Abstract
The performance of ultraviolet (UV) protection, antimicrobial activity, and self-cleaning characteristics of nano titanium dioxide (TiO2) with acrylic binder were assessed on the cotton fabric using pad-dry-cure method. Titanium iso-propoxide was used as precursor with two different mediums of water and ethanol to synthesize nano-sol by sol-gel technique. The synthesized nano-sol-gel was then characterized by using Fourier transform infrared (FTIR) spectroscopy, particle size analyzer (PSA), X-ray powder diffractometry (XRD), and scanning electron microscopy (SEM). The nano TiO2 finished cotton fabrics were tested for ultraviolet protection factor (UPF), antimicrobial activity, self-cleaning action, and physical properties. The wash fastness of TiO2 nano finished cotton fabrics for 5th, 10th, 15th, and 20th washes was assessed and also their ultra protection factor values and the percentage reduction in bacteria in each stage were reported. The self-cleaning activity was assessed for 12 hours, 24 hours, and 48 hours duration by exposing coffee stain on the specimen fabrics to sunlight. The TiO2 nanoparticles had 12 nm when ethanol medium was used and 7 nm for water. The smaller nanoparticles had showed better results regards antimicrobial activity and self-cleaning. In case of UV-protection function it was found that the fabrics treated with 12 nm nanoparticles exhibit higher UPF values than the fabric treated with 7 nm nanoparticles. The durability of the imparted function was in the range of 32–36 washes for antimicrobial activity and UV-protection property.
Keywords
Introduction
Nanotechnology is one of the key technologies of the 21st century. It is used in all fields including textile industries for medical, hygienic, and technical textiles. In the recent years, consumers have become increasingly aware of the need for sunrays protection. The ultraviolet (UV) radiation can lead to acute and chronic reactions and damage, such as acceleration of skin aging and sunburn. Approximately 10% of sun protection of the skin against the action of solar radiation is a relatively new objective of textile finishing, since the textile does not always guarantee adequate protection [1]. Thus, a special additional sun protection finish is applied in the form of UV stabilizers. Atmosphere absorbs most of the noxious radiations emitted by the sun, only 5% of the harmful radiations reach to the surface of the earth [2–4]. Sun emits three types of UV radiation UV-A, UV-B, and UV-C [5,6]. The visible spectrum is approximately of the range from 400 to 700 nm in wavelength. UV-B rays range from 280 to 320 nm and they are the primary concern for protection. The UV-A rays range from 320 to 400 nm. The actual damage to human skin from UV radiation is a function of the wavelength of the incident radiation and the wavelengths of maximum danger to skin are 305–310 nm. Therefore to be useful in protecting the wearers from solar UV radiation, textiles must demonstrate effectiveness in the 300–320 nm range [5]. The ozone layer blocks the sun output of UV-C and most UV-B radiations. The UV-B radiation that reaches the earth surface poses the greatest danger for sunburn and skin damages [7]. Personal risks can be largely averted by avoiding exposure to the sun during the middle of the day by covering up with appropriate clothing, headgear, and sunglasses, and by using sun protection factor (SPF) 30+ sunscreen. The UV absorbers are compatible with dyes and these finishes can be applied by normal pad-dry-cure method between 30 and 40 g/L depending on the type of fiber and its construction [8,9].
The awareness of UV radiation of health and hygiene has increased the demand for functional textiles or protective textiles. One of the approaches to improve the UV-blocking property, antimicrobial activity, and self-cleaning activity of fabrics is to coat the surface with nanoparticles [10,11]. Zinc oxide (ZnO) nanoparticles embedded in polymer matrices like soluble starch are a good example of functional nanostructures with potential for applications such as UV protection [12,13]. Metal oxides like titanium dioxide (TiO2) and ZnO UV blockers are more stable compared to organic UV-blocking agents [14]. TiO2 and other ceramic materials have an absorption capacity in the UV region of 280–400 nm and reflects visible and infrared (IR) rays [15,16]. Hence, a common nanosize TiO2 and ZnO will enhance the UV-blocking property due to their increased surface area and intense absorption in the UV region [17,18]. Moreover, treating the fabric with nanosized silver particles found to improve antibacterial properties [19,20]. To certain degree, sterilizing effect are displayed by metallic ions and metallic compound. A part of the oxygen in the air or water is turned into active oxygen by means of catalysis with the metallic ion, thereby dissolving the organic substance to create a sterilizing effect. The use of nanosized particles would help to maximize antibacterial activity as the number of particles per unit area is higher. TiO2 is a photocatalyst, when it is illuminated by light with energy higher than its band gaps, the electrons in titanium dioxide will jump from the valence bond to the conduction bond [21, 22]. Several articles have discussed the use of the photocatalytic property of TiO2 in the field of textiles. A fabric treated with TiO2 could provide effective protection against bacteria and the discoloration of stains due to the photocatalytic activity of nano TiO2.
In the present work, Titanium iso-propoxide is synthesized into nano-sol by sol-gel technique with water (T1) or ethanol (T2) as a medium. Two sets of nanoparticles were successfully synthesized. The synthesized nano-sol was then characterized using the typical methods of FTIR, PSA, XRD, and SEM. These nano-sols were then applied on to the sample fabrics using a pad-dry-cure method. Then, the treated fabric samples were tested for the functions of antimicrobial activity, ultraviolet protection factor (UPF), and self-cleaning activity as per American Association of Textile Chemists and Colorists (AATCC) standard tests. The treated samples were also tested for the durability of the effect after repeated washes.
Materials and methods
Materials
The bleached cotton plain woven fabric was fabricated with 40 s Ne count in both warp and weft, cloth set of 38 ends/cm and 38 picks/cm, fabric mass of 118 grams per square meter and 122 cm fabric width. Titanium iso-propoxide (AR Grade, molecular weight [mol. Wt]: 284.26) procured from M/s. Himedia Laboratories (P) Ltd, Mumbai, was used as precursor. Lissapol-N nonionic was used as surfactant for dispersion on nanoparticles in acrylic binder (Texacryl binder SLN). Deionized water was used for the hydrolysis of titanium iso-propoxide and for preparation of sol-gel solution.
Synthesis of TiO2 nano-sol using titanium iso-propoxide as precursor
Sol-gel nanosynthesis techniques

Procedure I
Procedure II: The TiO2 gel containing anatase particles was prepared as the 30 wt % hydrogen peroxide (H2O2) solution was added to the 10 mL of 1 mol/L ethanol solution of titanium iso-propoxide. The ratio of H2O2: TIP was kept as 12:1. The ethanol was added to the obtained solution and the total volume of the solution was adjusted to 100 mL, the solution was then heated at 75°C for 12 hours in a closed vessel.
Characterization of nanoparticles
The nanoparticles were characterized for their size, shape, chemical, and physical structures using the following techniques.
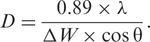
Where;
λ is the wavelength of the incident X-ray beam (1.54 Å for the Cu Kα),
θ is the Bragg's diffraction angle, and
Δ
Coating of nano-sol/nanoparticles on textile substrate
The bleached 100% cotton woven fabric of plain weave, 38 ends/cm, 38 picks/cm, and 118 g/m2 were used. Before coating, the fabric was dried at 100°C for 5 minutes by using oven to remove the moisture content present in the fabric. The cotton fabric was immersed for 1 minute in aqueous nano-sol with 1% acrylic binder in padding mangle.
Fabrics were coated with the nanosolution in different concentration levels such as 1.0%, 1.5%, and 2.0%. TiO2 nano-sol/nanoparticles were applied on the cotton fabric using pad-dry-cure method. The nanoparticle solution was evenly coated by using laboratory padding mangle machine, running at a speed of 15 m/min with a pressure of 15 kg/cm2. After padding the cotton fabric was dried at 70°C for 5 minutes and cured at 130°C for 3 minutes using curing chamber.
Evaluation of antimicrobial activity
The antimicrobial properties of treated cotton fabrics were evaluated by quantitative assessment method as per AATCC test method 100-2004. The test was carried out with A – the number of bacteria recovered from the inoculated, treated test specimen swatches in the jar incubated for over 24 hours, and B – the number of bacteria recovered from the inoculated, treated test specimen swatches in the jar immediately after inoculation (at ‘0’ contact time).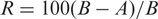
Evaluation of UV protection
The in vitro method is used for assessing the UV protection of the cotton fabric as per the AATCC-183 (2004) test method. It measures the transmittance or blocking of UV radiation through fabrics by UV-vis spectrophotometer. The make/model of the instrument used was Varian, Cary 5000, spectral range of 175–3,300 nm. The UV profiles of the untreated samples were compared to the spectra collected from the same fabrics treated with nanoparticles, and the effectiveness in shielding UV radiation was evaluated by measuring the UV protection, transmission, and reflection. Each measurement is the average of four scans obtained by rotating the sample by 90°. The transmission data was used to calculate the UPF, according to the following equation [3,8]:
Δλ – the band width in nm.
Washing fastness
The wash fastness of TiO2 nanocoating on fabric was tested by following AATCC method 61 (1996) test no. 2 A using Atlas Launderometer LEF instrument. One wash in the Atlas launderometer LEF is equivalent to five home machine launderings.
Evaluation of self-cleaning activity
The self-cleaning action/photoactivity of the TiO2-coated cotton fabric was investigated by a procedure of exposing the samples containing adsorbed coffee stain to visible irradiation. The measured quantity of 5% coffee solution was introduced on the cotton fabric and was allowed to spread. One half of each stain on the fabric was exposed to sun light for 12–48 hours, while the other half was covered with a black paper to prevent its irradiation from sunlight. The exposed part of the stain was compared with that of the covered part for self-cleaning action. Model/make of the instrument Premier colour scan SS 5100 A spectrophotometer was used to measure the photo degradation of coffee stain. The self-cleaning action was quantified by comparing K/S values of the exposed and unexposed portions of the same stain [10,14].
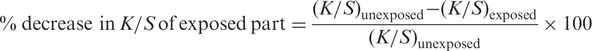
Physical testing of fabrics
The physical properties of the 100% cotton woven fabric such as fabric weight (American Society for Testing and Materials (ASTM) D3776:2009), tensile strength and elongation (ASTM D 5035:2006), crease recovery (AATCC 66:2008), stiffness (ASTM D1388:2008), air permeability (ASTM D737:2008), tearing strength (ASTM D1424:2009), drape coefficient, thickness (ASTM D1777:2007), abrasion resistance (ASTM D 3884:2009), and bursting strength (ASTM D3786:2009) of both untreated and treated samples were evaluated after conditioning the specimens at 65% Relative Humidity (RH) and 27 ± 2°C for 24 hours by bringing them to approximate moisture equilibrium in the standard atmosphere for preconditioning textiles as directed in Practice D 1776 in an environmental chamber (ASTM 2008).
Results and discussion
Characterizations of TiO2 nano-sol/nanoparticles
The results and interpretations of the various characterization tests of the two types of TiO2 nanoparticles synthesized using the procedures I and II were discussed. These analytical tests include: FTIR, XRD, PSA, and SEM.
Fourier transform infrared spectroscopy (FTIR).
The Figure 1(a) shows the FTIR spectra of untreated cotton fabric sample. The spectrum of interference pattern obtained for the (T1) TiO2 nanoparticles obtained by the synthesis procedure I (using the hydrolysis of titanium iso-propoxide with water at room temperature) is shown in Figure 1(b). The spectrum of interference pattern obtained for the (T2) TiO2 nanoparticles obtained by the synthesis procedure II (using the hydrolysis of titanium iso-propoxide with ethanol at 75°C for 12 hours) is shown in Figure 1(c). The Figure 1(b) and (c) clearly shows that the TiO2 absorption band is near to 432 cm−1. The Figure 1(a), FTIR spectra from untreated sample, did not show any peak at 432 cm−1, which confirm the presence of TiO2 at the treated samples. The peaks at 3,000 and 1,200 cm−1 indicate the presence of –OH and C = O residues, probably due to atmospheric moisture and CO2, respectively.
Fourier transform infrared (FTIR) spectrogram of (a) untreated fabric sample, (b) T1 nano-sol synthesized by procedure I, and (c) T2 nano-sol synthesized by procedure II.
Nanoparticles size analyzed by XRD
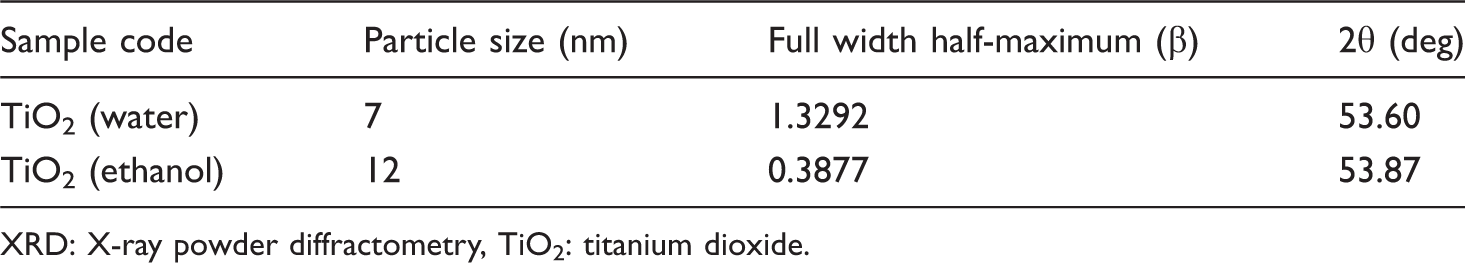
XRD: X-ray powder diffractometry, TiO2: titanium dioxide.

X-ray powder diffractometry (XRD) pattern of T2 nanoparticles synthesized by procedure II.
Particle size analysis of (a) T1 nanoparticles (water as medium) and (b) T2 nanoparticles (ethanol as medium).
Scanning electron microscope (SEM) analysis of (a) untreated 100% cotton fabric, (b)TiO2-treated 100% cotton fabric for T1 nanoparticle (procedure I), and (c) TiO2-treated 100% cotton fabric for T2 nanoparticle (procedure II).
Functional testing of antibacterial activity
The Figure 5 shows the comparison of antimicrobial activity of the treated fabrics as percentage reduction of bacteria (for the two representative types Antimicrobial activity of cotton fabric.
Antimicrobial activity of fabrics after washing.
Number of washes predicted using the forecast statistical analysis for antibacterial activity

Functional testing of UV protection (UPF)
Figure 7 shows the values of UPF for the fabric sample (treated with the nanoparticles of TiO2) for UV-A (315–400 nm) and UV-B (280–315 nm) radiations. These results show that the UPF values of the treated fabric had better UV protection than the untreated fabrics.
Ultraviolet protection factor (UPF) values for the fabric samples.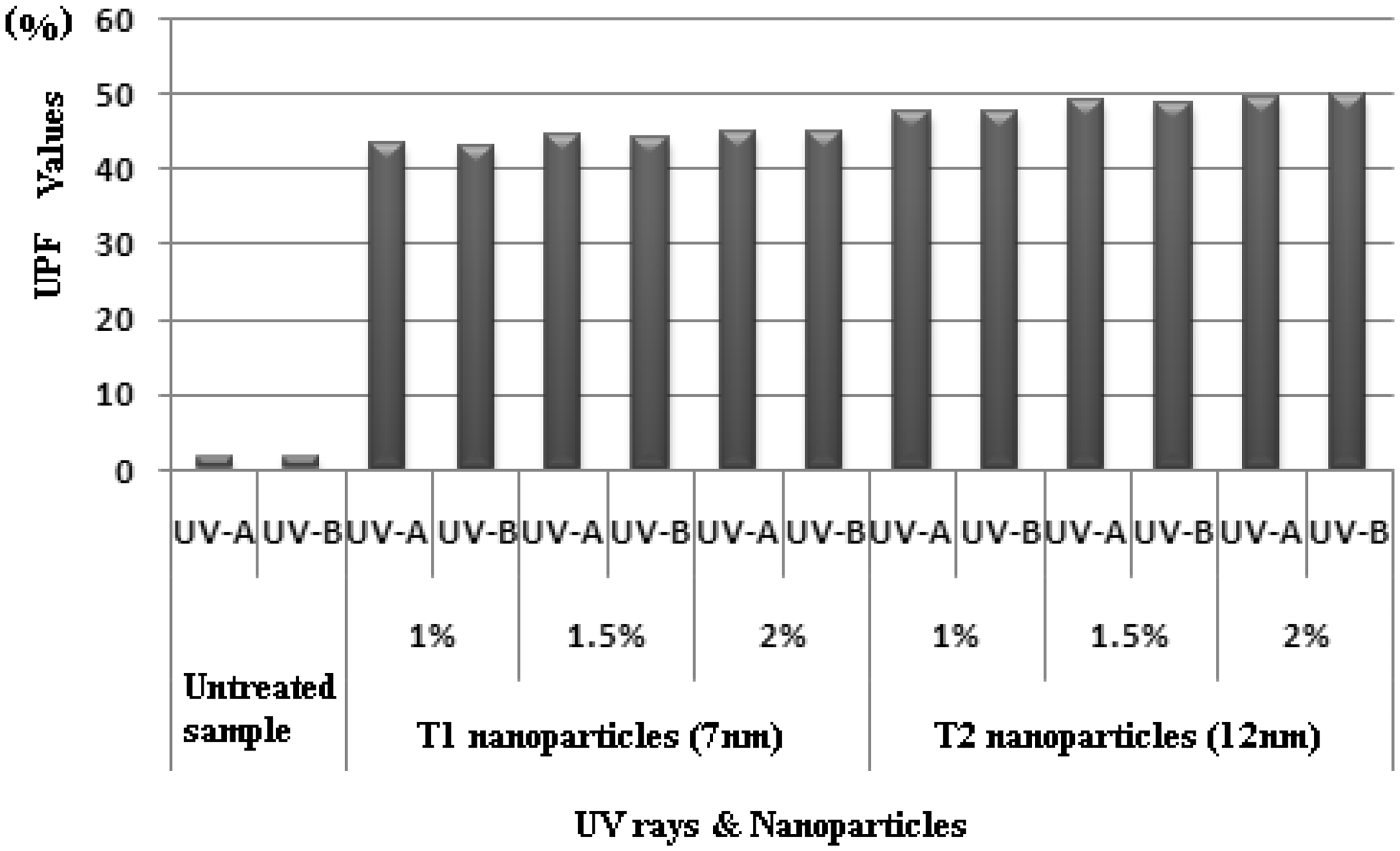
The 100% cotton fabrics treated with TiO2 nanoparticles of T1 shows lower UPF values than the same fabrics treated with TiO2 nanoparticles of T2. According to Raleigh's scattering theory, the scattering of light is inversely proportional to the fourth power of wavelength. The optimum particle size for scattering the radiation is calculated as 15–40 nm. This clearly indicates that nanoparticles nearer to the range of 15–40 nm exhibit better scattering of UV rays. Out of T1 (7 nm) and T2 (12 nm), T2 is closer to the above said range. And hence the UPF value for T2 is more than T1.
Ultraviolet protection factor (UPF) values after a number of washes.
Number of washes predicted using the forecast statistical analysis for UPF values

UPF: ultraviolet protection factor, TiO2: titanium dioxide.
Functional testing of self-cleaning activity
The self-cleaning property of TiO2-coated cotton textile is based on the highly oxidative intermediates generated at the cotton surface. The mechanism of photocatalysis reaction in presence of these particles is well established and is reported by many researchers. The K/S values (absorption to scattering coefficient) of exposed and unexposed parts of the samples were measured after 0, 12, 24, and 48 hours. The comparison of K/S values of different test sample are shown in Figure 9. The percentage decrease in K/S value for the exposed samples in comparison to unexposed sample is measured to quantify the self-cleaning activity. The rate of degradation of coffee stain is faster in first 12 hours of exposure. In the nanoparticles-coated samples, no visual deterioration in the properties of cotton fabric is observed up to 48 hours of exposure. The nanoparticles of smaller size have more surface area and exhibits superior self-cleaning action. The smaller nanoparticles of T1 (7 nm) show better self-cleaning activity than T2 (12 nm).The TiO2, which has a particle size below 10 nm, was found to have higher self-cleaning activity. The sample coated with nano-sol T1 shows a better photocatalytic activity then T2. As shown in Figure 9, after the first 12 hours of exposure of the sample, it was observed that the decrease in K/S value was nearly 68%.
Percentage decrease in K/S values of coffee-stained cotton fabric.
Physical properties
Physical properties of untreated and TiO2-treated cotton fabrics
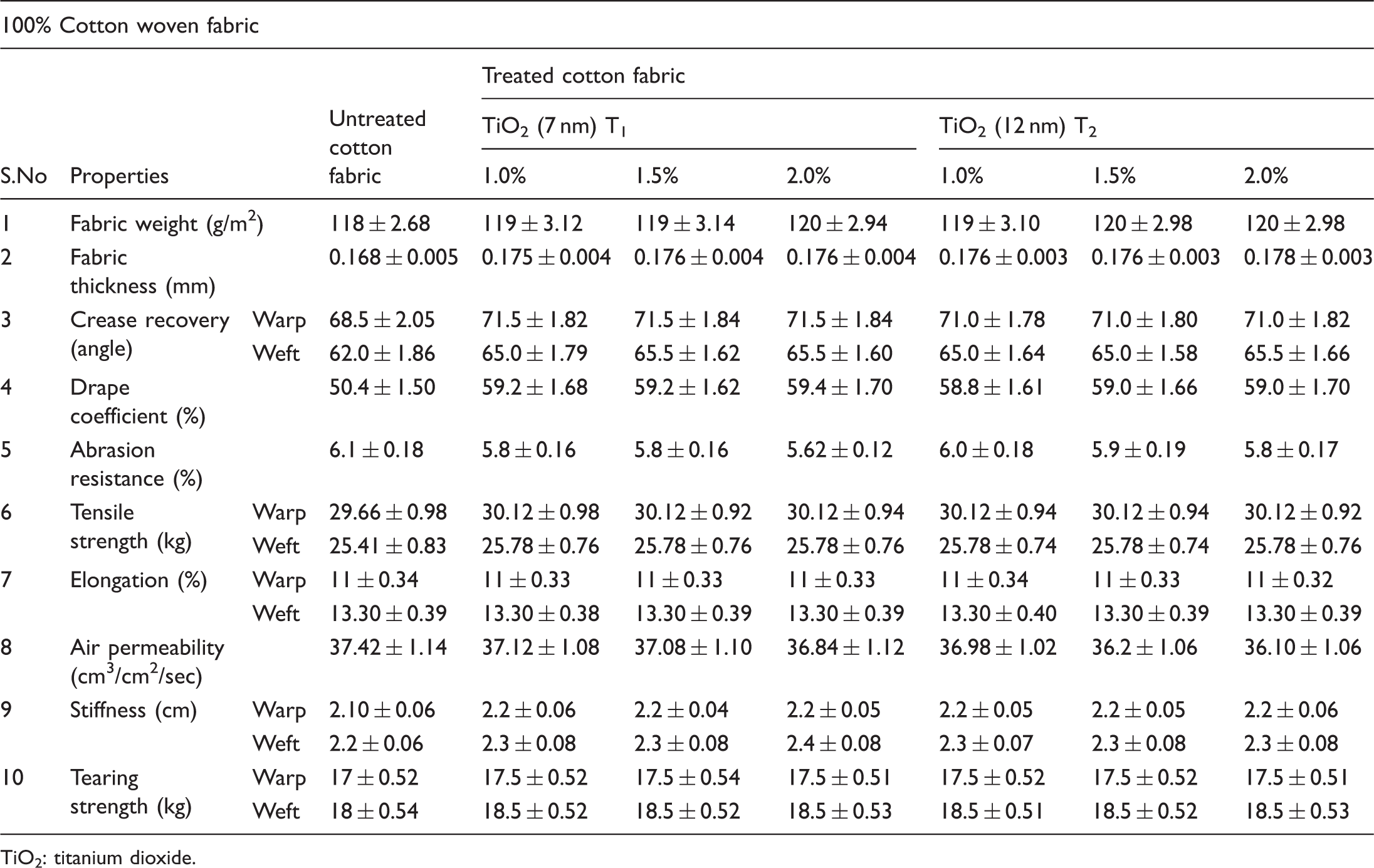
TiO2: titanium dioxide.
Conclusions
The nano TiO2-treated cotton fabrics were tested the important functional properties viz. antimicrobial activity, UV protection, and self-cleaning activity. The durability characteristics of the multifunctional treatments of the cotton treated fabrics were studied. The influences of the surface treatment of nanoparticles on the mechanical properties of the treated fabrics were examined.
The following conclusions have been drawn from the test results: In the synthesis of TiO2, water medium at 30°C yields nanoparticles of size 7 nm, whereas ethanol medium at 75°C yields nanoparticles of size 12 nm. The FTIR analysis was carried out to analyze the presence of TiO2 on the cotton fabric. The presence of an IR signature in the region 432 cm−1 in the finished product indicate the presence of TiO2 in the treated fabric. The PSA was used to analyze the nanoparticles, which conforms that water medium give nanoparticles of size 7 nm and ethanol medium of size 12 nm. The XRD spectra of the TiO2 nanoparticle were analyzed and the presences of well-defined peaks indicate the crystalline structure. The particle size plays a primary role in determining their adhesion to the fibers. It was observed that the larger particle agglomerates and were easily removed from the fabric surface, while the smaller particle penetrate deeper and adhere strongly into the fabric matrix. In the case of antimicrobial activity, the fabrics treated with smaller sized TiO2 nanoparticles (T1) 7 nm show better results than the fabrics treated with slightly large sized (12 nm) TiO2 nanoparticles (T2). These results are due to the fact that a decrease in the size of particles increases the surface area. In the case of UV-protection function, it was interesting to find that the fabrics treated with slightly larger sized (12 nm) TiO2 nanoparticles (T2) have higher UPF values than the fabrics that were treated with smaller sized (7 nm) TiO2 nanoparticles (T1). In the case of self-cleaning activity, the smaller nanoparticle size in situ coating of TiO2 derived using sol-gel technique; nano-sol TiO2 (T1) show better self-cleaning activity as compared to large nanoparticles of nano-sol TiO2 (T2). The UPF and antimicrobial activity increases enormously after TiO2 finishing on cotton, when compared to untreated fabric and is retained until 10 washes; beyond 15 washes there was deterioration in UPF values and antimicrobial value. The durability of the imparted functions was in the range of 32–36 washes for antimicrobial activity and UPF value.
Footnotes
Notes
So far, he has published 20 technical research articles in national journals and presented 5 technical articles in the national- and international-level technical conferences. He has also organized various national-level conferences and workshops. His areas of specialization are weaving, fabric structure and designing, and textile mill planning.
