Abstract
The performance of ultraviolet protection, antimicrobial activity, soil release action and self-cleaning characteristics of nano zinc oxide (ZnO) with acrylic binder were assessed on the cotton fabric using pad-dry-cure method. Different precursors such as zinc chloride and zinc nitrate were used to synthesize nanoparticles by wet chemical technique. The synthesized nanoparticles were then characterized using Fourier transform infrared spectroscopy, particle size analyser, powder X-ray diffractometry and scanning electron microscopy. The nano ZnO finished cotton fabrics were tested for ultraviolet protection factor, antimicrobial activity, soil release action and self-cleaning action. The wash fastness of nano zinc oxide (ZnO) finished cotton fabrics after 5th, 10th, 15th and 20th washes were assessed. Also the ultraviolet protection factor values, percentage reduction in bacteria and soil release percentage in each washing stage are reported. The self-cleaning activity was assessed for 12, 24and 48 h duration by exposing 6% of coffee stain on the specimen fabrics to sunlight. The zinc oxide nanoparticle size of 24 nm was obtained from synthesis of zinc chloride as a precursor, and another nanoparticle size of 38 nm were obtained from synthesis of zinc nitrate as a precursor. The smaller nanoparticles (24 nm) show better results in terms of antimicrobial activity, soil release and self-cleaning action. In case of ultraviolet protection function, it was found that the fabrics treated with 38 nm nanoparticles exhibit higher ultraviolet protection factor values than the fabric treated with 24 nm nanoparticles. The durability of the imparted functions was in the range 25–38 washes for antimicrobial activity, soil release action and ultraviolet protection, respectively.
Keywords
Introduction
Nanotechnology research in textile area focuses on creating unique properties in apparel fabrics like antibacterial activity, ultraviolet (UV) protection, self-cleaning, water and oil repellency, stain proof, anti-static, improved moisture regain and comfort in synthetic-based textiles without compromising the original hand, breathability and durability of fabric. It also shows promising applications in developing advanced textile materials such as nanocomposite fibres, nanofibres and other nanomaterial incorporated textiles for applications in medical, defence, aerospace and other technical textile applications such as filtration, protective clothing besides a range of smart and intelligent textiles [1]. In the recent years, consumers have become increasingly aware of the need for rays protection against sun rays. The UV radiation can lead to acute and chronic reactions and damage, such as acceleration of skin ageing and sunburn. Approximately, up to 10% protection of the skin against the action of solar radiation is a relatively new objective of textile finishing, since the textile does not always guarantee adequate protection [2]. Thus, a special additional sun protection finish is applied in the form of UV stabilizers. Atmosphere absorbs most of the obnoxious radiations emitted by the sun, only 5% of the harmful radiations reach to the surface of the earth [3, 4]. Sun emits three types of UV radiation UVA, UVB and UVC [5, 6]. The visible spectrum is approximately of the range from 400 to 700 nm in wavelength. UVB rays range from 280 to 320 nm and they are the primary concern for protection. UVA rays range from 320 to 400 nm. The actual damage to human skin from UV radiation is a function of the wavelength of the incident radiation and the wavelengths of maximum danger to skin are 305–310 nm. Therefore, to be useful in protecting the wearers from solar UV radiation, textiles must demonstrate effectiveness in 300–320 nm range [6]. The UV absorbers are compatible with dyes and these finishes can be applied by normal pad-dry-cure method between 30 and 40 g/L depending on the type of fibre and its construction [7, 8]. The awareness of UV radiation of health and hygiene has increased the demand for functional and protective textiles. One of the approaches to improve the UV blocking property, antimicrobial activity and self-cleaning activity of fabrics is to coat the surface with nanoparticles [9, 10]. Zinc oxide (ZnO) nanoparticles embedded in polymer matrices such as soluble starch are a good example of functional nanostructures with potential for applications such as UV protection [11, 12]. Metal oxides like ZnO as a UV blocker are more stable as compared to organic UV blocking agents. Hence, the nanoform of ZnO will really enhance the UV blocking property due to the increased surface area and intense absorption in the UV region [13, 14]. Nanotechnology offers a good range of efficient tool and technique to protect desirable fabric characteristics, most of these focusing on modification of the fabric surface using nanoparticles. Moreover, treating the fabric with nanosized silver particles is found to improve antibacterial properties [14, 15]. A fabric treated with zinc oxide could provide effective protection against bacteria and discolouration of stains due to the photocatalytic activity of nano zinc oxide [16, 17]. Several articles have discussed the use of the photocatalytic property of zinc oxide in the field of textiles [18].
In this study, two set of precursors such as zinc chloride and zinc nitrate were synthesized into nanoparticles by wet chemical technique. The synthesized nanoparticles were then characterized using the typical methods of Fourier transform infrared spectroscopy (FT-IR), particle size analyser (PSA), X-ray diffractometry (XRD) and scanning electron microscopy (SEM). These nanoparticles were then applied on to the cotton fabric samples using a pad-dry-cure method. Then, the treated fabric samples were tested for the functions of antimicrobial activity (AATCC 100-2004), UV protection (AATCC 183-2004), soil release action (AATCC 175-2008) and self-cleaning activity (ASTM D1925-1988). The treated samples were also tested for the durability of the effect after repeated washes.
Materials and methods
Materials
Bleached 100% cotton plain woven fabric was procured with 40s Ne (14.8 Tex) count in both warp and weft, cloth set of 38 ends/cm, 38 picks/cm and a mass of 118 g/m2. Zinc chloride (AR Grade) purchased from M/s Fisher Scientific, thermo electron LLS India (P) Ltd, Mumbai and zinc nitrate (AR Grade) sourced from M/s Merck chemicals (P) Ltd, Mumbai were used as precursors. Lissapol-N non-ionic was used as a surfactant for dispersion of nanoparticles in acrylic binder (Texacryl binder SLN). Deionized water was used for the hydrolysis of zinc oxide (ZnO) and for preparation of solution.
Synthesis of ZnO nanoparticles
Two different precursors, namely, zinc chloride and zinc nitrate were used to synthesize ZnO nanoparticles by adopting wet chemical method.
Synthesis of ZnO nanoparticles using zinc chloride as a precursor (procedure I)
The homogeneous phase reaction between zinc chloride and sodium hydroxide at high temperature yields ZnO nanoparticles. NaOH (pellet minimum 99% purity) and 2-propanol (minimum 99.5% purity) were used in this method. 5.5 g of zinc chloride was dissolved in 200 mL of water at 90℃. 16 mL of 5 M sodium hydroxide aqueous solution were added drop by drop to the zinc chloride solution. The mixture was stirred for 15 min at 95℃. The particles were separated by discarding the supernatant solution and sedimentation. It was then washed five times with distilled water for each wash, the ratio maintained between washing solution and the suspension was 1:10. Finally, this lowers the concentration of NaCl below 10−6 M. This is done for removal of NaCl from the suspension. The purified particles were peptized with 2-propanol in ultrasonic bath for 15 min at room temperature. The peptization process was necessary to disrupt the microagglomerates and release the nanounits of zinc oxide. This resulted in formation of nanoparticles of ZnO. These particles were centrifuged at 4000 r/min for 1 h and collected. Finally, they were treated at 250℃ for 5 h in an oven to obtain ZnO nanoparticles.
Synthesis of ZnO nanoparticles using zinc nitrate as precursor (procedure II)
The zinc oxide nanoparticles were prepared by wet chemical method using zinc nitrate, sodium hydroxide and soluble starch as stabilizing agent. Zinc nitrate of 29.747 g (0.1 mol) was dissolved in 500 mL of 1% soluble starch solution. Then, the solution was kept under constant stirring using magnetic stirrer at 800 r/min to completely dissolve the zinc nitrate. After complete dissolution of zinc nitrate, 0.2 mol of sodium hydroxide solution was added under constant stirring, drop by drop touching the walls of the vessel. The reaction was allowed to proceed for 2 h after complete addition of sodium hydroxide. After the completion of reaction, the solution was allowed to settle for overnight (12 h) and the supernatant solution was then discarded carefully. The remaining solution is centrifuged at 12,000 r/min (to remove moisture) for 10 min and the supernatant solution was discarded. The nanoparticles thus obtained were washed thrice using distilled water. Washing was carried out to remove the by-products and excessive starch that were bound with the nanoparticles. After washing, the nanoparticles were dried at 80℃ for overnight in an oven. While drying, the complete conversion of zinc hydroxide (Zn (OH)2) into ZnO occurs.
Characterization of nanoparticles
The nanoparticles were characterized for their size, shape, chemical and physical structures using the following techniques.
Powder XRD method
The crystallinity of the nanoparticles was determined by XRD using a SHIMADZU–XRD 6000, an advanced XRD equipped with a Cu-Kα radiation of wavelength λ = 1.5406 Å, applied voltage 30 kV and current 30 mA. The dried particles were deposited as a randomly oriented powder onto a plexiglass sample container, and the XRD patterns were recorded at angles between 10° and 80°, with a scan speed of 5°/min, sampling pitch of 0.02° and preset time of 0.24 s. The crystallite domain diameters (D) were obtained from XRD peaks according to the Debye–Scherrer’s equation [9]
Fourier transform infrared spectroscopy
SHIMADZU–FT-IR 8400S with a spectral range 4000–400 cm−1 was used. Spectra were collected with a resolution of 0.9 to 1.0 cm−1 and given as ratio of 200 single beam scans to the same number of background scans in pure KBr (IR grade supplied by Aldrich). KBr was ground to fine powder and mixed with samples (2% (w/w).
Particle size analyzer
The size distribution of nanoparticles was determined using PSA. The size of the nanoparticles was obtained through PSA, using a Sympatec GmbH, NANOPHOX (0143 P). The nanoparticles measuring duration 150.10 s, temperature 27℃, laser power 75% and measuring range 1–1000 nm.
Scanning electron microscopy
The fabric samples were treated with nanoparticles were mounted on a specimen stub with double-sided adhesive tape and coated with gold in a sputter coater and examined with a SEM (Jeol Model JSM-6360).
Coating of nanoparticles on fabric
Before coating, the fabric was dried at 100℃ for 5 min in an oven to remove the moisture present in the fabric. The cotton fabric was immersed for 1 min in aqueous nanosol with 0.5% acrylic binder in padding mangle.
Fabrics were coated with the nanosolution at different concentration levels, namely, 1.0%, 1.5% and 2.0%. Zinc oxide nanosolution was applied on the cotton fabric using pad-dry-cure method. The nanoparticle solution was uniformly coated using laboratory padding mangle, running at a speed of 15 m/min with a pressure of 15 kg/cm2. After padding, the cotton fabric was dried at 70℃ for 5 min and cured at 130℃ for 3 min in a curing chamber.
Evaluation of antimicrobial activity
The antimicrobial properties of treated cotton fabrics were evaluated by quantitative assessment as per AATCC test method 100-2004. The test was carried out with Staphylococcus aureus and Klebsiella pneumonia. The percentage reduction of bacteria by the fabric sample is reported as R
Evaluation of UV protection
In vitro method is used to assess the UV protection of the cotton fabric as per the AATCC-183 (2004) test method. It measures the transmittance or blocking of UV radiation through fabrics by UV-vis spectrophotometer (Varian, Cary 5000) with a spectral range 175–3300 nm. The UV profiles of the untreated samples were compared with the spectra collected from the same fabrics treated with nanoparticles, and the effectiveness in shielding UV radiation was evaluated by measuring the UV protection, transmission and reflection. Each measurement is the average of four scans obtained by rotating the sample by 90°. The transmission data was used to calculate the ultraviolet protection factor (UPF), according to the following equation [6]
Evaluation of soil release property
The soil release property was tested by modified stain release test. The stain profiles of treated and untreated samples were compared with AATCC 175 (2008).
Washing fastness
The wash fastness of nano ZnO coating on fabric was tested as per AATCC method 61(1996), using Atlas Launder-O-meter LEF instrument (one wash in the Atlas Launder-O-meter LEF is equivalent to five home machine launderings).
Evaluation of self-cleaning activity
The self-cleaning action of the ZnO coated cotton fabric was investigated by exposing the samples containing adsorbed coffee stain to visible radiation. Measured quantity of 6% coffee solution were introduced on the cotton fabric and it is allowed to spread. One half of each stain on the fabric was exposed to sun light for 12–48 h, while the other half was covered with a black paper to prevent its radiation from sunlight. The exposed part of the stain was compared with that of the covered part for self-cleaning action. Premier colour scan SS 5100 A–spectrophotometer was used to measure the photo degradation of coffee stain. The self-cleaning action was quantified by comparing K/S values of the exposed and unexposed portions of the same stain
Results and discussion
Characterization of ZnO nanoparticles
The results were analysed and interpretations of the various tests were used to characterize the two types of ZnO nanoparticles (Z1 and Z2) synthesized. The various analytical tests include FT-IR, powder XRD, PSA and SEM.
Fourier transform infrared spectroscopy
Figure 1(a) shows the FT-IR spectra of untreated cotton fabric sample. The spectrum of interference pattern obtained for the (Z1) ZnO nanoparticles obtained by the synthesis procedure I (using the hydrolysis of zinc chloride) is shown in Figure 1(b). The spectrum of interference pattern obtained for the (Z2) ZnO nanoparticles obtained by the synthesis procedure II (using the hydrolysis of zinc nitrate) is shown in Figure 1(c). Figure 1(b) and (c) clearly show the ZnO absorption band is near to 440 cm−1. In Figure 1(a), the FT-IR spectra from untreated sample, did not show any peak at 440 cm−1, which confirm the presence of ZnO in the treated samples. The peaks at 3450 and 2350 cm−1 indicate the presence of –OH and C = O residues, probably due to the atmospheric moisture and carbon dioxide, respectively.
FT-IR spectra of (a) untreated fabric sample; (b) zinc oxide (Z1) treated sample and (c) zinc oxide (Z2) treated sample.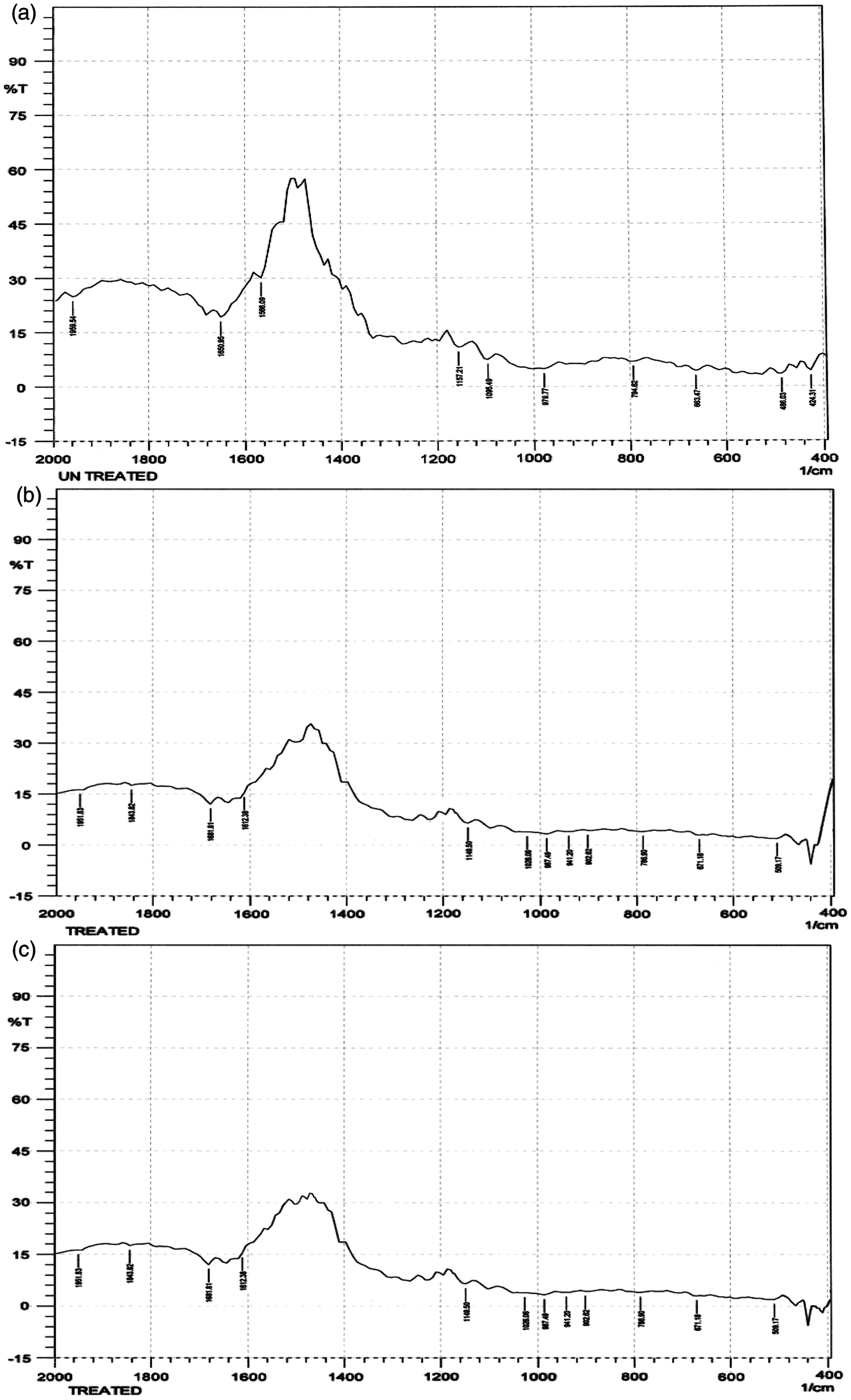
X-ray diffraction
The XRD spectrogram of ZnO nanoparticles synthesized using zinc chloride is shown in Figure 2(a). The distinctive ZnO peaks are at 32.90, 34.5, 36.40, 47.62, 57.28, 68.78, 72.26 and 82.10, respectively. The XRD spectrogram of ZnO nanoparticles synthesized using zinc nitrate is shown in Figure 2(b). The XRD patterns of these samples gave seven distinctive ZnO peaks at 32.90, 34.5, 36.40, 47.50, 57.0, 62.50 and 68.72, respectively. The XRD pattern for ZnO nanoparticles obtained shows sharp peaks. This means that the crystallinity is more for Z1 and Z2 nanoparticles.
XRD patterns of (a) Z1 and (b) Z2 nanoparticles synthesized by procedures I and II.
Particle size analyser
The particle size distribution of nano ZnO synthesized were determined using PSA and are shown in Figure 3(a) and (b), respectively. The nanoparticles had 24 nm for Z1 and 38 nm for Z2 and the density distribution for the maximum peak is 1.4.
Particle size analysis of (a) Z1 (procedure I) and (b) Z2 (procedure II) nanoparticles.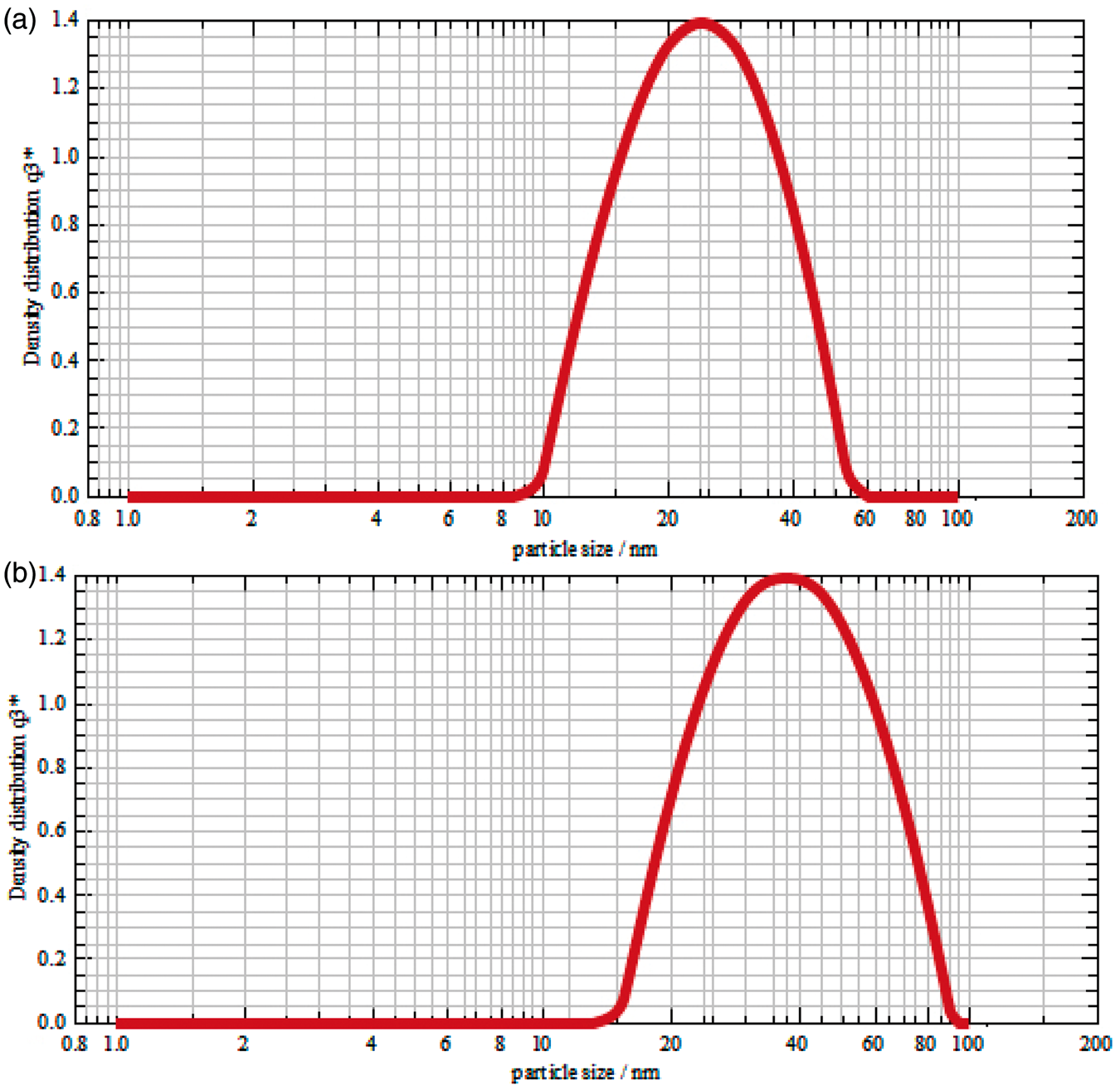
Zinc oxide nanoparticle size.

Scanning electron microscopy
Figure 4(a) shows the SEM micrograph of the untreated 100% cotton woven fabric sample. Figure 4(b) and (c) shows the SEM micrograph of nanoscaled ZnO nanoparticles on cotton fabric samples. The nanoparticles are well dispersed on the fibre surfaces in both cases. From Figure 4(b), it is possible to observe that Z1 nanoparticles were more uniformly distributed over the fabric surface than fabric coated with Z2 nanoparticles in Figure 4(c), where uneven and agglomerated patchy coating is seen due to the larger particle size. The particle size plays a primary role in determining their adhesion to the fibres: it is reasonable to expect that the larger particle agglomeration will be easily removed from the fibre surface, while the smaller particles will penetrate deeper and adhere strongly into the fabric matrix.
SEM analysis of (a) untreated 100% cotton fabric; (b) ZnO-treated 100% cotton fabric for Z1 nanoparticle (procedure I); and (c) ZnO-treated 100% cotton fabric for Z2 nanoparticle (procedure II).
Testing of antimicrobial activity
Antimicrobial activity of cotton fabric.

Testing of antimicrobial activity after laundering
Antimicrobial activity of fabrics after washing.

The linear regression analysis is used to predict the number of washes required to reach the untreated antimicrobial activity value of zero. (Z1) fabric are found to be 32 for 1%, 1.5% and 2% concentration levels and (Z2) fabric are found to be 25 for 1% and 26 for 1.5% and 2% concentration levels. The details of the prediction are given in Figure 5.
Number of washes predicted using linear regression analysis for Z1 and Z2 nanoparticles for antimicrobial activity.
Testing for UPF
UPF values for the fabric samples.
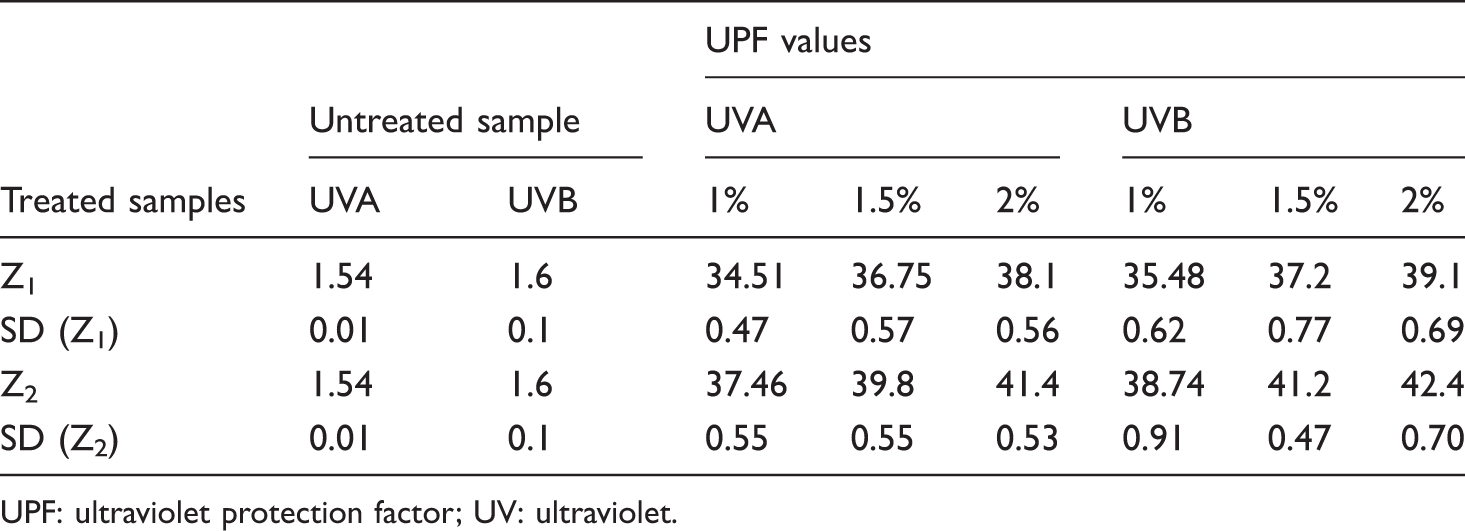
UPF: ultraviolet protection factor; UV: ultraviolet.
Z2 sample shows higher UPF value than the Z1. According to Raleigh’s scattering theory, the scattering of light is inversely proportional to the fourth power of wavelength. The optimum particle size for scattering the radiation is calculated as 15 to 40 nm. This clearly indicates that nanoparticles nearer to the range 20–40 nm exhibit better scattering of UV rays, and hence, the UPF value for Z2 is more than Z1.
Testing of UV protection after a series of washes
UPF values after a number of washes.
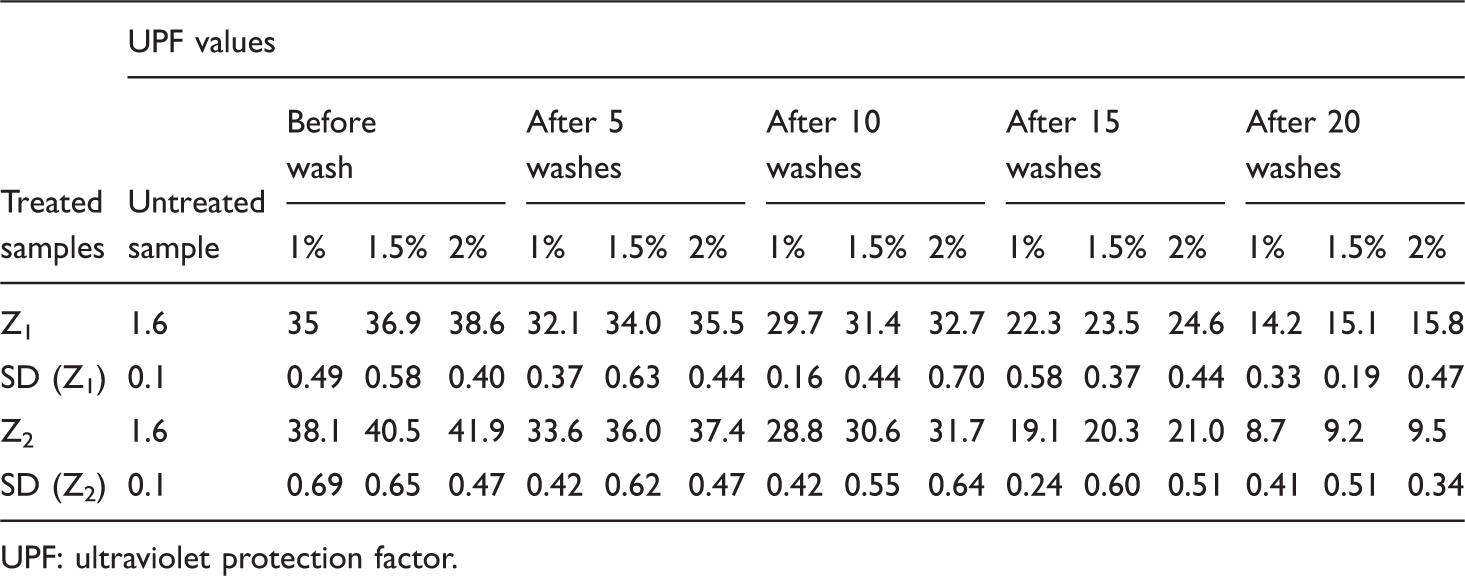
UPF: ultraviolet protection factor.
From Figure 6, it is clear that the predicted number of washes for (to reach untreated UPF value of 1.6) ZnO (Z1) nanoparticles treated fabrics are found to be 34 for 1%, 1.5% and 2% concentration levels and ZnO (Z2) nanoparticles treated fabrics are found to be 26 for 1%, 2% and 28 for 1.5% concentration levels.
Number of washes predicted using linear regression analysis for Z1 and Z2 nanoparticles for UPF values.
Testing for soil release action
Soil release rating values.

Fabrics treated with ZnO nanoparticles show significant improvement in soil release rating than untreated samples. Both Z1 and Z2 nanoparticles show better soil release ratings in the range of 6 at different concentration levels.
Testing the durability of soil release action at different wash levels
Soil release ratings at different wash levels.

From Table 7, it is clear that up to five washes there is a slight change in the soil release rating for both Z1- and Z2-coated fabrics. However, after five washes, there is a reduction of soil release rating by 1 rating for every five washes. The reduction in soil release rating is found to be gradual.
The linear regression analysis is carried out to predict the number of washes for which the fabric would have the soil release rating equivalent to the value of untreated sample. The size of ZnO nanoparticles influences the durability of the soil release property in terms of the number of washes.
From Figure 7, it is clear that the predicted number of washes for Z1 coated fabric sample, the predicted value was 38 for 1%, 1.5% and 2% concentration levels. For Z2 coated fabric sample, the predicted value was 32 for 1%, 1.5% and 2% concentration levels. This is only for the subjective method of evaluation used in testing the soil release ratings.
Number of washes predicted using linear regression analysis for Z1 and Z2 nanoparticles for soil release ratings.
Linear regression equation for Z1 and Z2 nanoparticle samples.

UPF: ultraviolet protection factor.
Testing for self-cleaning activity
The self-cleaning property of ZnO coated cotton textile is based on the highly oxidative intermediates generated at the cotton surface. The K/S values (absorption to scattering coefficient) of exposed and unexposed parts of the samples were measured after 0, 12, 24 and 48 h. The comparison of K/S values of different test samples are shown in Figure 8. The percentage decrease in K/S value for the exposed samples in comparison to unexposed sample is measured to quantify the self-cleaning activity. The rate of degradation of coffee stain is faster in first 12 h of exposure. In the nanoparticle coated samples, no visual deterioration in the properties of cotton fabric is observed up to 48 h of exposure. The nanoparticles of smaller size have more surface area and exhibits superior self-cleaning action. The smaller nanoparticles of Z1 (24 nm) shows better self-cleaning activity than Z2 (38 nm). The sample coated with nanoparticles Z1 shows a better self-cleaning activity then Z2.
Percentage decrease in K/S values of coffee stained cotton fabric.
The statistical significance at different concentration levels, namely, 1%, 1.5% and 2% and the statistical significance at different time duration levels such as, 12, 24 and 48 h, respectively, at 95% confidence limits. The results reveal that there is significant difference between different concentrations for a given duration and significant difference between different time durations with same concentrations levels.
Conclusions
The nano ZnO treated cotton fabrics were tested the important functional properties viz antimicrobial activity, UV protection, soil release action and self-cleaning activity. The durability characteristics of the multifunctional treatments of the cotton treated fabrics were studied.
The synthesis of ZnO as water medium yields nanoparticles of sizes 24 nm (Z1) and 38 nm (Z2) at different temperature levels. The FT-IR analysis was carried out to analyse the presence of ZnO on the cotton fabric. The presence of an infrared signature in the region 440 cm−1 in the finished product indicates the presence of ZnO in the treated fabric. PSA was used to analyse the nanoparticles which confirms that Z1 nanoparticles of size 24 nm, and Z2 nanoparticles of size 38 nm. The XRD spectra of the ZnO nanoparticle were analysed and the presence of well-defined peaks indicate the more crystalline structure. The particle size plays a primary role in determining their adhesion to the fibres. It was observed the larger particle agglomerates and was easily removed from the fabric surface, while the smaller particle penetrate deeper and adhere strongly into the fabric matrix. In the antimicrobial activity, the fabrics treated with smaller sized ZnO nanoparticles (Z1) 24 nm shows better results than the fabrics treated with large sized (38 nm) ZnO nanoparticles (Z2). These results, due to the fact that a decrease in the size of particles in increase of the surface area. In the case of UV protection function, it was interesting to found that the fabrics treated with slightly larger sized (38 nm) ZnO nanoparticles (Z2) have higher UPF values than the fabrics that were treated with smaller sized (24 nm) ZnO nanoparticles (Z1). In the case of soil release property, it is observed that the fabrics treated with ZnO nanoparticles (Z1 for 24 nm and Z2 for 38 nm) give better results when compared to untreated fabric sample. In the case of self-cleaning activity, the smaller nanoparticle size in situ coating of ZnO derived using wet chemical technique; nanoparticles ZnO (Z1) show better self-cleaning activity as compared to large nanoparticles of ZnO (Z2). The antimicrobial activity, UPF and soil release action increases enormously after ZnO finishing on cotton when compared to untreated fabric and retain until 10 washes; beyond 15 washes there was deterioration in antimicrobial activity, UPF and soil release action values.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.




