepared (a), after 10x washing (b), after 20x washing (c), and after 40x washing cycles (d). Washing was conducted as per ISO 6330:2021 protocol. NPs adhesion and distribution are visible even after excess washing, with noticing the slight reduction of NPs clusters on the fabric after washing.
The chemical composition of coated and uncoated cotton fabrics was analyzed by SEM/EDS in order to investigate and confirm the presence of TiO2 NPs and compare the results with the washed samples (Figure 7). The uncoated cotton showed the typical spectrum of raw cotton with its organic components, carbon (C) and oxygen (O). Au peak appeared due to the gold film coating on the samples. No other significant peaks were observed in the spectrum of the uncoated samples. The coated samples showed additional peaks of Ti, along with C and O, confirming the successful deposition of TiO2 nanoparticles on the surface of fabrics. It was observed that the treated and 40x washed fabrics also showed a peak of Ti, but with a slightly lower intensity, indicating the presence and durability of the NPs on the fabric with slight reduction in the concentration. SEM images and EDS analysis of (a) uncoated, (b) TiO2 coated, and (c) coated/40x washed fabrics.
Antibacterial properties assessment
The effectiveness of the coated fabric against microbes such as
The antibacterial properties for the TiO2 NPs-coated fabrics were tested following ISO 20743:2013 protocols. Samples of treated cotton fabric were exposed to Control versus treated fabrics exposed to 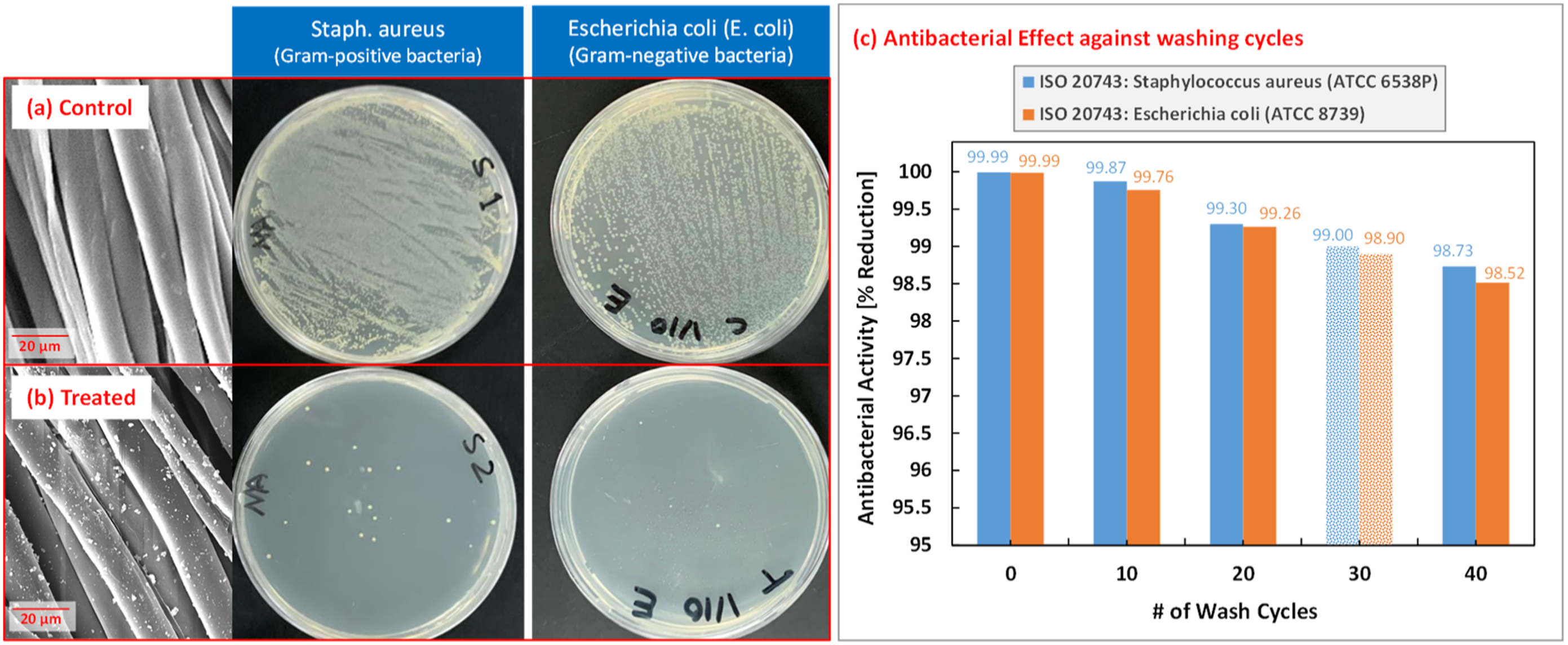
Discussion
The newly developed antibacterial textile coating process facilitated the attachment of the 25 nm photocatalytic TiO2 NPs on the fabric as the main active substance acting against microbes without using binding additives. The homogenous distribution of the NPs on the cotton fabric was successfully demonstrated and thoroughly characterized. The smaller the NPs (<50 nm), the coating coverage on the fabric becomes more efficient and the photocatalytic performance becomes more effective due to the high surface area to volume ratio, and hence exhibiting better antibacterial activity at low NPs concentration.11,12 Achieving coating consistency with high reliability over large fabric areas is critical for maintaining the outstanding antibacterial performance obtained from the tested samples to be suitable for applications in the healthcare sector. The presented process followed strict procedures to ensure proper NPs distribution in the coating solution as well as on both sides of the cotton fabric after coating.
The NPs coating resulted in an increase of fabric weight by less than 0.012% with the coating being in the form of separated NPs clusters on the fabric that are smaller than 2 µm in diameter and as small as 25 nm. In general, applied coating can influence the tensile strength and the flexibility of the fabric due to the added mass of the coating and the slight density increase. However, due to the small size of NPs in the reported coating process and the formed clusters that are separately attaching to the fabric, in addition to the fact that no binding layer was utilized, the effect on flexibility and tensile strength is expected to be minimal.
Epidemic species of Gram-negative and Gram-positive bacteria were tested in this study with the TiO2 coated fabric, and the results were found to be exceeding many of the growth inhibition percentages found in literature. Previous studies reported various antibacterial performance for TiO2 treated fabrics with up to 99.9% growth reduction percentage.26,32,42,43 The ISO compliant antibacterial testing in this work demonstrated significant growth reduction percentage of 99.99% against important pathogenic bacteria such as
NPs cluster management was also a critical aspect that was enhanced in the process by controlling the intermediate steps of the coating process that helped achieving cluster reduction such as the effective ultrasonication during solution preparation and during the coating of the fabric. SEM imaging results for the washed samples showed the likelihood of the large NPs clusters peeling off from the fabric with excessive use or when exposed to friction such as washing. This reduction in NPs concentration can lead to slight reduction in the durability of the coating and, hence, reducing the antibacterial effectiveness. Despite the outstanding performance presented in this work, further optimization is possible for durability enhancement by reaching sub-micron level NPs clustering with maintaining the homogenous distribution of the coating. Quality measures across the treated fabric were applied through visual SEM inspection as well as antibacterial testing to reach the presented parameters, and to set a baseline when moving towards scaling up the coating process to an industrial scale. Additional parameters and quality measures for the antibacterial coating relevant to the performance with colored fabrics, different fabric types, and different fabric characteristics are planned for investigation in future work.
The coating process demonstrated the potential for commercial utilization in the healthcare and hospitality sectors, among others. The outstanding and durable antibacterial performance of the treated cotton fabrics in addition to the scalability and cost effectiveness of the treatment process can lead to increasing the possibility of acquiring a significant market share of the ∼$10 billion global antimicrobial healthcare fabrics market, and ∼$18 billion global antimicrobial hospitality linen market, with >7% compound annual growth rate (CAGR) between 2020 and 2025.
Conclusion
In this study, a TiO2 nano-coating process for cotton fabrics was presented to achieve durable and highly effective antibacterial textiles for various applications including the healthcare and hospitality sectors. The coating process was applied on cotton fabric, and thorough characterization was conducted to analyze the properties of the utilized materials and investigate the quality of the NPs coating on the cotton fabrics. Reliable TiO2 NPs distribution on the surface of the fabric was achieved over large areas with minimizing clusters size and count. Coating durability analysis was also conducted by washing the coated fabrics using internationally recognized processes and repeating the characterization on the washed samples. Significant antibacterial performance was achieved for the TiO2 NPs coated textile, exhibiting 99.99% bacteria growth reduction for
Footnotes
Acknowledgments
The authors would like to thank the Ministry of Education of Saudi Arabia and the “Deanship of Scientific Research (DSR)” at Imam Abdulrahman Bin Faisal University (IAU) for the financial support to conduct this study (research project No. 2019-057-CPH).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Abdulrahman Bin Faisal University (IAU) for the financial support to conduct this study (research project No. 2019-057-CPH). The research project was granted as a US patent with number: US 11,072,884 B2 date July. 27, 2021.
