Abstract
The COVID-19 pandemic has led to a greater need for non-transmissible products like face masks, which are effective in preventing the spread of the virus but come with challenges such as reduced breathability and biohazard waste. This study addressed these issues by integrating green-synthesized zinc oxide particles and Azadirachta indica extract into nonwoven face masks. Eight samples were developed using the needle punching nonwoven method and then calendared. These samples were coated with varying levels of A. indica extract (0, 15, and 30 wt.%) and ZnO particles (0, 2, and 4 wt.%) using the dip and dry method. Comparative analyses were performed with market-available masks (MA) and untreated masks (US) for air permeability, thermal resistance, tensile behavior, and antimicrobial efficacy against Staphylococcus aureus. The untreated masks had the highest air permeability at 826 mm/s, which decreased with higher A. indica and ZnO concentrations. Scanning electron microscope (SEM) images confirmed even distribution of ZnO particles. Thermal resistance and tensile properties were directly linked to A. indica and ZnO concentrations. The sample treated with 30 wt.% A. indica and 4% ZnO showed the highest thermal resistance (0.042 m2K/W), elongation (92% CD, 84% MD), and tenacity (0.93 g/tex CD, 0.87 g/tex MD). Increased A. indica and ZnO concentrations demonstrated significant antimicrobial activity, with a maximum zone of inhibition of 8.03 mm recorded at 30 wt.% A. indica and 4 wt.% ZnO particles. These findings suggest potential applications for developing surgical facemasks with improved efficacy.
Introduction
The rapid spread of the COVID-19 pandemic has impacted over 14.9 million people across the globe. According to a 2020 report from the World Health Organization (WHO), the virus has led to the deaths of 618,017 individuals. The world must develop a practical and effective strategy to prevent the spread of future pandemics and avert similar global crises.1,2 The main way of spreading COVID-19 from person to person is through droplets when someone is within proximity (1 m) of an infected person. Even simple communication can lead to infection. Over the past 2 years, facemasks have been crucial in controlling COVID-19. Using facemasks as a non-pharmaceutical method is a cost-effective way to reduce respiratory infections, as they can block 68%–91% of microbe transmission. However, it’s important to safely discard used facemasks, as they can become a potential source of further infection. Researchers are working on enhancing the antimicrobial properties of masks to eliminate microbes and biohazard waste. Surgical masks are often treated with chemicals, antibiotics, and finishes to improve their antimicrobial effectiveness.3,4 The challenge with eliminating viruses using liquid-based chemicals, such as alcohols, is their lower boiling points, making it difficult to incorporate them into facemasks. On the other hand, antibacterial agents can be incorporated into facemask fibers to eliminate bacteria. However, many surgical masks currently available do not have antibacterial properties and may hurt the respiratory system, potentially causing toxicity and allowing harmful bacteria to enter the mouth. 5 Developing a surgical mask with enhanced antibacterial functions while upholding its air permeability and filtering qualities is of utmost importance. Considering the critical role of masks in safeguarding public health and contributing to sustainable development, it is imperative to pursue advancements in this area. Such improvements would enhance protection against bacterial threats and ensure the mask remains comfortable and functional for extended use.6–9
Facemasks are mostly made from synthetic materials such as polypropylene-based spun bond fabrics. These fabrics have low moisture interaction and smaller pore sizes, making them uncomfortable for everyday use. On the other hand, cotton fabric is widely used in clothing and textiles due to its flexibility and eco-friendly properties, and it is commonly used worldwide.10–13 Cotton is the most significant textile industry crop.14–16 It’s clear that while wool, silk, and nylon have been used in some regions for a long time, cotton has surpassed all other natural materials in terms of flexibility and comfort. Regarding face masks, using cotton-based, nonwoven fabrics can enhance comfort, especially considering their continuous interaction with environmental conditions.17,18
Nanotechnology is a field that enables the precise manipulation and construction of materials at the nanoscale level, typically ranging from 1 to 100 nm. Using advanced techniques and tools, such as molecular self-assembly and nanofabrication methods, nanostructures and microstructures with an exceptionally large active surface area can be developed. These structures possess unique chemical properties and exhibit remarkable reactivity, even at extremely low concentrations. This characteristic makes them particularly valuable for various applications, including drug delivery, environmental remediation, and advanced materials development. 19 Nano and microparticles are studied due to their high surface area/volume ratio, which makes nanomaterials suitable for filtration and biomedical products such as wound dressings, face masks, drug delivery systems, and surgical sutures.20–23 Many research studies have been conducted to improve the properties of facemasks, including the number of layers, different additives, functional nanofibers, and manufacturing techniques. Various methods have been used to apply these nanoparticles onto the substrate, such as dip coating, spray coating, ultrasonication, pad-dry curing procedure, in-situ chemical reduction, and covalent bonding procedures.17,24–26 In addition, metallic nanoparticles such as Ag, ZnO, Cu-based compounds, and TiO2 are utilized in producing antimicrobial textiles due to their potential antibacterial properties.12,15,27–32
ZnO particles are biocompatible, exhibit long-term stability, and possess great mechanical durability. Their simple production process has led to their development as biocidal materials.33,34 Numerous studies have demonstrated the effectiveness of Zinc Oxide (ZnO) as an antibacterial coating for surfaces that could be contaminated with bacteria or SARS-CoV-2.35,36 It is widely recognized that zinc (Zn) and zinc oxide (ZnO) are particularly beneficial as supportive treatments for COVID-19 infection. 37 Research has shown that using a variety of polymers electrospun with inexpensive ZnO NPs can be beneficial for creating protective apparel. ZnO-infused polyvinyl pyrrolidone (PVP) and PVA nanofibers have demonstrated antibacterial effectiveness against S. aureus, E. coli, K. pneumoniae, and S. aeruginosa strains. 38
Most antibacterial products are chemical-based, which can lead to toxicity and other negative effects. However, natural products can replace these chemicals in an environmentally friendly way that is compatible with human use. Azadirachta indica, also known as “Neem,” is a tree native to India and belongs to the Meliaceae family of plants, commonly known as the mahogany family. 39 Previous research on A. indica revealed that the whole plant contains active compounds with various therapeutic properties, including seeds, leaves, roots, bark, trunk, and branches. 40 Several therapeutic benefits are associated with the A. indica plant, including antibacterial, antioxidant, anti-inflammatory, anti-diabetic, anti-cancerous, anti-HIV/AIDS, and antihypertensive effects. 41 A. indica is a natural product that is mixed with different polymers. It has been widely known as a medicinal plant due to its excellent microbial resistance and is used by researchers for the green synthesis of nanoparticles.42–45
There is limited research on loading A. indica extract with Zinc oxide (ZnO) particles/nanoparticles onto cotton nonwoven fabric to improve its antibacterial properties. This study aims to develop nonwoven cotton fabric coated with A. indica extract and ZnO particles using needle punching and calendaring techniques. Here, we developed an antibacterial three-layer facemask by adjusting the concentrations of ZnO (0, 2, 4 wt.%) and the concentration of A. indica extract (0, 15, 30 wt.%). The properties of the samples were analyzed for air permeability, thermal resistance, and tensile strength. Additionally, the antimicrobial activity of the prepared samples was assessed.
Materials and methods
Materials
Azadirachta indica leaves were sourced from the botanical garden of the University of Agriculture in Faisalabad, Pakistan. Long staple American cotton was obtained from Suraj Cotton Mills in Shahkot. Lab-grade Zinc Acetate Dihydrate and Sodium hydroxide were purchased from Sigma Aldrich to synthesize Zinc oxide particles.
Methodology
Step I involved preparing the extract of A. indica leaves by washing 50 g with distilled water, drying them at room temperature, and heating them in 500 mL of distilled water at 95°C for 30 min in a closed steel jar. The resulting mixture was filtered using a vacuum filtration assembly to obtain the extract, which was then stored in a clean conical flask for further use.
In Step II, aqueous zinc acetate dihydrate 0.02 M was added to 200 mL of deionized water in a beaker and stirred at room temperature. Then, 3–5 drops of extract were added to the beaker and stirred for 15–20 min. The mixture was treated with 2.0 M aqueous NaOH and left for stirring until a white solution with a pH of 12 was obtained, which was then agitated for 2 h. The precipitates were filtered and washed with distilled water/ethanol, and the white powder of ZnO was obtained after drying at 60°C in a vacuum oven.
In Step III, nonwoven sheets were prepared using cotton through needle punching. Long-staple cotton was chosen due to its low density and good tensile properties. Aqueous solutions were formed by varying the ZnO concentrations (0, 2, 4 wt.%) while maintaining three concentrations of A. indica extract (0, 15, 30 wt.%).
Subsequently, the nonwoven sheet was soaked in ZnO/extract solutions prepared through the dip coating method. Eight different samples were prepared using varying concentrations of extract and ZnO. Table 1 displays the sample codes for the different plant extract and ZnO concentrations. The sample names consist of two parts: the first part is alphabetical and represents the additive name, while the numeric part indicates the concentration of the additive. In this case, A. indica concentration is denoted by N (based on the local name: Neem), and ZnO is denoted by Z. Finally, the coated nonwoven substrate was embedded into a Polypropylene surgical face mask with one layer of propylene above it, and the second layer below it. All three layers were sewn along the edges, as shown in Figure 1 (Step 3).
Sample codes with varying A. indica extract and ZnO concentrations.

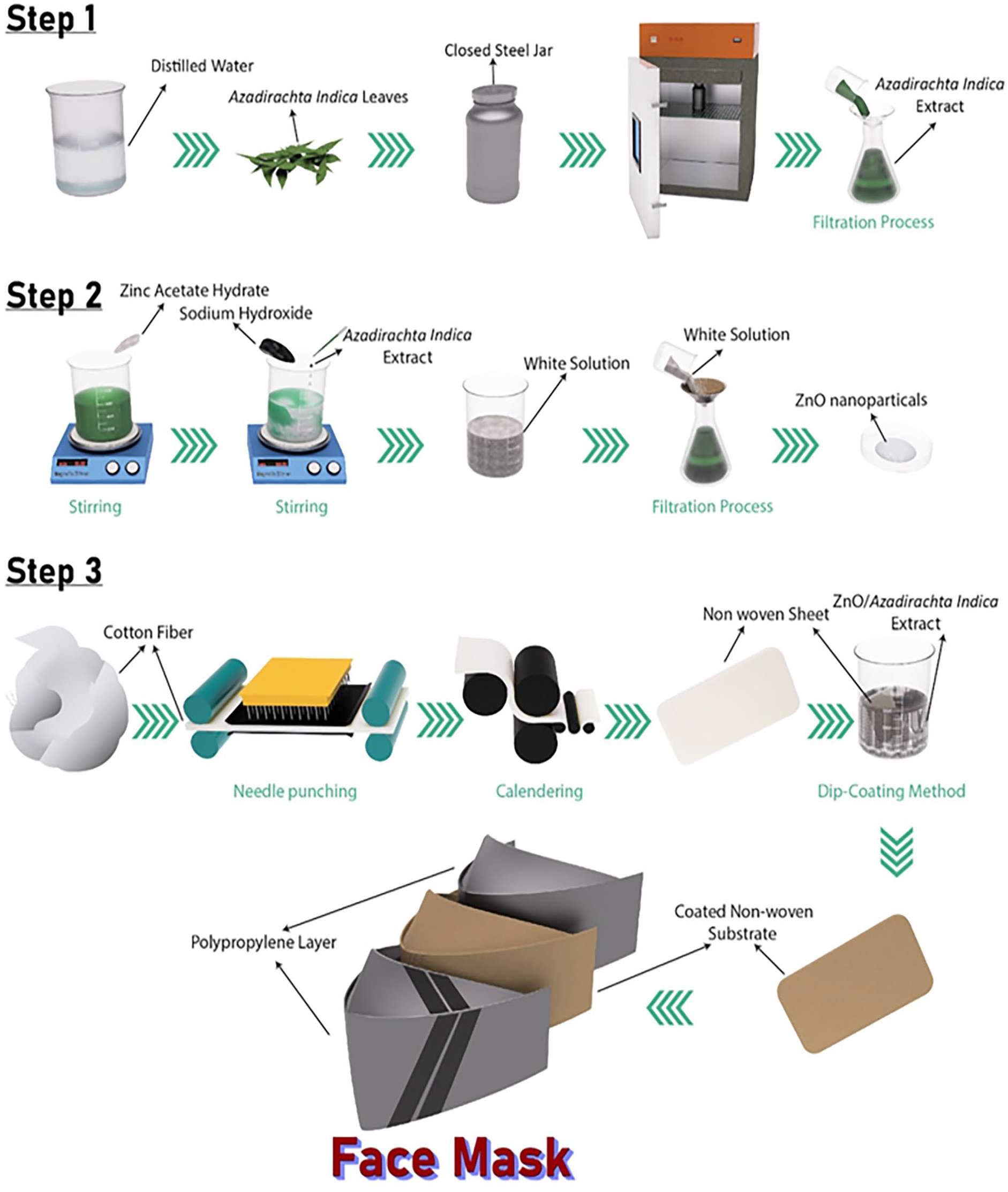
Development of sample facemasks with antibacterial properties.
Characterization
The SDL Atlas air permeability tester was used to measure air permeability following the ASTM D737 standard protocol in South Carolina, USA. The ATC 147 standard protocol was used to test the antibacterial activity of samples. In this method, the sample was placed on the Sabouraud agar plate, and a culture of S. aureus bacteria was grown on the plate. The plate was placed inside an incubator at 37°C for 24 h. After 24 h, the containers were checked, and their antimicrobial activity was studied. The transparent area, which indicated the inhibition of bacterial growth, was observed with the naked eye. This transparent region is called the zone of inhibition, and it determines the efficiency of the antibacterial activity. Scanning electron microscopy (SEM) produces images by illuminating a sample’s surface with a high-energy electron beam and then detecting signals from the interaction of the incoming electrons with the sample surface, which are translated to determine the morphology of nanostructures.46,47 The surface morphology and elemental analysis of the ZnO particles were done through a Hitachi S-3500-SEM-EDAX. The fabric’s thermal resistance was tested using PERMETEST, manufactured by Sensor Instruments & Consulting, following ISO 11092 standards.
The value of the zeta potential indicates the possible behavior of the dispersion. Coagulation is considered as good whose zeta potential is between −30 and +30 mV. 48 The surface zeta potential was measured according to a technique devised by Malvern Instruments (Zeta Sizer, model Nano-ZS; zeta potential = −42.0 ± 4.2 mV, pH = 9.2). The maximum sample size allowed for surface zeta potential measurement was 4 mm × 7 mm × 1.5 mm (L × W × H). 49 The ZnO powder (refractive index 2) was dispersed in the ethanol solution (refractive index 1.33) and kept in an ultrasound bath for about 2 h. Then, the particle size and distribution of the ZnO mixture were measured.
The tensile strength and elongation of the fabric were determined using the Lloyd LRX with ASTM D 5035-95 standard protocol. The environmental conditions in the testing area were maintained at 21 ± 1°C and relative humidity of 65% ± 2%. The standard test method for breaking force and elongation of textile fabrics was followed using the strip method. For each sample, a strip of size 200 mm × 50 mm (L × W) was tested in both the machine direction (MD) and cross direction (CD).
Results and discussion
Particle size distribution of ZnO particles
Figure 2 illustrates the particle size distribution of the as-prepared ZnO. The average size of the ZnO particles was approximately 638 nm, with relative intensities of 96.4%. There is a second peak at 5700 nm with a relative intensity of 3.6%, indicating that a small percentage (3.6%) of ZnO particles have sizes greater than most particles (96.4% having size of 638 nm). The average diameter of mature cotton fibers is approximately 10 µm. The ZnO particles are in the nanoscale range and can adhere to the ellipsoidal cotton fibers with diameters.

Size distribution chart of synthesized ZnO particles by Malvern Instruments Inc. Zeta Sizer, model Nano-ZS.
Table 2 displays the weight gain of different coated and uncoated nonwoven cotton samples with varying concentrations of A. indica extract and ZnO. Notably, when A. indica extract concentration is used, the samples experience greater weight gain compared to ZnO particles. This is because most of the neem extract is cellulose-based, which allows for better interaction with cotton through hydrogen bonding than that of ZnO particles.
The weight gain of coated nonwoven cotton samples with varying A. indica extract – ZnO concentrations.

Air permeability
Figure 3 illustrates the air permeability of samples from MA, US, and coated materials with varying concentrations of ZnO particles (0, 2, 4 wt.%) and A. indica extract (0, 15, 30 wt.%). The US sample, made with long staple cotton, exhibits the highest air permeability at 826 mm/s due to its cellulose-based cotton structure and lower-density fibers. The needle-based nonwoven structure also allows for better air passage than the MA 3-layer polypropylene facemask, which has a tightly spun bond nonwoven structure with better fiber packing, reducing air permeability at 525.2 mm/s. The N0/Z2 and N0/Z4 samples, treated with 2 and 4 wt.% ZnO particles, demonstrate reduced air permeability compared to the US sample, measuring 785 and 784 mm/s, respectively. 50 The use of A. indica extracts significantly impacts air permeability compared to the ZnO-treated samples. There is a notable reduction in air passage, decreasing from 785 to 750 mm/s in the N0/Z2 sample. Furthermore, as the amount of ZnO particles increases, the air passage is further reduced, with an air permeability of 734 and 732 mm/s for samples N15/Z2 and N15/Z4, respectively. Interestingly, the impact of ZnO particles on air permeability remains consistent when increasing their concentration from 2 to 4 wt.%. This behavior is observed even without the A. indica extract, as N0/Z2 and N0/Z4 have air permeability of 785 and 784 mm/s, respectively.

Air permeability of MA, US, and coated samples with varying concentrations of N: A. indica extract (0, 15, 30 wt.%) and Z: ZnO particles (0, 2, 4 wt.%).
These findings suggest that incorporating ZnO particles affects the arrangement of cotton fibers, but higher concentrations do not further impact air permeability. Additionally, the results indicate that increasing the neem extract concentration to 30 wt.% (samples N30/Z0, N30/Z2, N30/Z4) leads to a decrease in air permeability compared to an increase in ZnO alone. The minimum air permeability is measured in the N30/Z4 sample, with a value of 662 mm/s. This decrease in air permeability is attributed to the higher amount of A. indica extract and ZnO particles absorbed in the nonwoven cotton fabric, which blocks the fabric pores.
SEM analysis
The SEM micrographs in Figure 4 confirmed the morphology of ZnO. Three images were taken at different scales to evaluate ZnO particle shape and distribution comprehensively. An enlarged image captured at a scale of 500 nm (Figure 4(a)) revealed that the ZnO nanoparticles exhibited various shapes, including spherical, hexagonal, and irregular. The concept of particle distribution refers to their spatial arrangement. A uniform distribution of particles, characterized by even dispersion, guarantees consistent properties. The image in Figure 4(b) at a scale of 200 nm displayed well-dispersed ZnO particles without significant clustering, indicating homogeneous distribution. Figure 4(c), taken at a 1 μm scale, provided valuable insights about the surface coverage and density of the ZnO particles. The images captured at various scales facilitated the visualization of individual particles and their collective arrangement, contributing to the comprehension of the distribution patterns. The EDAX (Energy Dispersive X-ray Spectroscopy) spectrum in Figure 4(d) confirmed the sample’s elemental composition. The analysis identified two elements, zinc (Zn) and oxygen (O), confirming the presence of ZnO particles on the fabric’s surface.

(a–c) SEM images of ZnO particles at scales of 500 nm, 200 nm, and 1 μm, respectively and (d) EDAX of the sample shows elemental composition.
Thermal resistance test
The thermal resistance of the facemask samples, shown in Figure 5, includes samples coated with varying concentrations of ZnO particles and A. indica extracts. Thermal resistance is the ratio of the temperature difference between two faces of a material to the rate of heat flow per unit area. According to Figure 5, the US facemask exhibits a higher thermal resistance of 0.029 m2K/W than the MA facemask of 0.014 m2K/W. This higher thermal resistance is attributed to the fabric’s high gram per square meter and thickness, reducing the surface area for heat conduction. Additionally, cotton fibers are bulkier than polypropylene fibers, allowing for more trapped air inside the fabric, which hinders heat transfer. Furthermore, the heat conduction of cotton (0.026 W/mK) is lower than that of polypropylene (0.11 W/mK). However, the moisture regain of cotton is 8.5%, significantly higher than the 0.3% of polypropylene. This higher moisture regain of cotton provides better comfort properties than polypropylene, making the wearer feel more comfortable despite the higher thermal resistance of cotton. This advantage is particularly beneficial in colder climates.

Thermal resistance of MA, US, and coated samples with varying conc. of [N: neem extract (0, 15, 30 wt.%) and Z: ZnO Particles (0, 2, 4 wt.%)].
The N0/Z2 and N0/Z4 samples exhibit the same thermal resistance of 0.032 m2K/W, consistent with air permeability results. However, the thermal resistance increases by adding ZnO particles and neem extract to the facemasks. Specifically, N15/Z0 and N15/Z2 show a thermal resistance of 0.035 m2K/W, while N15/Z4 has a thermal resistance of 0.037 m2K/W. Moreover, N30/Z0, N30/Z2, and N30/Z4 demonstrate an increase in thermal resistance, measuring 0.039, 0.041, and 0.042 m2K/W, respectively. This increase can be attributed to the higher absorbed amount of A. indica extract and ZnO particles in nonwoven cotton, resulting in a thicker fabric and increased weight gain. Since all samples contain cotton, the heat conduction and resistance properties are the same. However, the thermal resistance is directly proportional to fabric thickness, indicating that treated samples resist more heat than untreated samples. The highest thermal resistance of 0.042 m2K/W is achieved by N30/Z4.
Antimicrobial activity
Studies have been conducted on the phytochemical composition and antibacterial capabilities of A. indica against various bacterial species. It has been revealed that the antibacterial activity of A. indica in the leaf, stem, root, and bark extract is due to the presence of phytochemicals such as phenolics, tannins, saponins, flavonoids, alkaloids, terpenoids, fatty acids, glycosides, etc. A. indica has been found to have antibacterial effects against a variety of bacteria (Gram positive and Gram negative) including K. pneumoniae, S. aureus, S. typhi, E. coli, S. pyogenes, etc. 51 The primary active ingredient in A. indica plants is called Azadirachtin, commonly used as a biological marker for this species. It is mainly seen in seeds and trace amounts in leaves. Azadirachta indica extract contains gedunin, nimbin, azadiractol, melianone, meliantrol, azadirone, vilosinin, and numerous other active ingredients. 51 Fabrics treated with zinc oxide (ZnO) nanoparticles demonstrate enhanced antibacterial activity as nanoparticle deposit increases and particle size decreases. Smaller particles are anticipated to be more effective against microbes due to their larger surface area-to-volume ratio. 52
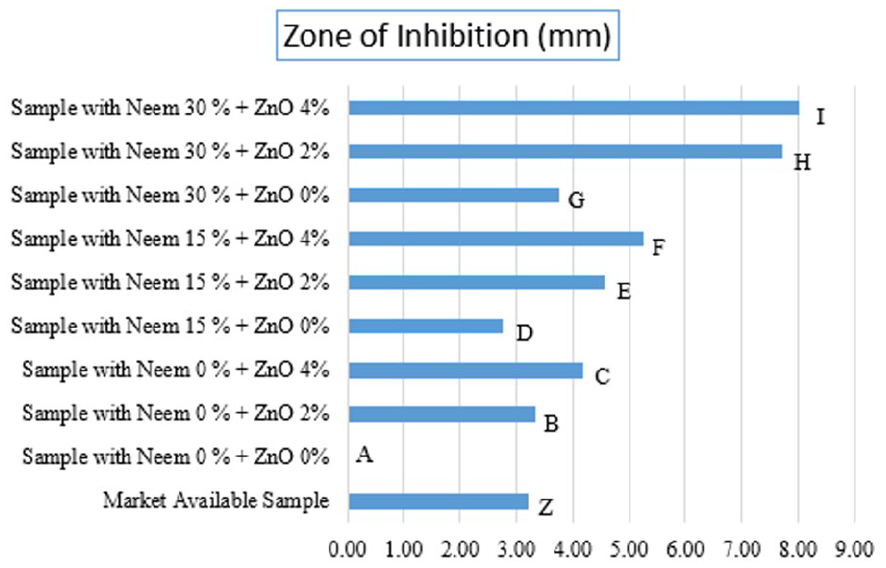
The zone of inhibition for MA, untreated (A), and coated samples with different concentrations of ZnO particles (0, 2, 4 wt.%) while maintaining the content of neem extract at 0, 15, and 30 wt.% against S. aureus. The untreated sample A does not show any zone of inhibition, while every other sample shows a clear zone of inhibition. However, the samples with a greater concentration of ZnO particles show higher antibacterial potential than the neem extracts. It is also interesting to note that the samples containing both ZnO and A. indica extract added up the zone of inhibition. Both agents eradicated the bacteria synergistically and enhanced the antibacterial activity.
Samples N0/Z4, N15/Z4, and N30/Z4 exhibited 4.19, 5.25, and 8.03 mm inhibition zones due to higher ZnO particle content. On the other hand, samples N0/Z2, N15/Z0, N30/Z0, N30/Z2, and N30/Z4 showed 3.35, 2.76, 4.56, 3.76, and 7.73 mm zones of inhibition, as illustrated in Figure 6. It was observed that ZnO particles, which have increased bacterial resistance, are safe to use in small amounts due to their photo-oxidizing and photocatalytic effects on chemical and biological entities. When these particles come into proximity with bacterial cell walls, they disrupt the structural stability of the cells, release antimicrobial ions (mainly Zn2+ ions), or generate reactive oxygen species, thereby enhancing the antimicrobial effect of ZnO particles compared to A. indica extract. 17
Antimicrobial potential of commercially available, untreated, and synthesized face mask.
However, the combination of A. indica and ZnO particles still performs better and significantly helps to stop bacterial growth, as shown in Figure 7. It is also interesting that the MA samples showed a zone of inhibition of 3.22 mm, while a similar outcome by N0/Z2 (minimum concentration of ZnO particles without A. indica) is 3.35 mm, and N15/Z0 (with minimum A. indica concentration and no ZnO) is 2.76 mm. This indicates that samples containing the minimum amount of ZnO and A. indica extracts are effective in antibacterial activity, similar to the MA. In addition, the N30/Z4 shows extraordinary antibacterial activity with an inhibition zone of 8.03 mm, and the rest of the samples fall in the same range, indicating unusual antibacterial activity.

Zone of inhibition of treated, untreated, and market-available samples.
Tensile properties
The results presented in Figure 8(a) and (b) depict the elongation (%) and tenacity (g/tex) of MA facemask samples, both untreated and treated with varying concentrations of ZnO particles (0, 2, 4 wt.%) and A. indica extract (0, 15, 30 wt.%) along the cross direction (CD) and machine direction (MD).

(a) Tensile elongation of MA, US, and coated sample and (b) tenacity graph of MA, US, and coated samples.
Figure 8(a) shows that the untreated sample (US) exhibits 84% elongation along the CD and 75% along the MD. Interestingly, the elongation increases with the concentration of A. indica extract, attributed to the natural compatibility of A. indica particles and cellulose molecules. This leads to physical crosslinking between cotton fibers and extended elongation. Conversely, the impact of ZnO on increasing elongation is limited, as it remains on individual fibers without making physical connections between them. Among all the samples, N30/Z4 shows the highest elongation % (92 along CD and 84 along MD). In contrast, MA shows 65% elongation along CD and 49% along MD due to its higher crystalline structure as a spun bond polypropylene fabric. Additionally, MA exhibits higher tenacity values (0.92 g/tex in CD and 0.90 g/tex in MD) than the US sample (0.74 g/tex in CD and 0.70 g/tex in MD).
Figure 8(b) shows that samples coated with lower ZnO particles and A. indica extract concentrations (N0/Z2, N0/Z4) have lower tenacity values in both CD and MD, which increase as the neem extract treatment concentration increases. The tenacity results suggest that A. indica extract causes some crosslinking between cotton fibers, resulting in better mechanical properties than the US sample. Notably, the MA facemask and N30/Z4 show similar tenacity values, indicating the effectiveness of the treatment.
Conclusions
The study aimed to develop an antibacterial facemask by adjusting the concentrations of ZnO particles (0%, 2%, 4%) and A. indica extract (0%, 15%, 30%) in the cotton nonwoven fabric. The fabric was prepared by needle punching and calendaring, dipping in ZnO and A. indica extract solutions, and drying. The mean diameter of the ZnO particles was 638 nm. SEM micrographs confirmed the presence of ZnO particles with varied shapes and their even distribution. The thermal resistance of facemasks increased with A. indica extract and ZnO particle concentrations due to the increased fabric weight, which restricted heat flow. Air permeability decreased with A. indica and ZnO concentrations due to better fiber interactions and filling the gaps with A. indica extract and ZnO. The sample treated with 30% A. indica and 4% ZnO exhibited the greatest values in terms of elongation (92% CD, 84% MD), tenacity (0.93 g/tex CD, 0.87 g/tex MD), and thermal resistance (0.042 m2K/W). The antimicrobial activity with 30% A. indica and 4% ZnO in nonwoven was excellent, with N30/Z4 showing the highest zone of inhibition (8.03 mm) against Staphylococcus aureus. Furthermore, the source materials were based on green synthesis, making them beneficial for preparing face masks against microbes. This work also suggests exploring other natural and green synthesis-based materials for future facemask development. This could involve studying different herbs and plants for manufacturing green nanoparticles and identifying more potent antimicrobial plants/materials. Changes in GSM, thickness, layers, and fabric compactness could also enhance breathability and comfort properties by altering thermal resistance and air permeability.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are thankful to the Pakistan Science Foundation (PSF) under the grant number (PSF/CRP/P-NTU-HELIX-194) and Higher Education Commission, Pakistan, under the grant code (20-16082/NRPU/R&D/HEC/2021-2020) for funding this work.
